Abstract
Background:
Insulin resistance (IR) is a well-known risk factor for cardiovascular complications. This study aimed to investigate the effect of a dietary model of IR in mice on cardiac remodeling, cardiac β-arrestin2 signaling, and the protective effects of carvedilol as a β-arrestin–biased agonist.
Methods and Results:
Insulin resistance was induced by feeding mice high-fructose/high-fat diet (HFrHFD) for 16 weeks. Carvedilol was adiministered for 4 weeks starting at week 13. At the end of the experiment, body weight, heart weight, left and right ventricular thickness, visceral fat weight, fasting blood glucose (FBG), serum insulin, IR index, and serum endothelin-1 were measured. In addition, cardiac tissue samples were histopathologically examined. Also, cardiac levels of cardiotrophin-1, β-arrestin2, phosphatidylinositol 4,5 bisphosphate (PIP2), diacylglycerol (DAG), and phosphoserine 473 Akt (pS473 Akt) were measured. Results showed significant increases in the FBG, serum insulin, IR index, serum endothelin-1, cardiac DAG, cardiac fibrosis, and degenerated cardiac myofibrils in HFrHFD-fed mice associated with a significant reduction in cardiac levels of cardiotrophin-1, β-arrestin2, PIP2, and pS473 Akt. On the other hand, carvedilol significantly reduced the heart weight, FBG, serum insulin, IR index, serum endothelin-1, cardiac DAG, left ventricular thickness, right ventricular fibrosis, and degeneration of cardiac myofibrils. In addition, carvedilol significantly increased cardiac levels of cardiotrophin-1, β-arrestin2, PIP2, and pS473 Akt.
Conclusion:
Carvedilol enhances cardiac β-arrestin2 signaling and reduces cardiac remodeling in HFrHFD-fed mice.
Introduction
Insulin resistance (IR) is a condition of inadequate insulin response usually associated with obesity, sedentary life style, and high-fat/high carbohydrate diets. 1 The prevalence of this condition increases rapidly around the world. One of the main complications of IR is an increased risk of cardiovascular disorders including cardiac remodeling and dysfunction. 2 The molecular mechanisms that link IR to cardiac remodeling are still not compeletely clarified.
Cardiac remodeling involves two main components: cardiac fibrosis and cardiac hypertrophy. Cardiac fibrosis commonly refers to the excess deposition of collagen and extracelluar matrix (ECM) components in the cardiac muscle. On the other hand, cardiac hypertrophy refers to the abnormal thickening of the heart muscle, which may result from increased cardiomyocyte size and deposition of ECM components. 3
One of the possible mechanisms by which IR mediates cardiac remodeling is the cardiac lipotoxicity. 4 During IR, the rate of lipolysis increases leading to excessive production of free fatty acids (FFAs). 5 Circulating FFAs can affect the heart leading to increased diacylglycerol (DAG) and reactive oxygen species (ROS) production in the cardiomyocytes 6 with subsequent impairment in calcium handling, contractile dysfunction, apoptosis, and remodeling. 7
Recently, specific FFAs receptors have been identified with a wide distribution in all organs. These receptors were known formerly as orphan Gprotein-coupled receptors (GPCRs or GPRs), however, they were renamed lately as FFA receptors (FFARs) 1 to 4. 8 FFARs 1 and 4 are Gαq protein–coupled receptors. 9 Activation of these receptors by circulating FFAs elevates intracelluar levels of DAG and protein kinase C (PKC), the later impairs insulin signaling and enhances inflammatory and apoptotic signals. 6
β-Arrestins are GPCRs desensitizing and scaffolding proteins. 10 Selective activation of β-arrestin signaling in the heart has been found to protect the heart and reduce cardiac dysfunction. 11 Furthermore, recent findings have identified a new promising role for β-arrestins in insulin signaling. β-Arrestins form a complex with insulin receptor substrate-1 (IRS-1), Akt, and Src to enhance insulin signaling. 12 On the other hand, IR is associated with severe downregulation of β-arrestin2, a subtype of β-arrestins. 13
Carvedilol is a third-generation β-blocker with a unique β-arrestin–biased agonistic activity. 14 Among other β-blockers, carvedilol possesses the highest cardioprotective efficacy against heart failure due to its unique effect on β-arrestin signaling. 14
This study aimed to investigate the effect of IR induced by feeding mice high-fructose/high-fat diet (HFrHFD) for 16 weeks on cardiac remodeling and cardiac β-arrestin2 signaling and the effect of carvedilol as a β-arrestin–biased agonist on these pathological changes.
Materials and Methods
Animals
Adult male Swiss albino mice (20 ± 5 g, 8 weeks old) were purchased from the Faculty of Veterinary Medicine, Zagazig, Egypt, and housed in plastic cages with wood shave bedding in the animal care unit of the Faculty of Pharmacy, Zagazig. The animals were kept under controlled temperature (23 ± 2°C), humidity (60 ± 10%), and 12-hour light/dark cycle. Mice were acclimatized for at least 2 weeks prior to the experiments and had access to standard pellet chow diet and tap water.
