Abstract
Background:
Coronary artery disease is the most prevalent manifestation among cardiovascular diseases. Despite modern treatment, risk of ischemic complications in patients with acute coronary syndrome (ACS) remains important. The late Na+ current blocker ranolazine has shown to reduce the risk of recurrent ischemia and worsening of angina in patients with non-ST-segment elevation ACS by possibly improving myocardial perfusion, but up to now no trial has addressed whether this enhanced perfusion also leads to a decrease in ischemic myocardium of patients with ACS. We designed a pilot trial (Reduction of Ischemic Myocardium with Ranolazine-Treatment IN patients with acute myocardial Infarction, ClinicalTrials.gov Identifier: NCT01797484) for feasibility and proof of concept that a 6-week ranolazine add-on therapy would reduce the area of ischemic myocardium in patients with ACS.
Methods and Results:
The trial was designed in a 2-armed, controlled and randomized way. Twenty participants with unstable angina, proof of acute cardiac ischemia, and myocardial dyskinesia by speckle-tracking echocardiography were included. Ten participants received the study drug ranolazine additionally to standard treatment. The control group received standard treatment without additional study medication. Speckle-tracking echocardiography was performed before coronary intervention, before the first dose of ranolazine, and after 6 weeks of ranolazine treatment. Ranolazine was administered safely during acute myocardial infarction. Speckle-tracking echocardiography proved to be suitable for evaluation of myocardial dyskinesia. Patients receiving ranolazine showed a trend to higher normal fraction of the cumulative global strain than patients in the standard treatment group (15% vs 11%). No major complications relating study medication were observed.
Conclusion:
In conclusion, in this preliminary hypothesis-driven study, 6-week ranolazine therapy was shown to decrease the area of dyskinetic myocardium in patients with ACS by trend. Global strain rate measurement using speckle-tracking echocardiography can be applied measuring those effects and is, compared to other techniques, safe and harmless. Our data provide a sound basis for a follow-up trial.
Keywords
Introduction
Coronary artery disease (CAD) is the most prevalent manifestation among cardiovascular diseases and is associated with high mortality and morbidity. 4 Fortunately, mortality associated with CAD has fallen steeply in the past decades, 5 which is partly due to the shift in the type of acute coronary syndrome (ACS), with a rise in non-ST-segment elevation ACS and a decline in ST-segment elevation myocardial infarction 6 and an improvement in medical treatment. Current pharmacological treatment consists of antiplatelet therapy, β-adrenoceptor and calcium channel antagonists, nitrates, and lipid-lowering drugs. 7 But despite modern treatment, the risk of ischemic complications in patients with ACS remains important. 8
Ca2+ removal from cardiomyocytes largely depends on the Na+/Ca2+ exchanger (NCX), which is driven by the Na+ gradient. Increased intracellular Na+ levels reduce the Na+ gradient and limit the ability of NCX to efficiently extrude cellular Ca2+ hindering the cell to fully relax. Ischemic myocardium shows an increase in the late Na+ current, which can worsen left ventricular function and predispose to arrhythmias via a Ca2+ overload. 1 The late Na+ current inhibitor ranolazine has been shown to increase myocardial blood flow in animal models, which is possibly due to a decrease in left ventricular stiffness. 9 –11 Even though treatment of patients with non-ST-elevation ACS with ranolazine produced no significant benefit of ranolazine concerning cardiovascular death, MI, or recurrent ischemia during a median of 1 year of treatment, deeper analysis revealed a reduction in the risk of recurrent ischemia, worsening of angina, and fewer increases in other antianginal therapy in patients. 1 –3 Another study in patients with stable CAD and myocardial ischemia examined the hypothesis that the improvement in myocardial blood flow with ranolazine could be detected by serial automated quantitative myocardial perfusion imaging using single-photon emission computed tomography. A short-term ranolazine therapy in patients with reversible perfusion defects on exercise treadmill improved myocardial perfusion. 12
Even though ranolazine has shown to reduce the risk of recurrent ischemia, and to worsen angina in patients with non-ST-elevation ACS by possibly improving myocardial perfusion, so far no trial has addressed whether this enhanced perfusion also leads to a decrease in ischemic myocardium or increase in left ventricular function of patients with ACS. We hypothesized that an early administration of ranolazine and improvement of microcirculation in patients with ACS (ie, directly after acute ischemia) should lead to a recruitment and reuptake of cardiac activity of hibernating myocardium. We therefore designed a pilot trial (Reduction of Ischemic Myocardium with Ranolazine-Treatment IN patients with acute myocardial Infarction [RIMINI-Trial]) for feasibility and proof of concept that a 6-week ranolazine add-on therapy would reduce ischemic myocardium in patients with ACS.
