Abstract
Objectives:
The endoplasmic reticulum (ER) stress-induced apoptotic pathway is associated with the development of acute myocardial infarction (AMI). Cortistatin (CST) is a novel bioactive peptide that inhibits apoptosis-related injury. Therefore, we investigated the cardioprotective effects and potential mechanisms of CST in a rat model of AMI.
Methods:
Male Wistar rats were randomly divided into sham, AMI, and AMI + CST groups. Cardiac function and the degree of infarction were evaluated by echocardiography, cardiac troponin I activity, and 2,3,5-triphenyl-2H-tetrazolium chloride staining after 7 days. The expression of CST, ER stress markers, and apoptotic markers was examined using immunohistochemistry and Western blotting.
Results:
Compared to the AMI group, the AMI + CST group exhibited markedly better cardiac function and a lower degree of infarction. Electron microscopy and terminal deoxynucleotidyl transferase dUTP nick end labeling confirmed that myocardial apoptosis occurred after AMI. Cortistatin treatment reduced the expression of caspase 3, cleaved caspase 3, and Bax (proapoptotic proteins) and promoted the expression of Bcl-2 (antiapoptotic protein). In addition, the reduced expression of glucose-regulated protein 94 (GRP94), glucose-regulated protein 78 (GRP78), CCAAT/enhancer-binding proteins homologous protein, and caspase 12 indicated that ER stress and the apoptotic pathway associated with ER stress were suppressed.
Conclusions:
Exogenous CST has a notable cardioprotective effect after AMI in a rat model in that it improves cardiac function by suppressing ER stress and myocardial apoptosis.
Introduction
Acute myocardial infarction (AMI) remains a leading worldwide cause of death and disability due to coronary heart disease. Unfortunately, AMI survivors impose a large economic burden on medical services. 1 Myocardial apoptosis is a critical form of cardiomyocyte death during AMI, 2 -4 and the surviving cardiomyocytes undergo additional progressive loss during heart failure after AMI. 5 Therefore, among terminally differentiated cardiomyocytes, 6 inhibition of myocardial apoptosis is an appealing therapeutic strategy for improving cardiac function, preventing ventricular remodeling, and decreasing the incidence of cardiovascular events after AMI.
The endoplasmic reticulum (ER) stress-induced apoptotic pathway is different from the death receptor pathway (extrinsic pathway) and the mitochondrially mediated apoptotic pathway (endogenous pathway). Thus, the ER is now recognized as an important organelle in determining cell death. In this context, stimuli such as ischemia, hypoxia, and disturbances in calcium levels lead to the accumulation of unfolded and aggregated proteins, which induce the ER to trigger the unfolded protein response (UPR) to maintain homeostasis and functional stability. However, if the stress response is excessively prolonged or too severe, the UPR can cause cells to undergo apoptosis. Recent groundbreaking studies have provided compelling evidence that ER stress is implicated in the pathophysiology of cardiovascular disease 7,8 and that the attenuation of ER stress-induced apoptosis can protect the heart against cardiomyocyte loss and improve cardiac function. 9 Furthermore, compared to other drugs, various bioactive peptides have high target specificity in the field of cardiovascular disease. 8,10 Thus, there is significant interest in the therapeutic potential of simulating the physiological effects of bioactive peptides in regulating the ER stress-induced apoptotic pathway.
Cortistatin (CST), named after its predominant expression in the cerebral cortex, is a novel cyclic bioactive peptide. It was identified in 1996 by Lecea et al who determined in their initial studies that it could reduce activity of the cerebral cortex. 11 Cortistatin was also demonstrated to have anti-inflammatory effects in some experimental models and endocrine activity in the human studies. 12 -14 Other studies have reported that CST and its receptors are widely distributed in systemic tissues, which include the heart, arterial blood vessels, and the immune system. 15,16 Interestingly, CST exhibits strong structural homology and shares 11 of the 14 residues with somatostatin (SST), which includes an FWKT (Phe-Trp-Lys-Thr) tetramer that is crucial for binding to SST receptors (SSTRs). Therefore, CST has a high affinity for all 5 recombinant SSTRs.
Yan et al demonstrated that the expression of CST and SSTRs is downregulated in human coronary artery endothelial cells after treatment with tumor necrosis factor α. 17 However, the functions of SST and CST may not be the same. Reports have demonstrated that CST binds to the ghrelin receptor (growth hormone secretagogue-receptor [GHS-R1a]), 18 which indicates that GHS-R1a may be able to protect the heart against ischemia or reperfusion injury by inhibiting ER stress and ER stress-mediated apoptosis. 19 Recent studies have also demonstrated that the plasma levels of CST are elevated among patients with coronary heart disease. 20 In a previous study, we found that CST directly reduced myocardial apoptosis by inhibiting ER stress in rats with sepsis, 21 and these results led us to hypothesize that CST may play a critical role in cardiovascular disease. However, given the status of CST as a novel bioactive peptide, there are limited data regarding its effects on myocardial apoptosis after AMI. Therefore, the present study aimed to evaluate the cardioprotective effects of CST treatment after AMI and to investigate whether its cardioprotective mechanism involves suppression of ER stress and myocardial apoptosis.
