Abstract
Atrial fibrillation (AF) is the most common rhythm disturbance seen in clinical practice, and its prevalence and incidence are rising rapidly as the population ages with its attendant complications. Management of AF involves anticoagulation, and fortunately new drugs for long-term anticoagulation are now available. Maintenance of sinus rhythm, though intuitively better than rate control strategy, has not been shown to offer mortality benefit. Still, maintenance of sinus rhythm is considered an appropriate therapeutic strategy when symptoms are not adequately controlled with rate control. Though significant advances have been made in ablation techniques for AF, pharmacological therapy is still the first line of treatment for rate control and maintenance of sinus rhythm, given ease of use, noninvasive nature, and limited experience with catheter-based ablation techniques. Class IC and III agents (Vaughan Williams classification) form the backbone for pharmacological maintenance of sinus rhythm. Dronedarone, a recently approved class III agent, provides a significant advance because of its relatively safe side effect profile. Currently drugs with selective atrial channels blocking properties, like Vernakalant, are being tested in trials and may provide an opportunity to maintain sinus rhythm with limited toxicity. Large trials are also being conducted to better define the efficacy of catheter-based ablation strategy as first-line treatment. Here, we review the current status of commonly used antiarrhythmic medications for the maintenance of sinus rhythm in AF.
Introduction
Atrial fibrillation (AF) is the most common clinically significant rhythm disturbance seen in clinical practice. Though, AF does not carry the immediate mortality associated with its ventricular counterpart, ventricular fibrillation, it carries clinical relevance because of its effect on long-term mortality, quality of life, and functional status of patient. Both the incidence and prevalence of AF are rising in epidemic proportions. Current estimates for the number of people afflicted with this disease in the United States are in the range of 2 to 3 million, 1 and this number is predicted to cross 10 million by the year 2050. 2 This alarming rise in the disease burden is likely to cause constraint on medical resources. No doubt that primary prevention strategy, targeting the population at risk will help in reducing the rising number of AF patients; at the same time, there is a requirement to address the treatment needs of an individual patient with AF.
Management of AF
Treatment of the AF involves a multifaceted approach. Three basic strategies involved are rate control, rhythm control, and therapeutic anticoagulation. In this article, we will focus on the pharmacological maintenance of sinus rhythm, and briefly mention the status of catheter-based ablation therapy as well as the current status of anticoagulation therapies. We review the status of commonly prescribed antiarrhythmic drugs (AADs) in current clinical practice for rhythm maintenance. Though same drugs can be used in specific circumstances for the conversion of AF rhythm to sinus rhythm, this topic is beyond the scope of this review article.
From the perspective of rhythm maintenance, it is useful to classify AF into paroxysmal, persistent, and permanent types. 1 Paroxysmal AF is defined as recurrent episodes of AF lasting less than 7 days with spontaneous termination. When AF lasts more than 7 days and/or an intervention is required to maintain sinus rhythm, it is designated as persistent AF. Permanent AF usually includes patients who have been in AF for more than a year and in whom all the interventions have failed to keep a patient in normal sinus rhythm.
While managing patients with AF, one has to choose between rate control and rhythm control as the primary strategy. This is an important issue since data from large randomized trials3–7 suggest that rhythm control strategy does not provide significant benefit over rate control strategy in terms of mortality. 3 However, maintenance of sinus rhythm is the conventional wisdom, and there are data showing that sinus rhythm is better than AF.
Evidence in Favor of Maintaining Sinus Rhythm
In the large observational Framingham Heart Study, AF was associated with increased mortality, especially in women. 8 In addition, in controlled trials, such as the studies of left ventricular dysfunction (SOLVD) trial, AF has been shown to be associated with increased mortality. 9 The estimated relative risk of death associated with AF is about twice that in the general population. 10 Though there are some preliminary data from catheter ablation studies, 11 and subgroup analysis of the atrial fibrillation follow-up investigation of rhythm maintenance (AFFIRM) trial12–14 to suggest survival benefit with maintenance of sinus rhythm, 11 till now none of the trials comparing rhythm maintenance with the use of AAD to rate control strategy has shown any mortality benefit with rhythm control strategy. A large ongoing trial, catheter ablation versus anti-arrhytmic drug therapy for atrial fibrillation (CABANA) comparing the use of AAD with catheter ablation with the primary end point of mortality is likely to provide valuable information in this regard (D L Packer, MD, personal communication).
