Abstract
Cangrelor is an intravenously administered P2Y12 receptor antagonist with very fast, potent, and quickly reversible action. In the CHAMPION PHOENIX trial, cangrelor provided an improved anti-ischemic protection compared with clopidogrel, without increasing the risk of severe bleeding. Cangrelor is currently approved by drug regulating authorities for patients undergoing percutaneous coronary intervention (PCI) without prior treatment with a P2Y12 receptor antagonist and not receiving a glycoprotein IIb/IIIa inhibitor, while its use is endorsed with a class IIb recommendation by the European Society of Cardiology guidelines. Several subanalyses of CHAMPION PHOENIX trial have tried to elucidate the role of cangrelor in PCI, including its usefulness during a 2-hour landmark analysis, impact on intraprocedural stent thrombosis, and reduction in myocardial infarction (MI) rate. The influence of gender, geographic region, access site, and bivalirudin use on cangrelor’s effects has also been reported. In patients with ST elevation MI and in clinical scenarios of disturbed absorption of oral antiplatelet agents or in need of an intravenous agent, cangrelor may surpass oral agents’ drawbacks. Transitioning to an oral agent is mandatory following cangrelor infusion discontinuation, although ticagrelor may be administered earlier without any pharmacodynamic interaction. Nevertheless, the clinical role of cangrelor in conjunction with administration of prasugrel or ticagrelor remains unclear. Accruing real-life experience is expected to improve our understanding of cangrelor’s role in everyday clinical practice.
Introduction
Dual antiplatelet treatment consisting of an oral P2Y12 receptor antagonist administered on top of aspirin represents a mainstay treatment for patients undergoing percutaneous coronary intervention (PCI). Clopidogrel remains the most broadly prescribed P2Y12 receptor antagonist. However, variability in the antiplatelet action of clopidogrel has been well described and high on-clopidogrel treatment platelet reactivity commonly occurs. The latter has been associated with a higher rate of adverse thrombotic events. 1 Prasugrel and ticagrelor are newer antiplatelet agents able to provide faster, more potent, and consistent P2Y12 antagonism compared with clopidogrel. In clinical outcome studies, these agents have led to an improvement in ischemic outcome, though with an increased bleeding risk compared with clopidogrel. However, a delay in the onset of antiplatelet action has been occasionally observed even when using novel agents, which is clinical setting dependent. 2 –4 Moreover, oral agents pose a relatively slow offset of activity, which could be problematic when surgery is urgently needed. The development and clinical testing of a potent P2Y12 receptor antagonist, which can be administered intravenously (IV) and has rapid onset and offset of action, could potentially overcome some of the drawbacks of the oral antiplatelet agents.
Cangrelor, a nonthienopyridine adenosine triphosphate analogue, is a reversible P2Y12 receptor antagonist. Cangrelor, administered as an IV bolus followed by infusion, is rapidly distributed, reaching C max within 2 minutes, and its concentration is consistently maintained for the duration of the infusion. Due to relatively rapid hydrolysis to its inactive metabolite, cangrelor has a half-time of 3 to 6 minutes. Adenosine diphosphate (ADP)-induced platelet aggregation is highly inhibited throughout the duration of cangrelor infusion. Once the infusion is stopped, there is rapid offset of effect and the platelet function returns to normal within 60 minutes. 5 The potential role of cangrelor as an adjunctive pharmacotherapy to PCI represents the focus of this review. Data obtained from randomized trials and their subanalyses, clinical scenarios in need of an IV agent, emerging real-life experience, and current perspectives to cangrelor use are presented.