Materials and Methods
Preparation of Animals and Reagents
All experimental procedures were approved by the Animal Care and Use Committee of Harbin Medical University and complied with the requirements of the Guide for the Care and Use of Laboratory Animals. A total of 36 adult male Wistar rats (specific pathogen free, weight: 220 g ± 20 g) were acquired from the Experimental Animal Center of The First Affiliated Hospital (Harbin Medical University). The rats were housed individually in clear plastic cages at a controlled temperature (22°C-25°C) and humidity (40%-50%), with a 12-hour light/dark cycle (light: 7
The rat CST-14 peptide and antirat CST-14 antibody (H-060-04) were obtained in their lyophilized forms from Phoenix Pharmaceuticals Inc (Belmont, California). The peptide was allowed to return to room temperature for 30 minutes before reconstitution. Based on the manufacturer’s protocols, we gradually reconstituted the peptide to the desired concentration (350 μg/mL) using sterile normal saline. The antibody was reconstituted using double-distilled water and was stored in aliquots (1 mL/vial) at −20°C. Rabbit polyclonal antibodies to GRP94 (ab13509), GRP78 (ab21685), caspase 12 (ab18766), and CCAAT/enhancer-binding proteins (C/EBP) homologous protein (CHOP; ab10444) were obtained from Abcam Inc (Cambridge, Massachusetts). The antibody to caspase 3 (19677-1-AP) was purchased from Proteintech Group Inc (Wuhan, China), and the antibodies to Bcl-2 (BA0412) and Bax (BA0315-2) were purchased from Wuhan Boster Biological Engineering Co (Wuhan, China). Antibodies to β-actin and all secondary antibodies were obtained from Santa Cruz Biotechnology (Santa Cruz, California).
Acute Myocardial Infarction Model
A total of 36 rats were randomly divided into 3 groups (n = 12 for each group): the sham group, AMI group, and AMI + CST group. The AMI rats were anesthetized using an intraperitoneal injection of 10% chloraldurate solution (0.3 mL/100 g), fixed on an operating table, intubated, and ventilated using a respirator pump. Then, their thoracic cavity was opened to expose the heart. The left anterior descending coronary artery was ligated from the pulmonary conus to the left atrial appendage (2-3 mm) using 5-0 silk sutures to establish the AMI model. Coronary artery occlusion was confirmed via epicardial blanching and ST-segment elevation in the limb leads on the electrocardiogram monitor. The sham group was subjected to the same surgical procedure, but the suture of the coronary artery was left untied. At 2 hours after the successful operation, rats in the AMI + CST group received an intraperitoneal injection of the CST solution (175 μg/kg/d), whereas rats in the sham and AMI groups received the same quantity of normal saline. These injections were given for 7 consecutive days, after which time, the evaluation described subsequently was performed. A total of 75% of the rats survived the AMI induction procedure.
Cardiac Function Analysis Using Echocardiography
Echocardiographic images were recorded with the anesthetized rats lying in a supine position using a Philips Sonos 7500 ultrasound system that was equipped with a 12-MHz transducer. Two-dimensional echo images in the apical 4-chamber view and left ventricular M-mode tracings immediately below the level of the papillary muscles were used to calculate the left ventricular end-systolic volume (LVESV, mL), left ventricular end-diastolic volume (LVEDV, mL), left ventricular end-systolic diameter (LVESD, mm), left ventricular end-diastolic diameter (LVEDD, mm), left ventricular ejection fraction; LVEF = (LVEDV − LVESV)/LVEDV × 100%, and left ventricular fractional shortening; LVFS = (LVEDD − LVESD)/LVEDD × 100%. All measurements were performed in triplicate during 3 consecutive cardiac cycles by an investigator who was blinded to the rats’ group assignments; the average values were used for our analyses.
Serum Testing
After the rats were anesthetized, 4 mL blood samples were collected from the abdominal aorta using a heparinized syringe, and each sample was immediately centrifuged at 3000 rpm for 10 minutes at 4°C. The plasma was removed (0.2 mL/vial) and frozen at −80°C until the samples were tested for cardiac troponin I (c-TNI). The c-TNI activity was measured using a sandwich enzyme-linked immunosorbent assay (ELISA)that calculates the change in absorbance in proportion to c-TNI activity, according to the manufacturer’s protocol (Rat Troponin I type 3, Cardiac ELISA; BlueGene Biotech Co, Ltd. Shanghai, China).
