Abstract
Basilar vascular smooth muscle cells (BASMCs) hyperplasia is a prominent feature of cerebrovascular remodeling and stroke during the development of hypertension. Tanshinone IIA (Tan) has been reported to exhibit a protective effect against the pathological features of hypertension. Previous studies have shown that phosphoinostitide-3 kinase (PI3K)/3′-phosphoinostitide dependent kinase (PDK1)/AKT pathway is involved in the regulation of proliferation of various cell types. Therefore, there may be a crosstalk between Tan antihypertension processes and PI3K/PDK1/AKT proliferative effect in BASMCs. To test this hypothesis, we used a 2-kidney, 2-clip hypertension model to examine the effect of Tan on PI3K/PDK1/AKT pathway by cellular, molecular, and biochemical approaches. Our results revealed that the abundance of PDK1 in plasma was paralleled with an increase in blood pressure and the cross-sectional area of basilar artery in hypertensive rats. Tan decreased blood pressure and hypertension-induced PDK1 phosphorylation but produced no effect on the phosphorylation of PI3K. Moreover, Tan attenuated endothelin 1 induced the activation of PDK1/AKT pathway in rat BASMCs. Tan could inhibit cell cycle transition by regulating the expression of cyclin D1 and p27, in turn, prevent proliferation of BASMCs. Our study provides a novel mechanism by which Tan prevents cerebrovascular cell proliferation during hypertension, and thus Tan may be a potential therapeutic agent for cerebrovascular remodeling and stroke.
Introduction
Basilar vascular smooth muscle cells (BASMCs) hyperplasia initially causes an increase in the cross-sectional area of the cerebrovascular wall. 1 Increasing evidence suggested that endothelin 1 (ET-1) plays a critical role in proliferation of BASMCs in the pathogenesis of hypertension (htn). 2
Tanshinone IIA (Tan) is an active ingredient extracted from the rhizome of the Salvia miltiorrhiza (Danshen), and sodium tanshinone IIA sulfonate is a water-soluble derivative of Tan after sulfonation (Supplemental Figure S1). Previous studies revealed that Tan has been used to prevent and treat cardiovascular diseases. 3 Its beneficial effects are mainly focused on vasodilatory, 4,5 antithrombotic, 6 anti-inflammatory, 7 free radical scavenging, 8 and mitochondria-protective effects. 9 Kim and colleagues found that Tan induced vasorelaxation and subsequently reduced blood pressure in htn rats, mainly through increasing expression of endothelial nitric oxide synthase. 10 It was also reported that Tan modulated pulmonary vasoreactivity through the inhibition of Ca2+ influx and release in hypoxic pulmonary htn rats. 5 Moreover, Wang et al found that Tan prevented cardiac remodeling by reducing the production of reactive oxygen species in 2-kidney and 2-clip (2k2c) hypertensive rats. 11 These findings have demonstrated the beneficial effects of Tan therapy in htn. However, the effect of Tan on cerebrovascular remodeling in the development of htn remains to be elucidated.
The phosphoinostitide-3 kinase (PI3K)/3′-phosphoinostitide dependent kinase (PDK1)/AKT pathway is involved in the regulation of multiple cellular processes including proliferation, apoptosis, differentiation, and transcription. 12 Once activated by the upstream signal molecules such as epidermal growth factor receptors, Ras, Src, and insulin receptor (IR), PI3K phosphorylates phosphatidylinositol-(3,4)-bisphosphate to generate phosphatidylinositol-(3,4,5)-trisphosphate (PIP3). This alteration results in recruitment of PDK1 and AKT to the membrane via interacting with their pleckstrin homology (PH) domains 13 and consequently triggers a cascade of cellular events. 14 Activation of PI3K/ PDK1/AKT promotes proliferation of smooth muscle cells, vascular remodeling, and the development of various vascular pathologies such as htn. 15
In the present study, we investigated the activation of PDK1 in the 2k2c htn model. Moreover, we examined the protective action of Tan on proliferation of BASMCs and the relationship between Tan-prevented BASMCs proliferation and PI3K/PDK1/AKT pathway.