Ethical Statement
All procedures were conducted in accordance with the national and international guidelines for care and use of laboratory animals and were approved by the Animal Ethics Committee of the Faculty of Pharmacy, Zagazig University, Egypt (Approval number: P2/12/2016).
Drugs
Carvedilol was obtained from Bio Pharma (Cairo, Egypt) and dimethyl sulfoxide (DMSO) was purchased from Sigma-Aldrich (St. Louis, MO, USA).
Experimental Design
After the acclimatization period, mice were randomly distributed into 3 experimental groups (n = 10 each). Group 1 (standard chow diet, SCD): includes mice that were fed SCD (CPC Co, Giza, Egypt) for 16 weeks. Groups 2 and 3 (HFrHFD): include mice that were fed HFrHFD composed of 155 g of chow diet, 200 g of beef tallow, 170 g of fructose, 320 g sweetened condensed milk, 100 g corn gluten (60% protein), 25 g of salt mixture, and 30 g of water per kilogram of diet and received fructose (20% wt/vol) in drinking water for 16 weeks. 15 All nutritional parameters of this diet meet or exceed the guidelines of National Research Council, Canada, for rats and mice (Table 1). Groups 1 (SCD) and 2 (HFrHFD) received only the vehicle. Group 3 (CARV) received carvedilol (10 mg/kg/d, IP). Carvedilol was dissolved in the vehicle, a mixture of DMSO, Tween 80, and water (1:1:8) and administered at a volume of 100 μL/40 g body weight. Carvedilol was administered for 4 weeks starting from week 13 of feeding. Carvedilol dose was selected based on previous publications. 16
Components of SCD and HFrHFD.
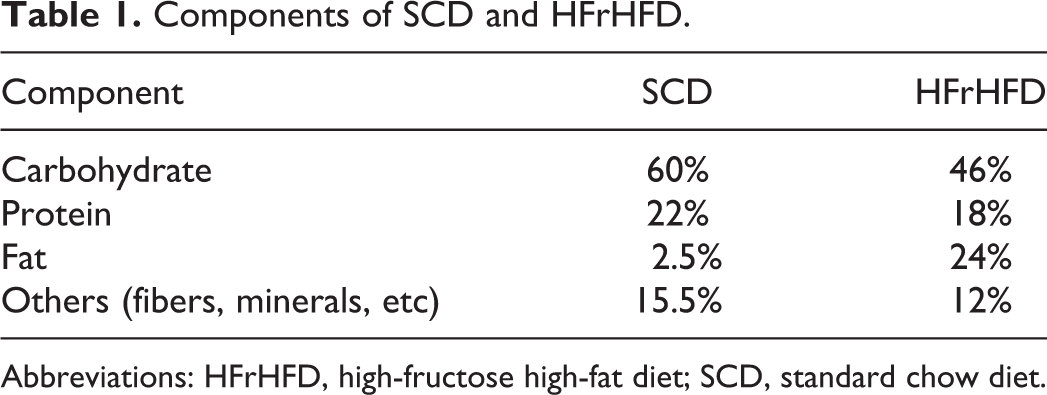
Abbreviations: HFrHFD, high-fructose high-fat diet; SCD, standard chow diet.
Blood and Tissue Sampling
At the end of experiments, mice were euthanized by decapitation. Trunk blood was collected from the site of decapitation and centrifuged (4000 rpm, 4°C, 15 minutes). Serum was stored at −80°C for subsequent analyses. Heart samples were collected and divided into 2 parts; one part was kept in formalin for histopathological examination, the other part was snap frozen into liquid nitrogen and stored at −80°C for subsequent biochemical analyses.
Measurement of Body Weight and Visceral Fat Weight
At the end of the experiments and before euthanasia, fasted mice body weights were measured. After euthanasia, visceral (epididymal) fat was dissected and weighed.
Measurement of Blood Glucose Level
Blood glucose level was measured from a blood drop obtained from mouse tail tip using an automated glucometer (GM100; Bionime GmbH, Berneck, Switzerland).
Determination of Serum Insulin and Endothelin-1 and the Calculation of IR Index
Serum insulin and endothelin-1 levels were measured by enzyme-linked immunosorbent assay (ELISA) technique using kits supplied by CUSABIO, Houston, Texas (Cat. No. CSB-E05071m) and Biovision, San Francisco, California (Cat No. k7429-100), respectively. Insulin resistance index was calculated using homeostatic model of assessment (HOMA) index for IR according to the following equation 17 :

Histopathological Examination
Tissue specimens from the heart of each animal were excised, directly fixed in 10% formalin solution, dehydrated in alcohol, cleared in xylene, and embedded in paraffin blocks. Sections of 5-µm thickness were obtained and stained with hematoxylin and eosin and Masson trichrome stain.
