Abstract
Atherosclerosis is a chronic inflammatory vascular disease characterized by lipid accumulation and endothelial dysfunction. Cytoglobin has been shown to exert protective effects under oxidative stress conditions. The aim of this study was to determine whether recombinant human cytoglobin (rhCYGB) has protective effects against atherosclerosis. We intraperitoneally injected rhCYGB (0, 4, or 7 mg/kg BW) into the atherosclerotic rats daily for 60 days. The rhCYGB injections reduced low-density lipoprotein cholesterol (LDL-C) levels and increased high-density lipoprotein cholesterol levels in a dose-dependent manner, rhCYGB (7 mg/kg) significantly attenuated atherosclerosis. Blood proteins were separated by 2-dimensional electrophoresis and analyzed by mass spectrometry, and the majority of the proteins in question were participated in oxidative stress pathways and cardiovascular diseases. Human hepatocellular liver carcinoma cell line (HepG2) cells were treated with oleic acid (0.3 mmol/L), and Human acute monocytic leukemia cell line (THP-1) cells were incubated with oxidized LDL (ox-LDL; 50 µg/mL) to induce foam cell (FC) formation in vitro. Treatment with different concentrations of rhCYGB (0, 5, 10, and 15 μg/mL) significantly decreased the lipid droplet levels in HepG2 cells and cholesterol ester levels in FCs. Moreover, rhCYGB significantly increased superoxide dismutase and glutathione peroxidase activity and decreased malondialdehyde and nicotinamide adenine dinucleotide phosphate oxidase activity in cells. In addition, rhCYGB decreased NO and Reactive oxygen species (ROS) levels in FCs by functioning as an NO dioxygenase enzyme and ROS scavenger. Taken together, our findings indicate that rhCYGB prevented atherosclerosis by regulating lipid metabolism and oxidative stress. Our study provides insights into the possible usefulness of rhCYGB as an antiatherosclerosis agent.
Introduction
Atherosclerosis is a chronic inflammatory vascular disease affecting large- and medium-sized arteries that are characterized by lipid accumulation and endothelial dysfunction. 1 Atherosclerosis is recognized as a leading cause of morbidity and mortality and has attracted attention from clinicians and researchers worldwide. Atherosclerotic lesions involving the arterial wall are characterized by hemorrhage, thrombosis, fibrous tissue proliferation, and calcium deposition, phenomena that lead to several adverse vascular events, such as angina, myocardial infarction, and stroke. 2,3 Hyperlipidemia and lipoprotein retention are crucial factors associated with the development of atherosclerosis and upregulate early atherosclerotic processes, including lipoprotein oxidation and cellular chemotaxis. 4 Arterial wall macrophages can take up excess low-density lipoprotein (LDL) and oxidized low-density lipoprotein (ox-LDL) molecules via scavenger receptors. Large amounts of ox-LDL induce macrophages to transform into foam cells (FCs). Lipoprotein retention and FC accumulation in the intimal layer of the arterial wall play vital roles in endothelial dysfunction and atherogenesis and thus initiate the early stages of atherosclerosis. 5,6 Moreover, lipid and protein oxidation in the vascular wall increases oxidative stress, thereby promoting the development of atherosclerosis.
Cytoglobin (CYGB), a member of the hexacoordinate globin superfamily (hxHb), is ubiquitously expressed in all tissues and has been reported to have protective effects in various cell types. Human CYGB weighs 20.9 kDa, comprises 190 amino acids, and is encoded by a gene located on chromosome 17q25. The CYGB participates in O2 sensing, transport, and storage. 7 Moreover, CYGB exerts enzymatic (peroxidase) effects and detoxifies ROS and nitric oxide (NO). 8,9 The CYGB has also been reported to act as a peroxide scavenger in fibrotic liver, pancreas, and kidney. 10 In addition, CYGB overexpression protected rat hepatic stellate cells against oxidative stress and thus prevented the cells from differentiating into myofibroblast-like cells. 11 Conversely, CYGB knockout in skeletal muscle or siRNA-mediated CYGB depletion in C2C12 myoblasts increased ROS levels and apoptosis, resulting in severe defects in muscle repair and regeneration. 12,13 Importantly, ferric CYGB was reported to bind lipid molecules with high affinity under oxidative stress conditions by transforming from a hexacoordinate to a pentacoordinate heme molecule. 14
Given the antioxidant activity and lipid-binding effects of CYGB, we hypothesize that the drug may have potential as a therapy for atherosclerosis in patients with lipid metabolic disorders. As CYGB is expressed at relatively low levels in cells, 11 we utilized recombinant bacteria to highly express recombinant human CYGB (rhCYGB) to investigate its effects on lipid metabolism and oxidative stress in high-fat diet (HFD)-induced atherosclerosis.
Materials and Methods
Reagents
Simvastatin was obtained from Sigma-Aldrich (St. Louis, Missouri). Vitamin D3 was obtained from Shanghai General Pharmaceutical Co, LTD (Shanghai, China). Alginic sodium diester (ASD; Hainan Helpson Medicine, Hainan, China), which was used as a positive control, is a commercial Chinese medicine used to modulate lipid metabolism. The indicated human monoblastic leukemia cell line (THP-1 cells) and HepG2 cells were purchased from the Chinese Academy of Science (Shanghai, China). Roswell Park Memorial Institute medium (RPMI-1640), Dulbecco's Modified Eagle Medium (DMEM), fetal bovine serum (FBS), penicillin, and streptomycin were obtained from Gibco BRL Life Technologies (Gaithersburg, Maryland); Ox-LDL was obtained from Zhongshan University (Guangzhou, China).
Preparation of rhCYGB
The rhCYGB was expressed using an Escherichia coli PET28a-rhCYGB-BL21 (DE3) plasmid, which was generated as described previously. 15 Proteins were renatured and preliminarily purified using Sephadex G-25 (GE Healthcare, Connecticut) and then further purified by affinity chromatography using the appropriate monoclonal antibody that was prepared in our laboratory. Purified rhCYGB was identified by sodium dodecyl sulfate polyacrylamide gel electrophoresis (12% acrylamide) and BandScan5.0 software. The protein concentration was determined by Bicinchoninic Acid Protein Assay Kit (BCA) assay, which was purchased from Pierce (Waltham, Massachusetts). The rhCYGB antioxidant activity was determined by Total antioxidant capacity assay kit (T-AOC) assay, which was obtained from Nanjing Jiancheng (Nanjing, China).
