Abstract
Aims:
Acute myocarditis is a potentially lethal inflammatory heart disease that frequently precedes the development of dilated cardiomyopathy and subsequent heart failure. At present, there is no effective standardized therapy for acute myocarditis, besides the optimal care of heart failure and arrhythmias in accordance with evidence-based guidelines and specific etiology-driven therapy for infectious myocarditis. Carvedilol has been shown to be cardioprotective by reducing cardiac pro-inflammatory cytokines present in oxidative stress in certain heart diseases. However, effects of carvedilol administration in acute myocarditis with its impact on matrix metalloproteinases’ (MMPs) activation have not been elucidated.
Methods and Results:
Carvedilol in 3 doses (2, 10, and 30 mg/kg) was given daily to 3 study groups of rats (n = 8) with experimental autoimmune myocarditis by gastric gavage for 3 weeks. In comparison to untreated rats (n = 8) with induced myocarditis, carvedilol significantly prevented the left ventricle enlargement and/or systolic dysfunction depending on the dose in study groups. Performed zymography showed enhanced MMP-2 activity in untreated rats, while carvedilol administration reduced alterations. This was accompanied by prevention of troponin I release and myofilaments degradation in cardiac muscle tissue. Additionally, severe inflammatory cell infiltration was detected in the nontreated group. Carvedilol in all doses tested, had no impact on severity of inflammation. The severity of inflammation did not differ between study groups and in relation to the untreated group.
Conclusions:
The protective effects of carvedilol on heart function observed in the acute phase of experimental autoimmune myocarditis seem to be associated with its ability to decrease MMP-2 activity and subsequently prevent degradation of myofilaments and release of troponin I while not related to suppression of inflammation.
Introduction
Acute myocarditis is an inflammatory disease of the heart caused by either infections (viral, bacterial, and fungal) or other noninfectious factors, including systemic diseases, autoimmune dysregulation, drugs, and toxins. It is characterized by mononuclear or mixed cellular infiltration associated with myocyte necrosis in the presence or absence of fibrosis. 1 Recently, thanks to the advancement in defining the etiology of myocarditis, etiology-driven therapy for infectious acute myocarditis is used more and more often, significantly improving the prognosis of the disease. For eosinophilic, granulomatous, giant cell myocarditis, and lymphocytic myocarditis associated with connective tissue disorders and with rejection of a transplanted heart, the efficacy of immunosuppression was widely investigated and confirmed by many research. 2 –6 Nevertheless, despite the advancement in therapy of myocarditis, a specific standardized treatment is not yet available mainly due to the fact that mechanisms of damage and inflammation in acute myocarditis are still not fully understood. 2,3,5 Also, in acute myocarditis, the presence of unscreened pathogens may limit effectiveness of therapeutic approaches interfering with the immune system carrying the risk of decreased pathogen eradication and subsequent enhanced myocardial damage. 2,3,5,7 Therefore, searching for new strategies of the management of acute myocarditis remains of great importance.
Carvedilol is a nonselective β- and α1-adrenoceptor antagonist with antioxidant properties. It was shown that carvedilol reduced the severity of experimental autoimmune myocarditis (EAM) in rats accompanied by suppression of the messenger RNA expression of inflammatory cytokines and interleukin 1 (IL-1) protein expression. 8 Moreover, it was found that carvedilol offered protection against acute EAM which was attributed to the suppression of IL-1β and tumor necrosis factor α and promotion IL-10 and IL-1Rα. 9 It was also shown that it may modulate production of IL-12 and interferon-γ in murine model of viral myocarditis. 10,11 Inhibitory potential of carvedilol on matrix metalloproteinase (MMP)-2 and MMP-9 expression was also observed in experimental periodontitis. 12
Although MMPs are known for their ability to cleave substrates in the extracellular matrix, it was proved that MMP-2 is activated in tissues undergoing oxidative stress, and intracellularly in cardiomyocytes, it is responsible for troponin I, myosin light chain 1, α-actinin, and titin degradation. 13 –20 Therefore, activation of MMPs may be partially responsible for the decrease of cardiac contractile function in acute myocarditis. In concordance with this hypothesis, Tang et al showed that MMPs such as MMP-3 and MMP-9 were upregulated in murine EAM. 21 Hishikari et al showed that early treatment with clarithromycin (MMP inhibitor) is effective to attenuate myocarditis and to prevent subsequent impairment of cardiac function mostly by MMP-9 suppression. 22 Guttierez et al showed increased transcripts levels, immunoreactivity, and enzymatic activity for MMP-2 and MMP-9 in heart tissue during the acute phase of experimental Trypanosoma cruzi infection, in contrast to significantly decreased heart inflammation, delayed peak in parasitemia, and improved survival rates in mice treated with an MMP inhibitor. 23 Together, these findings provide new insight into the molecular mechanism of myocarditis and a future therapeutic option in the acute phase of myocarditis.
