Abstract
Objective:
The effects of zedoary guaiane-type sesquiterpenes (ZGS)-based eluting stent (ZES) in accelerating reendothelialization and inhibiting neointimal hyperplasia were examined in a porcine coronary artery model.
Methods:
The ZES was prepared by polymer-free 316L stainless metal stents. Sirolimus-eluting stents (SES) and bare metal stents (BMS) with identical platforms were used as controls. Stents with 15 mm in length and 2.0 to 3.5 mm in diameter were implanted in porcine coronary arteries. Scanning electron microscopy (SEM) and histopathology were performed to assess the reendothelialization and neointimal hyperplasia. The 3-(4, 5-dimethylthiazol-2yl)-2, 5-diphenyl-2H-tetrazoliumbromide assay and flow cytometry were used to assess the influence of ZGS on human umbilical vascular endothelial cells (HUVECs).
Results:
At 7 days, SEM showed that percentage of endothelial coverage area was 94.04% ± 5.01% for ZES, 47.59% ± 19.91% for SES (P < .01 for ZES vs SES), and 59.58% ± 19.61% for BMS (P < .05 for ZES vs BMS). At 28 days, the percentage of coverage area was 98.51% ± 1.86% for ZES, 86.18% ± 8.16% for SES (P < .05 for ZES vs SES), and 94.26% ± 5.58% for BMS. Neointimal area and stenosis were significantly lower in ZES (1.07 ± 0.48 mm2, 27.66% ± 12.20%) compared to BMS (1.73 ± 0.69 mm2, 44.08% ± 15.03%, both P < .01, respectively), with no difference in SES (0.94 ± 0.12 mm2, 28.87% ± 6.00%, both P > .05, respectively). The ZGS also promoted HUVECs viability and improved HUVECs proliferation compared to sirolimus.
Conclusion:
The ZES accelerated reendothelialization and suppressed neointimal hyperplasia in a porcine coronary artery model, with beneficial effects on HUVECs.
Introduction
Drug-eluting stents (DES) have effectively suppressed neointimal hyperplasia in patients undergoing percutaneous coronary interventions. 1 However, concern about DES-induced late stent thrombosis has raised. 2 Pathological studies have implicated incomplete reendothelialization induced by DES as a potential mechanism for late thrombotic events. 2,3 The antiproliferative drugs (sirolimus and paclitaxel)-associated antihealing effect plays a crucial role in deendothelialization. 4 Therefore, looking for a kind of drugs to facilitate endothelial healing along with inhibiting neointimal hyperplasia is encouraged in next generation stents.
Curcuma zedoaria, a Chinese medicinal herb, has been used to treat cancers for more than 100 years, including gastric cancer, 5 hepatoma, 6 and uterine cervical cancer, 7 while it has low toxicity on normal cells, such as human umbilical vein endothelial cells (HUVECs), 8 gastric epithelial cells, 9 and fibroblasts of human mucus lineage. 10 Moreover, Curcuma zedoaria has been demonstrated to prevent inflammation, 11,12 oxidant, 13 and hepatic injury activities. 14 Zedoary guaiane-type sesquiterpenoids (ZGS) are bioactive constituents of Curcuma zedoaria. Our previous study has demonstrated their effects on inhibiting neointimal hyperplasia in a porcine model. 15 However, the influence of ZGS on early reendothelialization in vivo and in vitro remains unclear. Therefore, the effects of ZGS on endothelial healing were examined in a porcine coronary artery model and detected by HUVECs.
Methods
Stent Preparation
The stent platforms used were polymer-free 316L stainless metal with in situ nano/micropores on the surface. These nano/micropores ranged from 400 nm to 1 μm to release drugs steadily. The ZGS was kindly provide by Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian, China. The ZGS-eluting stents (ZES) were made by coating ZGS into the nano/micropores via freeze–drying technique. Sirolimus-eluting stents (SES; Lepu Medical Company, Beijing, China) and bare metal stents (BMS; Lepu Medical Company) with identical platforms were used as controls. All stents used in the experiments were 15 mm in length and 2.0 to 3.5 mm in diameter.
Stent Implantation
The process of experiment was conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication Number 85-23, revised 1996). The study protocol was approved by the Institutional Animal Care and Use Committee of Xiyuan Hospital.
Chinese minipigs (weight, 25-35 kg) were premedicated with aspirin (300 mg), clopidogrel (300 mg), and antibiotics (penicillin 1 g) orally 24 hours prior to stenting. Those animals were anesthetized with ketamine (20 mg/kg, intramuscularly [IM]), diazepam (0.4 mg/kg, IM), and 3% sodium pentobarbital via the marginal ear vein (25 mg/kg, intravenously). A 6F sheath was inserted into the right or left femoral artery under sterile conditions. Heparin (5000-10 000 U) was administered through the sheath to maintain an activated clotting time (250-300 seconds). Nitroglycerin was injected to avoid vasospasm. Coronary angiography was undertaken using Judkin-3.5 guiding catheter. The appropriate stent was delivered to the intended site with a stent to artery diameter ratio of 1.2:1.0. Continuous hemodynamic and electrocardiographic monitoring was performed throughout the procedure. In each animal, the stents were randomly deployed into 3 coronary arteries. During the follow-up period, animals received oral aspirin (100 mg) and oral clopidogrel (50 mg) daily with a standard diet in the animal laboratory center.