Animal Experimentation
All animal protocols were reviewed and approved by the Chinese Association of Laboratory Animal Care and the Institutional Animal Care and Use Committee of Southern Medical University, Guangzhou, China. Forty male Sprague-Dawley (SD) rats (aged 2-3 months and weighing 200-240 g) were purchased from the Experimental Animal Centre of Southern Medical University. The animals were housed under standard laboratory conditions (temperature, 18°C -20°C; humidity, 70%; 12 hours light/12 hours dark cycle), with free access to food and water. Acute rhCYGB toxicity was determined in vivo, according to a previously described protocol developed by our laboratory. 15 The atherosclerosis rat model was established by administering the animals an HFD and vitamin D3 injections (70 × 10 4 IU/kg, 4 times daily every 3 days). 16 The HFD diet comprised ordinary rat chow diet (85% w/w) supplemented with cholesterol (4% w/w), pig oil (10% w/w), propylthiouracilum (0.2% w/w), and bile salt (0.5% w/w). Forty male rats were divided into 5 groups. The control group (8 rats) was fed ordinary rat chow diet, and the other groups (32 rats) were fed an HFD for 30 days and intraperitoneally injected with vitamin D3 four times daily every 3 days to establish the atherosclerosis model. The 32 atherosclerotic rats were intraperitoneally injected with different concentrations of rhCYGB (0, 4, or 7 mg/kg BW/d) or ASD (6 mg/kg BW/d, positive control) for 60 days while receiving the HFD diet.
Measurement of the Lesions in Rats
At the end of the 90-day experiment, blood samples were collected from the abdominal arteries of the 40 rats, placed in tubes, centrifuged at 3000 rpm for 15 minutes to obtain plasma, and stored at −80°C. Triglyceride (TG), total cholesterol (TC), HDL-cholesterol (HDL-C), and LDL-cholesterol (LDL-C) levels were determined using an Abbott Aeroset TM Biochemical Analyzer (Abbott Laboratories, Illinois, USA). The liver (n = 8) and abdominal aorta (n = 8) were harvested and fixed in 10% formalin. The slides were stained with hematoxylin after being deparaffinized and rehydrated, after which they were subjected to eosin staining and dehydration. The thicknesses of the intimal and medial layers of the abdominal aorta (n = 8) were determined using an HP IAS-1000 image analysis system (Tongji thousand screen imaging company, Wuhan, China).
Two-Dimensional Electrophoresis (2-DE) Analysis
Serum samples (n = 8) were separated from blood and added to columns from a Protein Miner Protein Enrichment Kit to remove highly abundant proteins. The proteins were purified with a 2-D clean-up kit. The dry-strips (pH 5-8, 7 cm in length) were rehydrated for 16 hours with 120 μg of serum protein in 300 μL of rehydration buffer (7 mol/L urea, 2 mol/L thiourea, 4% and 40% CHAPS, and 0.3% ampholytes). Isoelectric focusing (IEF) was subsequently performed using a protein IEF cell system (Bio-Rad, California, USA). This procedure comprised the following 4 steps: 250 V for 1 hour, 500 V for 1 hour, 1000 V for 1 hour, and 10 000 V for 3-5 hours. Then, the dry strips containing the focused proteins were equilibrated using equilibration buffers containing 2% DL-Dithiothreitol (DTT) (w/v) and 2.5% iodoacetamide (w/v) for 15 and 20 minutes, respectively. Upon completion of equilibration, we transferred the strips onto a 12.5% polyacrylamide slab gel and continued 2D separation over 9.5 hours using a Protean II Xicell Electrophoresis System (Bio-Rad). Silver staining (Sigma-Aldrich, St Louis, Missouri) was used to stain the separated proteins. The 2D images were captured using a Umax Magic Scan System (Amersham Biosciences) and analyzed by PD quest 8.0.1 (Bio-Rad). The analyte and matrix were then mixed at a ratio of 6:1 (V/V), and mass spectrometric analysis was performed according to the manufacturer’s instructions. We acquired the mass spectral data and searched the NCBInr database with Mascot software.
RNA Extraction and Real-Time Polymerase Chain Reaction Analysis
Abdominal aorta tissues (n = 8) were harvested, and total RNA was prepared using TRIzol reagent (Invitrogen, Grand Island, New York), according to a previously described protocol. 17 Complementary DNA (cDNA) synthesis and real-time polymerase chain reaction (PCR; ABI Prism 7700 Sequence Detection System) were performed as described previously. 18 The experiments were performed in triplicate, and their results were compared to a standard curve. The sequences of the primers used for quantitative real-time PCR (qRT-PCR) are listed in Table 1.
Sequences of the Primers Used in the qRT-PCR Analysis.

Abbreviations: ApoE, apolipoprotein E; SOD, superoxide dismutase; Gpx, glutathione peroxidase; GST, glutathione S-transferase; LBP, lipopolysaccharide-binding protein.
Cell Culture and Treatment
HepG2 and THP-1 cells were cultured in DMEM and RPMI-1640, respectively, containing 10% FBS, 1% penicillin, and streptomycin at 37°C in a humidified atmosphere of 5% CO2. The cytotoxicity of rhCYGB was tested using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. HepG2 cells were incubated with rhCYGB (0, 5, 10, or 15 μg/mL) and oleic acid (OA) (0.3 mmol/L) for 24 hours. The ASD (1 μg/mL) was used as a positive control. THP-1 cells were incubated with propylene glycol methyl ether acetate (PMA) (100 ng/mL, Sigma) for 72 hours so that they would differentiate into macrophages. Then, the cells were treated with 50 µg/mL ox-LDL in serum-free RPMI 1640 medium containing 2% Bovine Serum Albumin (BSA) for 48 hours so that they would transform into FCs. 19 The FCs were subsequently treated with different concentrations of rhCYGB (0, 5, 10, or 15 μg/mL) or 1 μg/mL simvastatin (the positive control) for 24 hours. THP-1 cells treated with an equivalent volume of dimethyl sulfoxide (0.1%) without ox-LDL served as a normal control.