AFFIRM and related trials were inconclusive with regard to the effect of one strategy or the other on the quality of life. Data from recent studies, like sotalol amiodarone atrial fibrillation efficacy trial (SAFE-T) 15 and others,16,17 support the idea that maintenance of sinus rhythm is associated with marked improvement in functional capacity, over a 12-month period.18–20
In concurrence with improved quality of life, sinus rhythm is associated with improvement in left atrial size and function and left ventricular systolic function.21,22 This implies that maintenance of sinus rhythm may reduce remodeling in the atria and the ventricles.1,23
Indications for Rhythm Control
In general, pharmacological therapies designed to maintain sinus rhythm are preferred over catheter-based ablation and surgical therapies because of the ease of drug administration, lower cost, and absence of procedural complications associated with invasive procedures.
Though there are no clear guidelines to suggest the use of rhythm maintenance strategy in specific patient subgroups, rhythm maintenance is a preferred approach for patients with symptomatic AF, particularly in whom rate control did not control the symptoms. It is important to document that patients labeled as having “asymptomatic AF” do not have limitation in terms of exercise performance. The best time to assess symptoms is the time period immediately after cardioversion. The truly symptomatic patient would report a major improvement in his or her level of physical activity and fatigue while in sinus rhythm.
Because of the risk of tachycardia-induced cardiomyopathy in the long run and since AF begets AF, young patients with AF should also be considered for rhythm control. Per the American College of Cardiology/American Heart Association/European College of Cardiology (ACC/AHA/ESC) 2006 guidelines, the use of AAD for rhythm maintenance in these patients is a class IIa recommendation.
In patients with heart failure, although rhythm control strategy has not been shown to lower mortality as evident in the AF–congestive heart failure (CHF) study, 24 maintenance of sinus rhythm may reduce the incidence of hospitalization 25 and improve quality of life. 18 Though there are no specific guidelines specifically in the setting of CHF and AF, it is prudent to consider the use of AAD to decrease hospitalization and for symptom control.
Maintaining sinus rhythm may also help patients who are dependent on “atrial kick” and “AV synchrony” for appropriate left ventricular filling. These are patients with aortic stenosis, hypertrophic obstructive cardiomyopathy, mitral stenosis, and diastolic heart failure.
In general, cardioversion is best avoided in asymptomatic elderly patients as it may uncover sinus node dysfunction, potentially leading to pacemaker implantation. Because of this reason, ACC/AHA/ESC 2006 guidelines recommend against the use of AAD in the presence of severe sinus node or atrioventricular (AV) node disease until the patient has a functioning electronic pacemaker.
Drugs for Maintenance of Sinus Rhythm
For maintenance of sinus rhythm the first step is to choose between the drug therapy and ablation therapy. Atrial fibrillation ablation, usually catheter-based and rarely surgical, is almost never used as the first-line approach. This is likely related to the invasive nature, limited availability, cost, and small but definite risk of serious and life-threatening complications associated with ablation procedures. Currently, class IC and class III drugs form the backbone of AAD therapy in symptomatic patients for rhythm maintenance. Class IA agents such as quinidine are no longer used, because of the associated high risk of proarrhythmias and excess mortality (Tables 1 and 2 ).