What Is Known From Randomized Trials
In 2 phase III randomized clinical trials, cangrelor was compared with clopidogrel administered before PCI (cangrelor vs standard therapy to achieve optimal management of platelet inhibition—CHAMPION PCI) or after PCI (cangrelor vs standard therapy to achieve optimal management of platelet inhibition— CHAMPION PLATFORM). 6,7 In CHAMPION PLATFORM trial involving 5362 clopidogrel-naive patients, the composite of death, myocardial infarction (MI), or ischemia-driven revascularization (IDR) at 48 hours did not differ significantly between cangrelor (7.0%) and placebo (8.0%) arms, with odds ratio (OR) of 0.87 and 95% confidence interval (CI) of (0.71-1.07). Major bleeding was higher among cangrelor-treated patients (5.5% vs 3.5%, P < .001), a difference attributed mainly to a higher rate of hematomas at the access site. Moreover, in CHAMPION PCI trial comparing the use of cangrelor with 600 mg of clopidogrel given at the start of PCI among 8877 patients, the primary composite end point of death from any cause, MI, or IDR at 48 hours occurred with a similar frequency for the 2 arms, 7.5% vs 7.1%, with OR (95% CI) of 1.05 (0.88 to 1.24). Major bleeding—Acute Catheterization and Urgent Intervention Triage Strategy (ACUITY) criteria rate was 3.6% with cangrelor versus 2.9% with clopidogrel, OR (95% CI) of 1.26 (0.99 to 1.60), P = .06. Enrollment in both CHAMPION PCI and CHAMPION PLATFORM was terminated early based on the recommendation of the interim analysis review committee, because of a low likelihood of achieving the primary composite end point.
In CHAMPION PHOENIX trial, patients were randomized to receive a bolus and subsequently infusion of cangrelor or a loading dose of clopidogrel (Table 1). 8 Following prespecified adjustment for clopidogrel loading dose (300 vs 600 mg), the primary end point of death, MI, IDR, or stent thrombosis assessed at 48 hours was reduced by cangrelor over clopidogrel. Severe bleeding at 48 hours (primary safety end point) did not differ between cangrelor and clopidogrel groups. The effect of cangrelor appeared to be consistent in a variety of prespecified and other clinically important subgroups.
CHAMPION PHOENIX Trial Data. 8

Abbreviations: CABG, coronary artery bypass grafting; GUSTO, Global Use of Strategies to Open Occluded Coronary Arteries; IDR, ischemia-driven revascularization; MI, myocardial infarction; NSTE-ACS, non-ST elevation-acute coronary syndrome; PCI, percutaneous coronary intervention; ST, stent thrombosis; STEMI, ST elevation myocardial infarction.
A patient-level (n = 24 910) data analysis from CHAMPION program reaffirmed the superiority of cangrelor compared with clopidogrel or placebo for the reduction of periprocedural ischemic events. 9 The primary safety end point of non–coronary artery bypass graft (CABG) severe or life-threatening bleeding (according to Global Use of Strategies to Open Occluded Coronary Arteries [GUSTO] criteria) occurred at 48 hours with a similar frequency of 0.2% in both groups. It has been postulated that the inaccurate way of MI ascertainment in CHAMPION PLATFORM and CHAMPION PCI did not allow discrimination of reinfarction in patients undergoing PCI early following hospitalization. In contrast, in CHAMPION PHOENIX, the universal MI definition was implemented with careful assessment of baseline status, at least 2 samples of troponin, new ischemic symptoms, new ischemic electrocardiogram changes, and/or angiographic evidence.
CHAMPION PHOENIX Substudies
Several substudies of CHAMPION PHOENIX trial have been performed expanding our knowledge on the role of cangrelor in PCI (Table 2). In a landmark analysis, the rate of death, MI, IDR, or stent thrombosis at 0 to 2 hours was reduced by cangrelor over clopidogrel, with no significant differences between the 2 arms thereafter. 10 It is especially noteworthy that the early benefit observed with cangrelor represents the true impact of the IV drug. Généreux et al performed an independent, blinded analysis of intraprocedural stent thrombosis (IPST). 11 The latter predicted an increase in mortality, MI, IDR, and also definite or probable stent thrombosis at 48 hours and at 30 days. Cangrelor effectively reduced the rate of IPST. More bleeding was observed among IPST patients, most likely reflecting a higher rate of glycoprotein IIb/IIIa inhibitor (GPI) use in this cohort. A preliminary analysis based on the number of high-risk lesion characteristics on angiography has also been reported. 12 Cangrelor, compared to clopidogrel, reduced 48-hour ischemic outcomes consistently and irrespectively of PCI lesion complexity and clinical presentation, for example, acute coronary syndrome (ACS) versus stable ischemic heart disease. In CHAMPION PHOENIX, 462 patients (4.2%) suffered an MI; the incidence of MI was reduced by cangrelor compared with clopidogrel at 48 hours. 13 Regardless of the type, MIs remain associated with significant odds of death in the 30 days after the event. Moreover, cangrelor reduced MIs of different sizes, predominantly those MIs with high levels of biomarkers, as well as MIs associated with signs or symptoms of ischemia.
