Abstract
Acetylsalicylic acid, or aspirin, is perhaps the most well-studied drug in human history, but controversy persists regarding both optimal dose and its use in the primary prevention of atherothrombotic events. This article reviews the following: the effect of aspirin upon the cyclooxygenase pathway; clinical trials of aspirin for both secondary and primary prevention; prospective and retrospective studies of aspirin dose; the potential interaction between aspirin and ticagrelor; and the concept of aspirin resistance. It concludes with a review of major society guidelines regarding aspirin and offers a perspective on the evidence-based use of aspirin in clinical practice.
Mechanism of Action
Acetylsalicylic acid, or aspirin, is absorbed by the stomach and small intestine and hydrolyzed by esterases to salicylic acid. Bioavailability varies by preparation; it approaches 50% in uncoated aspirin preparations, and peak plasma levels are reached 40 minutes after an oral dose. Enteric-coated preparations, however, have lower bioavailability and a longer time to peak plasma levels. 1,2 Although aspirin is rapidly cleared from the circulation, with a half-life of 15 to 20 minutes, it irreversibly acetylates both of the platelet enzymes cyclooxygenase (COX)-1 and COX-2; this effect persists for the life span of the platelet, which is typically around 10 days. 3 Aspirin thus has a sustained pharmacodynamic effect upon platelets, despite its short half-life. It is notable that only low doses of aspirin are necessary to inhibit COX-1, whereas much higher doses are required to inhibit COX-2. 4 This accounts for the higher doses of aspirin required to attenuate the COX-2-dependent effects of hyperalgesia and inflammation. 3,5 In contrast, the administration of as little as 30 mg of daily aspirin results in near-complete suppression of platelet-derived thromboxane A2 (TXA2). 6
Both COX-1 (also known as prostaglandin H-synthase 1) and COX-2 catalyze the conversion of arachidonic acid, derived from cell membrane phospholipids, to prostaglandin H2 (Figure 1). Tissue-specific isomerases then convert prostaglandin H2 to prostanoids, the most important being TXA2 and prostaglandin I2 (also known as prostacyclin). 2 TXA2 is primarily generated by COX-1 within platelets, in response to platelet agonists like collagen, adenosine diphosphate, and thrombin. 7 The TXA2 then binds its receptor on the surface of platelets, thereby inducing irreversible platelet aggregation. 2 In addition, TXA2 causes vasoconstriction and proliferation of vascular smooth muscle cells and promotes atherosclerosis. 8

Aspirin mechanism of action. PL indicates phospholipid; COX, cyclooxygenase; PG, prostaglandin; TX, thromboxane; PG, prostaglandin.
In contrast to TXA2, prostacyclin is primarily produced by COX-2 within vascular endothelial cells. 9,10 The effects of prostacyclin are essentially opposite of TXA2, as prostacyclin inhibits platelet aggregation, causes vasodilation, and inhibits vascular smooth muscle cell proliferation and atherosclerosis by binding its receptor on the surface of platelets. 2 Importantly, COX-2 is less susceptible to inhibition by low doses of aspirin. 11 This explains why low doses of aspirin are thromboprotective, although it is unclear whether higher doses of aspirin are more thrombogenic due to inhibition of the prostacyclin pathway. It does appear, however, that selective inhibition of COX-2 increases the risk of myocardial infarction (MI) and stroke. 12
Aspirin and Secondary Prevention Clinical Trials
Although a precursor of aspirin was used as an analgesic and antipyretic by Hippocrates around 400
Cardiovascular Indications for Secondary Prevention With Aspirin Therapy.

Abbreviations: MI, myocardial infarction; CVA, cerebrovascular accident.