Material and Methods
Patients
The study was exercised according to the principles of the Declaration of Helsinki (revised in Tokyo 1975, Venice 1983, Hong Kong 1989, Sommerset West 1996) and the International Council for Harmonisation–based Good Clinical Practice Rules. The study protocol was approved by the local Review Board for Studies in Humans. After assessment of eligibility and written informed consent, 20 participants aged between 35 and 60 years with unstable angina pectoris, proof of acute cardiac ischemia (serum levels of troponin-T-hs >14 pg/nL), angina pectoris > Canadian Cardiovascular Society Classification of Angina Pectoris II, and proof of myocardial dyskinesia were included in the study. The main exclusion criteria were refusal to participate in the study and/or parts of the study, younger than 18 years, acute cardiopulmonary decompensation, middle- and high-grade liver insufficiency (Child-Pugh score B and C), high-grade renal insufficiency (creatinine clearance >30 mL/min), homeless and drug-addicted patients, pregnant and/or breastfeeding women, treatment with ranolazine prior to enrollment, allergy against ranolazine, and concomitant use of strong CYP3A4 inhibitors or of classes Ia and III antiarrhythmics (except amiodarone). A prior MI was no exclusion criterion. All patients included in the study received standard CAD treatment based on current guidelines. 13
Study Protocol
The trial was designed in a 2-armed, controlled and randomized way. Randomization was accomplished by a paper-based method. All patients with ACS meeting the inclusion criteria and consenting to the study were included (Table 1). After stabilization of the patients, the first speckle-tracking echocardiography (SPE) was performed. The second SPE was done after angioplasty. Ten participants then received the study drug ranolazine additionally to standard treatment (β-blockers, angiotensin-converting enzyme inhibitors/angiotensin II type 1 inhibitors, acetylic acid, adenosine triphosphate receptor antagonists, statins) and were instructed about the intake scheme. Ranolazine was given orally for 6 weeks (Figure 1). In the first week, 500 mg were given bidaily. After 7 days, the dose was raised (visit 1) and patients received 750 mg ranolazine bidaily until the end of the study period at the end of week 6. The control group consisted of 10 participants receiving standard treatment without additional study medication. At the end of study week 2, all patients presented at the University Heart Center Hamburg Eppendorf for physical examination, ECG, and special laboratory test (visit 2). At the end of study week 6, the patients presented again for a last physical examination, ECG, safety, special laboratory test (blood count, creatinine, glutamate oxaloacetate transferase, glutamate pyruvate transaminase, γ glutamyl transferase), and the final ECG with speckle-tracking technique (visit 3). At each visit, the patients were evaluated for possible cardiac complications and adverse events. It is important to note that the objective of this study was to evaluate the effects of the drug on ischemic myocardial area and not on symptoms. The measurement of the ischemic myocardial area was performed in all probands via 3 functional ECG examinations with speckle-tracking technique (SPE). 14 The first SPE was done before the coronary intervention, the second before the first dose of ranolazine or directly after coronary intervention in the control group, respectively, and the third after 6 weeks of ranolazine treatment (visit 3). During the time between hospital discharge and follow-up presentation, the patients were given information about how to contact an investigator of the RIMINI-trial 24-hour a day on every day of the week. Laboratory staff was blinded regarding specimen allocation of study groups. The primary end point was the global strain rate as an indicator and quantifier for functional or dysfunctional (ischemic) myocardium after 6-week treatment with or without ranolazine. The secondary end point was the incidence of cardiac complications after 6 weeks with or without ranolazine.
