Abstract
Background:
Apixaban is a direct oral anticoagulant, which inhibits factor Xa. It has demonstrated clinical efficacy in prevention of stroke and systemic embolism in adult patients with nonvalvular atrial fibrillation and a better safety profile compared to warfarin.
Objectives:
(1) To describe the characteristics of patients with nonvalvular atrial fibrillation beginning treatment with apixaban, (2) to analyze concomitant prescriptions of medications that could potentially interact with apixaban, (3) to evaluate the level of appropriate usage according to the recommended dosage, and (4) to estimate the level of apixaban persistence among naive and non-naive patients.
Methods:
Cohort study using data from primary care (System for Research in Primary Care database, users of the Institut Català de la Salut; Catalonia, Spain) from August 2013 to December 2015.
Results:
Mean age for apixaban-treated patients was 71.8 years (standard deviation = 11.1) and 55.6% were male. In all, 3.2% of patients receiving apixaban were taking drugs described as potentially related to either pharmacokinetic or pharmacodynamic interactions. According to the summary of product characteristics, 81.1% of patients with a recommended dose of 2.5 mg twice daily and 51.8% with a recommended dose of 5 mg twice daily actually took this dose. After 1 year of follow-up, 62.6% of the apixaban users showed good adherence.
Conclusion:
The prescribed dose of apixaban did not fully follow the recommended dose, particularly in patients who were treatment naive. Patients with a prior history of anticoagulant treatment were more likely to remain persistent to treatment with apixaban.
Introduction
Atrial fibrillation (AF) is the most common type of cardiac arrhythmia with a current estimated prevalence in the developed world of approximately 1.5% to 2% of the general population. It can cause symptoms (palpitations and dizziness) but is sometimes asymptomatic (episodes of “silent” AF). Anticoagulation therapy is critical for reducing the risk of consequences of AF, as patients with AF have a 5-fold risk of stroke and a 3-fold incidence of congestive heart failure and higher mortality. 1,2 Vitamin K Antagonists (VKAs) significantly reduce the risk of stroke and death in patients with nonvalvular AF (NVAF) and have long been the cornerstone of therapy for this condition. 3 From 2011, based upon randomized trials demonstrating their comparable or superior efficacy and safety relative to VKAs, direct oral anticoagulants (DOACs) became available. Unlike VKAs, DOACs do not require laboratory monitoring, do not have a narrow therapeutic index, and have fewer food and drug interactions. 2,4
Apixaban is one of the novel DOACs that, by inhibiting the factor Xa, has emerged as an alternative to VKAs in the prevention of stroke and systemic embolism (SE) in adult patients with NVAF. 5
The clinical efficacy of apixaban for the prevention of stroke and SE in adult patients with NVAF was demonstrated as well as an improved safety profile compared to aspirin, in 1 phase III clinical trial Apixaban Versus Acetylsalicylic Acid to Prevent Stroke in Atrial Fibrillation Patients Who Have Failed or Are Unsuitable for Vitamin K Antagonist Treatment (AVERROES), and these end points were compared to warfarin in another phase III clinical trial Apixaban Versus Acetylsalicylic Acid to Prevent Stroke in Atrial Fibrillation Patients Who Have Failed or Are Unsuitable for Vitamin K Antagonist Treatment (ARISTOTLE). 6,7 In Spain, the indication of apixaban for the prevention of stroke and SE in patients with NVAF started in August 2013. 8 The Spanish Agency for Medicines and Health Products (AEMPS) recommends DOACs in patients with NVAF and history of hemorrhagic stroke or high risk of intracranial hemorrhage, ischemic stroke with clinic, or neuroimaging of high risk of intracranial hemorrhage in patients with poor control of international normalized ratio (INR) 2-3 and in patients allergic or intolerant to VKAs. 8
Since apixaban commercialization was initiated in Spain, little is known about its adoption into daily clinical practice. By way of this real-world data drug utilization study, we aim to assess apixaban patients’ characteristics. The aim of this study was to characterize patients with NVAF using apixaban for stroke prevention as well as to analyze the comedications prescribed and evaluate the level of appropriate usage according to the dosage recommended in the summary of product characteristics (SmPC) and the adherence and persistence in this treatment.
Methods
We carried out a noninterventional postauthorization study on the use of apixaban in Primary Care Catalonia’s (Spain) public health-care system. The protocol of the present study has been published elsewhere (http://www.encepp.eu/encepp/openAttachment/fullProtocol/13179;jsessionid=IWKy4mJFYCC1a-TnyRqJJDh1o01ZLKLHOwliAY3aaVa0-Ui4ubQ0!1617953341).