The Antithrombotic Trialists’ Collaboration summarized this breadth of clinical trials involving aspirin for the secondary prevention of vascular events in a comprehensive meta-analysis of 195 randomized studies. The outcome of serious vascular events, which included vascular death, nonfatal MI, and nonfatal stroke, was examined in high-risk populations of patients with vascular disease. Overall, antiplatelet therapy (primarily aspirin) reduced serious vascular events by one-fourth (10.7% vs 13.2%, P < .0001). 21 There were also significant reductions in vascular events for patients with acute MI, history of MI, stable and unstable angina, acute stroke, history of stroke or transient ischemic attack (TIA), and peripheral arterial disease. A subsequent meta-analysis by the same group included only patients with a history of MI, stroke, or TIA, encompassing 16 trials comparing aspirin alone to placebo. Again, there was a significant reduction in serious vascular events with aspirin (6.7% vs 8.2% per year, P < .0001). 17
Aspirin and Primary Prevention
The role of aspirin for the primary prevention of cardiovascular events is more controversial, as a relatively small reduction in events must be weighed against an increase in bleeding. This was addressed by the Antithrombotic Trialists’ Collaboration in 2009, in a meta-analysis of 6 large randomized trials of aspirin and primary prevention. Among 95 000 patients without a history of occlusive vascular disease, there was a small but significant reduction in nonfatal MI with aspirin versus placebo (0.18% vs 0.23% per year, P < .001); there was no reduction in vascular death or stroke. Furthermore, there was a small but significant increase in gastrointestinal and extracranial bleeding with aspirin therapy (0.10% vs 0.07% per year, P < 0.001). 17 The authors also observed that risk factors for coronary disease were also risk factors for bleeding; in other words, even among patients at moderate risk of developing vascular disease, the benefit of aspirin might be offset by bleeding risk.
Two recent meta-analyses of aspirin for primary prevention added 3 trials and 7105 patients to the previous 6 trials but came to similar conclusions. Bartolucci et al found a small benefit with aspirin in regards to nonfatal MI (odds ratio 0.81, 95% confidence interval [CI] 0.67-0.99, P = .04) but no effect on stroke or vascular death. 22 Rates of gastrointestinal bleeding with aspirin ranged from 0.3% to 4.5% with aspirin, but the risk of bleeding versus placebo was not analyzed. Seshasai et al, analyzing the same population, found a reduction in nonfatal MI (odds ratio 0.80, 95% CI 0.67-0.96) but no effect upon stroke or vascular mortality. There was a significant increase in both total bleeding (odds ratio 1.70, 95% CI 1.17-2.46) and serious bleeding episodes (odds ratio 1.31, 95% CI 1.14-1.50). 23 In other words, the number needed to treat with aspirin to prevent 1 nonfatal MI was 162, and the number needed to harm in regards to nontrivial bleeding was 73. Randomized trials of aspirin for primary prevention are summarized in Table 2.
Recent studies have also examined the role of aspirin for primary prevention specifically in diabetes, which is often referred to as a coronary artery disease “risk equivalent.” Somewhat surprisingly, there has been no clear benefit demonstrated. Two relatively recent and large randomized studies, the Japanese Primary Prevention of Atherosclerosis with Aspirin study (2539 patients with type 2 diabetes) and the Prevention of Progression of Arterial Disease in Diabetes trial (1276 patients with type 1 or 2 diabetes), showed no reduction in vascular events or death, with 81 to 100 mg aspirin versus placebo. 30,31 Similarly, a meta-analysis of 6 randomized trials of aspirin for primary prevention in diabetes did not show reduction in cardiovascular events or mortality, although aspirin did appear to reduce MI among men (relative risk 0.57, 95% CI 0.34-0.94, P = .03). 33 Two ongoing randomized trials of aspirin in diabetics, A Study of Cardiovascular Events in Diabetes (ASCEND) 34 and Aspirin and Simvastatin Combination for Cardiovascular Events Prevention Trial in Diabetes (ACCEPT-D), 35 may provide further clarity.
