Abstract
Aim:
Analyze the relative risks of critical cardiovascular outcomes and mortality associated with adherence to statin treatment in a clinical setting in people with no history of prior cardiovascular disease (CVD).
Methods:
A systematic review of the literature was conducted up to December 2016. The outcomes of interest were cardiovascular fatal or nonfatal events and all-cause mortality.
Results:
A total of 17 articles were included in a qualitative synthesis. Four were case–control nested in a retrospective cohort design and the other 11 were a cohort design. Seven studies compared the best adherer patients with the worst adherers. In the 3 studies (317 603 participants) that considered ischemic heart disease in this group, the pooled reduction in risk was 18% (95% confidence interval [CI]: 14%-22%, I2 = 0%); for the CVD outcome, 2 studies (131 477 participants) showed a pooled reduction in risk of 47% (95% CI: 36%-56%, I2 = 84.7%) with 1 included study showing a much larger reduction than the others; for the cerebrovascular event (CeVD) outcome, 2 studies (155 726 participants) showed a pooled reduction in risk of 26% (95% CI: 18%-34%, I2 = 0%); and for mortality, the reduction in risk was 49% (95% CI: 39%-57%, I2 = 62.4%). The other 4 studies (147 859 participants) compared the most adherent group with the rest. These showed a pooled risk reduction of CVD of 22% (95% CI: 6%-27%, I2 = 0).
Conclusion:
Adherence to statins treatment is shown as a key element for primary prevention, although these are observational data and the risk of bias from confounding cannot be ruled out. Standardization of measures of adherence to treatment would improve comparability between studies. Further research is warranted to design effective interventions to improve patients’ adherence.
Keywords
Introduction
Cardiovascular diseases (CVDs) are 1 of the leading causes of death worldwide. 1 Behavioral risk factors such as unhealthy diet, physical inactivity, tobacco use, and harmful use of alcohol are thought to be responsible for about 68% (38 million) of death globally of the world’s 56 million deaths in 2012. More than 40% were premature deaths under age 70 years. 2 “Hydroxylmethyl glutaryl coenzyme-A reductase inhibitors” (statins) have been shown to be an effective class of pharmaceutical therapy to reduce the incidence of cardiovascular events and mortality in primary 3,4 and secondary prevention. 5,6
Although the literature defines various related concepts, such as persistence, compliance, or discontinuation, 7 –9 the most frequently used and accepted term is adherence. This term implies an active collaboration between the health professional and the patient on the decisions which affect the person’s health. 9
Various studies have found that adherence is suboptimal in chronic asymptomatic diseases. 10,11 Adherence to long-term cardiovascular and noncardiovascular treatments in the general population is around 50% in developed countries and is much lower in developing countries. 7 Naderi et al 11 found that adherence to statin in primary prevention was 57% (95% confidence interval [CI]: 51%-64%) and 76% (95% CI 70%-82%) in secondary prevention. A meta-analysis of nonadherence to statin therapy found that the percentage of adherent patients at 1 year of follow-up was 49.0% for observational studies and 90.3% for randomized trials. 12 Therefore, it is possible that randomized trials overestimate the benefits of statin that would be obtained in the real-world settings.
While several articles have reported some kind of relationship between statin adherence and clinical outcomes using diverse data and methods, 13 –16 the relationship has not been extensively studied in primary prevention. The objective of this study is to estimate the empirical relationship between statin adherence in primary prevention with critical cardiovascular outcomes and mortality from a systematic review of the literature.
Methods
A systematic review was performed in the MedLine database (through PubMed) and Trip Database was searched up to December 2016.
Search Strategy
In MedLine, the terms used in the search strategies were (“Hydroxymethylglutaryl-CoA Reductase Inhibitors” [Mesh] OR statin* OR hmg-coa) AND (“Primary Prevention” [Mesh] OR “primary therapy”) AND (“Medication Adherence” [Mesh] OR “Patient Compliance” [Mesh] OR “statins adherence” OR “patient adherence” OR “patient cooperation”). Trip Database was consulted using free terms: “statins adherence” and “statins discontinuation.” Finally, a search was conducted of the reference list of the articles identified. Only articles written in English or Spanish were included.
Inclusion and Exclusion Criteria.