The fact that downregulation of MMP-2 and MMP-9 didn’t decrease pathogen eradication provides the underlying pharmacologic rationale for the use of MMPs inhibitors to reduce contractile dysfunction also in acute infectious myocarditis both in the settings when infectious factor is properly diagnosed and when it remains unscreened.
The EAM in rats is a useful animal model of inflammatory heart disease, as it mimics human fulminant myocarditis in the acute phase. 24 The present study was undertaken to determine whether carvedilol inhibits MMPs activity and decreases the severity of EAM in rats.
Materials and Methods
Animals
The study was conducted on 40 (6-8 weeks old) Lewis strain female rats obtained from AnimaLab (Poznan, Poland). The animals were randomly divided into 5 equal groups and housed together in the same conditions in the Animal Research Section of the Department of Pathomorphology, Poznan University of Medical Sciences, Poland.
Induction of Active EAM
Rats were injected in 1 hind footpad with antigen-adjuvant emulsion in accordance with a procedure described by Kodama et al. 24 In brief, purified porcine cardiac myosin (Sigma-Aldrich, Poznan, Poland) was emulsified with an equal volume of adjuvant complete Freund (Difco, Warszawa, Poland) supplemented with Mycobacterium tuberculosis strain H37Ra (Difco) to a final concentration of 5 mg/mL. On days 0 and 7, rats received subcutaneous injections of 0.1 mL emulsion into the footpad.
Twenty-four rats with induced EAM were divided into 3 equal groups (n = 8). High-dosage (group H; 30 mg/kg/d), middle-dosage (group M; 10 mg/kg/d), and low-dosage carvedilol (group L; 2 mg/kg/d) were given orally by gastric gavage for 3 weeks, from day 0 to day 21 (Sigma-Aldrich). Next 16 rats of a control group were divided into 2, with induced EAM (group I, n = 8) and not immunized (group C, n = 8). They were given vehicle orally by gastric gavage for 3 weeks from day 0 to day 21.
Echocardiography
A transthoracic echocardiogram was performed on day 21 using the Esaote MyLab Delta imaging system equipped with a 12-MHz transducer (Esaote, Maastricht, Holland). Two-dimensional echocardiographic examination was carried out in the parasternal short-axis view. M-mode cursor was positioned perpendicular to the interventricular septum and the left ventricular (LV) posterior wall at the level of the papillary muscles and interventricular septum end-diastolic diameter, LV end-diastolic (LVEDD), LV end-systolic diameter (LVESD), and LV posterior wall end-diastolic diameter were measured. For each rat, 3 to 5 measurements for every parameter were done and averaged for evaluation. Fractional shortening (FS) of the left ventricle was calculated using formula: FS = [(LVEDD − LVESD) / LVEDD] × 100%.
Sample Collection
The study was approved by the animal ethics committee of the Polish Academy of Science (decision no 25/2012). All possible steps were taken to avoid animal suffering at each stage of the experiment. On the day 21 after the immunization, after echocardiography, all animals were subjected to anesthesia using ketamine (10 mg/kg body weight) and then sacrificed by decapitation. The hearts were collected and divided into 2 samples. Half of each heart was rapidly deep frozen and crushed into a powder within a mortar and pestle at liquid nitrogen temperature and stored at −80°C for subsequent biochemical analysis. Second half of each heart was processed for hematoxylin–eosin (HE) staining and transmission electron microscopy (TEM).