Data were obtained from the Information System for Research in Primary Care (SIDIAP) database that contains anonymized clinical information from 279 primary health-care (PHC) centers managed by the Catalan Health Institute , which covers more than 5.8 million patients (approximately 80% of the Catalan population, which represents more than 10% of the Spanish population). This information emerges from Estació clínica d'atenció primària (ECAP) (electronic health records in PHC), and it includes sociodemographic characteristics, health conditions registered as International Classification of Disease, Tenth Revision (ICD-10) codes, clinical parameters, toxic habits, laboratory data, and general practitioners’ prescriptions and their corresponding pharmacy invoice data identified through anatomical therapeutic chemical (ATC) codes.
The study cohort included all eligible participants from the source population who had a new prescription for apixaban from August 2013 until December 2015 and a previously recorded diagnostic of NVAF (classified by ICD-10 codes). These participants were divided into 2 cohorts: patients who have initiated with apixaban in the period August 2013 to December 2015 as treatment-naive (no prior prescription of VKAs in the 12 months before the index date), and non-naive, that is, patients who have been previously treated with VKAs or other DOACs (dabigatran or rivaroxaban) in the 12 months before index date.
Apixaban prescriptions were identified through ATC codes from ECAP prescriptions. 9 Patients were followed up until discontinuation of apixaban (patient discontinuation was defined as 2 or more consecutive months after the las supply).
Data on sex, age, weight, smoking and alcohol habits, Mortalidad en áreas pequeñas Españolas y Desigualdades socioEconómicas y Ambientales (MEDEA) socioeconomic index, 10 estimated glomerular filtration rate, and comorbidities (number and type by ICD-10 codes) were collected at the index date. Congestive heart failure, Hypertension, Age, Diabetes, Stroke, Vascular disease, Age 65-74, Sex category (CHADS2, CHA2DS2VASc,) and Hypertension, Abnormal renal or hepatic function, Stroke, Bleedng, Labile INR, Elderly, Drug or alcohol use predisposing to bleeding (HAS-BLED) score were calculated based on these data.
Data for relevant comedications were identified and collected from prescriptions at the index date and up to 1 month after this date.
Data on the dosage and frequency of apixaban, 2.5 mg twice daily and 5 mg twice daily, were collected from the invoice record. Only data until September 30, 2015 were analyzed for this objective, as the system has a 2 to 3 months delay to get the invoice data of the prescriptions and for those patients prescribed apixaban after September 30, 2015, data on the dosage would not have been registered.
To assess the adherence in those patients with data of at least 1 year, we used the medication possession ratio (MPR), defined as the ratio of the number of days of medication supplied within the refill interval to the number of days in the refill interval.
Both adherence and therapeutic persistence to apixaban were assessed through pharmacy invoice data for patients who initiated treatment between August 2013 and December 2014 (n = 1971), in order to analyze data of at least 1 year of follow-up after initiation. It was calculated based on the pattern of repeat dispensed prescriptions.
Statistical Analysis
Patient’s characteristics were described using frequencies and percentages for categorical variables and mean and standard deviation (SD) for continuous variables as appropriate.
Level of agreement between real and recommended dose according to SmPC was assessed through Cohen’s κ statistic with its 95% confidence interval (95% CI). Time to discontinuation was described using Kaplan-Meier curves. Regarding missing data, no imputations were carried out. Data analysis was performed using R Statistical Software (version 3.3.2; R Foundation for Statistical Computing, Vienna, Austria).
Results
A total of 6135 patients initiated treatment with apixaban from August 2013 to December 2015, with most (76.8%) being naive to anticoagulant treatment.
Among them, there were more women (55.6%). Apixaban nontreatment-naive patients (mean age: 78.1 years, standard deviation [SD]: 8.7) were older than naive ones (mean age: 71.8 years, SD: 11.1), had more comorbidities (any, 92.5% vs 78.4%), more cardiovascular comedications (95.9% vs 86.8%), and higher risks of stroke (CHA2DS2VASc mean, 4.4 vs 3.0) and bleeding (HAS-BLED mean, 2.7 vs 1.7). Body mass index (BMI) could not be calculated for more than a quarter of the apixaban patients; however, 31.3% of the remaining patients were overweight or obese (BMI ≥ 25). Generally, patients treated with apixaban were nonsmokers (66%), with a relatively good renal function (58.8% had over 60 mL/min per 1.73m 2 ), and were elderly (67.1% patients were 70 years and older; Table 1).