Drugs for Maintenance of Sinus Rhythm

Pharmacokinetic Properties of Major Antiarrhythmic Drugs Used in Atrial Fibrillation
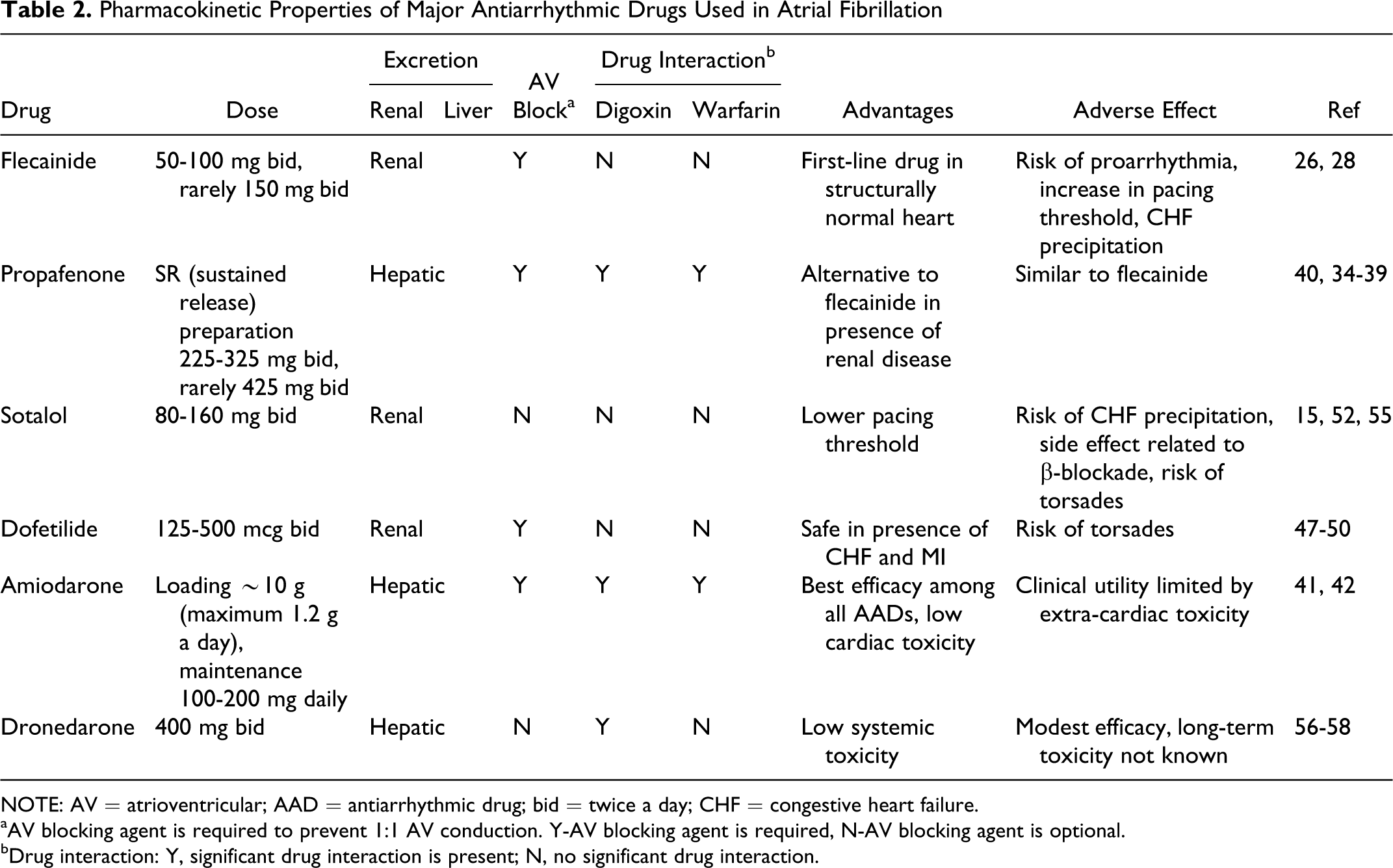
NOTE: AV = atrioventricular; AAD = antiarrhythmic drug; bid = twice a day; CHF = congestive heart failure.
aAV blocking agent is required to prevent 1:1 AV conduction. Y-AV blocking agent is required, N-AV blocking agent is optional.
bDrug interaction: Y, significant drug interaction is present; N, no significant drug interaction.