The possibility of gender-specific effects of aspirin for primary prevention was raised by a meta-analysis in 2006, which showed a 17% reduction (odds ratio 0.83, 95% CI 0.70-0.97, P = 0.03) in stroke for women without a reduction in MI or cardiovascular mortality; among men, there was a 33% reduction in MI without an effect upon stroke or mortality. 36 This was not entirely surprising, as the Women’s Health Study showed similar results and contributed the great majority of patients to the meta-analysis. 29 The apparent gender difference, however, may have arisen from the fact that most women enrolled in the Women’s Health Study were younger than 65; stroke is much more common in this population than MI. Furthermore, previous meta-analyses have shown no gender difference in regard to primary or secondary prevention with aspirin. 17,21
Although the risk to benefit ratio for aspirin in regards to secondary prevention is decidedly in the favor of treatment, decisions regarding aspirin for primary prevention must consider the small but significant increase in gastrointestinal bleeding with aspirin therapy. Such bleeding events are rare; in the 2009 meta-analysis by the Antithrombotic Trialists’ Collaboration, there was only 0.03% excess per year of extracranial bleeding in patients taking aspirin. Similarly, there was only a 0.05% reduction per year in nonfatal MI. 17 Therefore, when aspirin is used for primary prevention, less than 1 in 1000 patients would avoid a vascular event, and a similarly small number would suffer a major extracranial bleed. 3 It may be reasonable to consider primary prevention with aspirin for patients at moderate to high risk, defined as a 10% to 20% 10-year risk of cardiovascular events. In such a population, the reduction in nonfatal MI is closely balanced by the increase in major bleeding. 37 Such decisions, however, must also take into account the risk factors for gastrointestinal bleeding: advanced age, a history of ulcer disease, and concomitant nonsteroidal anti-inflammatory drugs. 38 In addition, clinical trials may underestimate the incidence of major bleeding; “real-world” rates of bleeding on aspirin may be as much as 5 times higher. 39
Effective Aspirin Dose
There is no question that low-dose aspirin (eg, less than 100 mg daily) is effective for the prevention of vascular events. A dose of 75 mg daily reduced MI and death in patients with stable 14 and unstable angina. 15 It was also effective for secondary prevention of cerebrovascular accident (CVA) in patients with TIA or minor CVA 40 and reduced CVA after carotid endarterectomy. 19 And aspirin 160 mg daily reduced death in patients with acute MI 16 and decreased recurrent CVA in patients with acute ischemic stroke. 41
The Antithrombotic Trialists’ Collaboration offered indirect comparisons of different aspirin doses ranging from 50 to 1500 mg daily for secondary prevention. There was a reduction in vascular death, MI, and CVA for all doses except those less than 75 mg daily, but this dose was studied in only 3 trials. 21 There did appear to be a trend for more benefit with a dose of 75 to 150 mg, although there was considerable overlap among CIs (Table 3). Similarly, a retrospective analysis of the Percutaneous Coronary Intervention–Clopidogrel in Unstable Angina to Prevent Recurrent Events (PCI-CURE) study examined the effect of aspirin dose among patients with acute coronary syndromes undergoing PCI. Patients receiving high (≥200 mg) and moderate (101-199 mg) aspirin doses had similar outcomes to low doses (≤100 mg) in regards to cardiovascular death, MI, and stroke. Major bleeding, however, was more frequent in the high- versus low-dose aspirin group: 3.9% versus 1.9% (hazard ratio 2.05, 95% CI 1.20-3.50, P = .009).
Trials of Aspirin for Primary Prevention.
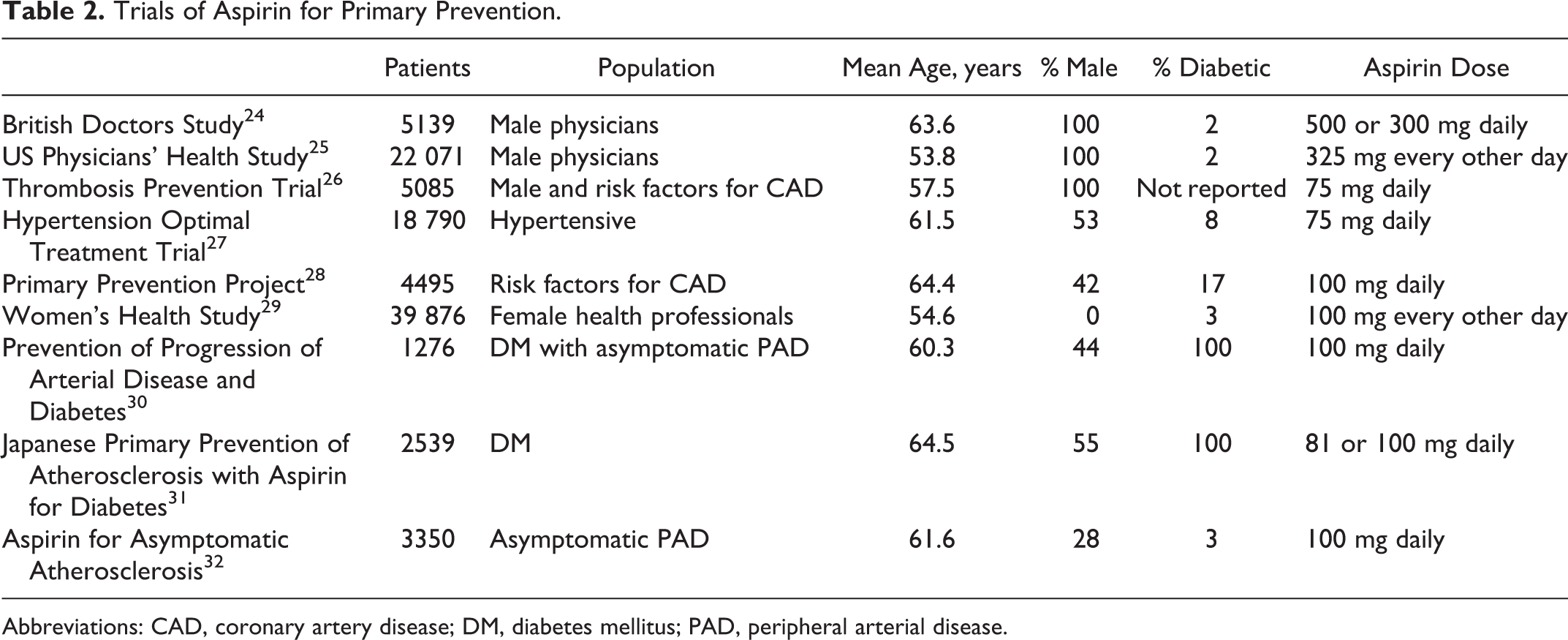
Abbreviations: CAD, coronary artery disease; DM, diabetes mellitus; PAD, peripheral arterial disease.