Sociodemographic Characteristics, Comorbidities, Risk of Stroke, and Hemorrhage in Patients Treated With Apixaban.
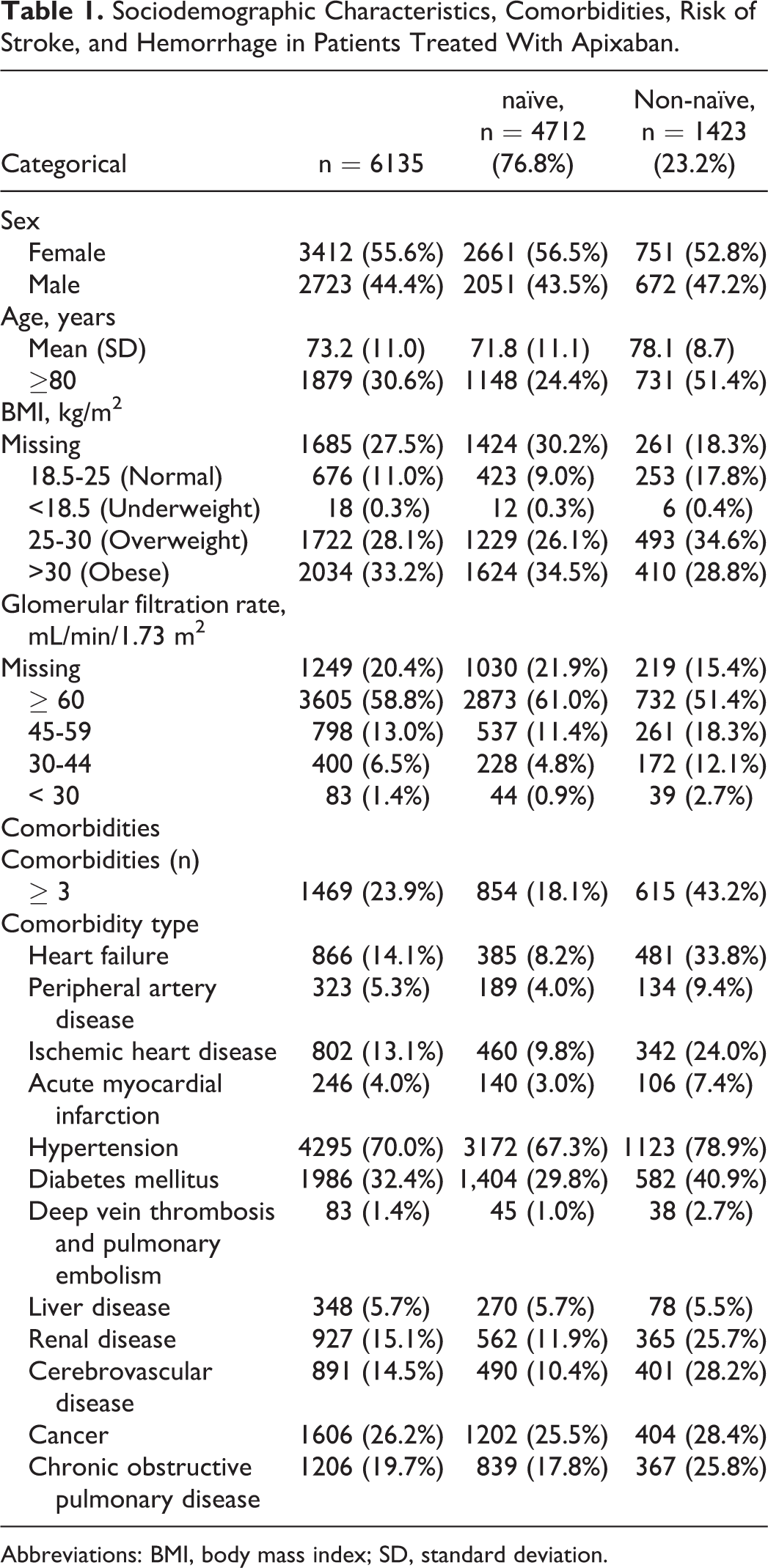
Abbreviations: BMI, body mass index; SD, standard deviation.