Calculation of the Percentage of Fibrosis Area Using Masson Trichrome Stain
Quantifying the percentage of collagen fibers area stained by Masson trichrome was done by selecting the blue color of fibers using Image J Software v.1.51d (National Institutes of Health, Bethesda, MD, USA). The area of fibers was selected and masked by red binary color. Then, the area of the red binary color was measured and expressed as average area in relation to the area of the standard measuring frame and expressed as area %. Fibrosis area was measured in transverse cross-sections of total heart samples, left ventricles, right ventricles, and perivascular area.
Measurement of the Thickness of the Right and Left Ventricular Walls
The measurement was carried out on the microscopic transverse cross-sections of total heart samples using Scion Image for Windows software (Meyer Instruments, Houston, TX, USA). 18 The highest thickness of either the right or left ventricle was recorded as a single read for each cross-section.
Determination of Cardiac Levels of β-Arrestin2 and Downstream Signals in the Heart
One hundred milligram was weighed from each cardiac tissue (total heart sample) and homogenized using Con-Torque Tissue Homogenizer supplied by Eberbach (Ann Arbor, MI, USA) in 500 μL phosphate-buffered saline. Each homogenate was centrifuged at 10 000 rpm and 5°C temperature for 10 minutes using a cooling centrifuge. The supernatant was collected into a new microcentrifuge tube (1.5 mL) and then used to assess all biochemical parameters. Units were expressed per milligram protein. Cardiac levels of β-arrestin2, phosphatidylinositol 4,5 bisphosphate (PIP2), DAG, phosphoserine 473 Akt, and cardiotrophin-1 were measured by ELISA technique using kits supplied by Nova Lifetech limited (Mongkok, Hong Kong; Cat. No. CELI-66111m), BlueGene Biotech (Shanghai, China; Cat. No. E03D0010), LifeSpan BioSciences (Seattle, WA, USA; Cat. No. LS-F18999), Abcam (Cambridge, United Kingdom; Cat No. ab176635), and Boster Biological Technology (Pleasanton, CA, USA; Cat. No. EK0564), respectively. Using ELISA technique for measuring β-arrestin2, PIP2, DAG, and phosphoserine 473 Akt was previously studied and referenced. 19 -22
Statistical Analysis
Data are presented as mean ± standard error of the mean. Statistical significance was tested using one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparisons test by GraphPad Prism v.6 (GraphPad Software Inc., La Jolla, CA, USA). P < .05 was considered significant.
Results
Carvedilol Reduced Heart Weight of HFrHFD-Fed Mice
Feeding mice HFrHFD for 16 weeks did not significantly affect body weight, heart weight, and visceral fat weight compared to SCD-fed mice. On the other hand, carvedilol treatment significantly reduced the heart weight (−22.8% and −23.6%, Figure 1B) and the heart weight normalized to tibial length (−28% and −25.7%, Figure 1E) compared to SCD and HFrHFD groups, respectively. However, carvedilol treatment did not significantly affect the body weight and the visceral fat weight compared to either SCD or HFrHFD groups.