Oil Red O Staining
Lipid droplets in HepG2 cells stained with Oil Red O appear red and can be observed by an ordinary optical microscope. Oil Red O (Sigma) staining was performed in accordance with the manufacturer’s instructions. The cells were fixed with 10% formalin for 1 hour and then incubated with 60% filtered Oil Red O solution at room temperature for 30 minutes before being washed with phosphate-buffered saline (PBS)3 times and stained with hematoxylin. The contents of the lipid droplets (n = 6) were measured on a microplate reader at a wavelength of 520 nm.
Determination of Cell Cholesterol and Cholesterol Ester Levels
After being treated with rhCYGB for 24 hours, the FCs were washed thrice with cold PBS and harvested in 1 mL of PBS with a rubber policeman. Protein content was measured by the Bradford method, and lipids were extracted by the Vallianou and Peroulis method. 20 Total cholsterol and cholesterol ester (CE) levels (n = 6) were determined by gas chromatography (1100 HPLC chromatographic column). Then, 100 μg of cholestane in chloroform was added to a cell suspension containing 1 mg of protein, which served as an internal standard. Hexane and isopropyl alcohol (4:1, V/V) were subsequently added to the suspension, which was sufficiently mixed before being centrifuged at 1500 rpm for 5 minutes, after which the upper organic phase was collected for cold drying. The dry sample was then dissolved in 100 μL of isopropyl alcohol/heptane/acetonitrile (35:12:52, V/V), centrifuged at 1500 rpm for 5 minutes after ultrasonic degassing, and characterized with a gas chromatograph.
Cellular Antioxidant Enzyme Activity Assay
After being at 24-hour treatment with rhCYGB, the HepG2 cells and FCs (n = 6) were washed thrice with cold PBS, harvested in 1 mL of lysis buffer with a rubber policeman, and incubated on ice for 30 minutes. The activity levels of 2 intracellular antioxidant enzymes, namely, superoxide dismutase (SOD) and glutathione peroxidase (Gpx) as well as the activity levels of 2 oxidative stress biomarkers, namely, reduced nicotinamide adenine dinucleotide phosphate oxidase (NADPH oxidase) and malondialdehyde (MDA), were determined by commercial kits from Nanjing Jiancheng (Nanjing, China). Briefly, SOD activity levels were determined by nitroblue tetrazolium assay, which measures the ability of the enzyme to produce superoxide (O2−) as part of the xanthine/xanthine oxidase system, 21 and GSH-Px activity levels were determined based on changes in the absorption peaks resulting from the oxidation of NADPH to NADP as previously described. 22 The MDA concentration was determined using thiobarbituric acid reactive substances assay, according to the manufacturer’s instructions.
Assessment of ROS and NO
Intracellular ROS and NO levels (n = 6) were measured by staining with a reactive oxygen species assay kit and 4-Amino-5-Methylamino-2,7-Difluorofluorescein Diacetate (DAFFM) diacetate from Beyotime (Jiangsu, China). Fluorescence images were taken at excitation and emission wavelengths of 488 and 525 nm and 495 and 515 nm, respectively, and analyzed by Image-Pro Plus software, Media Cybernetics, Maryland, USA. To ensure statistical significance, we counted more than 1000 total cells per group and performed the assessment in triplicate.
Statistical Analysis
All data were analyzed using SPSS 13.0. The data are presented as the mean (standard deviation). Statistical significance was determined by 1-way analysis of variance, and P < .05 was considered statistically significant.
Results
Purity and Acute Toxicity of RhCYGB
Human CYGB cDNA inserted into a PET28a plasmid was transfected into E coli BL21 (DE3) to express rhCYGB proteins, and rhCYGB purity and antioxidant activity of rhCYGB were subsequently assessed by our team. We found that rhCYGB is safe and nontoxic to rats even at high doses, such as 100 and 500 mg/kg. 15
RhCYGB Significantly Decreased the Lipid Content in the Liver and Blood in an HFD-Induced Atherosclerosis Rat Model
It is well known that hyperlipidemia and fatty liver disease play an important role in the early stages of atherosclerosis. To investigate whether rhCYGB moderates atherosclerosis resulting from fat metabolism, we performed blood biochemical examinations and evaluated liver samples by hematoxylin–eosin (HE) staining. As shown in Figure 1A, the number of fat droplets in rat hepatocytes in the HFD group was significantly higher than that in the control group. As expected, the number of fat droplets in the rhCYGB groups (4 and 7 mg/kg) was significantly reduced compared to that in the HFD group. The TC, TG, LDL-C, and HDL-C are commonly measured biomarkers used to evaluate the risk of cardiovascular disease. 5 The TC, TG, and LDL-C levels were significantly reduced and HDL-C levels were increased in the rhCYGB groups compared to the HFD group (P < .01; Figure 1B). At 60 days and 90 days post-HFD initiation, the weight of the rats in the HFD group was much lower than that in the rhCYGB-treated group (Table 2), and weight of the rats in the control group was the highest among all the groups.