We found that 5011 (81.7%) apixaban-treated patients had at least 1 comorbidity and 1469 (23.9%) had 3 or more comorbidities. Overall, in the studied population the most frequent comorbidities were hypertension (70%) and diabetes mellitus (32.4%). Cancer was the third more frequent comorbidity (25.5%) among the apixaban-naive patients and heart failure (33.8%) among the non-naive ones (Table 1).
The mean Congestive heart failure, Hypertension, Age, Diabetes, Stroke, Vascular disease, Age 65-74, Sex category (CHA2DS2VASc) score was 3.3 (SD = 1.8), and this was lower for the naive patients (3.0, SD = 1.7) compared to the non-naive ones (4.4, SD =1.6). The mean Hypertension, Abnormal renal or hepatic function, Stroke, Bleedng, Labile INR, Elderly, Drug or alcohol use predisposing to bleeding (HAS-BLED) score was 2.0 (SD = 1.1) for all patients, and 1.7 (SD = 1.0) and 2.7 (SD = 1.1) for the naive and non-naive cohorts, respectively (Table 2).
Risk Score for Stroke and Hemorrhage.
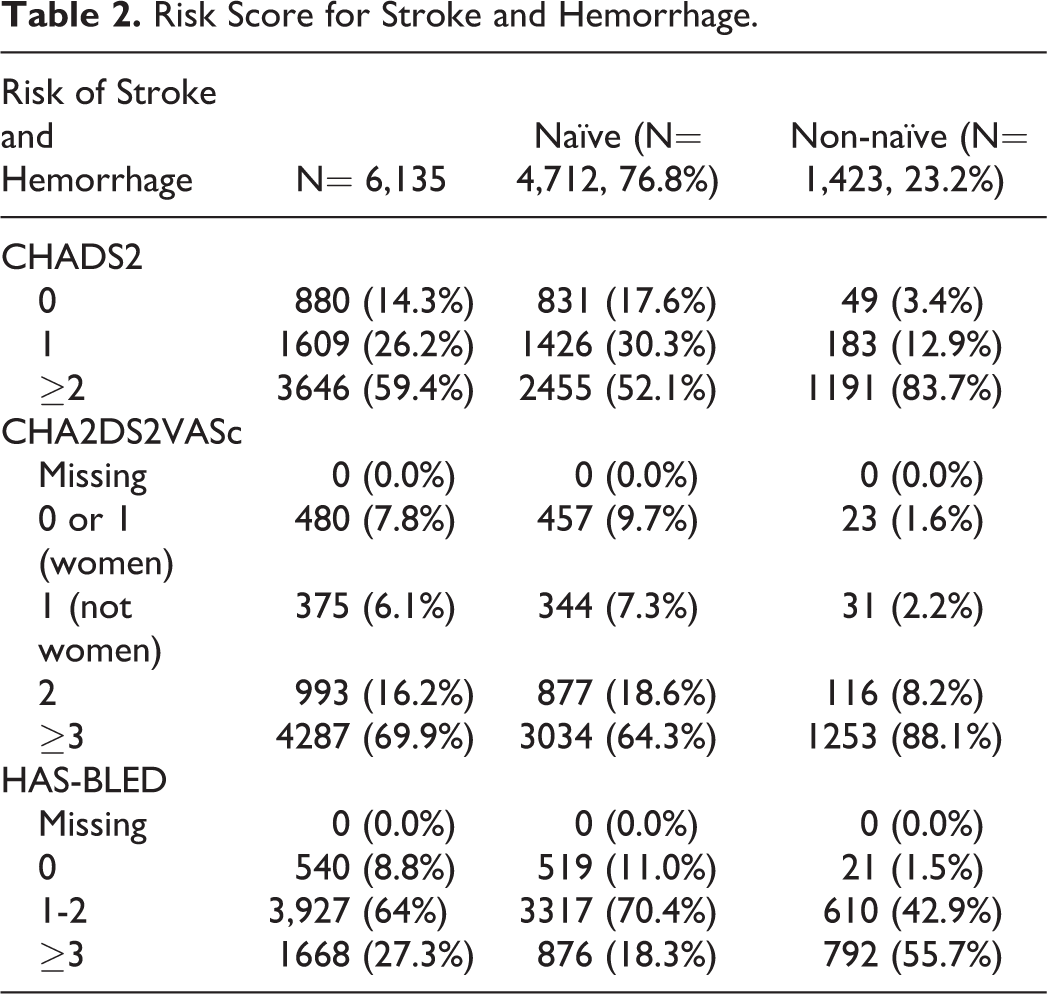
With regard to coprescribed medications in the overall apixaban-treated patient population, 86.1% had cardiovascular medications, 70.2% had proton pump inhibitors, 45.7% had lipid-modifying agents, 22.9% had drugs used in diabetes management, and 22.2% had antidepressants. Almost all patients taking apixaban had other medication prescribed concomitantly. Drugs described in the SmPC as potentially having a pharmacokinetic interaction with apixaban were prescribed in 4.3% of patients, and drugs described as potentially having a pharmacodynamic interaction were prescribed in 68.4% of patients (Table 3).