Subanalyses of the CHAMPION PHOENIX Trial.

Abbreviations: ACS, acute coronary syndromes; CI, confidence interval; IPST, intraprocedural stent thrombosis; MI, myocardial infarction; OR, odds ratio; SA, stable angina.
aefficacy end point: Death, myocardial infarction, ischemia-driven revascularization, or stent thrombosis at 48 hours; secondary end point: stent thrombosis at 48 hours.
bPrimary Ischemic outcome included new onset out-of-laboratory stent thrombosis (Academic Research Consortium).
cPrimary safety end point: Severe Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO)-defined non-CABG-related bleeding at 48 hours.
The benefits and risks of cangrelor remained consistent when its effects were analyzed separately in groups of patients with stable angina and ACS. 14 Among CHAMPION PHOENIX patients, 28% were female. 15 Although the reduction in the primary end point by cangrelor was 35% in women and 14% in men, P was not significant for interaction. Severe bleeding occurred with a similar frequency among women and men treated with either cangrelor or clopidogrel. Moderate bleeding was higher in women treated with cangrelor over clopidogrel (0.9% vs 0.3%, P = .02), while no such difference was observed among men (0.2% vs 0.2%, P = .68), with P for interaction = .04. In another analysis according to geographic region (US vs non-US sites), cangrelor consistently reduced the rates of ischemic end points compared to clopidogrel, without an excess of severe bleeding in both the US and non-US subgroups. 16 The reduction in the primary efficacy end point at 48 hours by cangrelor over clopidogrel was consistent and irrespective of the PCI access site. 17 In both cangrelor and clopidogrel arms, a femoral approach was accompanied by 2- to 3-fold higher rate of GUSTO severe/moderate bleeding and blood transfusions, compared with the radial approach. Major bleeding (ACUITY definition) occurred more frequently among cangrelor-treated patients in both femoral, OR (95% CI) of 1.69 (1.35-2.12), and radial cohorts, OR (95% CI) of 2.17 (1.02-4.62), P for interaction = .54. The impact of bivalirudin use was also separately analyzed in the context of CHAMPION PHOENIX. 18 Among patients receiving bivalirudin, the risk of primary efficacy end point at 48 hours was significantly reduced by cangrelor over clopidogrel.
In a substudy of pooled data from the CHAMPION program, patients with prior MI (n = 5699, 23%) had a higher rate of the primary end point versus those without prior MI: 4.9% versus 4.0%, P = .002. 19 The primary end point rate was reduced by cangrelor over clopidogrel from 5.7% to 4.2%, OR (95% CI) of 0.72 (0.57-0.92), in patients with prior MI and from 4.3% to 3.7%, OR (95% CI) of 0.85 (0.74-0.99) in patients without prior MI, P for interaction = .25. Severe bleeding as defined by GUSTO did not differ significantly according to prior MI status. The absolute benefit of cangrelor was greater in patients with than in those without prior MI. Analysis of pooled data also regarding the subgroup of patients with prior ischemic stroke or transient ischemic attack >1-year-old has also been reported. The rate of the primary efficacy end point was reduced with cangrelor versus clopidogrel, 4.3% versus 5.3%, OR (95% CI) of 0.80 (0.48-1.34), in consistency with the overall trials results and P for interaction = .97. Similarly, GUSTO severe bleeding rate was not differentiated by prior history of ischemic stroke or transient ischemic attack, P for interaction = .81. 20 Finally, in a very recent pooled, patient-level analysis of the 3 CHAMPION trials, cangrelor maintained its efficacy in reducing ischemic complications: among patients receiving GPI, the primary end point rate was 4.9% versus 6.5% in cangrelor versus clopidogrel arms, respectively, with OR (95% CI) of 0.74 (0.55-1.01), while in patients who did not receive GPI, 3.6% versus 4.4% in cangrelor versus clopidogrel arms, with OR (95% CI) 0.82 (0.72-0.94) and P for interaction = .55. Severe/life-threatening bleeding as defined by GUSTO did not differ according to the GPI use. Of note, a higher risk of bleeding was seen with GPI use regardless of randomization to cangrelor or clopidogrel. 21