Study Protocol and Data Acquisition at Each Visit.
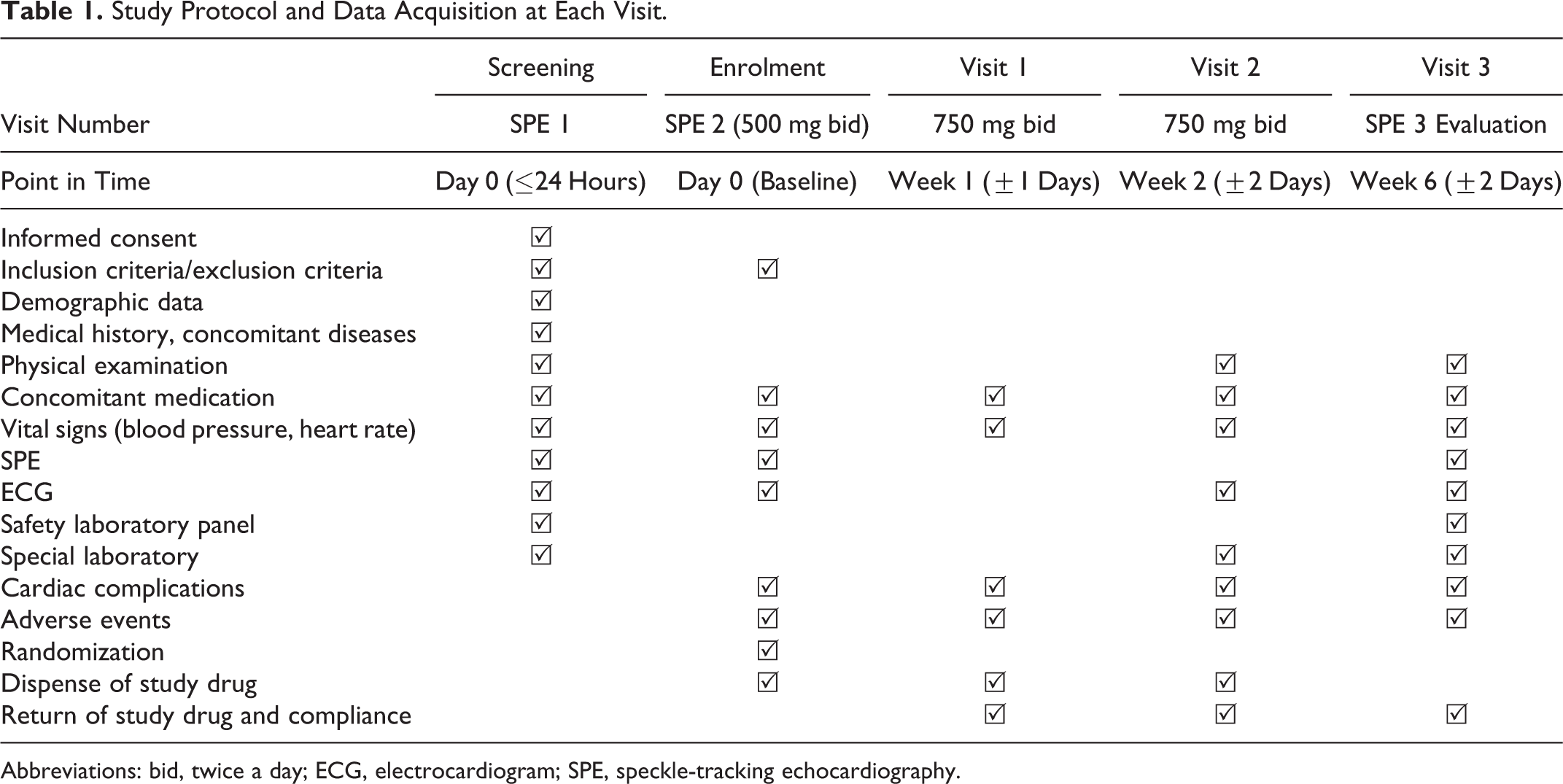
Abbreviations: bid, twice a day; ECG, electrocardiogram; SPE, speckle-tracking echocardiography.