Material and Method
Materials and Reagents
Dulbecco modified essential medium (DMEM)/F-12, Opti-MEMI medium, fetal calf serum (FBS), penicillin, streptomycin, and Lipofectamine2000 reagent were obtained from Invitrogen (Carlsbad, California). BX912 and LY294002 were purchased from Santa Cruz Biotechnology (Santa Cruz, California). Endothelin 1 enzyme-linked immunosorbent assay (ELISA) Kit was purchased from Abcam (Cambridge, Massachusetts). 3-Phosphoinositide dependent protein kinase 1 ELISA Kit was purchased from Global Biotech Co, Ltd (Shanghai, China).
Tanshinone IIA (sodium sulphonate, 99.0% purity assayed by high-performance liquid chromatography ) was purchased from Yunnan Plant Pharmaceutical Factory (Yunnan, China) and was prepared as 20 mmol/L stock solution in distilled water.
Male Sprague-Dawley rats were supplied by the Experimental Animal Central of the Second Military Medical University, Shanghai, China. All animal experiments were approved by the Committee on the Ethics of Animal Experiments of Shanghai Seventh People’s Hospital.
Induction of htn and Drug Administration
The 2k2c method operation was used to induce htn in male Sprague–Dawley rats (80-120g) as previously described. 16 In brief, under anesthesia with ketamine (100 mg/kg, intraperitoneally [ip]) and xylazin (5 mg/kg, ip), a median longitudinal incision on the abdominal skin was performed. The left and right renal arteries were occluded by placing silver clips with the internal diameter of 0.3 mm. The sham-operated rats were received the same procedure without the placement of renal artery clips and used as the sham group. In this model, 10 groups of animals were examined: sham and htn (0, 1, 4, 8, and 12 weeks). In experiments with the Tan-treated htn model, approximately 10 weeks later, only those rats with blood pressure ≥140 mm Hg and without stroke symptoms were selected for Tan treatment. The following 5 groups of animals were examined: sham, htn, htn + .05 mg/kg Tan, htn + 0.5 mg/kg Tan, and htn + 5 mg/kg Tan. Tan was administered via oral gavage once daily for 2 weeks at dose of 0.05, 0.5, or 5 mg/kg body weight. Systolic blood pressure was measured in conscious rats by tail-cuff plethysmography (BP-98A, Softron Co, Tokyo) as described previously. 17,18
Enzyme-Linked Immunosorbent Assay
The concentrations of ET-1 and PDK-1 in plasma were determined using ET-1 ELISA kit and PDK1 ELISA kit, respectively. All measurements were performed as recommend by the manufacturer’s protocol.
Cell Culture
Basilar artery smooth muscle cells (BASMCs) were isolated and cultured as described previously. 18 In brief, male Sprague-Dawley rats (80-120 g) were anaesthetized and decapitated. The cerebral basilar arteries were dissected rapidly and immersed in Kreb’s solution containing (in mmol/L, NaCl 137, KCl 5.4, CaCl2 2.0, MgCl2 1.1, NaH2PO4 0.4, glucose 5.6, and NaHCO3 11.9). After connective tissue and fat were carefully eliminated, the basilar arteries were cut into 1-mm strips and then were incubated in DMEM/F-12 supplemented with 10% FBS, 100 U/mL penicillin, and 100 U/mL streptomycin at 37°C in 5% CO2 atmosphere. The BASMCs were then migrated from the pieces about 8 to 10 days and identified by positive immunofluorescent staining using antibody against α-smooth muscle actin (Supplemental Figure S2). When reaching 70% confluence, cells were passaged with 0.25% trypsin with 0.02% EDTA. In this study, BASMCs were used at passages 8 to 14 and growth was arrested by replacing the medium with 0.2% FBS/DMEM/F-12 for 72 hours before corresponding treatments.