Potentially Interacting Medication With Apixaban in All Apixaban-Treated Patients, in Naive and Non-Naive Patients.

We analyzed which patients received the recommended dose according to the SmPC; however, there was an elevated number of missing data (39.7% [n = 2043] by the dispensed dose and 18.2% (n = 935) by the recommended dose) which complicated a proper assessment of this objective.
Therefore, we only had complete data to achieve this objective in 2546 (49.5%) patients. Among patients with recommendation for dose reduction (n = 360, 14.1%), a relatively low number of patients (n = 68, 2.7%) had 5 mg twice daily dose prescribed. Among those ones with recommendation for the standard 5-mg twice daily dose (n = 2186, 85.9%), 41.4% (n = 1053) had the 2.5-mg twice daily dose prescribed, despite not having met all necessary criteria for this dose reduction. This dosing pattern was more commonly associated with treatment-naive patients (n = 815, 51.7%) compared to nontreatment-naive patients (n = 238, 24.8%). The use of apixaban according to dose prescribed for naive and nonnaive patients (Table 4).
Use of Apixaban According to Dose Prescribed for Patients With Available Data in Naive (a) and in Non-Naive Patients (b).
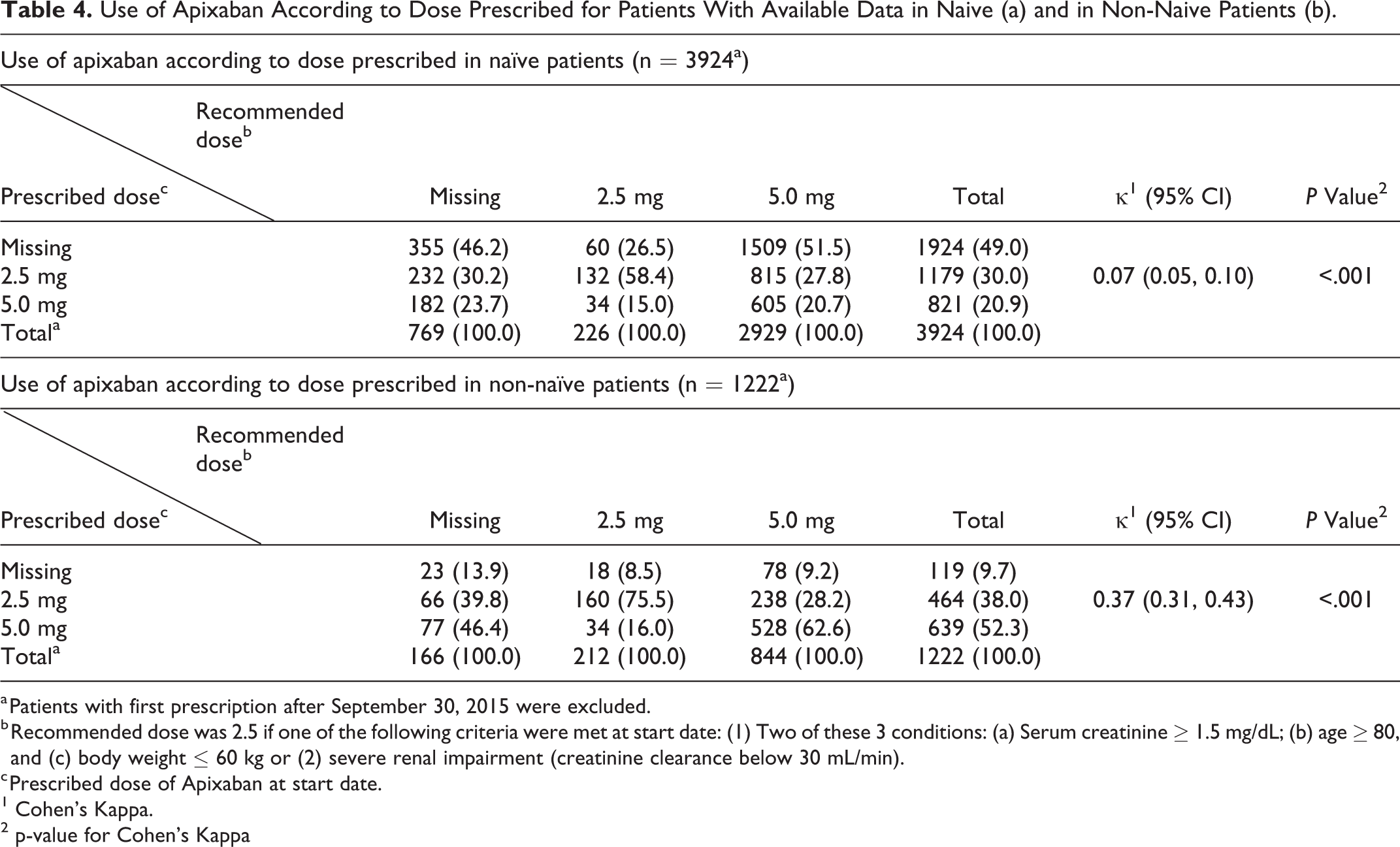
a Patients with first prescription after September 30, 2015 were excluded.