Meta-Analysis of Aspirin Dose in Secondary Prevention.a

a Adapted from Antithrombotic Trialists’ Collaboration. 21
There are relatively few clinical trials, however, that have directly compared different aspirin doses. The Dutch TIA study enrolled 3131 patients with TIA or minor CVA and randomized between 30 mg versus 283 mg of aspirin daily. There was no difference in regards to the primary end point of vascular death, CVA, and MI; minor bleeding was more common with the higher dose. 42 Similarly, the United Kindgom TIA study enrolled 2435 patients with TIA or minor CVA and randomized between placebo, 300 mg aspirin daily, and 600 mg aspirin twice daily. Again, there was no difference in regards to the primary end point of death, CVA, and MI, but upper gastrointestinal side effects like nausea and heartburn were more common with the 1200 mg than the 300 mg dose (odds ratio 1.54, 95% CI 1.25-1.89). 43 And the Aspirin and Carotid Endarterectomy trial randomized 2849 patients scheduled for carotid endarterectomy to aspirin 81 mg, 325 mg, 650 mg, or 1300 mg daily. When combining the 2 groups receiving lower doses, there was a benefit in regards to death, CVA, and MI at 3 months when compared to the high-dose groups: 6.2% versus 8.4% (P = .03, number needed to treat = 46). 44
More recently, the Clopidogrel and Aspirin Optimal Dose Usage to Reduce Recurrent Events–Seventh Organization to Assess Strategies in Ischemic Syndromes (CURRENT-OASIS 7) trial randomized 25 086 patients with an acute coronary syndrome to double-dose versus standard dose clopidogrel and high- (300-325 mg) versus low-dose (75-100 mg) aspirin. At 1 month, there was no difference in cardiovascular death, MI, or CVA between high- and low-dose aspirin (4.2% vs 4.4%, P = .61); there was a small increase in major gastrointestinal bleeding with high-dose aspirin (0.4% vs 0.2%, P = .04) but no difference in overall major bleeding. 45
Randomized trials of low- versus high-aspirin dose thus have shown no cardiovascular benefit to higher doses, with a trend toward harm in some instances. In addition, higher dose aspirin appears to increase gastrointestinal toxicity in the above trials. This is supported by epidemiological studies that show gastrointestinal side effects in patients taking aspirin are dose- dependent. 46 Although all aspirin doses increase gastrointestinal toxicity, this risk appears higher with doses greater than 300 mg. 47 In other words, there is possible cardiovascular harm and definite gastrointestinal toxicity with higher doses of aspirin. The lowest effective dose of aspirin, usually 81 mg daily, would appear most prudent.
Aspirin and Ticagrelor
Aspirin dose played a prominent role in the recent debate surrounding the Study of Platelet Inhibition and Patient Outcomes (PLATO). The trial was a randomized comparison of ticagrelor, a novel and reversible P2Y12 inhibitor, versus clopidogrel in 18 624 patients with an acute coronary syndrome. Ticagrelor reduced the primary end point of cardiovascular death, MI, and CVA: 9.8% versus 11.7% (hazard ratio 0.84, 95% CI 0.77-0.92, P > .001) 48 Post hoc subgroup analysis, however, revealed two surprising points: first, there did not appear to be a benefit of ticagrelor in patients in the United States; and second, this appeared to be linked to aspirin dose. 49 More specifically, there was an interaction between region and aspirin dose; it appeared that patients taking a maintenance dose >100 mg did not derive a benefit from ticagrelor (Table 4). Furthermore, systematic issues in clinical trial conduct were excluded.