Measurement of MMP-2 and MMP-9 by Gelatine Zymography
Prior to the biochemical analysis, the heart tissue powder was homogenized in the buffer containing 50 mmol/L Tris–HCl (pH 7.4), 150 mmol/L NaCl, 0.1% Triton X-100, and protease inhibitors Cocktail Set III (Sigma-Aldrich). In homogenates, protein content was analyzed using Bradford protein assay (Bio-Rad, Warszawa, Poland), and bovine serum albumin was used as a protein standard. The activity of MMP-2 and MMP-9 was assessed in heart extracts by gelatine zymography. Equal total protein samples were applied to 7.5% polyacrylamide gels copolymerized with gelatine (2 mg/mL), containing 0.1% sodium dodecyl sulfate (SDS). In following electrophoresis (100 V, 4°C), each gel was washed 3 times for 20 minutes in 2.5% Triton X-100 and then placed in incubation buffer (50 mmol/L Tris–HCl, 10 mmol/L CaCl2, 200 mmol/L NaCl, and 0.05% NaN3) at 37°C for 18 hours. After incubation, each gel was stained in 0.5% Coomassie Brilliant Blue R-250 (Bio-rad), 30% methanol, and 10% acetic acid for 2 hours and then destained in 30% methanol/10% acetic acid until the bands were clearly visible. Gels were scanned using GS-800 (Bio-rad) calibrated densitometer with Quantity One v4.6.9 software (Bio-Rad), and the relative MMPs activity was determined and expressed in arbitrary units calculated on the basis of recombined MMP standard activity.
Measurement of Troponin I by Western Blot
Samples of 20 µg of protein obtained from heart extracts were applied to 8% SDS-polyacrylamide gel electrophoresis gels under reducing conditions. In following electrophoresis (100 V, 20°C), samples were electroblotted onto a nitrocellulose membrane (by wet technique; 50 V, 30 minutes). A primary monoclonal mouse antibody against cardiac troponin I and secondary goat–anti-mouse conjugated with horseradish peroxidase were both used at 1:1000 dilution (Thermo Fisher Scientific, Waltham, Massachusetts, USA; Bio-Rad, respectively). The blot was developed using chemiluminescence assay (Clarity™ Western ECL Substrate, Bio-Rad). Membranes were scanned using ChemiDoc™ XRS+ System (Bio-rad) with Image Lab™ Software v5.2 for data analysis. Rat cardiac troponin I was used for standard curve preparation (Advanced ImmunoChemical Inc, Long Beach, California, USA).
Analysis of Inflammatory Process in Light Microscope
Tissue samples were fixed in 4% buffered formalin, dehydrated, and embedded in paraffin. For every studied sample, four 6-µm thick paraffin sections stained with HE were prepared. The severity of inflammation was evaluated under BX53 light microscope (Olympus, Tokyo, Japan) according to the scale of Godsel et al: absent (1 point; no inflammatory infiltration), mild (2 points; ≤10% of inflammatory infiltration in muscle tissue), moderate (3 points; >10% and ≤25% of inflammatory infiltration in muscle tissue), and severe (4 points; >25% of inflammatory infiltration in muscle tissue). 25 Images were made using BX41 microscope (Olympus) connected with camera Colorview IIIu (Olympus).
Ultrastructure of Cardiomyocytes in TEM
Small samples of the left ventricle myocardium of approximately 5 mm in diameter were fixed in 2.5% glutaraldehyde (Serva, Heidelberg, Germany). After 24 hours, samples were rinsed in 0.1 mol/L cacodylate buffer (Serva) and postfixed in 1% osmium tetroxide (Serva) for 1 hour. The specimens were dehydrated and embedded in epoxy resin (Epon 812; Serva). An ultramicrotome Power Tome XL (RMC, Tucson, USA) was used to obtain semithin (600 nm) and ultrathin (50 nm) sections. Ultrathin sections were counterstained with uranyl acetate and lead citrate (Serva) and visualized under the TEM JEM-1011 (Jeol, Tokyo, Japan). The electronograms were collected using imaging platform iTEM1233 equipped with the Morada Camera (Olympus), in order to assess the structure of myofibrils in cardiomyocytes under magnification from ×5000 to ×20 000.
Statistical Analysis
GraphPad Prism 7.0 was used for the statistical analysis (La Jolla, California, USA). All data are expressed as mean ± standard error of the mean. Comparisons between groups were tested by 1-way analysis of variance after assessment of normality of distribution. The post hoc analysis was done using Tukey test. P values <.05 were considered statistically significant.