b Recommended dose was 2.5 if one of the following criteria were met at start date: (1) Two of these 3 conditions: (a) Serum creatinine ≥ 1.5 mg/dL; (b) age ≥ 80, and (c) body weight ≤ 60 kg or (2) severe renal impairment (creatinine clearance below 30 mL/min).
c Prescribed dose of Apixaban at start date.
1 Cohen's Kappa.
2 p-value for Cohen's Kappa
For the 854 (43.3%) patients not discontinuing the medication and having at least 1 year of follow-up to calculate adherence, 62.6% of them showed good adherence (MPR between 80% and 120%). Regarding persistence, there were 652 (33.1%) patients discontinuing treatment in the first month (Table 5).
Descriptive of Medication Adherence and Discontinuation Rates in Patients Treated With Apixaban.

Abbreviation: MPR, medication possession ratio.
After 1 month of treatment initiation, almost half of the treatment-naive patients (45.7%, n = 596) discontinued treatment versus <10% of the non-naive patients (8.4%, n = 56; Table 5). When analyzing the characteristics among treatment-naive patients who discontinued after the first month of treatment versus treatment-naive patients who were persistent for 1 year, there were more women (66.9% vs 46.2%, mean difference: 10.7%, 95% CI: 4.3%-17.0%) who were younger (66.3 years old [±13.3] vs 74.6 years old [±10.5], mean difference: 7.6, 95% CI: 6.2-9.0) and had lower risk scores for stroke (In CHA2DS2VASc 2.4 [±1.6] vs 3.7 [±1.7], mean difference: 1.3, 95% CI: 1.1-1.5) and bleeding (In HAS-BLED 1.4 [±0.9] vs 2.1 [±1.0], mean difference: 0.7, 95% CI: 0.6 -0.8).
Discussion
During the study period, a total of 6135 patients with NVAF met inclusion criteria for this cohort study, of which 1423 (23.2%) patients were non-naive to oral anticoagulation treatment. We found more women than men were treated with apixaban, with a mean age of 73.2 years. Two studies carried out in United States had similar median ages as that reported in this study (73 and 70.9, respectively) but a higher proportion of men being treated with apixaban (53.1% and 59.7%). 11,12
Our results showed that 1469 (23.9%) patients had 3 or more associated comorbidities, with hypertension present in 70.0% of them, while US-based studies report more than 85% of the apixaban-treated patients had hypertension. 11,12 For diabetes mellitus, our results were similar (32.4% vs 25%-35% of the patients analyzed in the US-based studies), but in the case of the ischemic heart disease, only 13.1% of our patients who started apixaban had coronary artery disease versus 30% of patients in the study of Desai et al . 11 –13 It is important to note that in their study, only 20 patients initiated apixaban treatment but in patients who started other DOACs (rivaroxaban, dabigatran), higher rates of ischemic heart disease were also observed (30% from 821 patients and 28% from 1982 patients under treatment with rivaroxaban and dabigatran, respectively). In the study of Yao et al, the proportion of apixaban-treated patients with vascular disease was 28.3% (n = 7695) 11 , while in the study of Li et al they reported only 8.9% (n = 38 470) of patients with myocardial infarction. 12
In our study, the mean Congestive heart failure, Hypertension, Age, Diabetes, Stroke (CHADS2) score was not different from that in the ARISTOTLE trial or the study of Li et al (2.0 ± 1.4 vs 2.1 ± 1.1 and 2.1±1.3, respectively). 7,12 In the Desai study, patients starting treatment with apixaban had a CHA2DS2VASc mean score of 2.05 (± 0.94), while patients in our study had had a higher CHA2DS2VASc mean score of 3.3 (± 1.8), similar to the study of Li et al (3.2 ± 1.8) but not as high as that in the study of Yao et al (4 ± 1). 11 –13
With regard to bleeding risk, our patients’ mean HAS-BLED score was higher (2.0 ± 1.1) than for the patients in the Desai et al cohort (1.60 ± 0.82) but the same as in the study of Yao et al. 11,13 This higher mean of HAS-BLED score could be partly explained by incorrect dose prescription according to the SmPC as trying to minimize the risk of bleeding by dose reduction even not meeting all the criteria. In our population, treatment-naive patients had lower HAS-BLED scores than non-treatment-naive patients; however, the proportion of patients in the treatment-naive cohort receiving low dose of apixaban was even higher.