The choice of AAD for maintenance of sinus rhythm is based on whether the patient has structural heart disease or not. According to the ACC/AHA/ESC 2006 guidelines,
1
patients are subdivided into 4 categories: In patients with structurally normal heart, the drugs of choice are propafenone, flecainide, or sotalol. In case of failure or intolerance to these medications, dofetilide or amiodarone is recommended as a second-line drug treatment. In patients with coronary disease, the first-line treatment is dofetilide or sotalol. Class IC drugs are contraindicated in these patients. The second-line treatment is amiodarone. In patients with CHF or ejection fraction less than 35%, amiodarone or dofetilide is recommended. In patients with hypertension without substantial left ventricular hypertrophy, the drugs of choice are the same as in patients with structurally normal heart. In patients with substantial left ventricular hypertrophy, the drug of choice is amiodarone because of the higher risk of torsades de pointes, with other AADs in these patients.
Here, we briefly review the clinical pharmacology of the commonly used AADs. Importantly, prior to initiating any AAD therapy, the patient must have had at least 21 consecutive days of therapeutic international normalized ratio (INR), or a transesophageal echocardiogram documenting absence of left atrial or left atrial appendage clot in the setting of persistent or chronic AF or if the patient presents with first detected episode of AF and duration of AF is not known. In the setting of paroxysmal AF if the timing of onset of AF is known and the duration of episode is less than 48 hours, AAD therapy may be initiated without the need for therapeutic INR of 21 days, as the risk of embolic complications is low. In general, the chance of spontaneous or chemical cardioversion is low as the duration of episode of AF increases and DC cardioversion is generally needed for longer episodes of AF. Importantly, now with the advent of direct thrombin inhibitors, the recommendations regarding INR are bound to change.
Flecainide
Flecainide is a first-line class IC agent for the maintenance of sinus rhythm in patients with structurally normal heart. In the outpatient setting, flecainide is started at a dose of 50 mg twice a day, and 1 week later the dose is doubled to 100 mg twice a day if the QRS duration is within normal range. In the inpatient setting, flecainide can usually be started at a dose of 100 mg twice a day. In the presence of renal failure with creatinine clearance <35 mL/min, dose is reduced to 50 mg twice daily; however, propafenone is the preferred agent in the presence of chronic renal insufficiency. In rare cases, flecainide is used at the maximum dose of 150 mg twice a day, especially in obese patients.
Significant adverse effects associated with flecainide use are proarrhythmias, acute rise in pacing threshold, heart failure exacerbation, and conduction abnormalities. Heart failure exacerbation is more common in patients with known left ventricular systolic dysfunction 26 ; accordingly, flecainide use is not recommended in the presence of structural heart disease and its use should be discontinued if heart failure develops. Flecainide can increase pacing threshold, 27 making pacemaker check imperative after initiation of therapy. Among arrhythmias the most serious and life threatening is the wide complex tachycardia, 28 which occurs in association with the widening of QRS duration. 29 For this reason, we subject our patients to treadmill exercise 1 week after increasing the dose to 100 mg twice a day, although there are no guidelines on the timing of exercise test. Widening of QRS duration by 50% or more with exercise predicts the risk of proarrhythmias. 1 Utility of exercise test in the setting of AF is based on the property of use-dependent block of Na channel by class IC agents. 29
Precipitation of wide complex tachycardia usually requires withdrawal of the drug. In about 5% of cases, AF may convert to atrial flutter, 30 which is sometimes associated with 1:1 AV conduction due to atrial cycle length slowing. To prevent this, AADs with no significant β-blocker effect, such as flecainide, propafenone, and dofetilide, must always be used concomitantly with AV nodal blocking agents, such as β-blockers, diltiazem, verapamil, or digoxin.
Conduction abnormalities ranging from bundle branch block to AV block to symptomatic sinus bradycardia have been reported; however, this is rare and the incidence 31 is in the range of ∽2%. However, underlying bundle branch block is not a contraindication for use of flecainide. Among the noncardiac side effects, dizziness and blurred vision are quite common with the use of flecainide.