Western Blot Analysis
Basilar artery homogenates or cell extracts were subjected to Western blot analysis as described previously. 19 In briefly, basilar arteries or BASMCs were washed with cold phosphate-buffered saline (PBS) 3 times and then lysed in radioimmunoprecipitation assay buffer (Beyotime, Jiangsu, China) containing protease and phosphatase inhibitor cocktail (Merck, Darmstadt, Germany). After determining protein content by Bradford assay (Bio-Rad Laboratories, Hercules, California), equal proteins were separated by 8% to 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis and then transferred to a nitrocellulose membrane (Millipore, Billerica, Massachusetts). After blocking, the membranes were incubated with the following primary antibodies at 4°C overnight: p-PDK1 (Ser241), p-PI3Kp85 (Tyr458), PI3Kp85 and PI3Kp110α (diluted 1:500), PDK1, p-AKT (Thr308), and AKT (diluted 1:1000; Cell Signaling Technology, Danver, Massachusetts); cyclin D1, p27, Na+/K+ ATPase, and GAPDH (diluted 1:1000, Santa Cruz Biotechnology). After incubating with secondary antibodies including horseradish peroxidase -conjugated anti-rabbit, anti-goat, or anti-mouse (diluted 1:1000; Cell Signaling Technology) for 1 hour, detection was performed by a chemiluminescence system (Cell Signaling Technology) and image quantification was performed using ImageJ software (National Institutes of Health, Bethesda, Maryland).
Vector Construction and Transfection
The full-length PDK1 vector was obtained from Addgene (#20565, Cambridge, Massachusetts). The truncated PH domain of PDK1 mutant (ΔPH) was constructed by mutagenesis from the full-length PDK1 with QuikChange Lightning Site-Directed Mutagenesis Kit (Stratagene, California) and confirmed by sequencing. Vectors were transfected transiently with Lipofectamine2000 reagent in serum free-Opti-MEMI medium according to the manufacturer’s instructions.
Cell Cycle Analysis
After appropriate treatments, BASMCs were harvested by centrifugation and processed for cell cycle analysis as described previously. 18 DNA content was analyzed by flow cytometry (FACScalibur, Becton Dickinson, San Jose, California). The percentages of cells in different phases of the cell cycle were quantified.
Proliferation Assay
Cell proliferation was evaluated by cell counting and bromodeoxyuridine (BrdU) incorporation as described previously. 18 Cell number was examined using Cell Counting Kit-8 (Dojindo Molecular Technologies, Rockville, Maryland) according to the manufacture’s instruction. Incorporation of BrdU was measured according to the manufacturer’s instructions (BrdU kits from Calbiochem, San Diego, California). Absorbance was read at 450 to 540 nm using a microplate reader (Multiskan Spectrum, Thermo Scientific, Pittsburgh, Pennsylvania).
Cell Fractionation
Cytoplasmic and membrane fractions were separated by the Qproteome Cell Compartment Kit (Qiagen, Germany) according to the manufacturer’s protocol and were analyzed by Western blot.
Cell Treatment
The BASMCs were coincubated with ET-1 (10 nmol/L) and different concentrations of Tan (0.625, 1.25, 2.5, 5, 10, and 20 μmol/L), ET-1 and Tan (5 μM), ET-1 and LY294002 (LY, 10 μmol/L), or ET-1 and BX912 (BX, 5μmol/L) for indicated times, then cells were subjected to proliferation and Western blot assays.
In the experiments with PDK1 and ΔPH vectors, BASMCs were transfected with vectors for 48 hours prior to the coincubation with ET-1 and Tan or ET-1 and BX for indicated times. After treatment, BASMCs were used for Western blot, cell proliferation, and cell cycle analysis as described previously.
Statistical Analysis
All data were present as mean ± standard error of the mean. The n value represented the number of independent experiments on different batches of cells or different rats. The regression analysis was determined by the Pearson correlation test. 1-way analysis of variance followed by the Bonferroni multiple comparison post hoc test with a 95% confidence interval was employed in SPSS 16.0 system (SPSS Inc, Chicago, Illinois). A P value <.05 was considered to be statistically significant.