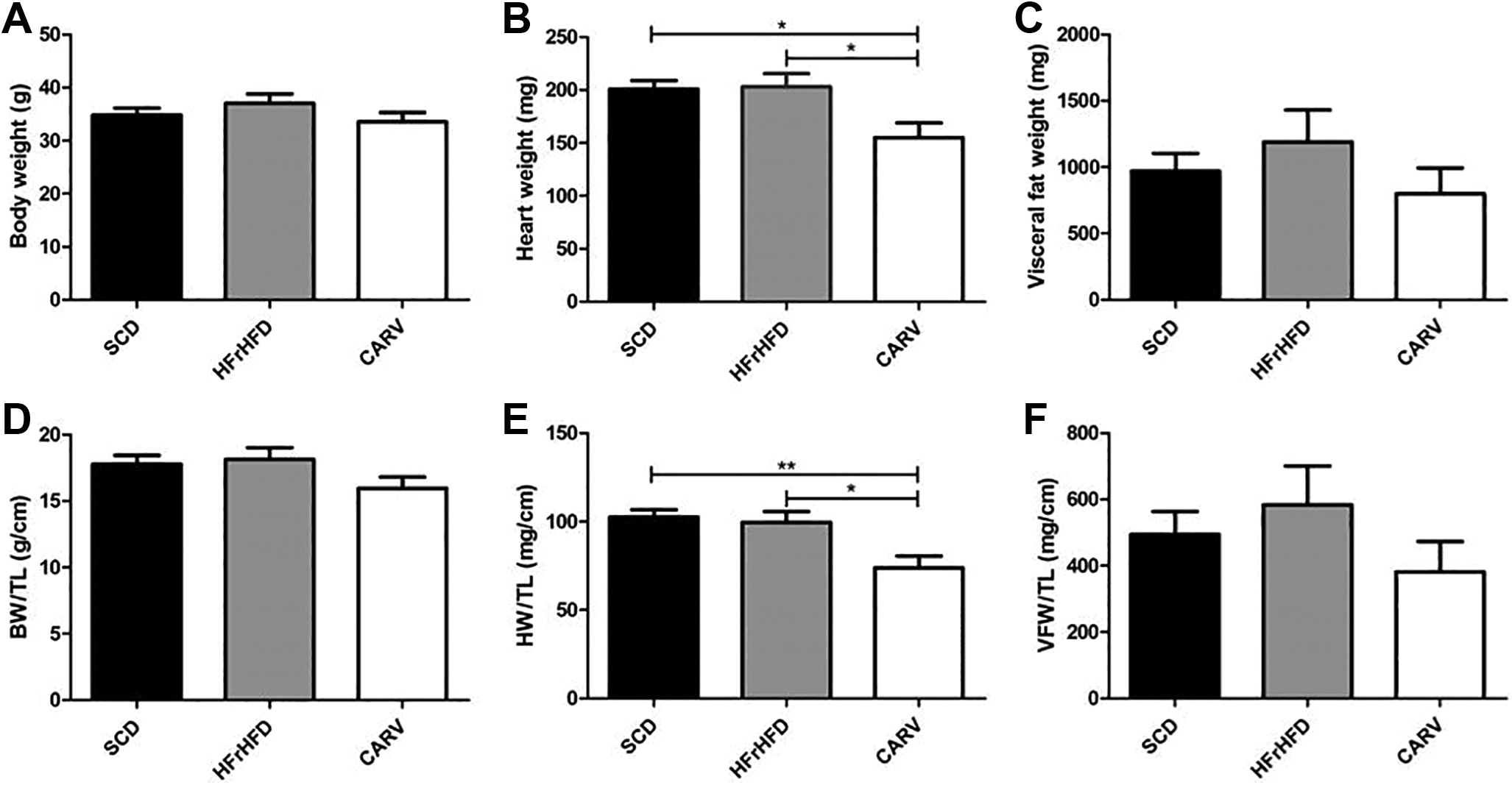
Changes in BW, HW, VFW, BW/TL, HW/TL, and VFW/TL. Graphical presentation of BW (A), HW (B), VFW (C), BW/TL (D), HW/TL (E), and VFW/TL (F). SCD: Mice were fed standard chow diet for 16 weeks. HFrHFD: Mice were fed high-fructose high-fat diet for 16 weeks. CARV: HFrHFD-fed mice were treated with carvedilol (10 mg/kg/d, IP) for 4 weeks starting at week 13. Statistical analysis was performed using 1-way analysis of variance (ANOVA) followed by Tukey post hoc test. Values are represented as mean ± standard error of the mean (SEM). n = 4 to 6. *P < .05. BW indicates body weight; BW/TL, body weight/tibial length; HFrHFD, high-fructose/high-fat diet; HW, heart weight; IP, intraperitoneal; SCD, standard chow diet; VFW, visceral fat weight.
Carvedilol Reduced IR in HFrHFD-Fed Mice
As depicted in Figure 2, feeding mice HFrHFD for 16 weeks significantly increased fasting blood glucose (FBG) levels (137.4 ± 1.5 vs 113.7 ± 5 mg/dL, Figure 2A), serum insulin levels (18.8 ± 0.75 vs 2.4 ± 0.24 IU/L, Figure 2B), and HOMA-IR (6.46 ± 0.56 vs 0.68 ± 0.09, Figure 2C) compared to SCD group. On the other hand, treating mice with carvedilol significantly decreased FBG levels (−17%, Figure 2A), serum insulin levels (−75%, Figure 2B), and HOMA-IR (−79%, Figure 2C) compared to HFrHFD-fed mice.

Changes in glycemic values. Quantitative analysis of fasting blood glucose (FBG) (A) and serum insulin (B). Graphical presentation of insulin resistance (C). SCD: Mice were fed standard chow diet for 16 weeks. HFrHFD: Mice were fed high-fructose high-fat diet for 16 weeks. CARV: HFrHFD-fed mice were treated with carvedilol (10 mg/kg/d, IP) for 4 weeks starting at week 13. HOMA-IR: Homeostasis model of assessment of insulin resistance. Statistical analysis was performed using 1-way analysis of variance (ANOVA) followed by Tukey post hoc test. Values are represented as mean ± standard error of the mean (SEM). n = 6 to 10. *P < .05, ***P < .001. HFrHFD indicates high-fructose/high-fat diet; HOMA-IR, homeostatic model of assessment-insulin resistance; IP, intraperitoneal; SCD, standard chow diet.
Carvedilol Reduced Cardiac Remodeling in HFrHFD-Fed Mice
Figure 3A and B depict representative camera and panoramic microscopic images of the hearts of the studied groups, respectively. Feeding mice HFrHFD for 16 weeks slightly increased both the left (5.5%, P > .05, Figure 4A) and right (26.6%, P > .05, Figure 4B) ventricular thickness compared to SCD group. On the other hand, carvedilol treatment for 4 weeks significantly decreased the left ventricular thickness (LVT, −20.6% and −16.2%, Figure 4A) compared to HFrHFD and SCD groups, respectively. No significant changes were observed in the right ventricular thickness (Figure 4B) in carvedilol-treated mice.