Marketing Authorization/Guidelines Recommendation
Cangrelor has been approved by the US Food and Drug Administration as an adjunctive therapy to PCI for reducing periprocedural thrombotic events in patients not pretreated with a P2Y12 receptor antagonist and in whom a GPI was not administered. 22 Slightly different is the approved indication by European Medicines Agency, where cangrelor use is intended for patients who have not received an oral P2Y12 receptor antagonist prior to PCI and in whom oral therapy with P2Y12 receptor antagonist is not feasible or desirable. 23 The latest European Society of Cardiology guidelines for the management of ACS in patients presenting without persistent ST-segment elevation propose cangrelor with a class IIb recommendation (level of evidence A). 24
The Issue of Pretreatment
As peri-PCI platelet inhibition appears to protect from ischemic complications and considering the delayed bioavailability of oral P2Y12 receptor antagonists, the practice of pretreatment is applied by many clinicians prior to a possible ad hoc PCI. However, such a strategy may delay CABG surgery or increase unnecessarily the risk of bleeding in patients who are managed medically. A meta-analysis of randomized controlled trials did not reveal mortality reduction by pretreatment with clopidogrel, OR (95% CI) 0.80 (0.57-1.11); however, the rate of major cardiac events reduced, OR (95% CI) of 0.77 (0.66-0.89), P < .001. Major bleeding rate was not affected by pretreatment, OR (95% CI) of 1.18 (0.93-1.50). 25 In addition, in patients undergoing PCI for non-STEMI, the rate of cardiovascular death, MI, stroke or urgent revascularization through 7 days from PCI was similar between pretreatment with prasugrel and no pretreatment arms (13.1% vs 13.1%, P = .93). However, the incidence of non-CABG major bleeding was 3-fold higher with pretreatment compared with no pretreatment (1.4% vs 0.51%, P = .02). 26 Among 28 306 patients undergoing PCI in a contemporary (2008–2013) US practice, the overall pretreatment rate (categorized as >2 hours before or 0-2 hours before PCI) was 23%. 27 Similarly, low rates of pretreatment have been described in recent European real-life experience, with apparent consequence in the great majority of patients to undergo PCI without adequate periinterventional platelet inhibition. 28 For such patients, cangrelor appears as the ideal antiplatelet agent to effectively protect from ischemic events during the periinterventional period with minimal excess of bleeding, due to its brief duration of action and rapid recovery of platelet activity shortly following infusion discontinuation.
Cangrelor: Why for STEMI?
A patient with STEMI represents a most challenging scenario, as the thrombotic potential is highest, and platelet inhibition, along with timely offered primary PCI, is of utmost importance and urgently needed. Apart from the inherently limited time for pretreatment, a delayed onset of antiplatelet action following loading with oral agents has been described in STEMI patients. 4 In the presence of nausea, vomiting, intubation, hemodynamic instability–cardiogenic shock, following resuscitation or therapeutic hypothermia, the bioavailability of oral P2Y12 receptor antagonists is reduced. 29 Moreover, in a pain-suffering patient, concurrent administration of oral antiplatelet agents and morphine is common and this appears to be a “dangerous liaison” with the absorption of the former being significantly diminished and/or delayed. 30,31 In a setting of primary PCI and regarding outcome, the magnitude of risk reduction with the use of the novel P2Y12 receptor antagonists compared with clopidogrel appears attenuated when compared with the use of these agents in the full ACS population, with possible reasoning including the delayed onset of antiplatelet action. 