Inclusion criteria:
Adult men or women in treatment with statins with no recent history of cardiovascular disease but at risk of developing cardiovascular disease (primary prevention, new statin users with no cardiovascular disease at baseline).
The assessment of exposure was the degree of adherence or noncompliance with statins.
The outcomes were relative risks of nonfatal or fatal cardiovascular or cerebrovascular events and overall mortality for adherent versus nonadherent patients.
Exclusion criteria:
Studies conducted only in populations with specific conditions, for example, only smoking participants, only people with obesity, diabetes, and so on, and studies that analyze risk factors associated with adherence.
Studies were excluded which use a comparison group that does not take statin, and studies that analyzed effects in patients who discontinue or are treated intermittently according to their health professionals’ instructions.
Excluded study designs: case reports, case series, review, and simulation and modeling studies.
Data extraction and management
Identification of the articles was carried out by 2 reviewers (E.M.R. and A.O.L.) who selected independently and any discrepancies were agreement with a third person (D.E.). Once the studies had been identified, and duplicate articles excluded, the title and abstract of each review was checked against the inclusion and exclusion criteria before requesting the full text. The review was managed using Reference Manager Software.
Two researchers carried out an independent evaluation of the methodological quality of each study (E.M.R. and A.O.L.) using the Newcastle-Ottawa Scale (NOS) 17 for assessing the quality and risk of bias of nonrandomized studies.
A data extraction form was developed to record bibliographic information, general characteristics of the study included, measure of exposure, and outcome data. The relative risk (RR) measures extracted from the articles were, where possible, those that had been adjusted for observed confounding variables. Relative risks were transformed if necessary so that a RR of less than 1 indicates that better adherence improves CVD outcoms.
Outcomes
Our main end points were CVDs, cardiovascular event, cerebrovascular event, disease-specific mortality, and all-cause mortality. International Classification of Diseases, Ninth Revision codes are diagnosis of chronic heart failure (CHF; codes 398 402 428) or complications of heart disease (codes 429), myocardial infarction or angina (codes 410-414), cerebrovascular events or stroke (codes 430-438), atherosclerosis arterial embolism and thrombosis (codes 440, 443-444), a medical procedure (coronary artery bypass grafting, angiography, angioplasty, stent), and/or coronary revascularizations.
Definition of Exposure
Exposure was the degree of adherence. Broadly, 2 types of comparison are reported in the literature. The first divides the cohort into 4 or more groups according to the degree of adherence (expressed in percentage terms). The worst adherers are taken to be the reference group, and RRs of CVD end points are then estimated for more adherent groups of patients compared to this reference group. In the meta-analysis, we then extract and pool the RRs for the most adherent patients compared to the least adherent, for each end point.
Other studies divide the cohort into 2 groups at some arbitrary cut point of adherence, for example, 60%, 75%, or 80%. The study then reports RRs comparing CVD end points for the more adherent group with the less adherent group. In the meta-analysis, we extract and pool these RRs for each end point.
Heterogeneity between studies was assessed using Higgin I2, where I2 values of >50% represent a substantial level of heterogeneity. 18 A random-effects model was used to combine study-specific RR. The analyses were performed with Stata.v13 software.
Results
Characteristics of Included Studies
The initial search identified 155 articles from which 113 studies were excluded as duplicates or not meeting the inclusion criteria from title and abstract. The most common reason for exclusion was that in spite of studying aspects relating to the research question, the articles focused on factors associated with adherence, or focused on secondary prevention, or familial hypercholesterolemia. After reading the full text, we excluded a further 30 articles (see Figure 1). Four of these excluded articles were reviews which were used only to identify relevant articles from their reference lists and for background information or discussion. 19 –22 Finally, 12 articles were included from the search plus a further 5 were found via cross references or hand searching. Of the 17 included articles, 4 were case–control nested in a retrospective cohort design and the other 11 had a cohort design.

Study flow diagram.