Outcomes in the PLATO Trial by Geographic Region and Aspirin Maintenance Dose.a

Abbreviations: CI, confidence interval; PLATO, Platelet Inhibition and Patient Outcomes.
a Adapted from Mahaffey et al. 50
Potential Mechanisms for Aspirin Resistance.a

Abbreviations: ADP, adenosine diphosphate; COX, cyclooxygenase; TX, thromboxane; NSAID, nonsteroidal anti-inflammatory drug; PGI2, prostaglandin.
A US Food and Drug Administration (FDA) Advisory Panel suggested, however, that the play of chance was a plausible explanation for this US paradox. 49 A subsequent analysis by the PLATO investigators quantified the probability of a particular region favoring clopidogrel by chance alone was 32%; the possibility of an interaction with aspirin maintenance dose, however, could not be excluded. 50 One possible biochemical explanation relates to the degree of P2Y12 blockade with ticagrelor versus clopidogrel. In a setting of near-complete P2Y12 inhibition, as with ticagrelor, TXA2 production is also inhibited; in this setting, aspirin may not enhance platelet inhibition. 51 This may then lead to inhibition of COX in vascular endothelium rather than platelets, with deleterious vascular effects akin to that of nonsteroidal anti-inflammatory drugs (NSAIDS). 52 Regardless of the potential mechanism, the FDA issued a boxed warning stating that aspirin doses greater than 100 mg decrease the effectiveness of ticagrelor. The PrEvention with TicaGrelor of SecondAry Thrombotic Events in High-RiSk Patients with Prior AcUte Coronary Syndrome–Thrombolysis In Myocardial Infarction (PEGASUS TIMI) 54 trial, a study of ticagrelor and aspirin maintenance doses between 75 and 150 mg in patients with a history of MI, will provide further insight regarding this potential interaction. 53
Aspirin Resistance
The concept of aspirin resistance emerged as a possible explanation for the occurrence of occlusive vascular events despite aspirin therapy. Such events, of course, are multifactorial and may occur despite appropriate platelet inhibition by aspirin. Aspirin resistance, therefore, is best defined as the inability of the drug to inhibit its target, COX-1, and subsequent TXA2 production. 54 As a result, a number of assays have emerged to approximate platelet function and aggregation. Patients with higher than expected platelet aggregation on aspirin have then been identified as having “high on-treatment platelet reactivity,” just as patients on clopidogrel have been similarly labeled. A meta-analysis of 15 studies confirmed that high on-treatment platelet reactivity in patients taking aspirin is associated with a 4-fold increase in cardiovascular events. 55
A wide variety of assays has been studied, including the VerifyNow Aspirin assay, the Platelet Function Analyzer (PFA)-100, serum levels of TXB2, urine levels of 11-dehydro TXB2, and light transmission aggregometry (LTA). 56 Not surprisingly, the prevalence of high on-treatment platelet reactivity varies widely: 2% with serum TXB2, 4% with LTA using arachidonic acid, 6.7% with VerifyNow Aspirin, 51.7% for LTA using 20 μmol adenosine diphosphate (ADP), and 59.5% for the PFA-100. 57,58 Measures that are more specific for the COX-1 pathway, such as serum TXB2 and LTA with arachidonic acid, suggest that high on-treatment platelet reactivity is more rare; in one study, the incidence was <1%. 59 Further complicating interpretation is that measurements of aspirin resistance may vary temporally in the same patient. 60 In addition, measurements of platelet reactivity are at least in part dependent upon patient-level comorbidities, regardless of aspirin therapy. 61
There are very few prospective studies, however, that have specifically examined the effect of increasing doses of aspirin upon measures of aspirin resistance. Gurbel and colleagues studied this in 125 patients with stable coronary artery disease; using a crossover design, the effect of aspirin doses of 81 mg, 162 mg, and 325 mg upon platelet function tests was examined. Aspirin resistance, as measured with LTA using arachidonic acid as the agonist, was rare at all doses. Other measures of platelet aggregation, however, appeared to decrease with increasing doses of aspirin; this included measurements of LTA with collagen, LTA with ADP, the PFA-100, and urinary 11-dehydro TXB2. 62 This suggested that inhibition of arachidonic acid-induced platelet aggregation was nearly complete at low doses of aspirin, but that aspirin might act upon other pathways of platelet aggregation in a dose-dependent manner. 3