Myocardial Scar Size Assessment
The rats were killed after undergoing the echocardiography and blood sampling, and the scar size was measured using 2,3,5-triphenyl-2H-tetrazolium chloride (TTC) staining. Briefly, the hearts were immediately excised and perfused with phosphate-buffered saline. Each heart was cut into 0.2-mm-thick transverse slices that were then incubated in a 1% TTC solution (Sigma-Aldrich, T8877-5G, Saint Louis, USA, pH 7.4) at 37°C for 15 minutes and fixed in 10% formaldehyde. In these slices, gray areas indicated myocardial scar size and red areas indicated normal tissue. Each slice was scanned and analyzed using Image J 1.44p software (NIH, Bethesda, Maryland). The relative scar size represented the degree of infarction.
Terminal Deoxynucleotidyl Transferase dUTP Nick End Labeling Assay for Myocardial Tissues
We used a terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) apoptosis assay kit (in situ cell death detection kit; Roche Applied Science, Shanghai, China) to evaluate the rate of apoptosis in the myocardial tissue. Based on the manufacturer’s instructions, myocardial cells were defined as being apoptotic if the entire nuclear area of the cell was positively labeled. The apoptotic cells were counted in 5 random fields at the border of the infarcted area, and the apoptosis index was calculated as the number of apoptotic cells divided by the total number of nucleated cells. The results were expressed as the average proportion of positive cells in each group.
Immunohistochemistry
Immunohistochemical staining for GRP94, GRP78, caspase 12, caspase 3, Bcl-2, Bax, and CST was performed using the routine immunohistochemistry streptavidin peroxidase method. The primary antibodies and dilutions were rabbit polyclonal GRP94 immunoglobulin G (IgG; 1:1500), anti-GRP78 IgG (1:2000), anti-caspase-12 IgG (1:1000), anti-caspase-3 IgG (1:600), anti-Bcl-2 IgG (1:400), anti-Bax IgG (1:400), and anti-CST-14 IgG (1:500). Nuclear counterstaining was performed using hematoxylin. Ten randomly selected fields near the infarct region from each heart section were examined at a magnification of 200× and were analyzed using Image-Pro Plus software (version 6.0; Media Cybernetics Inc, Silver Spring, Maryland). The positive staining results were analyzed using the integrated optical density value, which is a semiquantitative indicator of protein expression.
Electron Microscopy
Transmission electron microscopy was used to determine the presence of apoptotic cells. Rat myocardial samples were fixed in 2.5% glutaraldehyde, washed with Hank balanced salt solution, postfixed in 1% OsO4 in 0.1 mol/L cacodylate buffer, scraped off, and serially dehydrated using ethanol. Propylene oxide was used to dehydrate the samples, and the specimens were embedded in araldite. The samples were cut into ultrathin sections using an ultramicrotome, mounted on copper grids, and contrasted using lead citrate. The pathologists were blinded to the experimental samples and objectively described their findings.
Western Blot Analysis
After the rats were killed, the myocardial tissue was collected and processed according to the manufacturer’s instructions. 21 The primary antibodies and dilutions were anti-GRP94 (1:1000), anti-GRP78 (1:1000), anti-caspase-12 (1:500), anti-CHOP (1:300), anti-caspase-3 (1:400), anti-Bcl-2 (1:400), anti-Bax-2 (1:400), and anti-β-actin (1:500). Secondary anti-rabbit or anti-mice antibodies were used at a dilution of 1:2000. The results were visualized using enhanced chemiluminescence detection kits and were analyzed using the Quantity One software.
Statistical Analysis
GraphPad Prism (version 5.0) was used to analyze the results, and differences with a P value of <.05 were considered statistically significant. Data are expressed as the mean ± standard error of the mean. Differences between the 3 study groups were evaluated using analysis of variance.
Results
In the AMI and AMI + CST groups, 75% of the rats survived the infarct induction procedure. As shown in Figure 1, the coronary artery occlusion in our rat model of AMI caused marked ST-segment elevation in the limb leads (II, III, and aVF) and tachycardia, although there were no changes in the sham group (Figure 1A). These results indicate that our model provided an appropriate simulation of AMI.