32 Conceivably, several strategies have been tested to “bridge the gap” in antiplatelet activity observed in the early hours of STEMI when oral antiplatelet agents are administered. 33 Crushing ticagrelor or prasugrel tablets offers an earlier by approximately 1 hour antiplatelet activity compared with standard integral tablets loading, representing an improvement but not a clear solution for peri-PCI adequate platelet inhibition. 34,35 Prehospital, compared with in-hospital, administration of ticagrelor loading dose failed to improve the indices of coronary reperfusion at initial angiography, although it appeared to be safe and with a strong signal of acute stent thrombosis rate reduction. 36 Of note, most of the acute stent thrombosis cases occur within 2 to 3 hours from primary PCI, indicating a particularly “vulnerable” period, which can be ameliorated by cangrelor. 10,37 In the STEMI cohort of CHAMPION PHOENIX trial (n = 1991), cangrelor compared with clopidogrel, reduced the primary end point, with OR (95% CI) 0.75 (0.46-1.25) and in consistency with the overall trial results (P for interaction = .98). No difference in the rates of severe bleeding or transfusion was found, but moderate bleeding rate was higher among cangrelor-treated patients. 8
Beyond the randomized trials, 2 pharmacodynamic studies with cangrelor have been recently published. In a single-center, pharmacodynamic study, 32 STEMI patients were loaded with 180 mg of ticagrelor either within 1 hour before or at the beginning of primary PCI. Platelet reactivity, as assessed with the VerifyNow P2Y12 function assay, was 249 PRU (IQR 221-271) prior to cangrelor infusion, while 15 minutes after cangrelor infusion it was greatly decreased to 71 PRU (IQR 52-104), P < .001, with no cases of high platelet reactivity (Figure 1). 38 Moreover, in 21 P2Y12 receptor antagonist-naive ACS patients (including 10 STEMI cases), platelet reactivity measured by multiple electrode aggregometry (MEA) and thromboelastography prior to cangrelor and following PCI significantly reduced from 46.7 to 17.9 U and from 43.1 to 22.0 mm, respectively (P< .001). 39 At the end of PCI, cangrelor-treated patients presented with lower platelet reactivity, when compared to a matched cohort of oral agents only receiving patients. Furthermore, practice patterns, indications for use, and clinical events in 100 cangrelor recipients in a single-center have been retrospectively analyzed. 40 Cangrelor was used most commonly in the context of PCI for STEMI. Despite higher risk clinical presentations than in the CHAMPION program and the use of more potent oral P2Y12 receptor antagonists, cangrelor-related side effects were not observed and the rates of clinically significant ischemic or bleeding events were low.
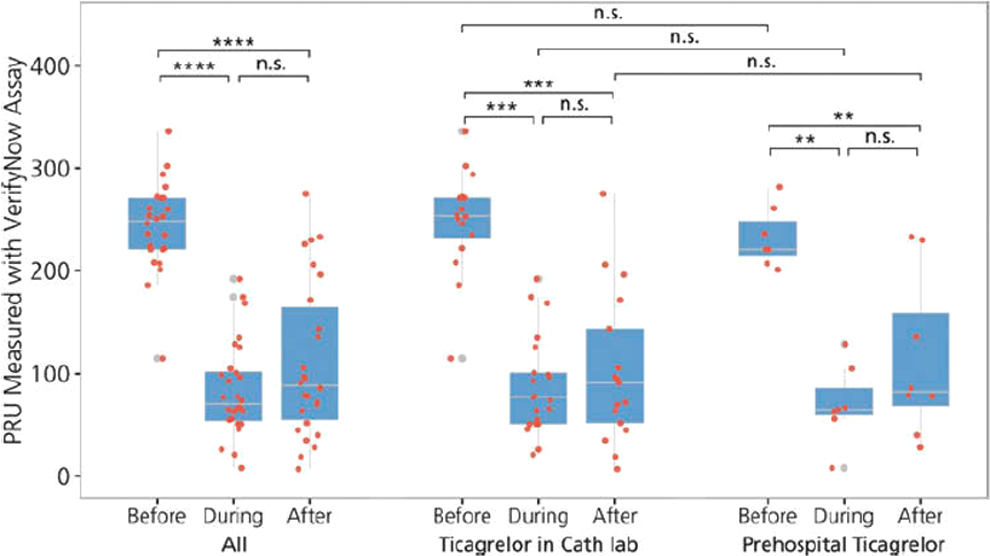
Box plot showing platelet reactivity units before, during, and after cangrelor infusion. Wilcoxon test was used for intraindividual comparison and Mann Whitney U test for comparisons between groups. PRU indicates platelet reactivity unit. **** P < .001, *** P < .01, ** P < .05, n.s. = not significant. Reprinted with permission from Mohammad et al. 38
Overall, the limitations of orally administered P2Y12 receptor antagonists may be surpassed by a rapidly acting IV agent, and the availability of cangrelor provides an important adjunctive treatment option for the prevention of periprocedural ischemic events when PCI is performed urgently and immediate platelet inhibition is required. Moreover, clopidogrel, ticagrelor, and cangrelor have been demonstrated to be cardioprotective. In a preclinical model of ischemia/reperfusion in rat, rabbit, and monkey, cangrelor administered a few minutes before reperfusion dramatically reduces MI. 41 –43 Although the precise mechanism of this salutary effect has not been clarified, it is clear that the P2Y12 antagonists act like conditioning mimetics. As agents like cangrelor must be present in the first minutes of reperfusion to have this infarct-sparing effect, this is easily achieved with an IV agent but can be problematic with oral agents.