Scanning Electron Microscopy Examination
Scanning electron microscopy (SEM) was carried out at 7, 14, and 28 days. The rate of reendothelialization was assessed by SEM (HITACHI S-3400N, Japan). Stented segments were opened longitudinally by fixing in 3% glutaraldehyde, then dried with liquid CO2, and coated with gold. The SEM photomicrographs of each specimen were examined using a computer-assisted digital assessment system to determine the percentage of vascular coverage area compared to the total luminal surface area. Rate of endothelialization (%E) = (1 − [nonendothelialized area/total stent area]) × 100. 16
Histomorphometry and Histopathology
Histological examination was performed at 7, 14, and 28 days. The hearts were harvested as described previously. 17 Stented segments were dehydrated in a graded series of ethanol, embedded in methylmethacrylate plastic, and stained with hematoxylin and eosin and elastic Van Gieson stains. Morphometric measurements were evaluated, including lumen area, stent area, neointimal area, and percentage of stenosis of each section, as described previously. 18
For the pathological assessment, scores for reendothelialization, inflammation, and microthrombi of each stent were evaluated. Reendothelialization score with endothelial cells adhesion was assigned according to a semi-quantitative grading scale 18 –20 : 0 for absent endothelium, 1 for present but <25% of the lumen circumference, 2 for between 25% and 75% of the circumference, and 3 for complete endothelialization. Strut-associated inflammation was graded as follows: 0 for none, 1 for scattered inflammatory cells, 2 for moderate to dense cellular aggregate surrounding the strut noncircumferentially, and 3 for circumferential dense inflammatory cells infiltration of the strut. The microthrombi were assessed semi-quantitatively by the following scale 21 : values of 0 corresponding to absence; 1 for focal findings involving any portion of the artery but <25% of the circumference of the artery; 2 for moderate accumulations involving <25% of the circumference of the artery; and 3 for severe, involving >25% of the circumference of the artery.
Cell Culture
The HUVECs were kindly provided by Women and Children’s hospital of Haidian (Beijing, China). Cells were routinely cultured in endothelial cell medium (ECM; ScienCell, America) in a humidified atmosphere of 5% CO2 at 37°C. Cells were subcultured once they reached 90% to 95% confluence. In all experiments, cells were used at passages 3 to 7.
Cell Viability Assay
The viability of cells was determined using the 3-(4, 5-dimethylthiazol-2yl)-2, 5-diphenyl-2H-tetrazoliumbromide (MTT) assay. The HUVECs were inoculated at a density of 5 × 104/mL in 96-well plates and treated with ECM containing different concentrations of ZGC (0, 10, 20, 40, 60, 80, 100, 200, 400, 600, 800, and 1000 µg/mL) for 24 and 72 hours. The original medium was removed, and 20 µL of 5% MTT was added to each well. Incubation of 4 hours required a humidified atmosphere containing 5% CO2 and 95% air at 37°C until MTT was taken up by active cells. Then, the medium was discarded, and the precipitated formazan was dissolved in 100 µL of dimethyl sulfoxide. The absorption value was measured at 490 nm.
Cell Cycle Assay
The cell cycle was analyzed by flow cytometry. The HUVECs were plated at a density of 1 × 105 cells/mL, treated with either ECM-containing sirolimus (1 µg/mL) or different concentrations of ZGS (0, 600, 800, and 1000 µg/mL) for 24 hours. After harvesting, the cells were washed and fixed with ice-cold alcohol (75%) for more than 24 hours. Then, the cells were incubated with phosphate-buffered saline (pH 7.4) containing RNaseA (5U) and propidium iodide (50 µg/mL) for 15 minutes at 37°C. All samples were analyzed on a flow cytometry system. The percentage of cells in each phase of the cell cycle was estimated using the program of Cell FIT cell cycle analysis (Beckman Coulter, Germany).
Statistical Analysis
All results are expressed as mean ± standard deviation or standard error of the mean. Statistical comparisons were performed by 1-way analysis of variance with post hoc analysis for multiple comparisons. A P value of <.05 was considered statistically significant. Statistical analyses were carried out using SPSS v16.0 software.
Results
There were 21 Chinese minipigs implanted with 63 stents (ZES = 21, SES = 21, and BMS = 21) in our experiment. All animals survived. One of the pigs in the BMS group had ventricular fibrillation while was saved by electrical defibrillation on time.
Scanning Electron Microscopy Findings
Seven days after implantation, 94.04% ± 5.01% of ZES was covered by neointima compared to either SES (47.59% ± 19.91%, P < .01) or BMS (59.58 ± 19.61, P < .05). At 14 days, the percentage of reendothelialization area was 96.37% ± 1.86% for ZES, 87.51% ± 8.58% for BMS, and 69.22% ± 16.44% for SES (P < .05 for ZES vs SES). At 28 days, the percentage of coverage area was 98.51% ± 1.86% for ZES, 86.18%± 8.16% for SES (P < .05 for ZES vs SES), and 94.26%±5.58% for BMS, as shown in Figure 1.