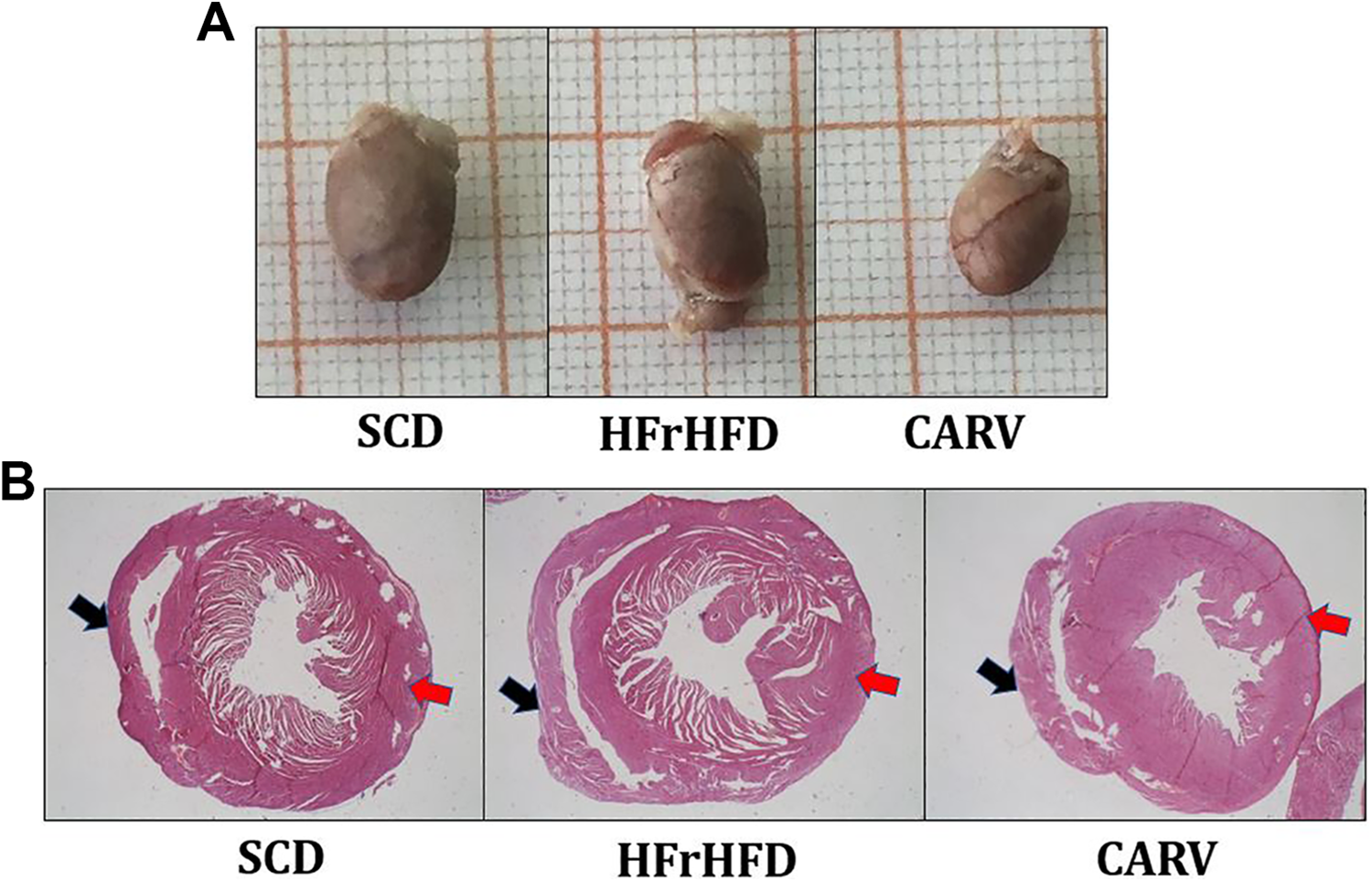
Cardiac morphological changes. Representative images of heart of different groups (A). Panoramic photomicrographs of cross-sections of the heart showing left ventricle (red arrows) and right ventricle (black arrows) (B). SCD: Mice were fed standard chow diet for 16 weeks. HFrHFD: Mice were fed high-fructose high-fat diet for 16 weeks. CARV: HFrHFD-fed mice were treated with carvedilol (10 mg/kg/d, IP) for 4 weeks starting at week 13. HFrHFD, high-fructose/high-fat diet; IP, intraperitoneal; SCD, standard chow diet.