Results
Improvement of Cardiac Function by Carvedilol
Echocardiographic results showed a significant (P < .05, n = 8) enlargement in LVEDD in untreated EAM rats (Figure 1A). Carvedilol (2 mg/kg/d) prevented the LV enlargement expressed as LVEDD (P < .05) and LV systolic dysfunction expressed as FS% (P < .05; Figure 1), whereas higher doses of carvedilol significantly improved either LVEDD (carvedilol 10 mg/kg/d, P < .05) or FS% (carvedilol 30 mg/kg/d, P < .05; Figure 1).

Effect of carvedilol on heart muscle. A, Effect of carvedilol on left ventricular end-diastolic diameter (LVEDD). B, Effect of carvedilol on cardiac mechanical function measured as the fractional shortening (FS%) of the left ventricle. L indicates low-dosage carvedilol experimental autoimmune myocarditis (EAM) group (2 mg/kg/d); M, middle-dosage carvedilol EAM group (10 mg/kg/d); H, high-dosage carvedilol EAM group (30 mg/kg/d); I, group with induced EAM without carvedilol; C, not immunized group without carvedilol. *P < .05.
Carvedilol Inhibits Only Activity of MMP-2
As a result of EAM, there was an enhanced activation of both 64 and 72 kDa MMP-2. Gelatine zymography showed that carvedilol in all doses tested significantly decreased 72 kDa MMP-2 activity in comparison to the control I group (P < .05), but only low dose of carvedilol (2 mg/kg/d) significantly inhibited 64 kDa MMP-2 activity compared to the control I group (P < .05; Figure 2). The activation of both 82 and 92 kDa MMP-9 did not differ significantly between groups treated and untreated by carvedilol (data not shown).
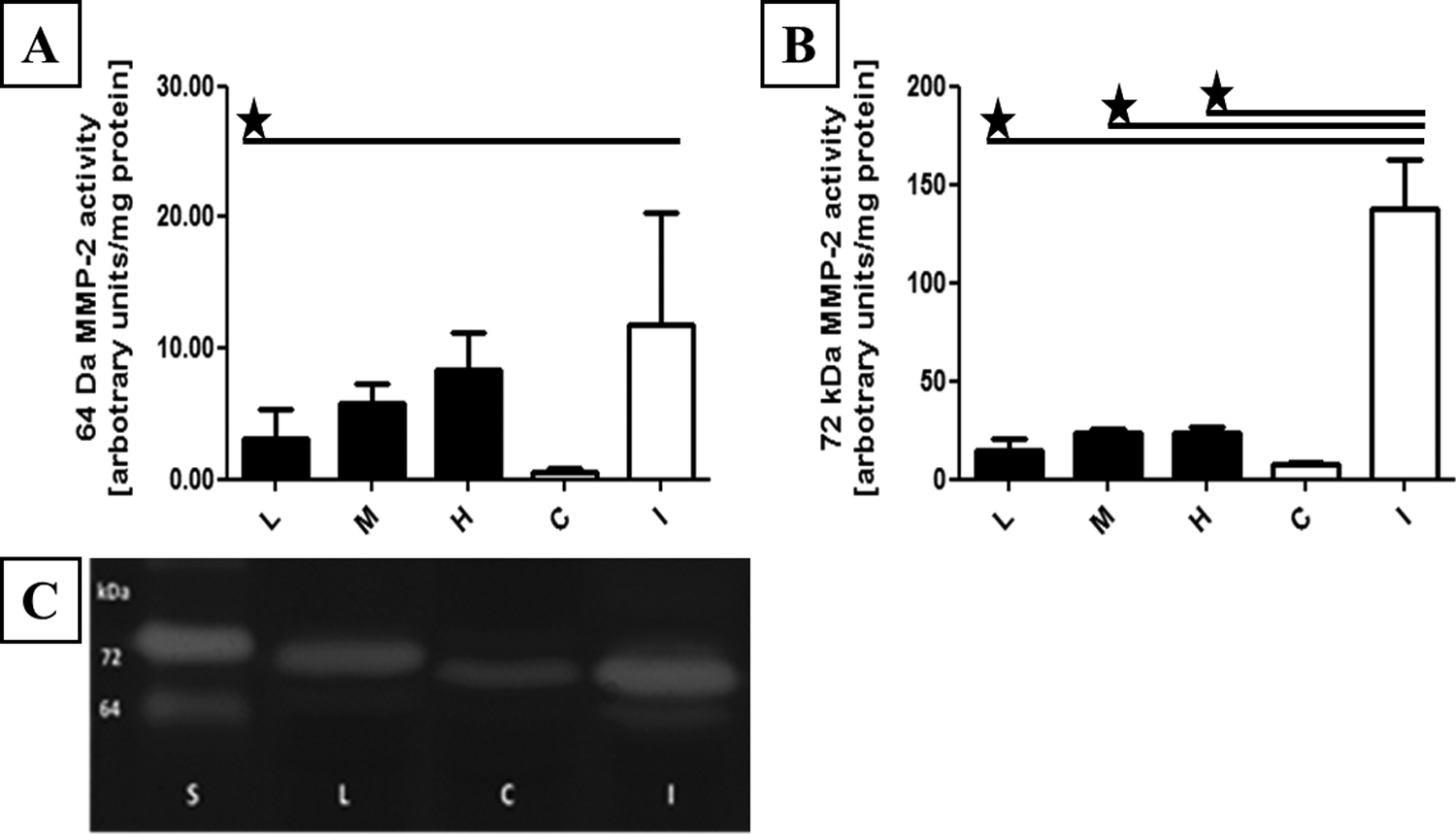
64-kDa MMP-2 activities in heart tissue. (A) 64 kDa and (B) 72 kDa MMP-2 specific activity in heart tissue samples was assessed by densitometric analysis. C, Representative zymogram showing gelatinolytic activities in heart tissue. MMP indicates matrix metalloproteinase; L, low-dosage carvedilol experimental autoimmune myocarditis (EAM) group (2 mg/kg/d); M, middle-dosage carvedilol EAM group (10 mg/kg/d); H, high-dosage carvedilol EAM group (30 mg/kg/d); I, group with induced EAM without carvedilol; C, not immunized group without carvedilol. *P < .05.
Carvedilol Prevents Troponin Degradation
Western blot showed that in carvedilol-treated groups (2 and 10 mg/kg/d), the level of troponin did not decrease and was comparable with the one observed in healthy rats (C group; P < .05; Figure 3).