Table 1 shows patient and study characteristics of included studies. One study (with results were published across 2 articles) recruited male participants as a follow-on study from a previous RCT. 30,31 A Finnish study published 2 articles, 34,35 one of which only included women. 34 The mean age within studies ranged from 51.4 years 32 to 66.5 years 24 . The mean time of follow-up ranged from 1.6 years 36 to 5.8 years. 28 Several articles required a minimum follow-up time as an inclusion criteria. 23,26,27,29,33 In 11 of the cohort studies, 23 –35 the minimum follow-up was 1 year, 33 and in 4 nested case–control studies, 36 –39 the range of follow-up was between 6 months and 6.5 years. 38,39
Characteristics Patients of Studies Included.

Abbreviations: CAD, coronary artery disease; CCB, calcium-channel blocker; CeVD, cerebrovascular events; CHF, chronic heart failure; CVD, cardiovascular disease; LDL, Low Density Lipoprotein ;MPR, medication possession rate; PDC, proportion of days covered; SD, standard deviation; SPAA, single-pill amlodipine/atorvastatin; WOSCOPS, West of Scotland Coronary Prevention Study.
aData referred to the first year if there is no express indication.
bNew users defined no prior statin prescription or lipid-lowering drugs filled during the 2 years preceding the enrollment date.
cNo history of CV events in 2 years previous to index date.
dNew users defined no prior statin prescription or lipid-lowering drugs filled during the 1 year preceding the enrollment date.
eNo history of CV events in 1 year previous to index date.
fSample size decreased after 2 years due to noncontinuous health plan enrollment.
gOnly patients surviving the 3-year follow-up period were included in the logistic analysis.
hNew users defined no prior statin prescription or lipid-lowering drugs filled during the 3 years preceding the enrollment date.
iNo history of CV events in 5 years previous to index date.
jNo claims for the index prescriptions for 6 months
kNew users defined no prior statin prescription filled during the 7 years preceding (when de register was established) and lipid-lowering drugs filled during the 3 year preceding the enrollment date. Patients who died or were institutionalized long term within 3 years prior to statin or had an outcome event within the first year after statin initiation were excluded.
lNo history of CV events in 3 years previous to index date.
mOnly information about primary prevention.
nOnly patients who initiated statin therapy for primary prevention of CVD.
Four case control studies nested in a cohort were found, 36 –39 and all of them were performed by the same Canadian research team (Table 1). In the nested case–control study, cases of a disease that occur in a defined cohort are identified and, for each, a specified number of matched controls is selected from among those in the cohort who have not developed the disease by the time of disease occurrence in the case. The cases were defined as those patients who have CeVD, 37 coronary artery disease (CAD), 36,38 or CHF. 39 Meanwhile, all controls (15 controls 37 –39 -20 controls 36 per case) were randomly selected from the source population (cohort) of the same age and follow-up using density sampling. All cohorts were included in the analysis as this study design should lead to independent sampling with little risk of duplication of cases or controls.
Statin Adherence
Studies were heterogeneous in the definition of adherence and cut points used to quantify the degree of adherence (Table 1). The most common definition of adherence was proportion of days covered (PDC). 23 –25,27,28,32 –35 This is calculated by dividing the quantity of statin dispensed by the total time interval from index date to events, deaths or cardiovascular events, whichever occurred earlier. Another indicator used was medication possession rate (MPR). 29,37 –39 This is defined as number of days of medication supplied within the refill interval divided by number of days in refill interval. In the West of Scotland Coronary Prevention Study (WOSCOPS) studies, 30,31 adherence was measured by whether a subject attended a scheduled trial visit (1 visit every 3 months) and was issued or not with a further supply of trial medication according to the doctor’s indication. Bouchard et al 36 used the percentage of the prescribed doses of medication actually taken by the patient over a specified period, and finally, Poluzzi et al 26 used the amount of statins received during the period (number of tablets purchased). The majority of studies, the denominator period used to calculate adherence was the total follow-up of the cohort, except for Chapman et al 33 and Rublee et al, 32 which categorized the patient into adherent or not over the first 6 months or used a “anniversary model”: “a 90-day period at the end of the 1-year assessment period was used as the measure of discontinuation, and patients who did not have pill coverage during these 90 days were considered nonadherent.” 32p.25 Lavikainen et al 34 and Rannanheimo et al 35 exposure was defined as baseline and time-varying cumulative adherence, and statins adherence was measured from as PDC at first-year intervals since statin initiation. Four studies required a minimum level of adherence to statin 28,34,36,33 in order to include the patient in the analysis: PDC >20%, 26 PDC >80% during first year, 24,25 or patients purchases amount of statins ≥85%. 28 Table 1 shows adherence rates as measured in each study.