Effect on cardiac remodeling and histopathology using hematoxylin and eosin (H&E) stain. Graphical presentation of left ventricle thickness (LVT) (A) and right ventricle thickness (RVT) (B). Representative photomicrographs of cardiac tissue stained with H&E stain, 200× (C). SCD: Mice were fed standard chow diet for 16 weeks. HFrHFD: Mice were fed high-fructose high-fat diet for 16 weeks. CARV: HFrHFD-fed mice were treated with carvedilol (10 mg/kg/d, IP) for 4 weeks starting at week 13. Statistical analysis was performed using 1-way analysis of variance (ANOVA) followed by Tukey post hoc test. Values are represented as mean± standard error of the mean (SEM). n = 3. **P < .01, ***P < .001. HFrHFD indicates high-fructose/high-fat diet; IP, intraperitoneal; SCD, standard chow diet.
In addition, histopathological examination of the cardiac tissue of HFrHFD-fed mice showed widespread and extensive Zenker necrosis (arrows) of degenerated myofibrils compared to SCD-fed mice (Figure 4C). On the other hand, carvedilol treatment markedly reduced these histopathological changes compared to HFrHFD-fed mice (Figure 4C).
Carvedilol Reduced Cardiac Fibrosis and Hypertrophy Markers in HFrHFD-Fed Mice
As depicted in Figure 5 and quantified in Figure 6, the cardiac tissue of HFrHFD-fed mice showed a significant increase in the myocardial interstitial fibrosis (blue stain, 209%, 160%, and 316%, Figure 6) in the total heart, left ventricle (red arrows), and right ventricle (black arrows), respectively, compared to SCD-fed mice. On the other hand, carvedilol treatment reduced the myocardial interstitial fibrosis (−41.7%, −26.7%, and −67.8%; Figure 6) in the total heart, left ventricle, and right ventricle, respectively, compared to HFrHFD-fed mice. However, the effect of carvedilol was significant only in the right ventricle. Notably, no significant changes were observed in the perivascular fibrosis area in all studied groups (Figure 7A). Furthermore, feeding mice HFrHFD for 16 weeks significantly decreased cardiac cardiotrophin-1 level (223.6 ± 10.46 vs 616.5 ± 17.39 pg/mg protein, Figure 7B), but it significantly increased serum endothelin-1 level (47.5 ± 4.9 vs 13.87 ± 0.98 pg/ml, Figure 7C) compared to SCD group. In addition, carvedilol treatment for 4 weeks significantly increased cardiac cardiotrophin-1 level (37.1%, Figure 7B), but it significantly decreased serum endothelin-1 level (−33%, Figure 7C) compared to HFrHFD group.

Effect on cardiac fibrosis. Representative panoramic photomicrographs of cardiac tissue stained with Masson trichrome stain (A). Representative photomicrographs of cardiac tissue stained with Masson trichrome stain, 200× (B). SCD: Mice were fed standard chow diet for 16 weeks. HFrHFD: Mice were fed high-fructose high-fat diet for 16 weeks. CARV: HFrHFD-fed mice were treated with carvedilol (10 mg/kg/d, IP) for 4 weeks starting at week 13. HFrHFD indicates high-fructose/high-fat diet; IP, intraperitoneal; SCD, standard chow diet.

Changes in the cardiac fibrosis area. Graphical presentation of fibrosis area percentage of cardiac tissue. SCD: Mice were fed standard chow diet for 16 weeks. HFrHFD: Mice were fed high-fructose high-fat diet for 16 weeks. CARV: HFrHFD-fed mice were treated with carvedilol (10 mg/kg/d, IP) for 4 weeks starting at week 13. Statistical analysis was performed using 1-way analysis of variance (ANOVA) followed by Tukey post hoc test. Values are represented as mean± standard error of the mean (SEM). n = 3. *P < .05, **P < .01. HFrHFD indicates high-fructose/high-fat diet; IP, intraperitoneal; SCD, standard chow diet.