Effect of carvedilol on troponin I (TnI) content in heart tissue. A, Densitometric analysis of TnI content in heart homogenates determined by Western blot. B, Representative TnI protein quantity in heart homogenates determined by Western blot. L indicates low-dosage carvedilol experimental autoimmune myocarditis (EAM) group (2 mg/kg/d); M, middle-dosage carvedilol EAM group (10 mg/kg/d); H, high-dosage carvedilol EAM group (30 mg/kg/d); I, group with induced EAM without carvedilol; C, not immunized group without carvedilol.
Carvedilol Did Not Reduce the Severity of Inflammation
In group I, there was the most severe inflammatory infiltration consisting of monocytes, lymphocytes, histiocytes, giant cells, and less frequently, neutrophils (Figures 4A and 5B). The number of giant cells ranged from 7 to 22 per whole paraffin section (Figure 4A). In all analyzed cases from studied groups (L, M, H), there was diffuse or multifocal inflammatory infiltration which covered the myocardium in varying degree; from severe through moderate to mild (Figure 4B-D). In all animals with the exception of control rats, inflammatory cells were present between cardiomyocytes (Figure 4B and C). Carvedilol in all tested concentrations had no statistically significant effect on the severity of inflammation in EAM rats (Figure 5A).

Severity of inflammation in cardiac tissue in experimental autoimmune myocarditis in hematoxylin and eosin staining. A, Giant cells (arrowheads) in the I group (without carvedilol). B, Mild, C, moderate, and D, severe inflammatory infiltration in the L group (with low-dosage carvedilol). Inflammatory cells located between cardiomyocytes (arrows).

Severity of inflammation in cardiac tissue in experimental autoimmune myocarditis in transmission electron microscopy. A, Severity of inflammation according to the scale of Godsel et al 25 described in “Material and Methods” section. B, Ultrastructure of giant cell (arrows) limited by the cell membrane in the I group. Ne indicates neutrophil; L, low-dosage carvedilol experimental autoimmune myocarditis (EAM) group (2 mg/kg/d); M, middle-dosage carvedilol EAM group (10 mg/kg/d); H, high-dosage carvedilol EAM group (30 mg/kg/d); I, group with induced EAM without carvedilol; C, not immunized group without carvedilol.