Results
Concentrations of ET-1 and PDK1 Were Elevated in Plasma in Hypertensive Rats
The body weight and blood pressure of sham and hypertensive rats were measured and compared weekly. Body weight gain exhibited a similar pattern between groups (Supplemental Figure S3A). However, at 1, 4, 8, and 12 weeks postoperatively, systolic blood pressure in the 2k2c group was significantly higher than those in the sham group (n = 40 in each group; P < .05, P < .01 vs corresponding sham group; Figure 1A). Consistent with a previous study, 19 the ET-1 concentration in plasma was gradually increased from 1 week after 2k2c surgery and paralleled with an increase in blood pressure in hypertensive rats (Figure 1B and C).

Concentrations of ET-1 and PDK1 in plasma were increased gradually during the development of hypertension. A, Systolic blood pressure (SBP) of 2k2c hypertensive (htn) and the corresponding sham rats were measured at 1, 4, 8, and 12weeks after operation (mm Hg) by tail-cuff plethysmography. *P < .05, **P < .01 vs corresponding sham groups, n = 40 in each group. B, ELISA assay showed that the ET-1 concentration was elevated in 2k2c hypertensive rats from 1 week after operation. C, Correlation between ET-1 concentration and blood pressure. D and E, The expression of p-PDK1 and PDK1 in basilar arteries isolated form sham and hypertensive rats was detected by Western blot using GAPDH as an internal control. F, The concentration of PDK1 in plasma was upregulated in hypertensive rats. G, H, and I, The PDK1 concentration was positively correlated with blood pressure, the ET-1 concentration, and the cross-sectional area of basilar arteries, respectively. *P < .05, **P < .01 vs corresponding sham groups, n = 6 in each group. ET-1 indicates endothelin 1; PDK, 3′-phosphoinostitide dependent kinase; ELISA, enzyme-linked immunosorbent assay; GADPH, glyceraldehyde 3-phosphate dehydrogenase.
We next determined the activation of PDK1 in hypertensive rats. Our results showed that PDK1 phosphorylation in basilar arteries did not change in the sham-operated rats during the time course (Figure 1D). However, PDK1 phosphorylation was remarkably increased in basilar arteries of hypertensive rats. At 1, 4, 8, and 12 weeks postoperatively, p-PDK1/PDK1 ratio was elevated to 1.59 ± 0.19, 2.38 ± 0.31, 3.36 ± 0.59, and 4.37 ± 0.41, respectively (Figure 1E). This result was further supported by the PDK1 concentration in plasma (Figure 1F). Of interest, the PDK1 concentration was positively correlated with blood pressure and the ET-1 concentration, respectively (Figure 1G and H). Notably, we found that the abundance of PDK1 in plasma was positively correlated with the cross-sectional area with the correlation coefficient of 0.7059 (Figure 1I). These results suggested that the activation of PDK1 is involved in vascular remodeling during htn.
Tan Suppressed htn/ET-1 Induced the Activation of PDK1
Compared with htn group, administration of Tan by gavage at dose of 0.5 or 5 mg·kg−1·d−1 for 2 weeks reduced systolic blood pressure from 172.65 ± 15.92 mm Hg to 139.87 ± 12.52 mm Hg or 137.68 ± 13.69 mm Hg, respectively (P < .01, n = 10 in each group; Figure 2A). However, 0.05, 0.5, or 5 mg·kg−1·d−1 Tan treatment produced no effects on rat weight (Supplemental Figure S3B). We next examined the effect of Tan on ET-1-induced proliferation of BASMCs. Compared to control, ET-1 led to 1.32 ± 0.08-fold and 1.45 ± 0.11-fold (P < .01, n = 6) increments in cell number (Figure 2B) and BrdU incorporation (Figure 2C), respectively. Treatment of BASMCs with Tan (5 μmol/L) almost abolished the effect of ET-1 on cell proliferation. Furthermore, we found Tan produced no significant cytotoxicity in BASMCs at concentration less than 10 μmol/L. Therefore, the concentration of 5 μmol/L was used in the following experiments.