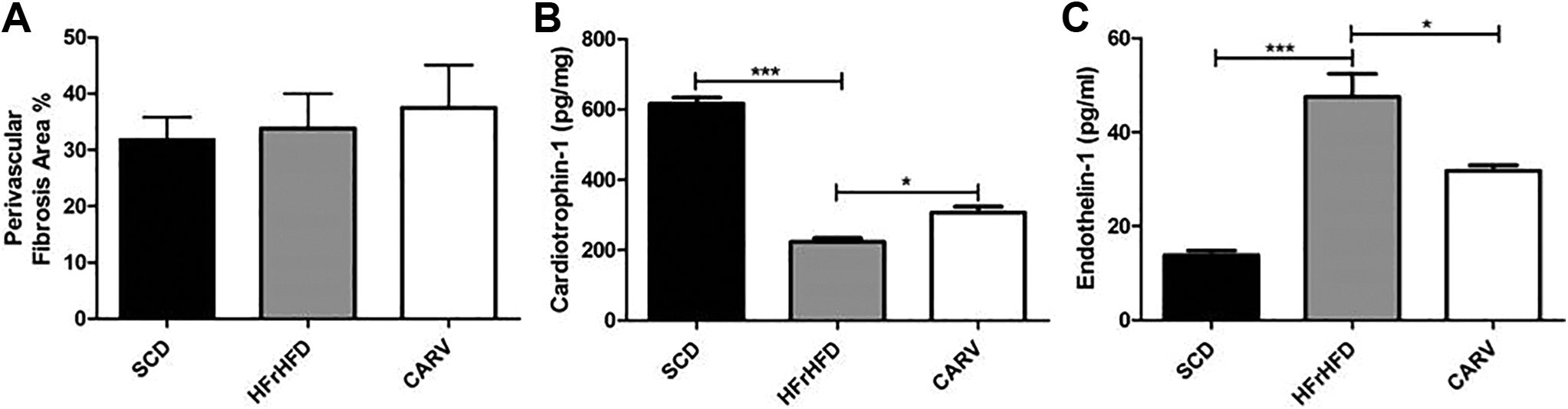
Changes in the cardiac perivascular fibrosis area and cardiotrophin-1 and serum endothelin-1 levels. Graphical presentation of cardiac perivascular fibrosis area percentage (A), Quantitative analysis of cardiac cardiotrophin-1 (B), and serum endothelin-1 (C). SCD: Mice were fed standard chow diet for 16 weeks. HFrHFD: Mice were fed high-fructose high-fat diet for 16 weeks. CARV: HFrHFD-fed mice were treated with carvedilol (10 mg/kg/d, IP) for 4 weeks starting at week 13. Statistical analysis was performed using one-way analysis of variance (ANOVA) followed by Tukey post hoc test. Values are represented as mean ± standard error of the mean (SEM). n = 3. *P < .05, ***P < .001. HFrHFD indicates high-fructose/high-fat diet; IP, intraperitoneal; SCD, standard chow diet.
Carvedilol Increased Cardiac β-Arrestin2 Signaling in HFrHFD-Fed Mice
Feeding mice HFrHFD for 16 weeks significantly decreased cardiac levels of β-arrestin2 (37.30 ± 2.66 vs 191.1 ± 10.89 ng/mg, Figure 8A), PIP2 (2.8 ± 0.21 vs 4.9 ± 0.47 ng/mg, Figure 8B), and AKT phosphorylation at serine 473 (2.50 ± 0.41 vs 16.80 ± 1.6 pg/mg, Figure 8D) compared to SCD group. Moreover, it significantly increased cardiac DAG level (13.73 ± 0.79 vs 9.3 ± 0.75 ng/mg, Figure 8C) compared to SCD-fed mice. On the other hand, carvedilol treatment for 4 weeks significantly increased cardiac levels of β-arrestin2 (117.5%, Figure 8A), PIP2 (47.5%, Figure 8B), and AKT phosphorylation at serine 473 in heart (176.8%, Figure 8D) compared to HFrHFD group. In addition, carvedilol treatment significantly decreased cardiac DAG level (−55.8%, Figure 8C) compared to HFrHFD-fed mice.

Changes in cardiac β-arrestin2 signaling. Quantitative analysis of β-arrestin2 (A), phosphatidyl inositol 4,5 bisphosphate (PIP2) (B), diacylglycerol (DAG) (C), phospho serine 473 of protein kinase B (AKT) (D). SCD: Mice were fed standard chow diet for 16 weeks. HFrHFD: Mice were fed high-fructose high-fat diet for 16 weeks. CARV: HFrHFD-fed mice were treated with carvedilol (10 mg/kg/d, IP) for 4 weeks starting at week 13. Statistical analysis was performed using one-way analysis of variance (ANOVA) followed by Tukey post hoc test. Values are represented as mean ± standard error of the mean (SEM). n = 3. *P < .05, **P < .01, ***P < .001. *a P < .05 using unpaired Student t test. HFrHFD indicates high-fructose/high-fat diet; IP, intraperitoneal; SCD, standard chow diet.
Discussion
The prevalence of IR increases every year in both developed and developing countries. Insulin resistance is a well-known risk factor for cardiovascular complications. 23 In this study, we investigated the effect of carvedilol as a β-arrestin–biased agonist on cardiac remodeling induced by feeding mice HFrHFD focusing on the changes in β-arrestin2 downstream signals.