Carvedilol Decreases Degradation of Myofibrils
The influence of carvedilol on myofilaments degradation varied in different groups of animals from L to H. Regenerating connective tissues (granulation), which surrounded the cardiomyocytes and inflammatory cells infiltration between the heart cells, were conspicuous (Figure 6A). Defects in the system of myofibrils were observed. There were damaged intercalated discs (Figure 6B), significant loss of myofilaments (Figure 6B and C), and swollen mitochondria (Figure 6D). However, in each group of carvedilol-treated EAM rats, examples of normal myofibrils system (Figure 6D) and correct intercalated discs absent in I group were described.

Ultrastructural lesions in experimental autoimmune myocarditis. A, Inflammatory cells located between cardiomyocytes. B, Damaged intercalated disc (arrowheads) and destroyed myofilaments (arrow). C, Significant loss of myofilaments and degenerating mitochondria (arrows). D, Swollen mitochondria located between normal cardiomyofibers. Ne indicates neutrophil; Z, Z line.
Decreased MMP-2 Activation Correlates With Prevention of Troponin Degradation and LVEDD
The results revealed that low levels of 64 kDa MMP-2 correlated significantly with high troponin levels and low LVEDD (r = −0.38, P = .03 and r = 0.58, P = .0005, respectively). Moreover, low levels of 72 kDa MMP-2 correlated with low LVEDD and better systolic function of LV expressed as FS% (r = 0.39, P = .026 and r = −0.49, P = .005, respectively). Analyzed associations are presented in Figure 7.

Pearson correlation coefficients between analyzed parameters. Statistically significant correlations are given in boldface.
Discussion
The present findings suggest cardioprotective properties of carvedilol in EAM rats directly associated with its ability to prevent MMP-2 activation. The EAM in rats mimics human fulminant myocarditis in the acute phase. Therefore, our findings are of great importance, as they provide an underlying pharmacologic rationale for the use of carvedilol to reduce cardiac mechanical dysfunction in acute myocarditis in clinical practice. To the best of our knowledge, no data on the effect of carvedilol on MMPs activity in EAM currently exist.
The MMPs are expressed in normal cardiomyocytes; however, their activity increases in heart subjected to oxidative stress. Particularly, they are upregulated in myocarditis, contributing to changes in the LV structure and function. Cheung et al found elevated activation of MMP-2 and MMP-9 following CVB3 infection. 26 Hishikari et al showed enhanced activity of MMPs in the inflammatory area in EAM rats. Moreover, they proved that early treatment with clarithromycin attenuated the MMP-9 activity in EAM hearts and improved cardiac function. 22
Carvedilol, a nonselective α1- and β-adrenoceptor blocker was shown to reduce the severity of EAM via suppression of pro-inflammatory and promotion of anti-inflammatory cytokines. It was also proved that carvedilol inhibited MMP-2 and MMP-9 in experimental periodontitis. However, there were no data about the effect of carvedilol in myocarditis via MMPs. In our study, we showed that carvedilol in all doses tested significantly decreased 72 kDa MMP-2 activity in comparison to the control C group. Moreover, low dose of carvedilol (2 mg/kg/d) also significantly inhibited 64 kDa MMP-2 activity compared to the control C group. It was accompanied by prevention of the LV enlargement expressed as LVEDD and/or LV systolic dysfunction expressed as FS% in all groups receiving carvedilol and prevention of troponin I degradation in heart muscle in L and M groups. Carvedilol has no effect on the severity of inflammation, but TEM revealed protective influence of this drug on the degree of degradation of myofilaments as in each group of carvedilol-treated rats, there were examples of normal myofibrils system not seen in I group. These data support the hypothesis that beneficial influence of carvedilol on inflamed heart muscle is not due to its influence on the severity of inflammation, but, at least partially, due to the decrease in MMP-2 activity leading to subsequent reduction in troponin and myofilaments degradation and prevention of mechanical dysfunction of the heart. These data correspond with the findings of Hishikari et al who showed that inhibition of other members of MMPs family such as MMP-9 with the use of clarithromycin in EAM rats had beneficial influence on cardiac function. 22