Tan suppressed the activation of PDK1 induced by hypertension/ET-1. A, Administration of Tan (0.5, 5 mg/kg) for 2 weeks reduced systolic blood pressure compared with 12-week hypertensive rats. B and C, BASMCs were stimulated with ET-1 (10 nmol/L) in the presence of different concentrations of Tan (0.625, 1.25, 2.5, 5, 10, and 20 μmol/L) for 48 hours, then cell proliferation was examined by CCK-8 kit and BrdU incorporation, respectively. D, PI3Kp85 phosphorylation remained unchanged in hypertensive rats after administration of Tan (0.05, 0.5, 5 mg/kg). E, BASMCs were coincubated with ET-1 and Tan (5 μmol/L) or ET-1 and LY294002 (LY, 10 μmol/L) for 1 hour, the phosphorylation level of PI3Kp85 was detected by Western blot. F and G, The expression of PI3Kp110α in basilar arteries and BASMCs was analyzed by Western blot, respectively. H, Tan attenuated hypertension-induced phosphorylation of PDK1. I, Tan or BX912 (BX, 5 μmol/L) was added to BASMCs in the presence of ET-1 for 1 hour. Western blot showed that Tan and BX reversed ET-1-induced PDK1 phosphorylation. Values are mean ± SEM from 6 to 10 rats of each group or 6 independent cell experiments. *P < .05, **P < .01 vs sham group or control group;
To understand the signal transduction pathway by which Tan regulated BASMCs proliferation, we examined PI3K/PDK1/AKT pathway, which has been documented to play a critical role in cell proliferation. Western blot showed that PI3Kp85 phosphorylation in basilar arteries was significantly increased at 12 weeks after operation. However, Tan produced no effect on htn-induced PI3Kp85 phosphorylation (Figure 2D). In addition, similar tendency was observed when BASMCs were treated with ET-1 in the presence or absence of Tan in vitro. LY294002, an inhibitor of PI3K, obviously inhibited ET-1-induced PI3Kp85 phosphorylation (Figure 2E). We found PI3Kp110α expression remained unchanged both in basilar arteries and in BASMCs after corresponding treatment (Figure 2F and G).
The PDK1 concentration was elevated in hypertensive rats and correlated positively with the ET-1 concentration. To investigate whether PDK1 was a molecular target for Tan in htn, we examined the effect of Tan on htn/ET-1-induced PDK1 phosphorylation. We found Tan significantly inhibited the upregulation of PDK1 phosphorylation in basilar arteries of hypertensive rats (Figure 2H). After administration of Tan at dose of 0.05, 0.5, and 5 mg·kg−1·d−1, p-PDK1/PDK1 ratio was decreased by 14.3%, 62.5%, and 26.1%, as compared to the htn group, respectively. The inhibition reached the maximal level at 0.5 mg·kg−1·d−1. Moreover, ET-1-evoked PDK1 phosphorylation in BASMCs was markedly suppressed by Tan or BX912 (an inhibitor of PDK1; Figure 2I), indicating Tan may target PDK1 as a signaling element.
Tan Inhibited ET-1-Induced Proliferation and G1/S Cell Cycle Transition Through PDK1/AKT Pathway
We used the full-length PDK1, the truncated PH domain of ΔPH, and a PDK1 inhibitor (BX912) to study whether PDK1 is involved in Tan-inhibited BASMCs proliferation. The CCK-8 assay revealed that BX912 significantly attenuated ET-1-induced BASMCs proliferation, similar to what was observed in the Tan-treated group. On the contrary, transfection of PDK1 or ΔPH restored Tan-inhibited proliferation of BASMCs (Figure 3A). Similar result was obtained in BrdU incorporation (Figure 3B). Furthermore, we found Tan, as well as BX912, significantly inhibited ET-1-induced AKT phosphorylation in BASMCs. However, the effect of Tan on AKT phosphorylation was almost abolished after transfection of PDK1 or ΔPH (Figure 3C).