The efficacy and safety of the DOACs might be compromised by coadministration of other drugs, 14,15 and dose adjustment may be needed when using certain DOACs concomitantly with other drugs. 16
In this study, 86.1% of apixaban-treated patients were taking cardiovascular drugs. Diltiazem has been described as a drug with interaction potential, and it was prescribed to a 3.8% (n = 236) of the patients of our study. However, the apixaban SmPC does not recommend a dose adjustment with drugs that are weak-to-moderate CYP3A4 or P-gp inhibitors. In the ARISTOTLE trial, 30.1% of patients received a calcium channel blocker as a prescribed co-medication. 7
With regard to the potential pharmacodynamic interaction with NSAIDs, the apixaban SmPC warns of its use concomitant with NSAIDs, although 29.4% (n = 1805) of our studied patients used both drugs. A previous study has reported an increase in apixaban exposure because of augmented bioavailability when administered concomitantly with naproxen 17 It is well known that anticoagulants may increase the risk of hemorrhage, including GI hemorrhage, especially with NSAIDs with a long half-life. However, apixaban has shown lower gastrointestinal (GI) bleeding rates in comparison to VKA and also with other DOACs. 7,11,12 In our study, 16.1% (n = 985) in the overall population had a platelet aggregation inhibitor coprescribed in 18.2% of treatment-naive patients and 9.1% of non-treatment-naive patients, which is consistent with the number of patients with coronary artery disease (CAD; Table 3).
Close monitoring for signs of bleeding is recommended when an anticoagulant is being used with other platelet aggregation inhibitors, especially when dual antiplatelet therapy is going to be considered, as it has been described as increasing the risk of major hemorrhagic complications in patients treated after and acute coronary syndrome. 16,18,19 Recent ESC guidelines on dual antiplatelet treatment (DAPT) in CAD recommends, in patients receiving oral anticoagulation concomitantly, to shorten the DAPT as much as possible as well as discontinuation of antiplatelet treatment at 12 months. 20
When a new drug enters the market, and in particular, a DOAC, physicians could prescribe lower doses because of its particular drug profiles, characteristics, and different dosages and also patients comorbidities. 21 –23 When we analyzed the first doses of apixaban prescribed at baseline, non-naive patients had better rates of correct dosage, although in both groups when the initial recommended dose was 5 mg twice daily, it was correctly prescribed to only around half the patients (42.6% in the naive group and 68.9% in the non-naive group). 24
Therapeutic adherence was assessed through pharmacy invoice data for patients initiating treatment in 2013 and 2014 (n = 1971). Among these patients, therapeutic adherence was measured in 43.3% of the apixaban cohort (n = 854) that had at least 1 year of follow-up, representing 32.5% of the naive and 64.6% of the non-naive population. The adherence was considered good in 61.1% of the naive and 64.2% of the non-naive patients. In 1 US-based study, Yao et al found a similar result with 61.9% apixaban adherence (PDC ≥ 80%) within 6 months of follow-up, with apixaban demonstrating the highest adherence rates among the DOAC studied. 25 We used the threshold of 80% MPR for good adherence as defined in the literature, but we also wanted to see the percentage over 120% as it could be a limitation of the MPR formula used to calculate it. 26
Persistence at 1 year was 43.3% for the whole cohort and was being higher for the non-naive patients (64.6%). The overall discontinuation rate for the first month was 33.1%, which was higher than those observed during the same time period in clinical trials (30). The discontinuation rate for apixaban in the phase III clinical trial was 25.3% versus 27.5% for warfarin. 7 There is not a clear explanation for this finding, and this study cannot help in providing potential reasons for it though similar patterns have been observed in other DOAC studies. 27
The rate of persistence in our study, 43.3%, is much lower than that from a population cohort study from the Stockholm region, which was 85.9% after 1 year. 28 Similarly, in another study performed in general practices in the United Kingdom using the Clinical Practice Research Datalink (CPRD) database, treatment persistence with apixaban was also higher, reaching 82.8% at 12 months. 29 When compared to warfarin, patients treated with DOACs generally tend to demonstrate better persistence, although more studies on this topic should be performed. 30