Depiction of the study scheme. Patients with ACS were screened for meeting the inclusion criteria. After stabilization of the patients, the first speckle-tracking echocardiography (preoperation) was performed. The second speckle-tracking echocardiography was done after angioplasty (postoperation). Ten participants then received the study drug ranolazine additionally to standard treatment. The control group consisted of 10 participants receiving standard treatment without additional study medication. Ranolazine was given orally for 6 weeks. The first week 500 mg were given bidaily. After 7 days, the dose was raised (visit 1) and patients received 750 mg ranolazine bidaily until the end of the study period at the end of week 6. At the end of study week 2, all patients presented at the study center for physical examination, ECG, and special laboratory test (visit 2). At the end of study week 6, the patients presented again for a last physical examination, ECG, safety, special laboratory test, and the final ECG with speckle-tracking technique (visit 3). At each visit, the patients were evaluated for possible cardiac complications and adverse events. ACS indicates acute coronary syndrome; ECG, echocardiography.
Speckle-Tracking Echocardiography
Speckle-tracking echocardiography was performed before coronary intervention, before the first dose of ranolazine, and after 6 weeks of ranolazine treatment. The functional ECG with speckle tracking (or 2-dimensional strain) is a noninvasive imaging technique that detects single reflexion signals (“speckles”) in the myocardium in a 2-dimensional image. The speckle movement was tracked and evaluated image-wise in a recorded loop via computer-based analysis. The summarized results were depicted in standardized segment-based projections as a global strain rate. The calculation was done in a computer-based way 14 using TomTec 4D echocardiography software (Version 5.0). (Figure 2).

Representative left ventricular longitudinal strain analysis, visualized by 2-dimensional speckle-tracking echocardiography. Deformation imaging of the left ventricle was performed in the same patient after coronary intervention (A) and at the end of the study (B). Ant indicates anterior; GLS, global longitudinal Strain; ES, end-systolic strain; Inf, inferior; La, lateral; Pk-peak, percentage shortening of longitudinal strain value of respective segment; Sept, septal; TTP, time to peak in milliseconds.
Statistical Analysis
The groups were compared using a baseline-adjusted multilevel mixed-effects linear regression with difference to baseline as dependent variable and to treatment, as well as the different areas of measurement within each patient as potential predictor. Where necessary, a transformation of the outcome variable was performed to satisfy the model assumptions. Treatment groups were tested at the 2-sided 5% significance level. Data are expressed as percentages for discrete variables. Comparisons were performed using StataCorp (2015 Stata Statistical Software: Release 14, College Station, Texas: StataCorp LP). A P value <.05 was considered as significantly different. The study was not designed for statistical power.
Results
Baseline Clinical and Laboratory Variables
Twenty patients were enrolled and completed the study. Participants’ characteristics at baseline and during the study period are presented in Table 2. Mean age was 64.9 and 95% confidence intervals (CI).
Patients of the ranolazine group presented a lower systolic blood pressure throughout the study, whereas the diastolic blood pressure was not different to control group patients. There was no difference in heart rate or glomerular filtration rate. The percentage of smokers was higher (80% vs 20%) in ranolazine patients, whereas patients in the control group more frequently had hyperlipidemia (10% vs 70%). This was by chance and not due to selection bias. The frequency of diabetes and obesity did not differ between the groups. Concerning the localization of coronary artery stenosis, patients most often presented a stenosis on the left anterior descending (LAD; 60%) vessel, followed by stenosis in the circumflex artery (30%). There was no significant difference in coronary artery stenosis localization between the groups. In each group, 1 case of Takotsubo cardiomyopathy occurred and was defined as acute cardiac ischemia according to current guidelines. No major cardiac events or re-events were recorded during the course of this trial. Relevant adverse (21 in 11 participants) or serious adverse (2 in 2 participants) events were recorded without causal relationship to the study. No participant dropped out of the study.