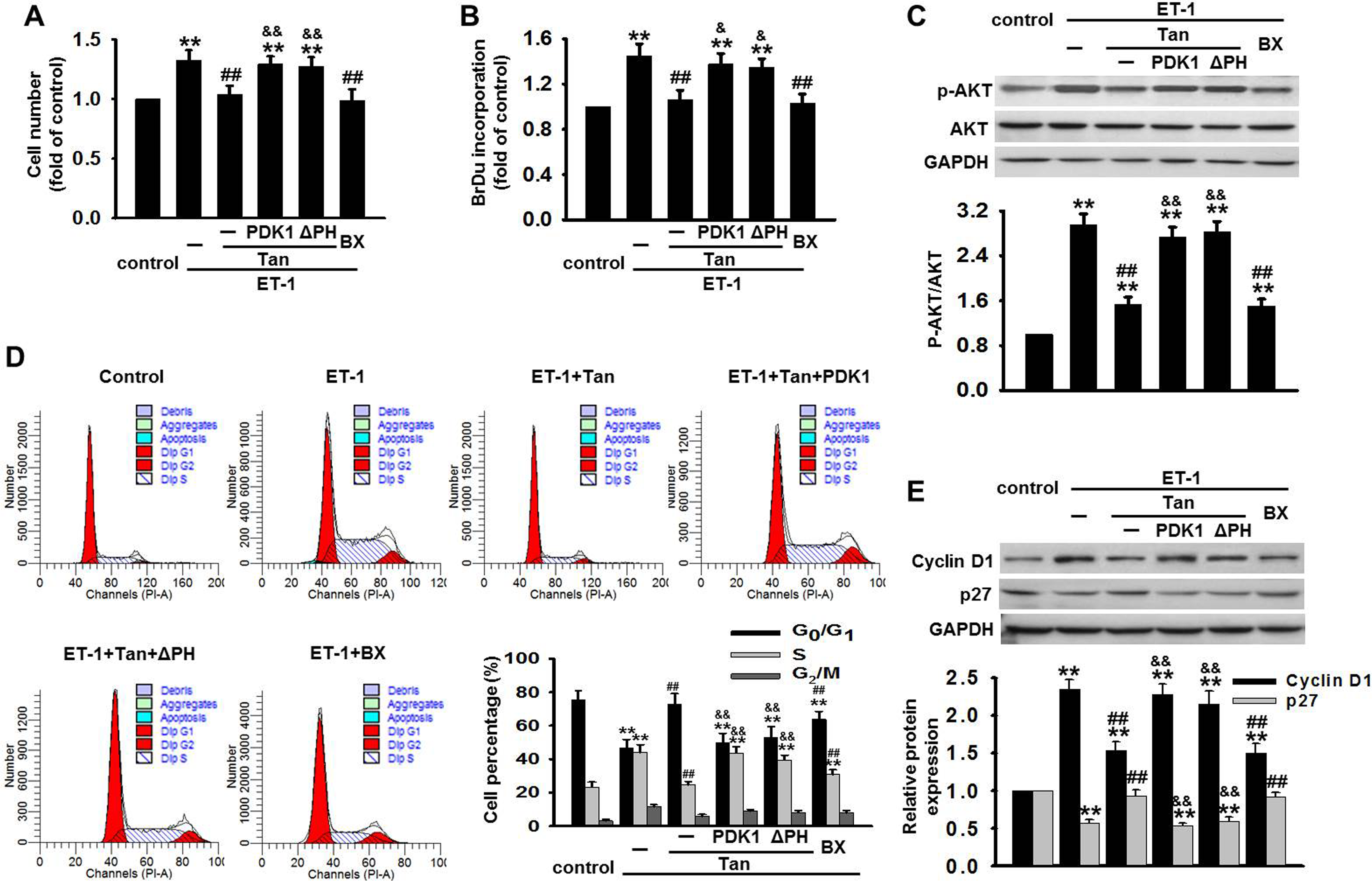
Tan inhibited ET-1-induced BASMCs proliferation and cell cycle transition. A and B, BASMCs were transfected with the full-length PDK1 or the truncated pleckstrin homology domain of PDK1 mutant (ΔPH) for 48 hours first, followed by coincubation with ET-1 and Tan, or ET-1 and BX for another 48 hours. Cell proliferation was tested by CCK-8 kit and BrdU incorporation, respectively. C, After transfection with PDK1 or ΔPH for 48 hours, BASMCs were coincubated with ET-1 and Tan, or ET-1 and BX for 1 hour. The phosphorylation level of AKT was determined by Western blot. D, BASMCs were treated as described in (A and B), cell cycle transition was examined by flow cytometry. E, After treatment of BASMCs described in (C), the expression of cyclin D1 and p27 was analyzed by Western blot, respectively. **P < .01 vs control group;
We next examined the effect of Tan on cell cycle progression by flow cytometry. As shown in Figure 3D, ET-1 resulted in a decrease from 75.57% to 46.46% in G0/G1 phase and an increase from 23.35% to 43.96% in S phase, as compared to control group, suggesting ET-1 promotes cell cycle progression and enhances cells’ entrance into S phase. Pretreatment with Tan and BX912 dramatically inhibited the effect of ET-1 on cell cycle with an increase in cell percentage in the G0/G1 phase and a decrease in cell population in the S phase. On the contrary, overexpressing PDK1 or ΔPH significantly attenuated Tan-suppressed cell cycle transition. To further investigate the molecular mechanism by which Tan arrested G1/S transition, we analyzed the expression of cyclin D1 and p27 in BASMCs. As shown in Figure 3E, ET-1 markedly increased cyclin D1 expression and decreased p27 expression. However, Tan and BX912 almost abolished the effect of ET-1 on cyclin D1 and p27 expression. Expectedly, the effect of Tan was attenuated when BASMCs were transfected with PDK1 or ΔPH.
Tan Impaired htn/ET-1-Induced Translocation of PDK1 and AKT