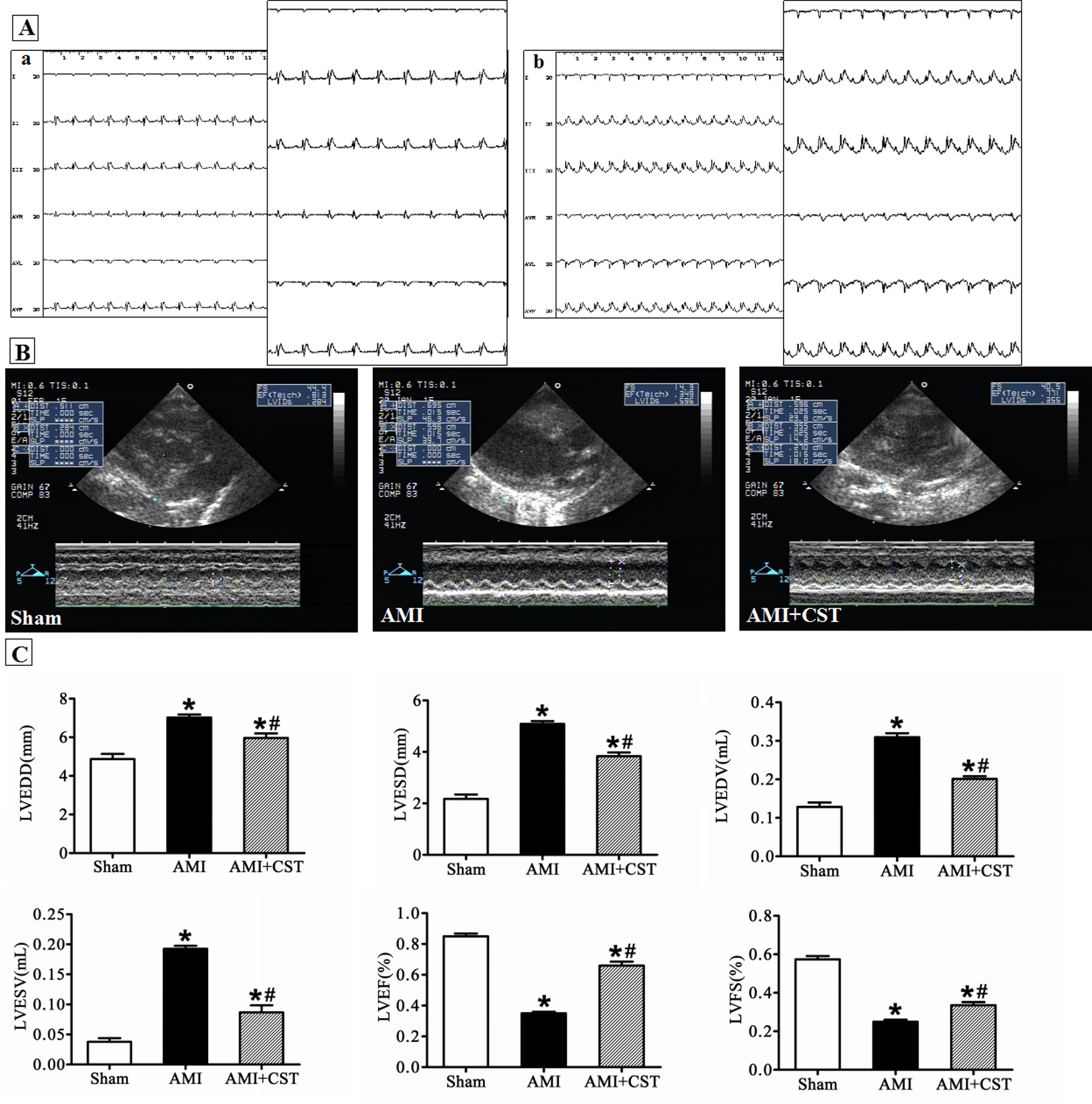
A, Electrocardiographic (ECG) monitoring. Electrocardiographic recordings were obtained from all experimental groups at a paper speed of 50 mm/s. Compared to the normal ST-segment height in the sham group (a), the success of the left anterior descending artery ligation in the AMI group was confirmed by ST-segment elevation in limb leads II, III, and aVF (b); n = 12 per group. B, Transthoracic echocardiography. Typical echocardiographic images from the sham, AMI + saline, and AMI + CST groups; n = 5 per group. C, Cardiac function parameters. LVESD: left ventricular end-systolic diameter, LVEDD: left ventricular end-diastolic diameter, LVESV: left ventricular end-systolic volume, LVEDV: left ventricular end-diastolic volume, LVEF: left ventricular ejection fraction, LVFS: left ventricular fractional shortening, n = 5 per group. Data are presented as the mean ± standard error of the mean. *P < .05 versus the sham group; # P < .05 versus the AMI group. AMI indicates acute myocardial infarction; CST, cortistatin.
Cortistatin Improves Myocardial Function in Rats Wth AMI
Transthoracic echocardiography was used to evaluate the protective effect of CST after AMI using various cardiac functional parameters, including LVESV and LVEDV, LVESD and LVEDD, and LVEF and LVFS (Figure 1B) as a loss of contractility and ventricular chamber enlargement are powerful predictors of cardiac dysfunction. The AMI group exhibited markedly reduced LVEF and LVFS and significantly greater LVEDD, LVESD, LVEDV, and LVESV compared to the sham and AMI + CST groups (Figure 1C).