Our results showed that non-naive patients, who mostly received VKA before starting a DOAC, have better treatment persistence than naive patients. This finding suggests that anticoagulant-experienced patients may be more aware of the importance of treatment adherence and, if they were persistent with VKA, which require a frequent INR monitoring, they would be persistent with a “less strict” anticoagulant treatment follow-up, as it has also been reported recently in an US-based study 31 . It also raised an important issue, as a poor adherence and even persistence to anticoagulant treatment in patients with NAVF would be translated into higher stroke risk. 32
We also think that future interventions on patient education for long therapies on asymptomatic diseases, such as DOAC for stroke prevention in patients with NVAF, will have to be developed, especially in naive patients as the under use/under dose .
Strengths and Limitations
The strengths of our study are representativeness for the general population, with a database that covers almost the 80% of the Catalonian population, with complete sociodemographic and health records, long follow-up, and real clinical practice data.
Some specific limitations in our database are the lack of association between GP’s prescriptions and dispensing associated with these prescriptions, and the high number of missing values of the first dose prescribed.
We do not have access to electronic health records from the hospitals and some first prescriptions of DOAC are filled by specialist physicians from the hospitals (mainly Cardiologists).
One specific limitation of the current study is that the HAS-BLED score might be underestimated as we equal the result for the item “Labile INR” to score 0, when no INR was registered.
In Spain, DOACs are less frequently prescribed than in the rest of European countries due to the more restrictive recommendations for their use established by the AEMPS. 8
Because of the high percentage of discontinuation rates, we are now studying the discontinuation rates in the rest of DOAC and comparing them with VKA.
This study has missing data from pharmacy claims and for some variables as it is common in observational studies using electronic databases (information bias). According to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE), we have reported the missing data and as we were not making any association we have just handled with these data by reporting them without performing any statistical inference. 33
Conclusion
Almost all patients treated with apixaban had at least 1 comorbidity. Patients who were treatment-naive had lower risks of stroke and bleeding than patients who were not treatment-naive. In the patient population assessed, <5% of patients were concurrently receiving a medicine potentially associated with a pharmacokinetic drug interaction. The prescribed dose of apixaban did not fully follow the recommended dose, particularly in patients who were treatment-naive. Patients with a prior history of taking oral anticoagulants were more likely to remain persistent to treatment with apixaban.
Footnotes
Authors’ Note
IDIAP Jordi Gol is the institution where the work reported has been done.
The study protocol was approved by the Ethics Committee of the ‘IDIAP Jordi Gol’ and classified by the Spanish Agency of Pharmacy and Medical Products (AEMPS). No inform consents were compile from the patients as the characteristics of the study did not required it.
Data can be accessed through request to the corresponding author.
Acknowledgments
The authors thank María Aragon and Darío García from SIDIAP for data management. We also would like to acknowledge Jose Chaves, from Pfizer, for his helpful comments and global revision of this manuscript.
Authors’ Contribution
J. Cortés contributed to design; contributed to acquisition, analysis, and interpretation; crtically revised the manuscript; gave final approval. M. Giner-Soriano contributed to design; contributed to interpretation; drafted the manuscript; critically revised the manuscript and gave final approval. M.A. Quijada-Manuitt contributed to design; critically revised the manuscript, and gave final approval. R. Morros contributed to conception and design; contributed to interpretation; drafted the manuscript; critically revised the manuscript and gave final approval. All author agree to be accountable for all aspects of work ensuring itegrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Pfizer SLU Spain, and was funded by the Alliance of Pfizer-Bristol-Myers-Squibb.