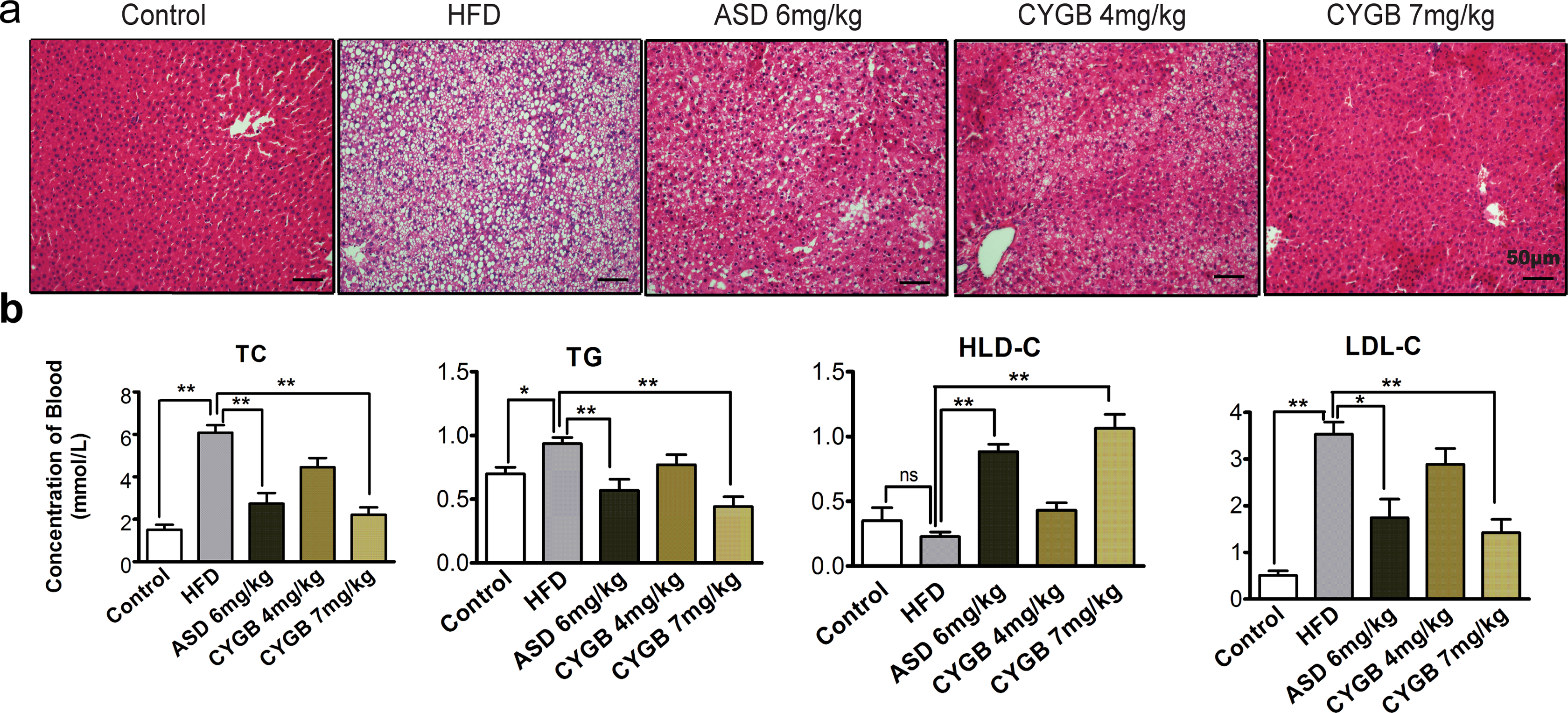
RhCYGB significantly decreased the lipid contents in the liver and blood of the HFD-induced atherosclerotic rats. A, HE-stained lesions in the liver. Representative images of 1 of 8 mice in each group are shown. Scale bars: 50 µm. B, TC, TG, HDL-C, and LDL-C levels in blood were determined by a biochemical analyzer. Means ± SEM. *P < .05, **P < .01. RhCYGB indicates recombinant human cytoglobin; HE, hematoxylin-eosin; HFD, high-fat diet; TC, total cholesterol; TG, triglycerides; LDL-c, low-density lipoprotein cholesterol; HDL-c, high-density lipoprotein cholesterol.
RhCYGB Changes the Weight of Experimental Rats.a

Abbreviations: HFD, high-fat diet; ASD, alginic sodium diester; rhCYGB, recombinant human cytoglobin.
aData are presented as means ± SD (n = 8).
bCompared to the control: P < .05.
cCompared to the HFD group: P < .05.
dCompared to the ASD group: P < .05.
RhCYGB Reduced the Thicknesses of the Atherosclerotic Lesions in the Arterial Wall in the HFD-Induced Atherosclerosis Rat Model
To test the ability of rhCYGB to prevent the development of atherosclerotic lesions, we stained serial sections of the abdominal aorta with HE and measured the thicknesses of the atherosclerotic lesions in these sections (Figure 2). The aorta is composed of the following 3 layers: the intima, the media, and the adventitia. The intima mainly comprises a layer of endothelial cells lining a basement membrane, the media comprises connective and muscle tissues, and the adventitia comprises connective tissue that protects the outer wall of the vessel. 23 HE staining revealed the presence of degenerating smooth muscle cells and speckled calcium deposits in the medial layer and FCs in the intimal layer of the abdominal aorta in the HFD group. These findings indicated that the atherosclerosis model had been successfully established. However, the atherosclerotic lesions in the abdominal aortas of the rhCYGB groups were much smaller after the rhCYGB treatment than before treatment and were even smaller than the lesions in the ASD group (the positive-control group; Figure 2A). Based on these results, rhCYGB exerted significant therapeutic effects by inhibiting the development of atherosclerosis and was slightly more effective than ASD in SD rats.
RhCYGB decreased the thicknesses of the atherosclerotic lesions in the arterial wall
of the HFD-induced atherosclerotic rats. A, HE-stained arterial wall lesions in the
rats. Representative images of one of eight mice in each group are shown (scale bars:
50 µm). The pictures in the square frame (upper panel) are shown in higher
magnification images (lower panel; scale bars: 20 µm). The various arrows point to
different lesions:  : intima
injury;
: intima
injury;  : lipid deposit;
: lipid deposit;
 : calcium accumulation; and
: calcium accumulation; and
 disordered arrangement of
fibers. B, Measurements of arterial wall thickness in each group of rats (n = 8; left)
and intimal-to-medial thickness ratios (n = 8; right). Means ± SEM.
*P < .05, **P < .01. RhCYGB indicates
recombinant human cytoglobin; HE, hematoxylin-eosin; HFD, high-fat diet.
disordered arrangement of
fibers. B, Measurements of arterial wall thickness in each group of rats (n = 8; left)
and intimal-to-medial thickness ratios (n = 8; right). Means ± SEM.