Patients’ Characteristics.a

Abbreviations: BMI, body mass index; BP, blood pressure; CAD, coronary artery disease; CFX, circumflex artery; GFR, glomerular filtration rate; LAD, left anterior descending; M1, marginal branch 1 of CFX; RCA, right coronary artery.
a Data are given as mean (SD) and 95% confidence intervals (CI) or number and percentages. Comparisons were performed by paired (baseline vs study end) or unpaired (control vs ranolazine) Student t test, 2 sided, using GraphPad Prism 6.
b P < .01 versus ranolazine baseline.
Speckle-Tracking Echo
Primary end point of the study was the global strain rate after 6-week treatment with or without ranolazine. Quantification was performed using SPE (Figure 1). Global strain rate of the left ventricle was evaluated at admission (screening), directly after coronary intervention (postoperation), and 6 weeks after initial event of ACS (end of the study; Figure 1). Numeric data of segment base measurements are listed in Supplemental Table 1. At the end of the study, 8 of 17 segments demonstrated improved global strain rates in the control group. In ranolazine-treated patients, 7 of 17 measured segments showed an improvement in the global strain rate (Figure 3). Listing global strain rate measurements in segments paired to affected coronary vessels and corresponding myocardial areas illustrated an improvement particularly in formerly ischemic or ACS affected segments which were perfused by LAD (Figure 4).

Speckle-tracking echocardiography. The functional echocardiography with speckle tracking (or 2-dimensional [2D] strain) detects single reflexion signals in the left ventricular (LV) myocardium in a 2-D image. The speckle movement was tracked and evaluated image-wise in a recorded loop via computer-based analysis. The summarized results were depicted in standardized segment-based projections (n = 17) as a global strain rate (solid line for controls, dashed line for ranolazine-treated patients). The calculation was done in a computer-based