Cortistatin Reduced Myocardial Scar Size and c-TNI Activity in Rats With AMI
As shown in Figure 2, the AMI + CST group exhibited a small scar size (left ventricular dilatation and ventricular wall infarction thinning) compared to the AMI group (scar size: 14.50% ± 2.55% vs 24.75% ± 4.47%, P < .05; Figure 2B). Furthermore, the c-TNI level was lower in the AMI + CST group (c-TNI: 5.82 ng/mL ± 2.11 ng/mL vs 2.87 ng/mL ± 0.67 ng/mL, P < .05; Figure 2C), indicating that CST treatment significantly reduced the severity of myocardial infarction. To identify the relationship between CST and AMI in rats, we used immunohistochemistry to detect changes in CST expression among the sham, AMI, and AMI + CST groups (Figure 2D). The expression of CST was elevated in the AMI and AMI + CST groups, although the AMI + CST group exhibited slightly lower CST expression (Figure 2E). This finding indicates that the expression of CST increased after AMI but decreased after treatment with exogenous CST.

A, 2,3,5-triphenyl-2H-tetrazolium chloride (TTC) staining of myocardial tissues. Representative images of TTC-stained sections were used to assess the effect of CST treatment on scar size. B, The percentage of scar size (n = 3 per group). C, The changes in c-cardiac troponin I in each group (n = 7 per group). D, The expression of CST in heart tissue. Magnification: ×400, n = 5 per group. Data are presented as the mean ± standard error of the mean. *P < .05 versus the sham group; # P < .05 versus the AMI group. AMI indicates acute myocardial infarction; CST, cortistatin.
Cortistatin Suppressed Myocardial Apoptosis in Rats With AMI
Apoptosis is critical to the development of AMI. 2 Myocardial apoptosis leads to a decrease in the number of cardiomyocytes, which are replaced by fibrous tissues. Therefore, we analyzed myocardial apoptosis in the heart tissues from the sham, AMI, and AMI + CST groups using transmission electron microscopy and the TUNEL assay. Surviving cardiomyocytes were rarely observed in the infarct area, while substantial apoptosis was observed at the border of the infarct area. 3,22 The TUNEL assay revealed a greater amount of positive nuclear staining in the left ventricular myocardial tissue in the AMI group (Figure 3A) and a higher percentage of apoptotic cells in the AMI group compared to the sham and AMI + CST groups (AMI vs sham: 60.64% ± 10.62% vs 5.21% ± 1.35%; AMI vs AMI + CST: 60.64% ± 10.62% vs 46.82% ± 10.25%, P < .05; Figure 3B). Transmission electron microscopy revealed biological markers of apoptosis in the nuclei, mitochondria, and myofibrils of the cardiac muscle tissues (Figure 3C). Karyopyknosis, vacuole degeneration in the mitochondria, and dissolution of myofibrils were clearly observed in the AMI group, and minor vacuole degeneration in the mitochondria was observed in the AMI + CST group. Western blot and immunohistochemistry analyses revealed increased expression of Bax (proapoptotic protein) and decreased expression of Bcl-2 (antiapoptotic protein; Figures 4A and B and 5A and B). As caspase 3 is the main activator of apoptosis via the death receptor, mitochondrially mediated, and ER stress-induced apoptotic pathways, 23 we considered caspase 3 activity and cleaved caspase 3 activity to be markers of apoptosiss. The expression of caspase 3 and cleaved caspase 3 was greater in the AMI group than in the AMI + CST and sham groups (Figures 4A and B and 5C and D). These results indicate that CST suppressed myocardial apoptosis, prompting us to perform additional experiments to examine the pathway through which this cardioprotective effect may be exerted.

A, Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) of myocardial tissues. The TUNEL assay was used to evaluate apoptosis (n = 5 per group). The TUNEL stains the entire nuclear area of apoptotic cells brown (positive), whereas normal cells are stained blue (negative; magnification: ×400). B, The apoptotic index (AI) of myocardial tissues. The apoptotic cells were counted in 5 random fields at the border of the infarcted area. AI = number of apoptotic cells/total number of nucleated cells. Data are presented as the mean ± standard error of the mean. *P < .05 versus the sham group; # P < .05 versus the acute myocardial infarction (AMI) group. C, Electron microscopy. Representative electron micrographs show the apoptotic architecture of the mitochondria, nucleus, and myocardial filaments (n = 3 per group): red indicates karyopyknosis, blue indicates vacuole degeneration in the mitochondria, and purple indicates myofibril dissolution (magnification: ×10 000).

A, Representative photomicrographs of immunohistochemical staining for apoptotic markers. The expression of Bcl-2, Bax, and caspase 3 was examined in the sham, AMI, and AMI + CST groups (magnification: ×400). Brown indicates positive staining (n = 5 per group). B, Quantitative comparison of Bcl-2, Bax, and caspase 3 positive cells in the myocardial tissue of the sham, AMI, and AMI + CST groups. Data are presented as the mean ± standard error of the mean. *P < .05 versus the sham group; # P < .05 versus the AMI group. AMI indicates acute myocardial infarction; CST, cortistatin.