It is noteworthy that translocation of PDK1 from cytoplasm to membrane is an essential step in AKT activation. Therefore, we investigated the effect of Tan on the subcellular localization of PDK1 and AKT, respectively. Western blot showed that PDK1 and AKT were mainly distributed in cytoplasm in basilar arteries isolated from sham-operated rats. At 12 weeks after 2k2c operation, the translocation from cytoplasm to membrane of PDK1 and AKT was significantly enhanced, suggesting that the enrichment of PDK1 and AKT in membrane may be one of the contributors for the activation of PDK/AKT pathway during htn. However, we found administration of Tan (0.5, 5 mg/kg) remarkably impaired htn-induced translocation of PDK1 and AKT (Figure 4A). Moreover, similar results were obtained in vitro that Tan inhibited ET1-induced translocation of PDK1 and AKT in BASMCs (Figure 4B), indicating the impairment of translocation underlies, at least partially, the inhibitory effect of Tan on PDK1 and AKT activation.

Effects of Tan on the subcellular localization of PDK1 and AKT. A, The content of PDK1 and AKT in cytoplasm and membrane in basilar arteries was detected by Western blot. Na+/K+ ATPase was used an internal control of membrane. B, BASMCs were stimulated with ET-1 in the presence or absence of Tan for 48 hours. Western blot showed that Tan inhibited ET-1-induced translocation of PDK1 and AKT. Values are mean ± SEM from 6 to 10 rats of each group or 6 independent cell experiments. **P < .01 vs sham group or control group;
Discussion
It is well established that BASMCs hyperplasia contributes to vascular remodeling during htn. 19 ET-1 induces vascular remodeling in the development of chronic htn mainly through its proproliferative and profibrotic effects. 2 Numerous studies have demonstrated that the concentration of ET-1 and the expression of ETA receptor are elevated in many htn models such as 1k1c Goldblatt rats, angiotensin II-infused rat, and deoxycorticosterone acetate salt rats. 20 In agreement with previous studies, we found ET-1 concentration was gradually increased in 2k2c hypertensive rats during the time course.