way using TomTec 4D echocardiography software. Strain rate was evaluated at admission, directly after coronary intervention (postoperation), and 6 weeks after initial event of acute coronary syndrome. Numeric data of segment base measurements are listed in Supplement Table 1. Positive values indicate a worsening, whereas negative values signify an improvement in contractility. The groups were compared using a baseline adjusted multilevel mixed-effects linear regression with difference to baseline as dependent variable and to treatment, as well as the different areas of measurement within each patient as potential predictor.
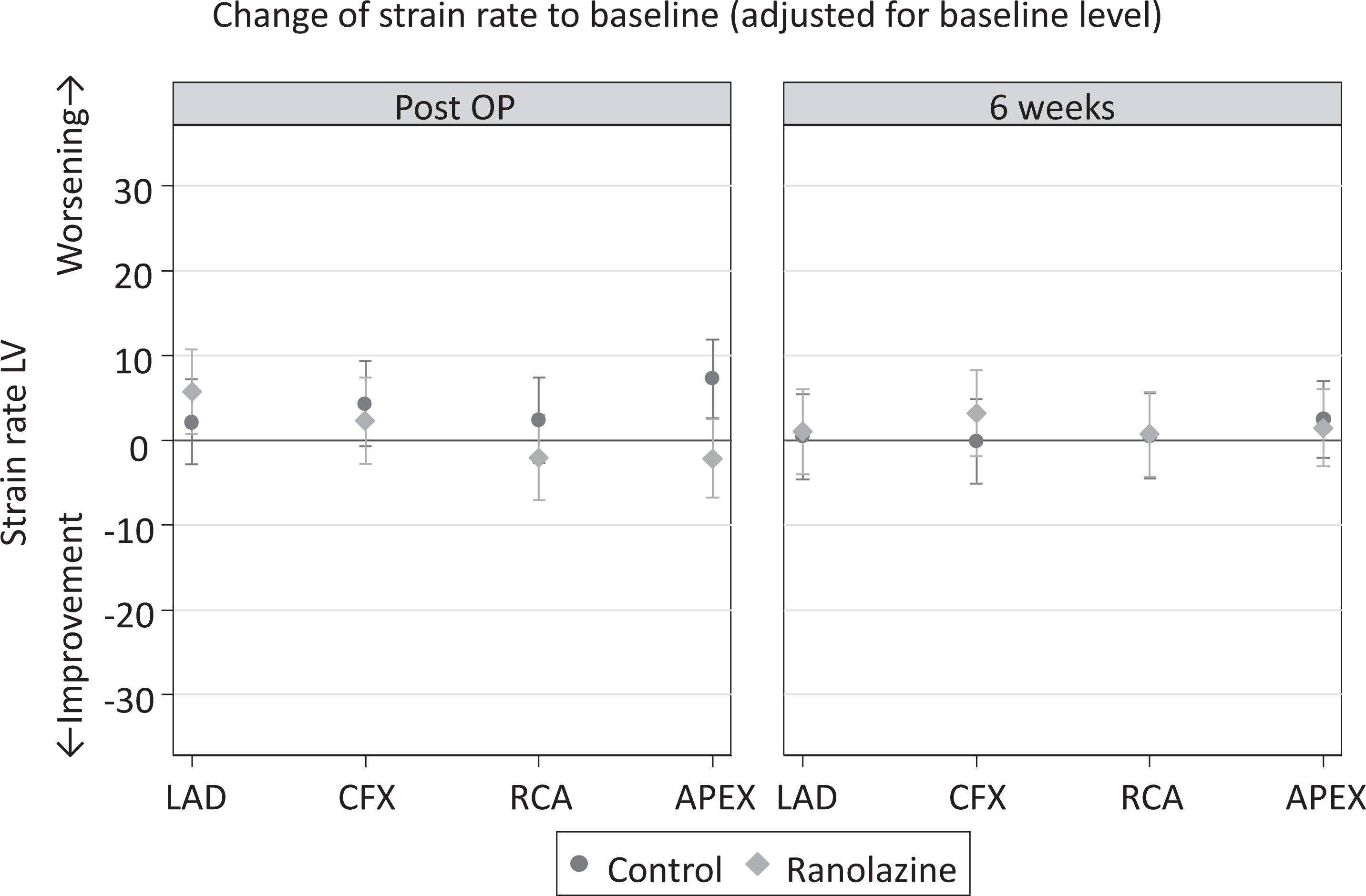
Global strain rate measurements in segments paired to affected coronary vessels. Global strain rate values matched with affected coronary vessels after coronary intervention (postoperation) and after 6 weeks of treatment with ranolazine (ranolazine) or only standard (control) medical treatment. Positive values indicate a worsening, whereas negative values signify an improvement in contractility. Strain rates were adjusted for baseline level before coronary intervention.
However, 6 weeks after initial cardiac ischemia, the vast majority (cumulative) of myocardium remained with an impaired global strain rate (Figure 5, abnormal cumulative global strain rate: 89% standard treatment group, 85% ranolazine group). Additionally, the abnormal global strain rate was higher than after admission (preoperation 79% in both groups). Interestingly, 6 weeks after coronary intervention, the normal fraction of the cumulative global strain rate was higher in the ranolazine (15%) than in the standard treatment group (11%). Statistical analysis showed no significant differences between the groups.

Cumulative global strain rate. Percentage of normal and abnormal cumulative global strain rate in control and ranolazine-treated patients before (preoperation), directly after (postoperation), and 6 weeks after coronary intervention (end).