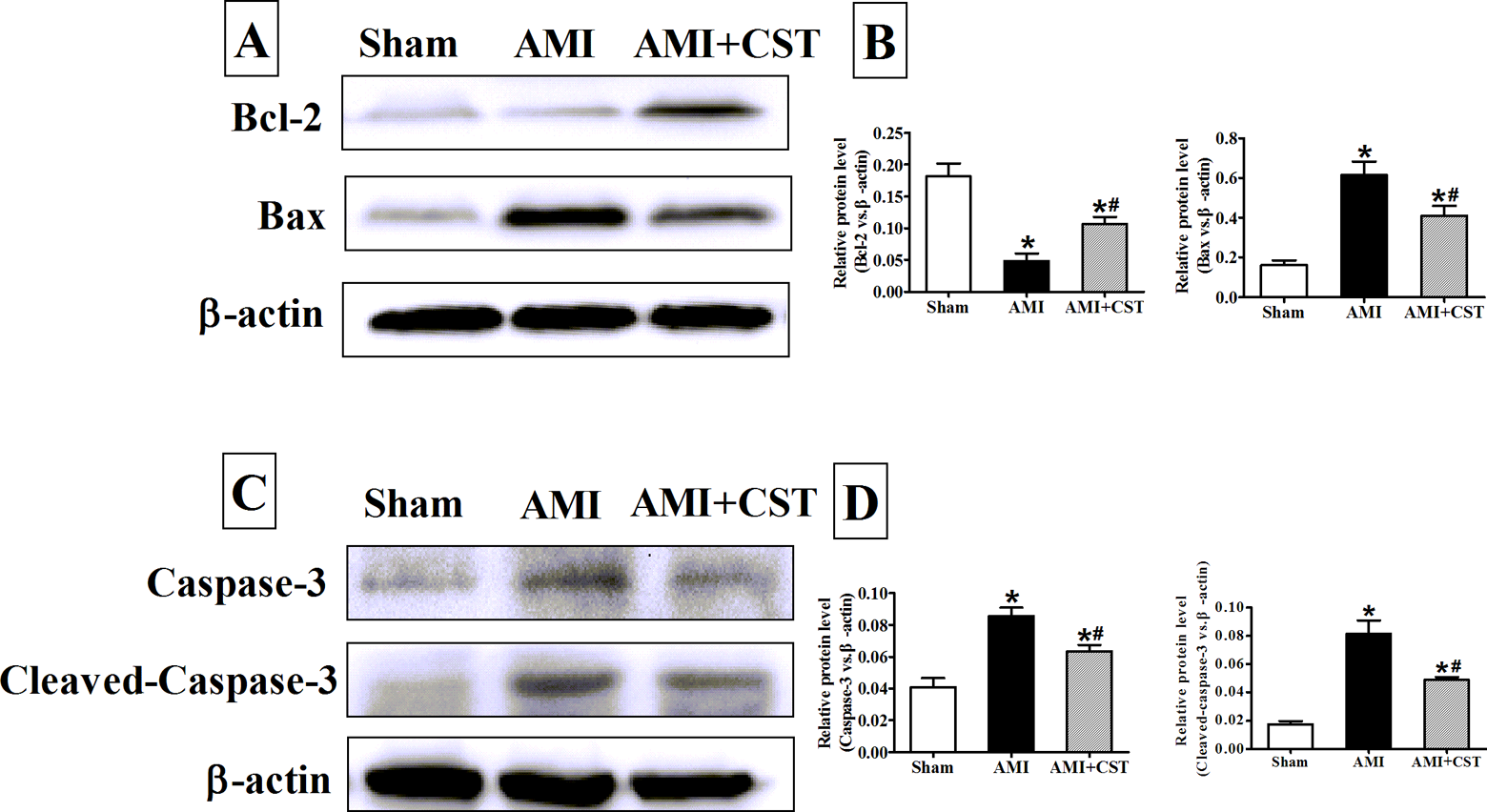
A and C, The expression of Bcl-2, Bax, caspase 3, and cleaved-caspase-3 proteins in myocardial tissues. Western blot analysis was used to detect the expression of Bcl-2, Bax, caspase 3, and cleaved-caspase-3 in myocardial tissues from the sham, AMI, and AMI + CST groups after 7 days. B and D, The density of each band was measured and compared to that of β-actin. β-actin was used as a protein loading control. Data are presented as the mean ± standard error of the mean. *P < .05 versus the sham group; # P < .05 versus the AMI group. AMI indicates acute myocardial infarction; CST, cortistatin.