In 4 case–control studies nested in a cohort, adherence for cases was calculated from the cohort entry date to the date of cardiovascular event, while for the controls it was from the cohort entry date to the date of selection.
Risk of Bias in Included Studies
In general, we judged the individual studies to have a low risk of bias, with similar scores in the NOS scales (Table 2). The WOSCOPS study 30,31 recruited patients as a follow-on study from a previous RCT, and this study was judged as the higher risk of bias in terms of patient selection. Most studies included patients with no history of CVD events in given period prior to the index date, except in 4 studies. 24,27,30,31 None of studies reported the number of participants lost to follow-up. With the exception of 2 studies, 30,31 all included studies had more than 19 000 participants and used a pharmacy claims database as the source of data on adherence and outcome.
Methodological Quality Summary.

Abbreviation: WOSCOPS, West of Scotland Coronary Prevention Study.
aCohort Studies: (1) representativeness of the exposed cohort; (2) selection of nonexposed cohort; (3) ascertainment of exposure; (4) outcome not present at beginning; (5) comparability of cohorts; (6) assessment of outcome; (7) follow-up long enough; (8) adequacy of follow-up.
bNested case–control studies: (9) is the case definition adequate? (10) Representativeness of the cases; (11) selection of the controls; (12) definition of the controls; (13) comparability of cases and controls; (14) ascertain of exposure; (15) same method of ascertain for cases and control; (16) nonresponse rate.
Effects of Adherence on Risk of Developing Critical Cardiovascular Outcomes and Mortality
Tables 3 and 4 show the effects of adherence to statin on clinical outcome (heart diseases, cardiovascular events, and mortality).
Effects of Adherence on Critical Cardiovascular Outcomes and Mortality: Cohort Studies.

Abbreviations: AMI, Acute Miocardial Infarction; ATC, Anatomical Therapeutic Chemical classification system; CCB, calcium-channel blocker; CHD, Coronary Heart Disease; CI, confidence interval; CV, cardiovascular; HR, Hazard Ratio; ICD, International Classification of Diseases; IHD, ischemic heart disease; MPR, medication possession rate; OR, odds ratio; PDC, proportion of days covered; SPAA, single-pill amlodipine/atorvastatin.
Effects of Adherence on Critical Cardiovascular Outcomes: Nested Case–Control in the First Year and After First Year of Follow-Up and Their Controls.
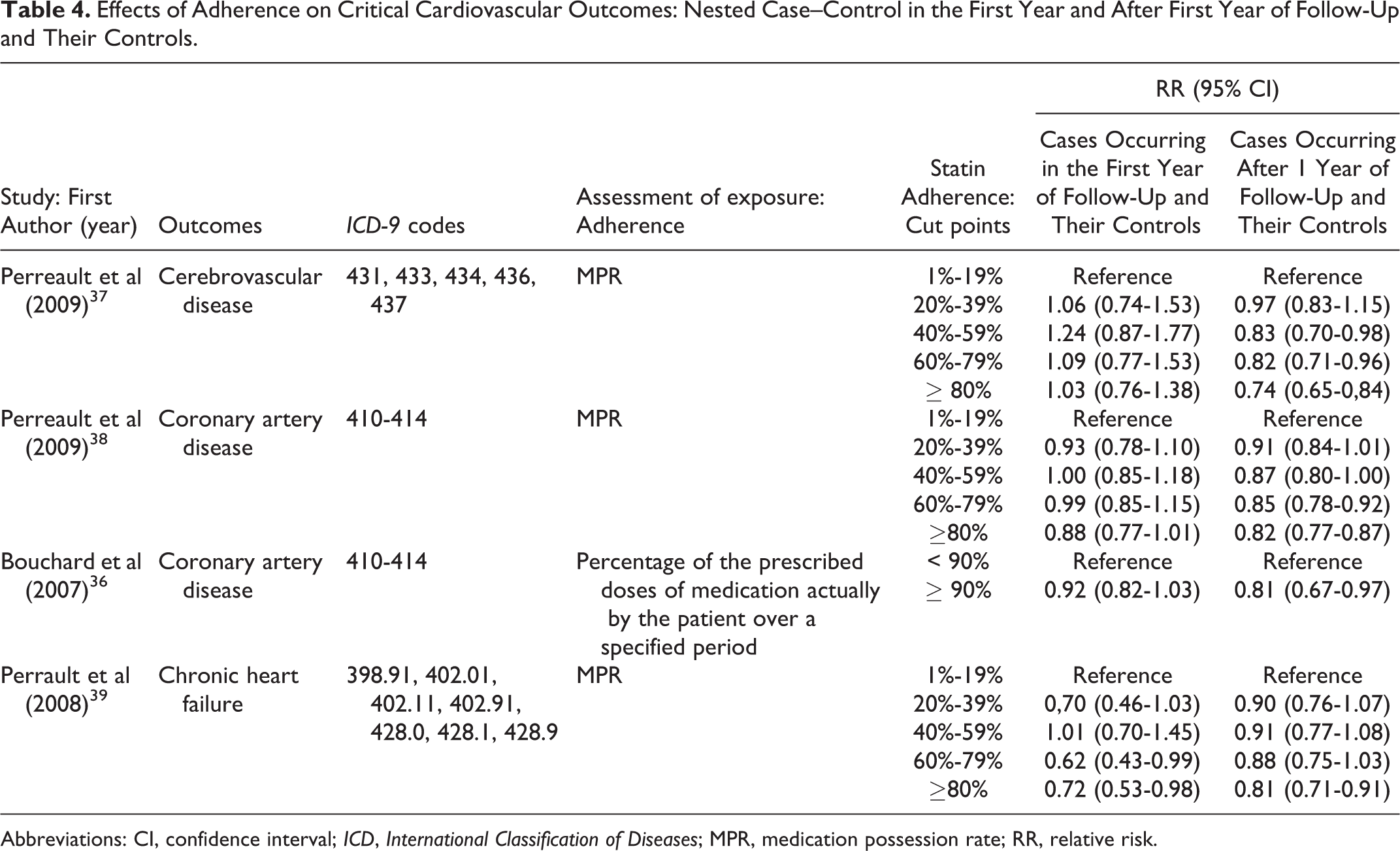
Abbreviations: CI, confidence interval; ICD, International Classification of Diseases; MPR, medication possession rate; RR, relative risk.