*P < .05, **P < .01. RhCYGB indicates
recombinant human cytoglobin; HE, hematoxylin-eosin; HFD, high-fat diet.
Pathological intimal thickening is the first manifestation of atherosclerosis, and the intimal-to-medial thickness ratio is used as a quantitative index of atherosclerosis development and progression. 24 -26 Consistent with our findings regarding atherosclerotic lesions, we found that the intimal thickness of the HFD group was much greater than that of the control group (P < .05). After rhCYGB treatment, the intimal-to-medial thickness ratio decreased from 58.82 ± 1.205 in the HFD group to 40.865 ± 1.374 in the 7-mg/kg group rhCYGB (P < .01; Figure 2B). Thus, rhCYGB decreased the intimal thickness at a dose of 4 mg/kg; rhCYGB also decreased the intimal thickness and the intimal-to-medial thickness ratio in a dose-dependent manner.
2-DE Analysis of the Differential Expression of Blood Proteins, a Phenomenon Accompanied by Differences in Gene Expression
To examine the effects of rhCYGB on the proteome of HFD-induced atherosclerosis, we performed 2-DE analysis to isolate and identify the blood proteins constituting the proteome. The 2-DE analysis revealed the presence of significant separation among the blood proteins. A total of 1322 proteins were in the control group, 1239 proteins were in the HFD group, and 1477 proteins were the 7-mg/kg rhCYGB group. The electrophoresis images of the serum proteins in the 3 groups revealed that 27 proteins were differentially expressed among the 3 groups and that these proteins were upregulated or downregulated by more than 1.5-fold in the treated groups compared to the control group (Supplemental figure). Ten peptide mass fingerprints were matched and identified by MALDI-TOF-MS and the abovementioned protein database. The expression levels of 5 of the 10 proteins were increased 3-fold, and the expression levels of the other 5 proteins were reduced 3-fold in the 7-mg/kg rhCYGB and control groups compared to those in the HFD group (P < .05). The functions of these proteins are listed in Table 3. These dysregulated proteins were predicted to be involved in an oxidation–reduction imbalance and cardiovascular disease. Specifically, apolipoprotein E (ApoE), beta-enolase (β-enolase), proteasome subunit alpha type-1 (PSMA1), creatine kinase M-type (CKM), and lipopolysaccharide-binding protein (LBP) were associated with lipid metabolism and atherosclerosis, and preprohaptoglobin, glutathione peroxidase 1 (GPx1), and glutathione S-transferase (GST) were associated with redox reactions. ApoE, GPx1, CKM, and LBP expression levels were decreased, and β-enolase, PSMA1, preprohaptoglobin, and GST expression levels were upregulated in the 7-mg/kg rhCYGB group compared to the HFD group. The expression levels of the genes encoding these proteins were validated by qRT-PCR, and the trends in the levels were the same as those in the protein expression levels (Figure 3).
Comparison of the Serum Levels of the Identified Proteins in the RhCYGB 7-mg/kg Group With Those in the HFD Group.
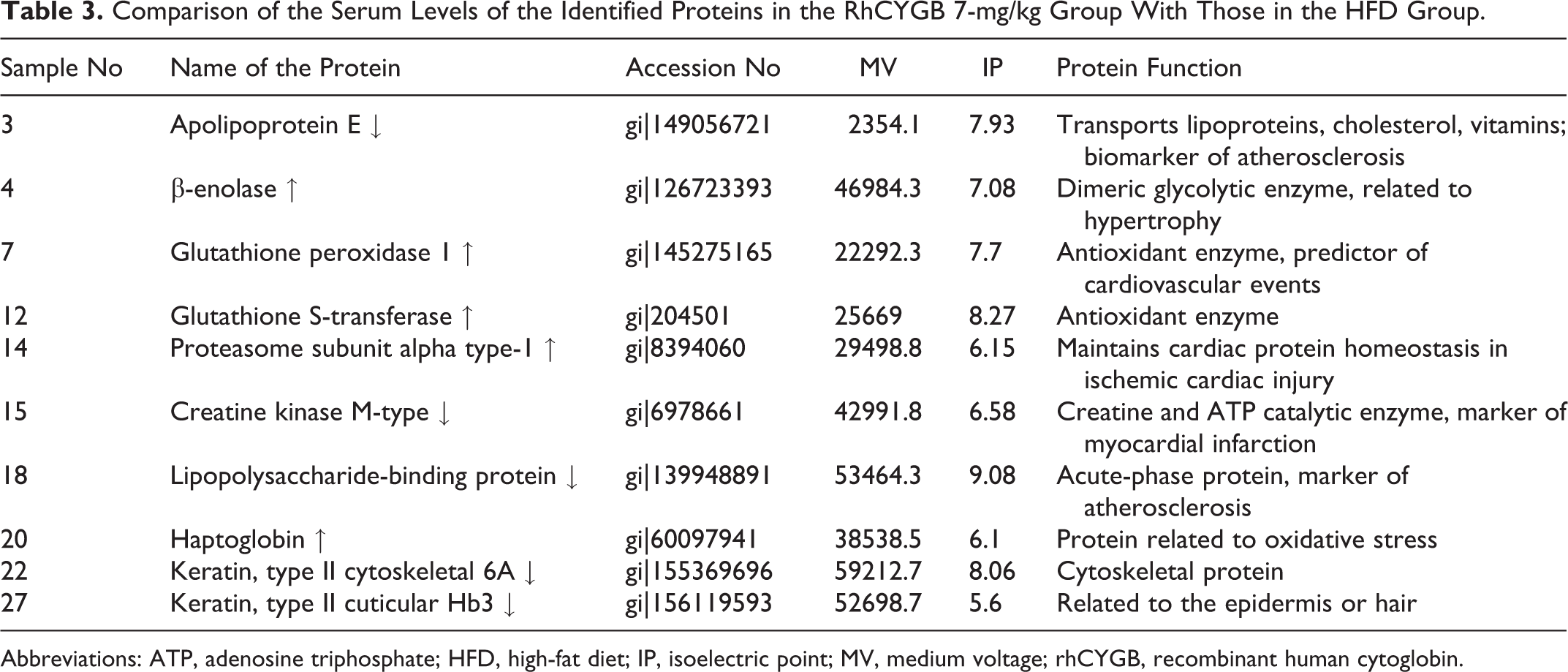
Abbreviations: ATP, adenosine triphosphate; HFD, high-fat diet; IP, isoelectric point; MV, medium voltage; rhCYGB, recombinant human cytoglobin.