Cortistatin Suppressed ER Stress in Rats With AMI
Endoplasmic reticulum stress is involved in the pathophysiological process of many cardiovascular diseases, 7,8 and recent studies have confirmed that suppressing ER stress can prevent myocardial apoptosis. 9 Therefore, we examined the expression levels of GRP94, GRP78, CHOP, and caspase 12 using immunohistochemistry and Western blotting. Compared to the AMI group, the AMI + CST group exhibited lower expression levels of GRP94 and GRP78, which indicated a reduction in ER stress (Figures 6 and 7). We also observed a simultaneous increase in the expression of caspase 12 and CHOP in the AMI group, although the AMI + CST group exhibited suppressed expression of CHOP and caspase 12. These results indicated that exogenous CST inhibited ER stress and the related apoptotic pathway.

A, Representative photomicrographs of immunohistochemical staining for ER stress markers. The expression of GRP94, GRP78, and caspase 12 was examined in the sham, AMI, and AMI + CST groups (magnification: ×400). Brown indicates positive staining (n = 5 per group). B, Quantitative comparison of GRP94, GRP78, and caspase 12 positive cells in the myocardial tissue of the sham, AMI, and AMI + CST groups. Data are presented as the mean ± standard error of the mean. *P < .05 versus the sham group; # P < .05 versus the AMI group. AMI indicates acute myocardial infarction; CST, cortistatin; ER, endoplasmic reticulum.

A and C, The expression of GRP94, GRP78, caspase 12, and C/EBP homologous protein (CHOP) in myocardial tissues. Western blot analysis was used to detect the expression of GRP94, GRP78, caspase 12, and CHOP in the myocardial tissues from the sham, AMI, and AMI + CST groups for 7 days. B and D, The density of each band was measured and compared to that of β-actin. β-actin was used as a protein loading control. Data are presented as the mean ± standard error of the mean. *P < .05 versus the sham group; # P < .05 versus the AMI group. AMI indicates acute myocardial infarction; CST, cortistatin; C/EBP, CCAAT/enhancer-binding proteins.
Discussion
Myocardial apoptosis plays an essential role in the myocardial remodeling and subsequent healing that are involved in multiple pathological processes after AMI. 2,22 Our results demonstrate that the development of myocardial apoptosis was associated with decreased myocardial contractility and increased expression of myocardial necrosis markers in a rat AMI model. However, exogenous CST showed a notable cardioprotective effect after AMI, possibly mediated by the inhibition of ER stress and myocardial apoptosis. We also observed that CST was elevated in the myocardial tissue after the induction of AMI, although exogenous treatment reduced the expression. This finding indicates that further studies are needed to elucidate the mechanisms by which CST exerts its cardioprotective effects.
Despite remarkable advances in medical and device treatments, myocardial apoptosis remains an important form of myocardial cell death due to the myocardial ischemia and hypoxia that occur after occlusion of a coronary artery. Myocardial apoptosis occurs in the border zone during AMI and was originally defined according to its morphological characteristics. 3 Furthermore, myocardial apoptosis results in the release of cellular contents, which amplifies the damage to neighboring cells 6 and can cause further left ventricular dysfunction. Our study is the first study to use CST to treat an animal model of AMI. Our findings confirmed the presence of cardiac dysfunction and myocardial apoptosis in the AMI group and demonstrated that exogenous CST treatment improved myocardial contractility and reduced myocardial apoptosis. Furthermore, the results from our TUNEL staining and electron microscopy revealed that CST ameliorated the clear changes in the apoptotic cell morphology and apoptosis index seen in the AMI group. In combination with our echocardiographic and TTC staining findings, these results indicate that CST treatment markedly reduced scar size and cardiac function.
Caspase 3 is an “executioner” caspase in the mammalian apoptotic pathway and can be activated by initiator caspases. 23 Activated caspase 3 cleaves several crucial substrates, which include the DNA repair enzyme poly (adenosine diphosphate-ribose) polymerase. In this context, Bcl-2 is an antiapoptotic protein that localizes to the ER membrane and protects against ER stress. Bax (another member of the Bcl-2 family) is a proapoptotic protein that forms dimers and induces apoptosis when it is overexpressed. 24 In our model, CST downregulated the expression of caspase 3 and Bax and upregulated the expression of Bcl-2. Thus, CST appears to be able to exert antiapoptotic effects after AMI, which reduce myocardial loss and myocardial scar size. Nevertheless, further studies are needed to improve our understanding of how ER stress interacts with its downstream apoptotic pathway.