The nested case–control studies stratified their results by outcomes in the first year of statin use and outcomes in subsequent years (Table 4). Figure 2 shows the increase or reduction in CVD risk in nested case–control studies comparing highest and lowest levels of adherence, stratified by time since starting statins. The relationship between the level of adherence and clinical outcome tends to be stronger after the first year for CeVD, and CAD, although not apparently for CHF.
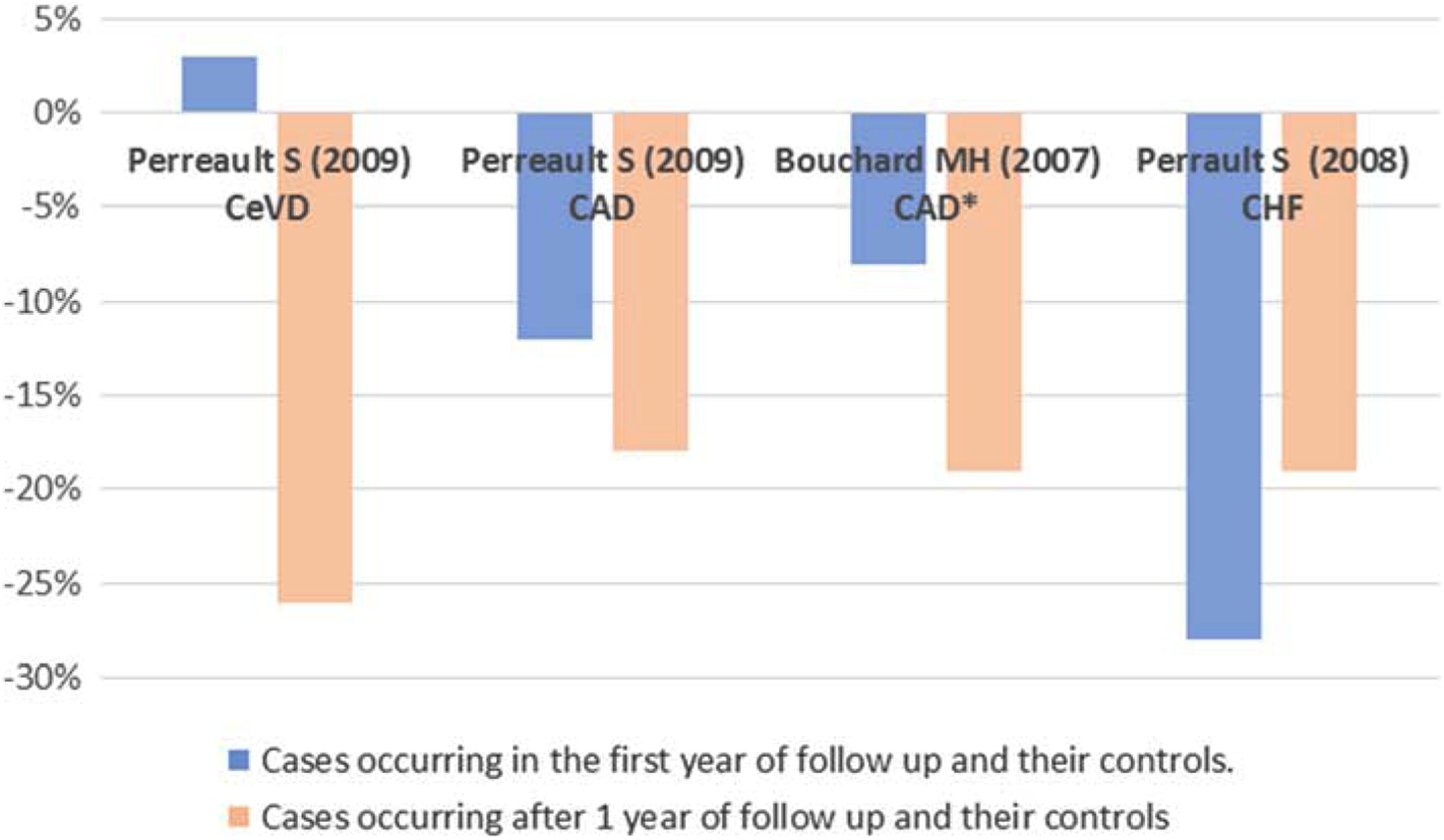
Adherence and relative risk clinical outcomes: nested case–control in the first year and after first year of follow-up and their controls.
Pooled Comparison of Best Adherer Versus Worse Adherer Patients
Figure 3 shows the forest plots of pooled RRs for outcomes of ischemic heart disease (IHD), cardiovascular events, cerebrovascular events, and mortality for studies that compared the best adherer patients (≥75%) with the worst adherers (<40%). For the 3 studies (n/N = 21 050/317 603) that considered IHD in this group, the pooled reduction in risk was 18% (95% CI: 14%-22%, I2 = 0%). This means that best adherers have an 18% lower risk of IHD events than the worst adherents. For the CVD outcome, 2 studies (n/N = 10 987/182 661) showed a pooled reduction in risk of 47% (95% CI: 36%-56%, I2 = 84.7%), with 1 study showing a much bigger effect than the other. 25 For the CeVD outcome, 2 studies (n/N = 4511/131 477) showed a pooled reduction in risk of 26% (95% CI: 18%-34%, I2 = 0%). A pooled risk reduction of 49% (95% CI: 39%-57%, I2 = 62.4%) was observed for mortality in 2 studies (n/N = 5024/155 726).