Transitioning to Oral P2Y12 Receptor Antagonists
The way of transitioning from cangrelor to an oral P2Y12 receptor antagonist has been extensively analyzed. The thienopyridines clopidogrel and prasugrel are prodrugs, which require metabolism to their respective active metabolite (AM); the latter binds irreversibly to the P2Y12 receptor. Clopidogrel-AM and prasugrel-AM levels peak after 1 to 2 hours, while their plasma half-life is approximately 30 minutes. 44 If circulating in the blood AMs find the P2Y12 receptor occupied by cangrelor, they cannot bind to the receptor and, therefore, cannot exert their action. In healthy volunteers, platelet inhibition provided by clopidogrel did not occur if cangrelor had been concurrently administered. 45 In cases when clopidogrel was administered following the end of cangrelor infusion, platelet function recovered transiently with cangrelor offset, with subsequent decrease reflecting the clopidogrel-AM onset of action. In stable coronary artery disease patients, initiation of clopidogrel prior to discontinuation of the cangrelor infusion increased the recovery of platelet function. 46 It is therefore conceivable that clopidogrel administration after the end of cangrelor infusion results in more sustained platelet P2Y12 inhibition.
Transitioning from cangrelor to prasugrel has also been reported. 47 Platelet reactivity was significantly reduced during cangrelor infusion, while following the end of infusion, platelet function transiently recovered. This phenomenon was less pronounced when prasugrel 60 mg loading dose was given at 30 minutes before cangrelor discontinuation. In a recent in vitro study, investigators directly measured the number of functional (unblocked) P2Y12 receptors using a 33P-2MeSADP binding assay following co-incubation with cangrelor and either clopidogrel or prasugrel AM. 48 The thienopyridine AMs had only limited ability to compete with cangrelor for binding to P2Y12.
Ticagrelor is a reversible, noncompetitive P2Y12 receptor antagonist. In stable coronary artery disease patients receiving aspirin, transitioning from cangrelor to ticagrelor has been analyzed. 49 Platelet reactivity increased modestly for the first hour following cangrelor discontinuation, reflecting offset of cangrelor; this increase was attenuated by earlier administration of ticagrelor, indicating the onset of ticagrelor action. Ticagrelor can therefore be administered prior, during, or following cangrelor infusion with no considerable consequences on the pharmacodynamic effects of cangrelor. On the other hand, the pharmacodynamic effects of ticagrelor appear to be greater when it is administered earlier. This issue may be particularly important in STEMI cases.
The US labeling suggests the administration of ticagrelor at any time during or immediately after the end of cangrelor infusion, and administration of prasugrel or clopidogrel immediately after cangrelor discontinuation. 22 European labeling proposes the administration of loading dose of any of the 3 oral agents following cangrelor discontinuation or alternatively, a loading dose of ticagrelor or prasugrel, but not clopidogrel, up to 30 minutes prior to the end of the infusion. 23 Importantly, it is unclear whether there is any clinical relevance to different transitioning strategies.
Is Cangrelor Beneficial in the Era of Prasugrel/Ticagrelor?