We found that low-dose carvedilol (2 mg/kg/d) improved heart function and reduced MMP-2 activation and troponin degradation more effectively than high doses of carvedilol. This dose is equivalent to 25 mg of carvedilol in humans when one compares the dosage corrected by using body surface area. 27 This dose is used to treat heart failure in humans. 28 –30 Moreover, it was shown that treatment with carvedilol (mean maintenance dose of 25 mg) in patients with congestive heart failure resulted in reduced oxidative stress without restoration of the chronotropic responsiveness index. 28,29 Although the reasons for the dose-unrelated cardioprotective action are not clear, the data suggest that the most effective dose should not exert any significant effects on hemodynamics of heart with decreased contractile function and, therefore, could be used in acute phase of myocarditis. Therefore, the therapeutic potential of low-dose carvedilol is very promising therapeutic strategy which, opposite to symptomatic treatment, targets mechanisms, leading to degradation of myofilaments and depletion of heart function in the course of acute myocarditis. Our findings strictly correspond with the findings by Watanabe et al who showed that a low dose of carvedilol (2 mg/kg/d) has beneficial effects on development of dilated cardiomyopathy induced by autoimmune giant cell myocarditis, although they did not investigate the influence of carvedilol on MMPs. 27
In acute myocarditis, initial cardiac inflammation helps to eliminate the pathogen and to prevent irreversible myocardial injuries. Therefore, anti-inflammatory or immunosuppressive therapy might favor infectious agent persistence causing worsening the patient’s outcome. 2,3,5,7 It may also refer to the drugs interfering with the immune system or endogenous homeostatic systems, that is, MMPs. Our present study shows that carvedilol had no effect on the severity of inflammation. Therefore, it should have no significant influence on pathogen clearance by inflammatory processes. The fact that downregulation of MMP-2 and MMP-9 during the acute phase of experimental myocarditis driven by T Cruzi delayed parasitemia peak suggests that inhibitors of MMPs are safe in administration in acute infectious myocarditis. 23
In conclusion, our present results afford the first evidence that carvedilol reduces mechanical dysfunction of heart muscle along with attenuation of MMP-2 activity and both troponin and myofilaments degradation in hearts subjected to EAM. This effect might provide a future therapeutic option in the acute stage of human fulminant myocarditis.
Study Limitations
First of all, the area of our interest seems to be a serious research problem from some time. New possibilities of clinical applications for carvedilol as well as new therapeutic strategies for myocarditis treatment are nowadays excessively investigated. Carvedilol was shown to reduce the severity of experimental myocarditis via suppression of pro-inflammatory and promotion of anti-inflammatory cytokines. It was also proved that carvedilol inhibited MMP-2 and MMP-9 in experimental inflammation and autoimmune diseases. However, our study reveals a cardioprotective action of carvedilol in EAM, what seems to be due to its ability to prevent MMP-2 activation and subsequent degradation of troponin, myofilaments, and depletion of heart function. Moreover, the most effective carvedilol dose used in our experiments corrected by usage of body surface area is equivalent to the dose used in human heart failure treatment. Additionally, it seems to be safe in the settings of acute contractility dysfunction related with myocarditis. Therefore, our findings are of particular importance because they provide an underlying pharmacologic rationale for the use of low-dose carvedilol and possibly other MMPs inhibitors in clinical practice in the acute stage of human fulminant myocarditis.
The second limitation regards the type of echo transducer used to assess LV diameter and function. Recent advancements in ultrasound technology include the development of high-frequency transducers for animal studies. Resolution defined as the ability to distinguish 2 closely situated structures is an important concern for all imaging methodologies especially when imaging very small targets. Because axial and lateral resolution improves with increasing frequency, higher frequency transducers are generally preferred for small rodent organs, but they are not always necessary and optimal. The theoretical resolution at the frequencies used in our study (12 MHz) is 0.128 mm, allowing precise analysis of heart diameters. 31 Moreover, for some researchers, M-mode imaging a 12-MHz probe is believed to be favorable for rat hearts, as higher frequencies tend to give unsatisfactory ultrasound penetration through the chest wall. 32 Therefore, in spite of availability of higher frequency probes, 12-MHz transducers are still utilized by most of scientists. 33 –35
Footnotes
Acknowledgments
The authors would like to thank veterinarian Grzegorz Dziwak and technical personnel of Department of Pharmacology as well as Department of Histology and Embryology for their assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the scientific grant of the Wroclaw Medical University (ST