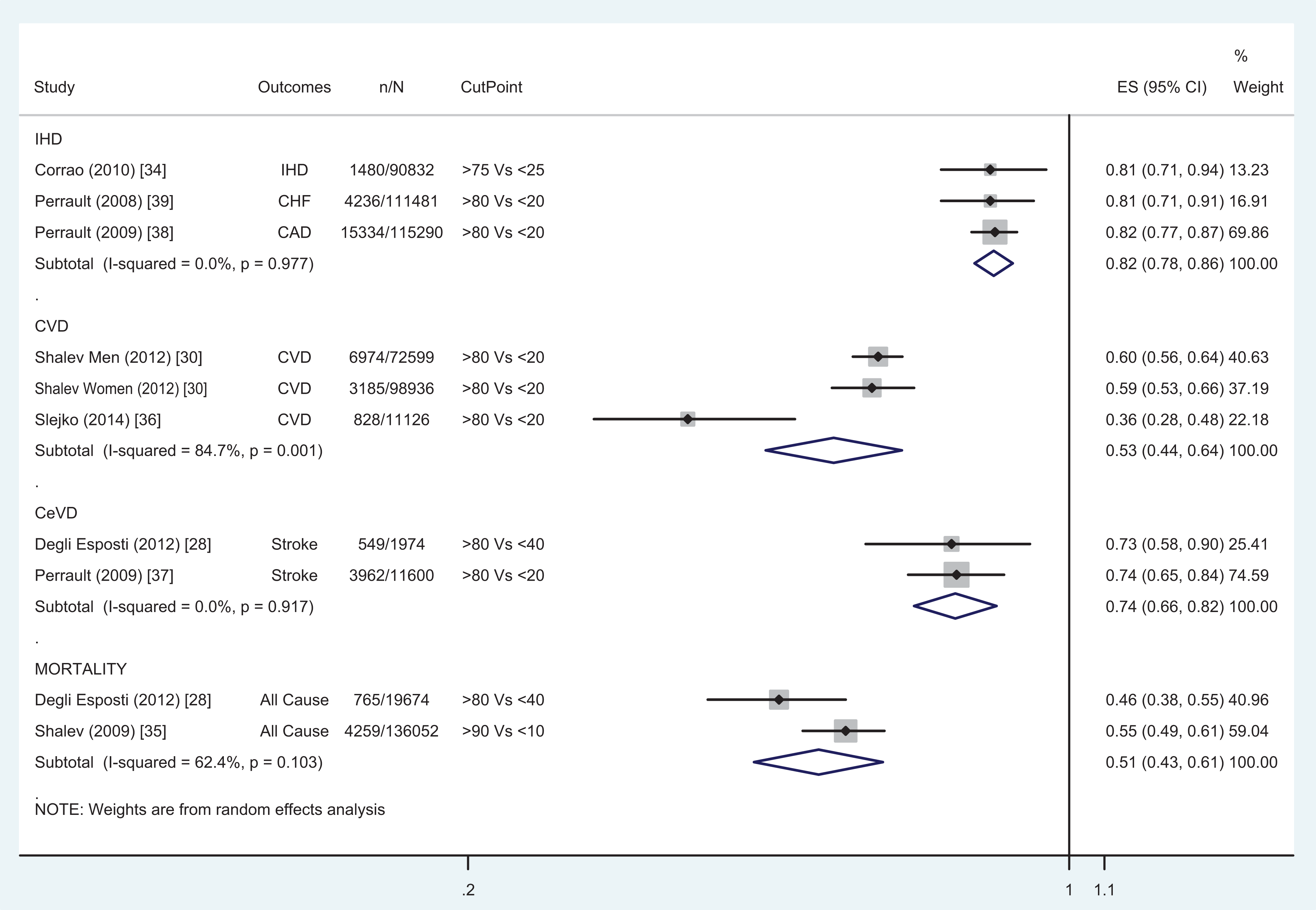
Meta-analysis 1: Comparison of relative risk of cardiovascular events between best adherent group and worst adherent group.
Pooled Comparison of Best Adherers Versus All Other Patients
Figure 4 shows a pooled risk reduction of 22% (95% CI: 16%-27%, I2 = 0%) for all CVD events, for 4 studies (n/N = 5474/147 859) that compared the most adherent group (≥60%) with the rest (<60%).

Meta-analysis 2: Comparison of relative risk of cardiovascular events between patients above a given cut point of adherence with those below.
Discussion
The results of this review show the benefits of good adherence to statin medication on primary prevention of CVD. On average over all end points, the most adherent patients had 32% fewer mortality, IHD, stroke, or cardiovascular events than the least adherent, and the most adherent had 22% fewer CVD events when compared with the rest of the sample.