Feeding mice HFrHFD for 16 weeks induced IR and cardiac remodeling. 15 Induction of IR in this model is attributed mainly to hypertriglycerdemia and increased activity of PKC with subsequent deactivation of IRS-1 leading to impaired insulin signaling. 24 To maintain blood glucose levels around normal values, insulin secretion usually increases leading to hyperinsulinemia. 25 Another possible cause for IR and hyperinsulinemia in this model is increased circulating FFAs levels, which activate FFARs in the pancreas leading to increased insulin secretion and FFARs in peripheral tissues leading to IR. 26
In accordance with previous studies, our results showed significant increases in FBG and serum insulin level in addition to significant increase in HOMA-IR in HFrHFD-fed mice. 27
Cardiac remodeling in HFrHFD-fed mice was previously reported in many studies. 15,27,28 It involves both diastolic and systolic dysfunctions, hypertrophy, cardiac fibosis, and endothelial dysfunction. 29 Molecular mechanisms underlying these pathological changes include decreased insulin signaling in cardiomyocytes leading to decreased Akt activity with subsequent increase in cardiomyocytes apoptosis. 6 Also, the elevated circulating levels of FFAs increase cardiomyocytes DAG levels with subsequent increase in oxidative stress and production of ROS. 6 Oxidative stress in turn impairs calcium handling and activates inflammatory, apoptotic, and fibrotic pathways in the cardiomyocytes leading to cardiac hypertrophy and fibrosis. 7,30 Oxidative stress also enhances the release of endothelin-1, which further contributes to cardiac hypertrophy and remodeling possibly by increasing cardiotrophin-1 expression. 31
In accordance with previous studies, our results showed significant increases in cardiac fibrosis, serum endothelin-1, and cardiac DAG levels in HFrHFD-fed mice compared to SCD-fed mice. In the same context, cardaic Akt activity significantly decreased in HFrHFD-fed mice compared to SCD-fed mice.
In contrast to previous studies, our results showed significant decreases in cardiac cardiotrophin-1 level in HFrHFD-fed mice compared to SCD-fed mice. Actually, the currently available information about the role of cardiotrophin-1 in cardiac remodeling is controversial. Some studies support its cardioprotective effects, 32 while others support its cardiac remodeling effects. 33 Furthermore, the majority of studies that examined the role of cardiotrophin-1 in cardiac remodeling were performed in end-stage heart failure, 34 a condition which is completely different from that of the current study.
On the other hand, β-arrestin2 role in cardiac protection is well established. 11 β-Arrestin2 can enhance the production of PIP2 and the activity of phosphatidylinositol 3 kinase (PI3K). 35 PI3K converts PIP2 to PIP3, which then activates Akt that in turn inhibits cardiomyocytes apoptosis and promotes their survival. 36 -38 Recently, it has been found that β-arrestin2 is also an essential protein for insulin signaling. Studies have revealed severe downregulation of β-arrestin2 in the liver and skeletal muscles of insulin-resistant mice. 13 Actually, β-arrestin2 forms a complex with IRS-1, Src, and Akt proteins promoting Akt phosphorylation and activation. 12
To our knowledge, no previous study examined the effect of IR on cardiac β-arrestin2 level. We showed here, for the first time, that feeding mice HFrHFD for 16 weeks potently decreased cardiac β-arrestin2 level. This may represent a new mechanism by which IR can mediate cardiac remodeling and cardiac complications. In harmony with decreased cardiac β-arrestin2 level, we showed also significant reduction in cardiac PIP2 level. Cardiac PIP2 regulates actin dynamics and T-tubule functions in the cardiomyocytes. 39 Phosphatidylinositol 4,5 bisphosphate deficiency has been found to be associated with T-tubule disruption and remodeling and defects in calcium cycling. 40
Carvedilol is a third-generation β-blocker which showed β-arrestin–biased agonistic activity. 14,41 Therotically and based on the findings of the recent studies that have shown a crucial role for β-arrestin2 in insulin signaling, we assumed that carvedilol might reduce IR and its related cardiac remodeling.
In accordance with our assumptions, carvedilol significantly reduced IR, heart weight, LVT, and right ventricular fibrosis in HFrHFD-fed mice. These changes were associated with significant reduction in serum endothlein-1 and cardiac DAG level, indicating improved endothelial function and cardiac insulin signaling, respectively. Also, carvedilol caused significant elevation in cardiac levels of β-arrestin2, PIP2, pS473 Akt, and cardiotrophin-1. These findings show that carvedilol can reverse IR, upregulate cardiac β-arrestin2 downstream signals, reduce cardiac apoptosis, and ameliorate cardiac remodeling in HFrHFD-fed mice.
Notably, this study has some limitations. We did not investigate the effects of carvedilol on SCD-fed mice; this would clarify whether the reduction in the heart weight and LVT is IR-dependent or not. Also, we did not measure the blood pressure changes, which may provide better understanding of the carvedilol cardioprotective effects.
However, this study reported several new findings; feeding HFrHFD to Swiss albino mice for 16 weeks caused IR and cardiac fibrosis associated with reduced cardiac β-arrestin2 and cardiotrophin-1. Furthermore, this study showed that carvedilol cardioprotective effects in insulin-resistant mice may be attributed, at least partially, to the upregulation of cardiac β-arrestin2 level and its downstream signals.
In conclusion, β-arrestin2 signaling is a promising target for the management of IR-induced cardiac remodeling.
Footnotes
Author contribution
WSI performed the experiments, all authors contributed equally to all other parts of this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