Relative gene expression levels of the blood proteins evaluated via 2-DE analysis. QRT-PCR was used to analyze ApoE, SOD, GST, GPx, and LBP expression in the serum of rats with HFD-induced atherosclerosis (n = 8). Means ± SEM. *P < .05, **P < .01. 2-DE indicates two-dimensional electrophoresis; ApoE, apolipoprotein E; SOD, superoxide dismutase; Gpx, glutathione peroxidase; GST, glutathione S-transferase; LBP, lipopolysaccharide-binding protein.
rhCYGB Suppressed Lipid Uptake and Enhanced Lipid Redox Reactions in HepG2 Cells
The in vivo studies demonstrated the hypolipidemic activity and antiatherogenic effects of rhCYGB. To study the effects of rhCYGB on lipid intake and redox reaction further, we used the HepG2 cell line as an in vitro model of lipid metabolism. As shown in Figure 4A, we analyzed the content of the lipid droplets stained by Oil Red O after incubating the cells with rhCYGB for 24 hours in DMEM containing 1% FBS. The administration of 15 μg/mL rhCYGB significantly reduced lipid droplet accumulation in the CYGB group compared to the OA group. In addition, intracellular TG and MDA concentrations were also significantly reduced in the rhCYGB group compared to the OA group (Figure 4A). Furthermore, 5, 10, and 15 μg/mL rhCYGB improved SOD and GPx activity in the CYGB groups compared to the OA group (SOD activity in the OA group 21.83 ± 1.024 vs SOD activity in the 15 μg/mL CYGB group 56.52 ± 0.5436, GPx activity in the OA group 22.93 ± 0.7464 vs GPx activity in the 15 μg/mL CYGB group 71.68 ± 1.03; Figure 4B).

RhCYGB suppressed lipid uptake and enhanced lipid redox reactions in HepG2 cells. A, Representative photographs of HepG2 cells stained with Oil Red O (left) and lipid levels in HepG2 cells (right; n = 6). Scale bars: 100 µm. B, Measurement of TG and MDA concentrations and SOD and GPx activity in HepG2 cells. The results are from 3 independent experiments performed in triplicate. *P < .05, **P < .01. RhCYGB indicates recombinant human cytoglobin; TG, triglycerides; MDA, malondialdehyde; SOD, superoxide dismutase; Gpx, glutathione peroxidase.
RhCYGB Attenuated Oxidative Stress-Induced Injury in FCs
To elucidate the mechanism underlying the antioxidant effects of rhCYGB on atherosclerosis, we examined THP-1 macrophage-derived FCs. These cells can serve as a reasonable cell model mimicking the in situ changes experienced by macrophages in atherosclerotic lesions. 27 The THP-1 macrophages can be triggered by β-very low-density lipoprotein (β-VLDL), ac-LDL, or ox-LDL endocytosis, which leads to CE accumulation within the cells and results in their having an FC-like appearance. 28 It has been reported that CEs are the main lipid component of these cells and that the CE–TC ratio is higher than 50% in FCs in atherosclerotic lesions. 29 As shown in Figure 5A, after incubating with rhCYGB (5, 10, and 15 μg/mL) and simvastatin (1 μg/mL) for 24 hours, FCs in the indicated groups displayed both reduced CE levels and reduced TC levels compared to those in the ox-LDL group. Specifically, the CE–TC ratio decreased to 38.04% and 39.44% in the CYGB (10 μg/mL) and simvastatin groups, respectively, compared to the 50 μg/mL ox-LDL group (P < .01). The data showed that the levels of MDA, a product of lipid peroxidation in FCs, decreased from 89.37 ± 1.594 nmol/mgpro in the ox-LDL group to 44.97 ± 1.377 nmol/mgpro in the CYGB group (15 μg/mL; P < .01). In contrast, SOD and GPx activity levels increased significantly after rhCYGB treatment in FCs in the CYGB group compared to those in the ox-LDL group. Accordingly, NADPH oxidase activity decreased in FCs in the CYGB group compared to those in the ox-LDL group (15 μg/mL; ox-LDL group, 0.505 ± 0.011 U/mgpro vs 15 μg/mL CYGB group, 0.135 ± 0.076 U/mgpro; P < .01; Figure 5B). These findings were confirmed by experiments utilizing an ROS fluorescent probe. These experiments showed that ROS content in the 15 μg/mL CYGB group was significantly reduced compared to that in the ox-LDL group (the fluorescence value was 157.6 ± 3.14 in the ox-LDL group vs 93.86 ± 1.52 in the 15-μg/mL CYGB group; P < .01; Figure 6B). The NO content displayed a similar trend in the corresponding groups (Figure 6A).

RhCYGB decreased lipid accumulation and lipid peroxidation in FCs. A, CE and TC contents in THP-1 cells were quantified by gas chromatography (left). The CE/TC ratio is shown (right; n = 6). The results are from 3 independent experiments performed in triplicate. B, Similar to Figure 4B, MDA content and SOD, GPx and NADPH oxidase activity levels in FCs. *P < .05, **P < .01. RhCYGB indicates recombinant human cytoglobin; FC, foam cell; TC, total cholesterol; CE, cholesteryl ester; MDA, malondialdehyde; SOD, superoxide dismutase; Gpx, glutathione peroxidase; NADPH, nicotinamide adenine dinucleotide phosphate.