The medical community has been apprehensive about the use of class 1C AADs after the results of the cardiac arrhythmia suppression trial (CAST) trial. However, the use of flecainide for rhythm control in AF is associated with low risk of proarrhythmia, 32 if appropriate exercise testing is done. In addition, flecainide is one of the AADs approved by the US Food and Drug Administration (FDA) for the management of supraventricular tachycardias, such as AF and atrial flutter, in patients without the evidence of structural heart disease. 33
Propafenone
Propafenone is another class 1C drug, which is used in a similar subset of patients as flecainide. Propafenone is efficacious in preventing relapse of AF.34–37 In recent studies, sustained release (SR) preparation has been found to be effective for delaying the recurrence of AF,38,39 though immediate release preparation is likely to have similar efficacy for rhythm maintenance even though comparative efficacy has not been studied. Starting dose of propafenone SR is usually 225 mg twice a day in the outpatient setting and increased to 325 mg twice a day in 1 week if tolerated. Rarely, it is used at 425 mg twice a day dose, especially in obese patients.
In contrast to flecainide, propafenone has primarily hepatic metabolism and no renal clearance 40 ; thus, it can be used safely in the setting of renal insufficiency but is contraindicated in the presence of severe liver disease. Propafenone also has a propensity to cause use-dependent proarrhythmia, occurring as wide complex tachycardia. 40 Propafenone use is associated with QRS prolongation, which can vary from 15% to 25%, with normal therapeutic concentrations. 40 Dose adjustment needs to be done if QRS duration becomes prolonged more than 50% of the baseline, either at rest or with exercise stress test.
Amiodarone
Amiodarone has the best efficacy in maintaining sinus rhythm and prevents recurrence of AF in about two third of patients 41 (Table 3 ). Amiodarone is classified as a class III agent because of its K+ channel blocking properties; however, it also has class I, II, and IV electrophysiological properties. Amiodarone is available for both oral and intravenous administration. Amiodarone usually requires a loading dose, about 10 g, which can be given in divided doses over 1 to 2 weeks, with a daily maximum dose of 2.2 g. However, a patient should not receive more than 400 mg/d as an outpatient, unless the patient has a pacemaker or implantable cardioverter defibrillator (ICD). Once the loading phase is over, daily maintenance dose of 200 mg/d is recommended. 42 Amiodarone loading can be initiated in the ambulatory setting, at a dose of 400 mg/d for 1 month and then 200 mg daily. A 12-lead electrocardiogram should be done 1 week after starting the drug to assess QT prolongation.
Efficacy of Commonly Used AAD for Rhythm Maintenance a

NOTE: Afib = atrial fibrillation; AAD = antiarrhythmic drug; bid = twice a day.
aα included patients with paroxysmal Afib and flutter, and β 54% participants in this trial had persistent Afib.
Clinical use of amiodarone is at times limited by its multisystem extra-cardiac toxicity. Amiodarone is safe to use in the presence of renal failure, but its use should be avoided in the presence of liver disease. Amiodarone interferes with hepatic metabolism of digoxin and warfarin, and thus the dose of these medications when used concomitantly should be reduced. Excessive sinus bradycardia is the most common cardiac side effect and usually responds to dose reductions and avoidance of other sinus node suppressing agents. 43 QT prolongation, though seen in a majority of patients, is associated with a low incidence of torsades de pointes, with the reported incidence of <0.5%, which is less than the other pure class III agents like sotalol and dofetilide.44,45 Amiodarone use is associated with hypothyroidism in about 20% of patients. 42 Drug-induced hypothyroidism is easy to treat and usually requires levothyroxine supplementation 46 and generally does not require discontinuation of amiodarone. Hyperthyroidism is less common with a reported incidence of about ∽3%; but this is difficult to treat and often requires discontinuation of amiodarone therapy. 42 To monitor for thyroid and liver toxicity, thyroid function tests and liver function tests (aspartate aminotransferase [AST] and alanine aminotransferase [ALT] levels only) should be assessed at baseline, at 6 weeks, and then every 6 months.
Because of the concern for irreversible pulmonary toxicity with amiodarone’s long-term use, baseline pulmonary function tests with diffusion capacity assessment and a chest X-ray should be done in every patient and repeated yearly. Amiodarone should be stopped only if a severe decrease in diffusion capacity occurs or amiodarone toxicity is clinically suspected. Eye examination should be done only if symptoms develop during treatment with amiodarone. Severe sinus node dysfunction may preempt the use of amiodarone for rhythm control because of negative chronotropic action of amiodarone. In certain patients, especially the elderly, dose can be reduced to 100 mg or even to 50 mg/d to decrease the incidence of severe sinus slowing. Amiodarone should not be used in the presence of advanced lung disease; however, chronic obstructive lung disease in itself is not a contraindication for its use.