In CHAMPION PHOENIX, the advantages for cangrelor were obtained when this agent was compared with clopidogrel. One cannot speculate about the relative benefit of cangrelor if the comparator was a prasugrel or ticagrelor. Both these latter agents are more potent and faster acting P2Y12 receptor antagonists than clopidogrel. A randomized, clinical comparison of cangrelor plus an oral novel P2Y12 receptor antagonist versus an oral novel P2Y12 receptor antagonist alone in a broad spectrum of clinical settings, including STEMI, would undoubtedly add to our understanding of the additional impact cangrelor may have had on peri-PCI events. Of note, a randomized, pharmacodynamic study of ticagrelor loading plus cangrelor versus ticagrelor loading alone in STEMI patients is currently underway (ClinicalTrials.gov Identifier: NCT02943369). In another study also in STEMI patients, platelet reactivity at infarct vessel open time is being compared between cangrelor switched to ticagrelor versus ticagrelor alone arms (ClinicalTrials.gov Identifier: NCT027333).
Unsettled Issues
An alternate way to achieve rapid and effective platelet inhibition is by IV GPI administration. However, following GPI discontinuation, antiplatelet activity persists for hours (in the case of eptifibatide and tirofiban) or days (in the case of abciximab). These GPI properties could constitute an advantage for cangrelor, as in case of a bleeding event, the rapid offset of cangrelor activity would aid in minimizing bleeding severity. In a recent exploratory analysis of pooled patient-level data from the 3 CHAMPION trials, cangrelor alone and clopidogrel-GPI (matched cohorts) were associated with similar ischemic risk at 48 hours. Of importance, and following propensity score matching, the rate of major or minor bleeding (Thrombolysis in Myocardial Infarction definition) was lower in the cangrelor alone group, 0.7% versus 2.4%, OR (95% CI) 0.29 (0.13-0.68), P = .004. 50 As an increasing number of patients may undergo PCI while on chronic treatment with an oral P2Y12 receptor antagonist, the question arises whether cangrelor could provide additional platelet inhibition in such patients. In an in vitro study, when cangrelor was added to the platelet-rich plasma of clopidogrel-pretreated patients, it resulted in further platelet inhibition. 51 In patients on prasugrel maintenance therapy, in vitro incubation with cangrelor provided more rapid than prasugrel reloading platelet inhibition. 52 Furthermore, maintenance treatment with ticagrelor for 5 days did not affect the inhibitory effects of cangrelor. 49 Nevertheless, the use of cangrelor in oral agent-receiving patients and aiming for further platelet inhibition is currently “off-label” and of unclear clinical significance. Although cangrelor with concomitant bivalirudin has been analyzed, the optimal antithrombin regimen to combine with cangrelor remains unknown. 18 Finally, cost differential between additional cangrelor versus prasugrel or ticagrelor alone is substantial. If a cangrelor strategy proves to be cost-effective, this will be a major determinant of how widely this agent will be used.
Practical Considerations
Cangrelor, when compared to no preloading with oral agents, decreases stent thrombosis and MI in all patients undergoing PCI. In stable patients, a cangrelor-based strategy compared to preloading with oral agents avoids unnecessary exposure in patients not requiring PCI, with subsequent decrease in bleeding. In nonST elevation ACS patients, a cangrelor-based strategy compared to preloading with oral agents avoids unnecessary exposure in medically treated patients and allows CABG to be performed early if needed, instead of waiting for 5 to 7 days. In the clinical scenario of STEMI and within the first few hours—when oral agents are not active—cangrelor decreases stent thrombosis and MI. Moreover, decreases in stent thrombosis and MI in all patients undergoing PCI unable to take oral medications are expected with the use of cangrelor.
In conclusion, in patients undergoing PCI cangrelor is a valuable tool to achieve potent P2Y12 receptor antagonism, which is reflected into a reduction in ischemic periinterventional events in most clinical settings, without concomitant increase in severe bleeding. Further real-life experience with cangrelor, outside the randomized trials, is eagerly needed to improve our understanding of its role in our armamentarium of adjunctive pharmacotherapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Alexopoulos—advisory board: AstraZeneca, Boeringer Ingelheim, Bayer, The Medicines Company; speaker honoraria: AstraZeneca. Dr Lekakis-advisory board fees and lecturing honoraria: Astrazeneca, Actelion, and MSD. Other authors have no conflicting interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