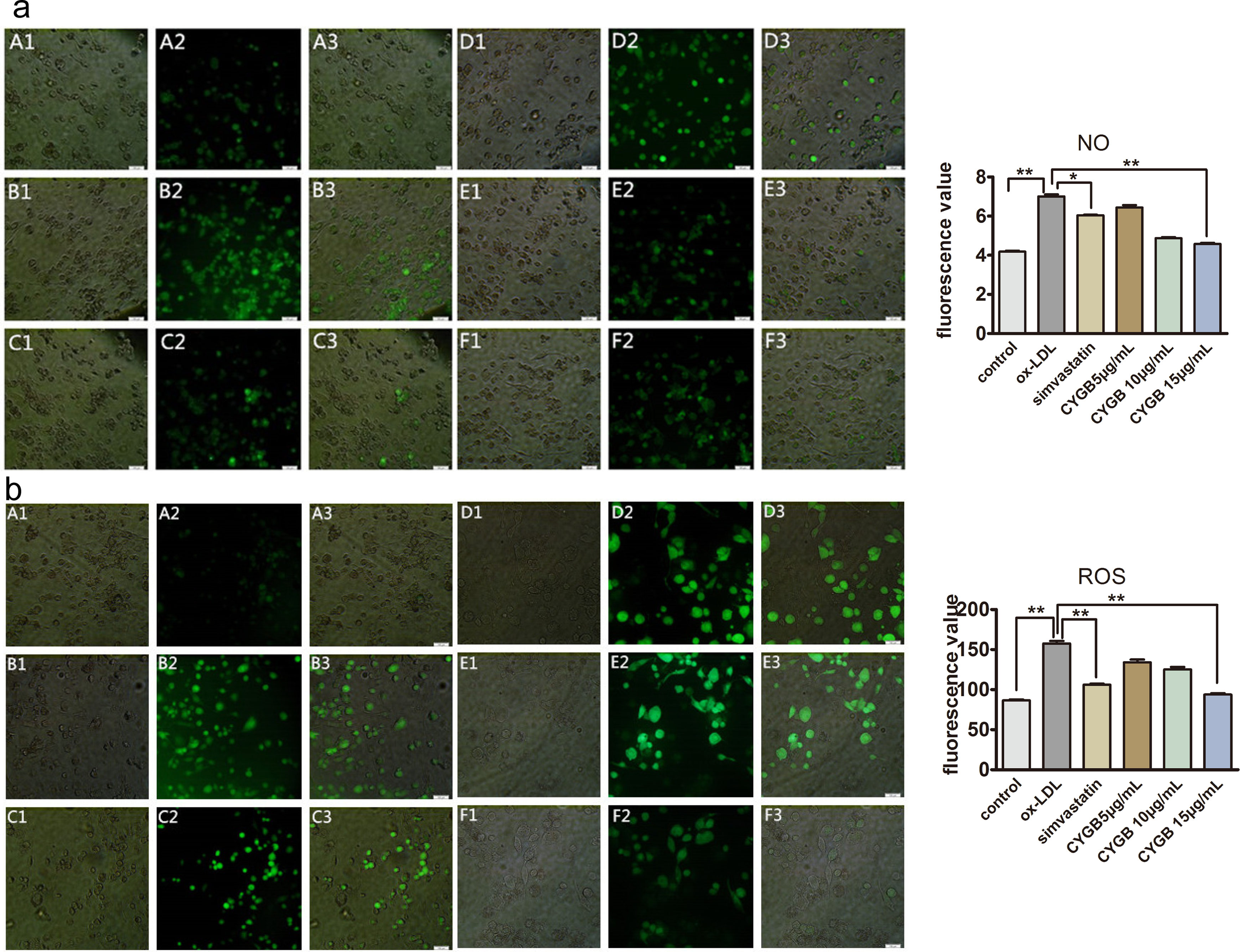
RhCYGB decreased NO and ROS levels in FCs. A, Representative images of NO fluorescent probes in FCs (left). Fluorescence values are shown (right; n = 6). B, Similar to (A), images of ROS fluorescent probes in FCs are shown (left), and fluorescence values were determined (right). Scale bars: 100 µm, *P < .05, **P < .01, ***P < .001. RhCYGB indicates recombinant human cytoglobin; NO, nitric oxide; FC, foam cell.
Conclusions
Many experimental studies have reported that CYGB has various biological activities. The CYGB scavenges toxic species, 11 protects cells from oxidative stress and oxidative DNA damage, 30 and exerts tumor suppressive effects under normal conditions. 12 Many studies have demonstrated that abnormal lipid metabolism and lipid peroxidation-induced oxidative stress are important factors in the occurrence and development of atherosclerosis. 31 Hence, the aim of the present study was to assess the antiatherosclerotic effects and antioxidant capacity of rhCYGB.
In this study, we used an early atherosclerosis rat model induced by an HFD and vitamin D3 injections. 32 The HE staining of the abdominal aorta showed that FCs had accumulated in the intima and that remodeling of the media had occurred after 30 days of HFD administration and vitamin D3 injection, indicating that the atherosclerosis rat model had been successfully established (Figure 2A). Evidence indicates that the liver is a center of lipid metabolism and that lipid and apolipoprotein disorders are associated with cardiovascular diseases. Our results demonstrated that rhCYGB decreased TC, TG, and LDL-C levels and increased HDL-C levels in blood. The rhCYGB also decreased the numbers of fat droplets in hepatocytes in atherosclerotic rats (Figure 1A, B), indicating that rhCYGB may be helpful for treating lipid metabolism disorders and, consequently, decreasing the risk of cardiovascular diseases. Interestingly, the weight of the HFD group was lighter than that of the other groups, but the weight of the rhCYGB-treated group was closer to that of the control group than those of the other groups (Table 2). We hypothesized that excess lipid accumulation in the rat liver in the HFD group influenced the normal metabolism of the liver and eventually caused digestive system dysfunction. The rhCYGB treatment modulated lipid metabolism, which resulted in the weight returning to a normal level in the corresponding group, indicating that rhCYGB had hepatoprotective effects, findings consistent with those of previous studies. 33
Evidence indicates that vascular remodeling is the main pathological change underlying the development of atherosclerosis. 34 Intimal–medial membrane thickening and blood vessel narrowing are caused by FC formation and vascular smooth muscle cell proliferation. Macrophages became trapped in the intima and transform into FCs after taking up ox-LDLC during atherogenesis in the HFD group. However, rhCYGB treatment attenuated FC accumulation and decreased intimal thickness (Figure 2). The CYGB can interact with lipids and convert them into various oxidized products by transforming from a hexacoordinate heme to a pentacoordinate heme under oxidative stress conditions 14 ; thus, CYGB can decrease lipid accumulation in FCs and enhance peroxidative activity to effectively prevent the development of atherosclerosis.