Amiodarone is not approved by FDA for rhythm control, probably because of its extra-cardiac toxicity. Amiodarone is recommended only after therapy with class 1C or other class III drugs has failed in patients without structural heart disease. Due to its long-term toxicity, in younger patients (<70 years of age) with symptomatic AF, who have failed other class IC and class III agents, amiodarone may be offered as an alternative to ablation therapy. In patients with significant structural heart disease in whom sotalol and dofetilide are not contraindicated, amiodarone should still be reserved as the last resort, except in elderly patients (>70 years), where long-term toxicity is less of a concern and the risk of torsades de pointes with pure class III drugs is higher. Only in patients with left ventricular wall thickness >1.5 cm, should amiodarone be used as a first-line therapy because all other antiarrhythmics with the possible exception of dronedarone are contraindicated due to the increased risk of torsades de pointes in this population. 1
Dofetilide
Dofetilide is a pure class III K+ channel blocking drug. 47 In European and Australian multicenter evaluative research on atrial fibrillation dofetilide (EMERALD) 48 and the symptomatic atrial fibrillation investigative research on dofetilide (SAFIRE-D) 49 trials, dofetilide was shown to be effective in the maintenance of sinus rhythm. Also based on the subset analysis of patients with AF in the Danish investigators of arrhythmia and mortality of dofetilide (DIAMOND) trial, 50 dofetilide was found to have efficacy in maintaining sinus rhythm. Dofetilide is administered orally in doses varying from 125 to 500 µg twice daily. Dofetilide is almost completely absorbed with reported bioavailability of ∽90%. It is cleared by the kidneys and therefore requires dose adjustment in the presence of renal failure. 51
We utilize the following formula for measuring creatinine clearance to prevent overestimation of creatinine clearance and thus potential toxicity with the use of dofetilide and sotalol in obese patients:
Sotalol
Sotalol is another class III antiarrhythmic drug, which in addition to blocking K+ channel also has nonspecific β-blocking effect. Sotalol has 100% bioavailability and since it is almost entirely excreted by the kidneys, it requires cautious use in the presence of renal failure. It can be safely used in the presence of liver failure and is the preferred drug in the presence of ICD since it does not raise the defibrillation threshold. 52 In addition, because of its β-blocking activity, it provides excellent ventricular rate control during recurrence of AF episodes. 52 Further, sotalol does not have any significant drug–drug interactions, 52 making it relatively easy to use, particularly when patients are taking many drugs.
Side effect profile is related to β-blockade, and more importantly the risk of torsades due to reverse use dependency. Risk of torsades is dose related and is higher in the presence of renal dysfunction and has been reported in the range of 2% to 3%. Therefore, patients who are started at the dose of 120 or 160 mg twice a day must be admitted to the hospital for at least 48 hours. Sotalol has been shown to have fair efficacy in maintaining sinus rhythm in placebo controlled studies.53,54 In the SAFE-T trial comparing sotalol with amiodarone, amiodarone was superior to sotalol for maintenance of sinus rhythm, though in subgroup analysis, patients with ischemic heart disease had similar response to sotalol and amiodarone for rhythm maintenance. 15 Sotalol 55 should not be used in patients with left ventricular ejection fraction <25%, as it may lead to acute decompensated heart failure.