The ER is very sensitive to environmental changes that disrupt its normal homeostasis. In this context, AMI describes a clinical state that is characterized by the lack of sustained coronary blood flow, reduced levels of myocardial oxygen and nutrition, and the accumulation of unfolded or misfolded proteins. Together, these factors can significantly affect ER homeostasis and can activate ER stress, which induces the related apoptotic pathway. The molecular chaperones GRP94 and GRP78 are homologs of the HSP protein and mainly mediate the UPR under physiological conditions. Their expression levels are directly correlated with the ability of the ER to manage unfolded and misfolded proteins. Therefore, GRP94 and GRP78 are widely regarded as markers of ER stress. 7 Furthermore, severe ischemia and hypoxia may lead to an increased expression of GRP94 and GRP78 after AMI. These findings are consistent with our experimental data, as we observed that the expression of GRP94 and GRP78 was increased in the AMI group but was lower in the AMI + CST group. Thus, our results indicate that CST treatment can help suppress ER stress after AMI. Furthermore, caspase 12 is located in the outer membrane of the ER, 25 where it acts as a master enzyme that is specifically involved in the ER stress-induced apoptotic pathway. Together with caspase 9, which in turn activates caspase 3, 26 caspase 12 mediates a novel intrinsic apoptotic pathway that is independent of other caspase-dependent pathways, and the activation of caspase 12 is regarded as a trigger for ER stress-induced apoptosis. Moreover, the expression of CHOP/GADD153 (growth arrest and DNA damage-inducible 153, part of the family of C/EBPs) is related to growth arrest and DNA damage, and this protein is a vital transcription factor that is responsible for ER stress-induced apoptosis. 8 The CHOP may also downregulate the expression of Bcl-2, 27 reduce the depletion of glutathione, and activate caspase 3, which ultimately leads to apoptosis. In addition, reports have suggested that deletion of the CHOP gene leads to an attenuation of ER stress-induced apoptosis. 28,29 We also observed that the expression of CHOP and caspase 12 was upregulated in the AMI group, which indicates that they participate in activating ER stress and the related myocardial apoptotic pathway. However, exogenous CST was able to suppress this ER stress and reduced the induction of myocardial apoptosis by downregulating the expression of CHOP and caspase 12.
Our immunohistochemical findings indicate that CST expression is markedly increased after AMI, although the expression decreases after exogenous treatment. Thus, CST may be a useful bioactive peptide for treating the pathophysiological processes that occur after AMI. The rat CST-14 peptide shares 13 of the last 14 residues with human CST and possesses similar biological activities that are activated by the cleavage of pre- and pro-CST. 30 Furthermore, there is accumulating evidence indicating that CST has functions that are distinct from those of its natural analogue (SST), 31 especially in various immune cells such as monocytes, macrophages, and dendritic cells. 15 Moreover, the expression of CST is more widespread than that of SST, which is not found in many tissues. Thus, it is likely that CST is an endogenous regulatory peptide that has multiple biological activities in the cardiovascular system. A recent study also reported that CST plays a distinct functional role in preventing and treating vascular calcification by binding to GHS-R1a instead of SSTRs. 32 As a postinfection treatment for Klebsiella pneumoniae infection, CST can also significantly attenuate apoptotic cell injury in the cortex and hippocampus. 33 However, there is no in vitro evidence or findings in the clinical setting of the effect of CST on myocardial apoptosis after AMI. Zhang et aldemonstrated that CST can exert cardioprotective effects by suppressing the ER stress-induced apoptotic pathway. 21 Ischemia and hypoxia (AMI) can lead to myocardial apoptosis via the ER stress pathway. 34 Reports have demonstrated that troponin T and N-terminal pro-B-type natriuretic peptide are increased in patients with sepsis, 35,36 and they are recognized as important biomakers in myocardial injury in AMI. These findings indicate that there is a correlation between AMI and sepsis. Although the results from our animal model suggest that CST is a bioactive peptide that provides a cardioprotective effect in AMI by suppressing ER stress and the related myocardial apoptotic pathway, further studies are needed to confirm our findings and to evaluate the usefulness of this peptide in the clinical setting.
Conclusion
In summary, our findings indicate that CST protects against myocardial apoptosis and improves cardiac function in rats after AMI. These effects may be due to its inhibition of ER stress and myocardial apoptosis. Our results suggest a novel therapeutic strategy for improving cardiac function and for reducing myocardial scar size after AMI. In addition, CST is able to bind to GHS-R1a, which can also protect against AMI-induced cardiac dysfunction. We plan to conduct further research to investigate this relationship.
Limitation
There are several limitations to this study. First, the border zone between infarct and normally perfused tissue is very sharp. Therefore, we will perform Evens blue and TTC staining to distinguish this zone. Second, to detect the cardioprotective mechanism of CST in myocardial apoptosis, we will use the CST receptor inhibitor and silence the ER stress-induced apoptosis pathway in vitro. Third, the expression of CST is greater in the AMI group and lower in the treated group, that is, only a phenomenon and cannot directly show CST improves remodeling. Thus, more research is needed to confirm the effect of CST in the future.
Footnotes
Author Contributions
Zhi-Yu, S contributed to conception and design, acquisition analysis, and interpretation; drafted the manuscript; and gave final approval. Yue, L contributed to conception and acquisition, critically revised the manuscript, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Li, D contributed to acquisition and interpretation. Bo, Z; Meng, Z; Wen-Xiu, L; and Xin, Z contributed to acquisition. Xin-Hua, Y contributed to conception and design, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (no. 81370319 and 81200235).