Previous studies have demonstrated the antioxidative and cellular protective capacity of rhCYGB. 30,35 After analyzing the effects of rhCYGB on the serum proteome in the HFD-induced atherosclerosis rat model, we determined that 10 proteins were differentially expressed among the 3 groups via comparative proteomics analysis. The gene ontology enrichment analysis showed that these proteins were involved in oxidative stress pathways and cardiovascular diseases. For example, ApoE is involved in lipid transport and cardiovascular disease. 36 The overexpression and accumulation of ApoE appear to cause hypertriglyceridemia by stimulating very LDL (v-LDL) triglyceride production and impairing v-LDL lipolysis. 37 Excessive amounts of plasma ApoE induce proatherogenic processes by mediating the retention of lipoproteins that contribute to FC formation in the vascular wall. 29 Our data suggest that rhCYGB attenuates atherosclerosis by keeping ApoE at a normal concentration. We will examine polymorphisms in the ApoE gene or use the ApoE-deficient mouse to further study the effect of rhCYGB on atherosclerosis in the future. The LBP is an acute-phase hepatic protein but is also synthesized in other cells. The LBP is a widely used atherosclerosis marker, and serum LBP levels are associated with carotid intima–media thickness. 38 Moreover, LBP participates in innate immune mechanisms in atherosclerosis. 39 β-enolase, PSMA1, and CKM are linked to ischemic injury, myocardial infarction, cardiac hypertrophy, and atherosclerosis, 40,41 and GPx-1 plays a vital role in antioxidant defense in the vascular wall. 42 The risk of cardiovascular events is inversely associated with increases GPx-1 activity, 43,44 suggesting that GPx-1 may be an important antiatherogenic therapeutic target in patients at risk of diabetic macrovascular disease. 45 The GSTs, phase II enzymes, are intimately involved in combating oxidative stress and detoxifying xenobiotics. 46 Haptoglobin is also an antioxidant protein that plays a key role in preventing free radical- and free hemoglobin-induced oxidative stress. 47
HepG2 cells were treated with OA (0.3 mmol/L) to simulate hepatocyte lipid uptake and accumulation in vitro. Large amounts of extracellular lipids entered the cells, resulting in the development of a metabolic disorder in the hepatocytes in the HFD group; however, rhCYGB reverse-transported the lipids, decreased intracellular lipid levels and enhanced SOD and GPx activity. It has been reported that CYGB can bind to oleate and transform from a hexacoordinate heme to a pentacoordinate heme. Lipid-induced CYGB transformation is a unique process that does not occur in similar hexacoordinate hemoglobins. Lipid-derived CYGB transformation may participate in cell signaling pathways and enhance the peroxidatic response to oxidative stress environments. 14
The CYGB has been reported to be involved in oxygen transport, storage, and sensing 48 as well as the cellular responses to oxidative stress and hypoxia. 30,49,50 The THP-1 cells were used to study the mechanism by which rhCYGB protects macrophages from oxidative stress in vitro. The THP-1 cells were converted into the FCs by ox-LDL, a well-known marker of lipoprotein-related oxidative stress in the arterial wall. 51 The rhCYGB reduced the production of MDA and NADPH oxidase (biomarkers of lipid peroxidation) and increased that of GPx and SOD in FCs. Similarly, rhCYGB decreased NO content and ROS in FCs, thereby protecting them against oxidative stress. The CYGB is a ubiquitously expressed hemoprotein that reversibly binds O2, NO, and CO. 52,53 Studies have shown that CYGB can function as an NO dioxygenase at the molecular level in rat hepatocytes. 54 The NO has been shown to protect against hydrogen peroxide-induced cell death at low concentrations and to enhance ROS toxicity at higher concentrations. 55 Large amounts of NO can be produced by chemically or immunologically activated macrophages, and this NO is oxidized to nitrates or nitrites within a few seconds its production. 56 Inflammatory NO is usually generated along with excessive O2−, which can form peroxynitrite (ONOO−), a compound that regulates the following cytotoxic and physiologic processes: LDL oxidation, DNA damage, and mitochondrial respiration. 57,58 Our results indicate that rhCYGB may participate in cell-mediated NO consumption and have NO dioxygenase-like effects, thereby protecting THP-1 cells from NO-induced oxidative injury.
Collectively, atherosclerosis has caused patients to suffer from multiorgan damage and has seriously impacted patient quality of life. Established lipid-lowering drugs (statins and fibrates) are helpful in the treatment of atherosclerosis; however, these drugs have many side effects in affected patients. 59 The animal and cell experimental results presented herein demonstrate that rhCYGB prevents atherosclerosis development by regulating lipid metabolism and protecting cells from oxidative stress-induced injury. This study not only confirmed that rhCYGB plays critical roles in atherosclerosis and lipid metabolism but also showed that rhCYGB may be useful as a therapy for atherosclerosis and lipid disorders.
Supplementary Material
Supplemental Material for Recombinant Human Cytoglobin Prevents Atherosclerosis by Regulating Lipid Metabolism and Oxidative Stress
Supplemental material for Recombinant Human Cytoglobin Prevents Atherosclerosis by Regulating Lipid Metabolism and Oxidative Stress by Lingling Ou, Xin Li, Baihong Chen, Zhenhuang Ge, Junyi Zhang, Ye Zhang, Gaotai Cai, Zhen Li, Ping Wang, Wenqi Dong in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
This research was supported by the School of Laboratory Medicine and Biotechnology of Southern Medical University. We appreciate the assistance provided by Dr Zhenhua Chen.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangdong Provincial Medical Research Foundation (No A2016360), the Guangdong Province Science and Technology Plan Project (No 2013A022100027), and the Guangzhou Science and Technology Project (No 201510010104).
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