There has been only one prospective study, however, of the use of testing for aspirin resistance to tailor therapy. The Aspirin Nonresponsiveness and Clopidogrel Endpoint Trial (ASCET) randomized patients on aspirin 160 mg daily to either continuation of the same dose or clopidogrel 75 mg daily. High on-treatment platelet reactivity in the aspirin group, as measured by the PFA-100, was present in one-fourth of patients treated with aspirin; but it was not associated with the composite end point of death, MI, ischemic CVA, and unstable angina. 63 Similar to prospective trials utilizing high on-treatment platelet reactivity to tailor clopidogrel therapy, like GRAVITAS, 64 ASCET was hindered by a very low cardiovascular event rate. In addition, the most recent prospective study of aspirin dose, CURRENT OASIS 7, 45 did not show a benefit with higher aspirin doses in acute coronary syndromes as detailed previously. It therefore appears that routine measurements of platelet reactivity for aspirin resistance, regardless of the assay, should not be utilized outside of research studies.
The concept of aspirin resistance, however, is nevertheless useful as it informs our understanding of mechanisms for atherothrombosis. Platelet COX-1-dependent TXA2 production is of course not the only relevant pathway in platelet activation; other receptors (eg, P2Y12 and glycoprotein IIb/IIIa), agonists (eg, ADP and thrombin), and pathways play prominent roles. Drug–drug interactions may decrease the effectiveness of aspirin; this has been described with the nonselective NSAIDS naproxen and ibuprofen. 65,66 In addition, there may be upregulation of COX-2-dependent TXA2 production, by endothelial cells, monocytes, and macrophages. 56,67 Potential mechanisms for the phenomenon of aspirin resistance are summarized in Table 5.
Guideline-Based Recommendations
There is little controversy regarding the use of aspirin for secondary prevention in patients with a history of cardiovascular disease. But given the wide-ranging and often complicated evidence in regards to both aspirin dose and its role in primary prevention, it is not surprising that major society guidelines are somewhat disparate. The American College of Cardiology Foundation/American Heart Association (ACCF/AHA) guidelines for ST-segment elevation MI, unstable angina/non-ST-segment elevation MI, and elective percutaneous coronary intervention (PCI) all state that it is reasonable to use an aspirin dose of 81 mg daily after PCI; this is given a class IIa, level of evidence B recommendation. 68–70 In general, it appears that a consensus is emerging that low-dose (<100 mg) aspirin is preferable for chronic therapy in most instances.
The use of aspirin for primary prevention is supported by the ACCF/AHA and American Diabetes Association with specific caveats: only in diabetic patients with a 10-year risk of vascular events greater than 10% without an increased risk of bleeding (class IIa, level of evidence C). 71 The American College of Chest Physicians suggests aspirin for primary prevention only in those aged 50 or older, at a dose of 75 to 100 mg; this is, however, only a class IIb recommendation. The US Preventive Services Task Force (USPSTF) recommends aspirin when the reduction in MI (for men) or ischemic stroke (for women) exceeds the risk of gastrointestinal bleeding (grade A); no comment was made regarding aspirin dose. 72 The European Society of Cardiology states that aspirin cannot be recommended in patients without cardiovascular or cerebrovascular disease because of the increased risk of major bleeding (class III, level of evidence B). 73 In other words, there is no consensus regarding the use of aspirin for primary prevention; but at the very least, it should involve careful assessment of both cardiovascular and bleeding risks.
Conclusion
Although aspirin has now been used for the evidence-based treatment of cardiovascular disease for almost 40 years, our understanding of both its pharmacologic effects and clinical role continues to evolve. Aspirin for the secondary prevention of atherothrombotic vascular events is one of the great success stories of modern medicine; but like all drugs, it has important limitations. The indiscriminate use of aspirin for primary prevention (even in diabetics) appears to be an unfounded strategy, as is the routine use of aspirin doses greater than 100 mg. Future research will inform the use of aspirin in the setting of newer and more potent antiplatelet agents. The story of aspirin to this point, however, is best summarized as more is not necessarily better.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