Cerebrovascular remodeling is an important determinant of stroke with an increase of blood pressure. 21 Tan is one of the active components extracted from the rhizome of the Salvia miltiorrhiza (Danshen), which has been reported to be able to prevent htn traditionally in Asia. 22 Consistent with a previous study in 2k1c hypertensive hamsters, 10 we found Tan at dose of 0.5 or 5 mg·kg−1·d−1 could reduce blood pressure in 2k2c hypertensive rat. The concentrations of Tan in this study were selected according to previous publications 23,24 and has been proved to be safe for rats. Here, we further investigated the effect of Tan on cerebrovascular cell proliferation during htn. Our data showed Tan arrested cell cycle transition from G1 to S phase through regulating the expression of cyclin D1 and p27, and it in turn prevented BASMCs proliferation.
Accumulating evidences suggest that PI3K/PDK1/AKT pathway plays a critical role in cell proliferation, survival, and intracellular trafficking. 25 ET-1 could induce smooth muscle cells proliferation by activating PI3K, 26 resulting in PDK1 recruitment and phosphorylation in arterial htn. 27 Our study is the first to show that the concentration of PDK1 was paralleled with an increase in blood pressure, ET-1 concentration, and the cross-sectional area of basilar artery, indicating PDK1 may be involved in the pathogenesis of htn. PDK1 is a regulator protein kinase containing 2 major domains: a large catalytic domain in N-terminal and a PH domain in C-terminal. 28 Upon stimulation, PIP3 generated by PI3K binds directly to PH domains of PDK1 and AKT, driving a translocation from cytoplasm to membrane which enables AKT to be phosphorylated by PDK1 at Thr308. 13 We found Tan suppressed htn/ET-1 induced phosphorylation and translocation of PDK1 and AKT, but produced no effect on PI3K phosphorylation, indicating PI3K may be not a target for Tan in the regulation of BASMCs proliferation. The PH domain plays a vital role in the regulation of PDK1 localization through interacting with PIP3. 27,28 However, in this study, transfection of the truncated PH domain of ΔPH reversed the effects of Tan on proliferation, cell cycle transition, and AKT phosphorylation, similar to what was observed in BASMCs transfected with full-length PDK1. These data suggested PH domain may be not necessary for PDK1 activation. Our findings were similar with a prior study, 29 which showed that PDK-1 with a deleted PH domain still underwent insulin-dependent membrane association. In addition to PIP3-mediated localization of PDK1 in membrane, several proteins have been reported to serve as a kind of scaffold proteins that interact with PDK1 directly, including Freund-1, Grb14-IR, and Grb2. 27,29 For example, a proline-rich sequence of PDK1 interacted with a Src homology 3 domain of Grb2, resulting in the recruitment and activation of PDK1. These findings suggested that this kind of scaffold proteins rather than PH domain may be a molecular target for Tan in the regulation of PDK1 activation. Clearly, further experiments are needed to verify this notion.
Based on these findings, we propose a signaling mechanism explaining how Tan prevents BASMCs proliferation, as illustrated in Figure 5. PI3Kp85 was phosphorylated in response to ET-1, resulting in the recruitment and activation of PDK1 and AKT. This led to AKT (Thr 308) phosphorylation and cell proliferation, which induced cerebrovascular remodeling during htn. Our present study demonstrated that Tan acted downstream of PI3K to interact directly with PDK1 and suppressed the activation of PDK1 and AKT. It may thus represent a novel mechanism by which Tan inhibits cerebrovascular cell proliferation during htn.

A proposed model explaining how Tan prevents BASMCs proliferation during cerebrovascular remodeling. BASMCs indicates basilar vascular smooth muscle cells.
Footnotes
Acknowledgments
This work was supported by Academic Leader Training Program of Pudong Health Bureau of Shanghai (PWRd 2010-08).
Author Contributions
Zhi-Liang Yu and Jie-Ning Wang: Conception and design of the study: Xiao-Hua Wu, Hui-Jun Xie, and Ying Han: Substantial contribution to data acquisition. Yang-Tai Guan and Yong Qin: Analysis and interpretation of the data. Zhi-Liang Yu and Jian-Ming Jiang: Drafting of the manuscript. Zhi-Liang Yu: Critical revision of the manuscript for intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant support: Academic Leader Training Program of Pudong Health Bureau of Shanghai (PWRd 2010-08).