Dronedarone
Dronedarone is a relatively new AAD approved for rhythm control in AF. Dronedarone is structurally similar to amiodarone with the notable exception of a lack of iodine on the side chain, which results in much fewer noncardiac side effects as compared with amiodarone. Like amiodarone, it has all 4 classes of action. It is recommended in oral dose at 400 mg twice daily and can be started in the outpatient setting. 56
Dronedarone use has been shown to result in lower rates of hospitalization,57,58 driven largely by lower incidence of hospitalization for AF recurrence. Even though it does not influence the overall mortality, 58 its use results in a significant decrease in death from cardiovascular causes, mainly because of a decrease in death from arrhythmia. Dronedarone also has efficacy in controlling ventricular rate during the recurrence of AF episodes. 59
Though dronedarone may not represent the “ideal rhythm” control therapy, it does represent a significant advance in terms of a decrease in systemic toxicity associated with drug therapy of AF. Given the current clinical data, dronedarone may be used as a first-line agent in patients with structural heart disease with ejection fraction >35% and no evidence of advanced heart failure (based on the results of European trial of dronedarone in moderate to severe congestive heart failure [ANDROMEDA] trial), 60 especially in young patients where systemic toxicity may be a concern with long-term amiodarone use. It also represents a good first-line alternative to dofetilide and sotalol in patients who do not have advanced heart failure and have ejection fraction >35%.
Guidelines for Long-Term Maintenance of Sinus Rhythm
Per the ACC/AHA/ESC 2006 guidelines, it is a class IIa recommendation to target maintaining sinus rhythm and to prevent tachycardia-induced cardiomyopathy. Though the guidelines address use of flecainide, propafenone, and sotalol, giving them class IIa recommendation for use in appropriately selected patients, amiodarone and dofetilide use is based on expert consensus. Since dronedarone has been recently approved by FDA for rhythm maintenance, the 2006 guidelines do not specifically address its use for AF.
Recently published European Society of Cardiology clinical practice guidelines for AF give class I recommendation for use of flecainide, propafenone, amiodarone, dronedarone, and sotalol for rhythm maintenance in appropriately selected patients. 61 Notable exception is the dofetilide, the use of which has not been addressed in these guidelines as this drug is not available in Europe.
Role of Ablation Therapy in AF
Conceptually ablation of AF represents an ideal therapy as it would be a one time procedure with curative intent and thus avoid the risk of toxicity associated with AAD use. Atrial fibrillation ablation procedure can be either surgery based if the patient is undergoing open heart surgery for some other reason or catheter based done via endovascular approach. Catheter ablation for AF has not yet become the first-line therapy for AF because of serious procedure-related morbidity and mortality. In addition, success rates for conversion of patients to sinus rhythm have been variable, in the range of ∽60% to 80% in multiple trials, 62 and similar results were reported in a recent randomized trial of paroxysmal AF. 63 Current guidelines give class IIa recommendation for catheter-based ablation for AF. In general, ablation is offered as a therapy for patients who have failed or cannot tolerate at least one first-line antiarrhythmic drug therapy. Future studies will need to address the long-term efficacy and safety concerns of catheter-based procedures, as all the randomized trials done till date have reported data up to 2 years. The details of ablation therapy are beyond the scope of this review.
Anticoagulation in AF
Therapeutic anticoagulation based on the risk stratification scoring (CHADS2) is in fact the only therapeutic intervention that has been shown to have mortality benefit in long term. 1 Oral vitamin K antagonist, warfarin, forms the backbone of this therapy till date. The use of oral warfarin is a resource-intensive strategy as it involves regular monitoring of the level of therapeutic anticoagulation. Clinical practice of oral anticoagulation may change if the results of a recent trial with oral direct thrombin inhibitor, dabigatran, 64 can be reproduced in clinical practice. In this trial, dabigatran was shown to be noninferior to warfarin. Major benefit of this medication is that it does not require regular monitoring, has minimal drug–drug and drug–food interactions, and has more consistent therapeutic action as compared to warfarin. Dabigatran was recently approved by FDA for prevention of thromboembolic events in patients with AF.
Conclusion
Atrial fibrillation as a clinical entity represents a heterogeneous disease. Wherever symptoms from AF do not respond to the rate control, rhythm control should be the preferred strategy. Nonetheless, each patient with AF needs to be individualized with regard to the treatment goals and choice of therapy. Newer drugs, like ranolazine and vernakalant, 65 are being actively investigated for their potential use in the treatment of AF. Our efforts should aim at deriving maximum benefit from the therapies we already have, and at the same time devote effort and time to discover and develop newer treatments for this highly prevalent disease.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article