Discussion
Ranolazine has shown to reduce the risk of recurrent ischemia and worsening of angina in patients with non-ST-segment elevation ACS by possibly improving myocardial perfusion, 1 –3 but up to now no trial has addressed whether this enhanced perfusion also leads to a decrease in ischemic myocardium or increase in left ventricular function of patients with ACS. We, therefore, designed a pilot trial (RIMINI-Trial, ClinicalTrials.gov Identifier: NCT01797484) for feasibility and proof of concept that a 6-week ranolazine add-on therapy would reduce the area of ischemic myocardium in patients with ACS.
The most important finding of this study are the following: (1) ranolazine was administered safely during acute MI in this trial, (ii) SPE proved to be suitable for evaluation of myocardial dyskinesia, and (iii) patients receiving ranolazine showed a trend to higher normal fraction of the cumulative global strain than patients in the standard treatment group at the end of the study. No major complications relating study medication were observed. The study protocol was well implemented and did not adversely influence guideline-based treatment.
In contrast to the study of Venkataraman et al, 12 which made use of radiation-based single-photon emission computed tomography, SPE was appropriate for evaluation of myocardial dyskinesia in this trial suggesting a noninvasive, radiation less, and objective method of follow-up in such patients. Six weeks after coronary intervention, the cumulative global strain rate was higher in the ranolazine than in the standard treatment group. This is in line with results from other studies showing a reduction in the risk of recurrent ischemia, worsening of angina and fewer increases in other antianginal therapy in non-ST-segment elevation myocardial infarction, 1 –3 and improved myocardial perfusion in patients with stable CAD on exercise treadmill. 12 Obtained results were not significantly different; however, this study was not powered for statistical significance, but rather a pilot trial to create a follow-up trial with enough statistical power. Our study does not provide enough data for the answer whether ranolazine has a beneficial effect on acute ischemic myocardium, but it seems not to cause harm in ischemic myocardium (which might also be true for Takotsubo cardiomyopathy).
Study Limitations
Our study should be interpreted in the context of several potential limitations. Most importantly, our study is a small, single-center study with concomitant administration of standard-of-care CAD therapy; nevertheless, our study should be considered a proof-of-concept approach, and more definitive studies with larger numbers of patients are needed. Our patients are likely to be representative of other patients with ACS in terms of dyskinetic myocardium.
Conclusion
In conclusion, in this preliminary hypothesis-driven study, 6-week ranolazine therapy was shown to decrease the area of dyskinetic myocardium in patients with ACS by trend. Global strain rate measurement using SPE can be applied measuring those effects and is, compared to other techniques, safe and harmless. Our data provide a sound basis for a follow-up trial.
Supplemental Material
Supplemental Material, Supplemental_Table_1_RIMINI_Schwemer_-_Copy - Effect of Ranolazine on Ischemic Myocardium IN Patients With Acute Cardiac Ischemia (RIMINI-Trial): A Randomized Controlled Pilot Trial
Supplemental Material, Supplemental_Table_1_RIMINI_Schwemer_-_Copy for Effect of Ranolazine on Ischemic Myocardium IN Patients With Acute Cardiac Ischemia (RIMINI-Trial): A Randomized Controlled Pilot Trial by Tjark F. Schwemer, Lukas Radziwolek, Navina Deutscher, Nadine Diermann, Susanne Sehner, Stefan Blankenberg, and Felix W. Friedrich in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
All named authors have seen and approved the final version of the manuscript.
Acknowledgments
The authors thank the patients who participated in this study.
Author Contributions
All authors have made substantial contributions to the manuscript. Tjark F. Schwemer and Felix W. Friedrich contributed to conception and design of the study; acquisition, analysis, and interpretation of data; and conception of the manuscript. Lukas Radziwolek contributed to acquisition, analysis, and interpretation of data. N. Deutscher and N. Diermann contributed to analysis and interpretation of data. Susanne Sehner and Stefan Blankenberg contributed to analysis and interpretation of data and critical revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Tjark F. Schwemer had financial support from BerlinChemie/Menarini/Gilead for the submitted work.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