Other studies have reviewed adherence to statins in primary and secondary prevention, 19 –21,40,41 but as far as we know, this is the first systematic review conducted in primary prevention that summarized results using meta-analysis. We only included articles in English and Spanish, and we did not search the “grey” literature (ie, non-peer-reviewed articles in unconventional information channels). We may therefore have missed some relevant studies, though the risk of bias is likely to be small.
There was considerable heterogeneity in the definition of the variables across the included studies, in terms of the measures of adherence used, the cut points, the study design, and the CVD outcome. Most studies classified adherence according to behavior during the entire study prior to the CVD event, and only 2 studies classified patients according to their level of adherence prior to the index date. 27,36 We controlled for this heterogeneity to some extent by stratifying the analysis by CVD end point, type of study design, and cut point. While the quality of these studies was considered generally reasonable, these are observational data and hence have all the corresponding caveats and confounders. For example, patients who adhere to treatment also may be more likely to adhere to other aspects of healthy lifestyle. Under these conditions, the observed effect would be picking up the impact of healthy lifestyle on CVD, not just adherence to statin therapy. The majority of the studies performed a Cox regression to adjust for differences in observed variables at baseline, which controls at least for observed confounders. Other factors such as intensity of statin, 42,43 which we were unable to take account of in this study, may influence both the effectiveness of statins and the degree of adherence.
The articles included in this review evaluated the degree of adherence by indirect methods based on the analysis of the number of units of statin dispensed (the majority of them based on refill claims data over long time periods). This is more properly described as “persistence” than adherence. The advantage is that these data can be collected objectively at low cost and represent the “real-world” settings. However, the measure lacks the patient’s perspective as it does not take account the patient’s acceptance of the physician’s advice nor compliance with the dosing, timing, and so on. Most studies categorized patients as adherent according to PDC or MPR calculated from the start of follow-up to the time of a cardiovascular events, disenrollment from health system, death, or end of time of follow-up, whichever occurred first, 23 –29,36 –38 and used this as the “exposure” variable. An exception was Slejko et al 25 who estimated adherence in yearly segments from the index statin prescription. Furthermore, Lavikainen et al 34 and Rannanheimo et al 35 calculated the statins adherence in intervals of 1 year from statin initiation, then adherence models were used in order to estimate each individual’s probability of having the observed adherence at 3 time points: 12, 24, and 36 months from statin treatment initiation. Another difference observed in the measurement of adherence was some studies have suggested that the best results are obtained after a full year at good levels of adherence, and they only included the results are obtained before this time. 29,39 –41,44 Despite the different definitions, and cut points used in the measure of adherence to statins, our meta-analytic approach, by stratifying for these factors, may be considered a reasonable approximation. 41 These results reinforce the recommendation about the need for standardization of measures of adherence and the common data elements involved in key cardiovascular end point events for future research. 42,43
The primary motivation for this study was to estimate how effective statins might be for primary prevention of CVD in the real-world settings. Self-selection into RCTs may end up recruiting patients who are most likely to be adherent, and this may imply that RCTs overstate the benefits of statins. 45,46 Furthermore, patient adherence in the real world is not as easily monitored or evaluated. 44 Investigation is needed into interventions that improve adherence in the real world. Improved adherence might increase pharmaceutical costs to payers (if more statins are dispensed) and may imply the need for more intensive follow-up in primary care for some patients but should also improve health outcomes and could be highly cost-effective, 29,47,48 though there are unlikely to be easy solutions. 48,49
This study at least highlights the need for health professionals to identify and work with patients at risk of poor adherence. 15,40 Health professionals themselves may need to develop or to be trained in this area. World Health Organization 7 promote an “adherence counseling toolkit” adaptable to different socioeconomic settings and covering patients’ information needs, clinical decision making, and behavioral tools for health professional.
Footnotes
Acknowledgments
The authors are grateful to Simon Thompson, Stephen Kaptoge, Daysi Yoe Ling Chang Chan for advice and support, and Camila Higueras, librarian EASP.
Author Contributions
Martin-Ruiz and Antonio Olry-de-Labry Lima contributed to design; acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Ricardo Ocaña-Riola contributed to analysis and interpretation, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Epstein, D contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed by the EPIC-CVD project under the European Commission. Directorate General for research & Innovation FP 7 Health 2011. Grant Agreement number 279233.