Scanning electron microscopy showed re-endothelialization of different stents after implantation (×50). (A), (D), and (G) Great endothelial coverage were observed in ZES. (B), (E), and (H) Uncovered struts were seen in SES. (C), (F), and (I) Different degree of endothelial coverage were showed in BMS. Although complete coverage was observed in BMS at 28 days, whereas mass of inflammatory cells were adhered. Red arrow indicated naked struts, black arrow indicated platelets, and arrowhead indicated inflammatory cells. ZES: ZGS-eluting stents, SES: sirolimus-eluting stents, BMS: bare-metal stents.
Histomorphometric Analysis
Histomophometric analyses at different times are illustrated in Tables 1 to 3. Average injury score among the groups was of no difference (P > .05). At 7 days, there were no differences in the stent and lumen areas between ZES (3.96 ± 0.36 mm2, 3.32 ± 0.67 mm2), SES (3.50 ± 0.28 mm2, 2.72 ± 0.25 mm2), and BMS (3.62 ± 0.49 mm2, 2.64 ± 0.57 mm2), whereas percentage of naked struts was significantly less in ZES (11.39% ± 5.58%) when compared to SES (51.82% ± 16.70%, P < .01) or BMS (47.79% ± 12.87%, P < .01). At 14 days, nearly complete endothelial coverage was observed in ZES. Both SES and BMS showed naked struts. Moreover, SES exhibited stent thrombosis at 14 days. At 28 days, neointimal area, lumen area, and stent area were similar between ZES and SES, while percentages of naked struts per section were lower in ZES (2.50% ± 3.75%) than SES (22.10% ± 24.24%, P < .05). Furthermore, neointimal area was much lower in ZES (1.07 ± 0.48 mm2) than in BMS (1.73 ± 0.69 mm2, P < .01), as shown in Figure 2.
Histological Results 7 Days After Implantation.a
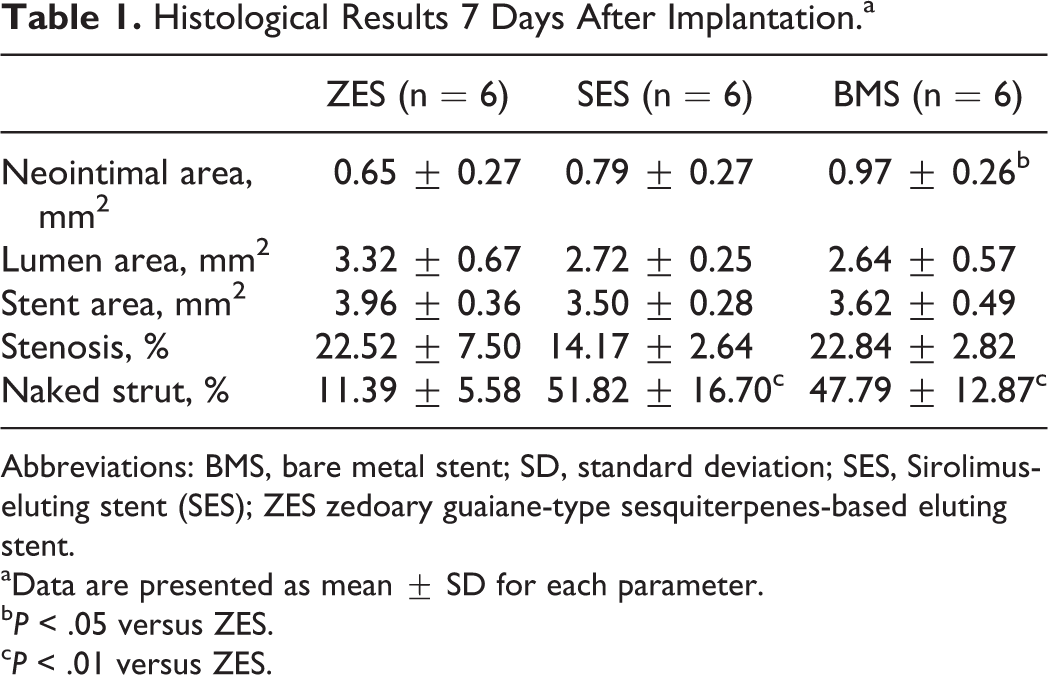
Abbreviations: BMS, bare metal stent; SD, standard deviation; SES, Sirolimus-eluting stent (SES); ZES zedoary guaiane-type sesquiterpenes-based eluting stent.
aData are presented as mean ± SD for each parameter.
b P < .05 versus ZES.
c P < .01 versus ZES.
Histological Results 14 Days After Implantation.a

Abbreviations: BMS, bare metal stent; SD, standard deviation; SES, Sirolimus-eluting stent (SES); ZES zedoary guaiane-type sesquiterpenes-based eluting stent.
aData are presented as mean ± SD for each parameter.
b P < .01 versus ZES.
c P < .05 versus ZES.
Histological Results 28 Days After Implantation.a
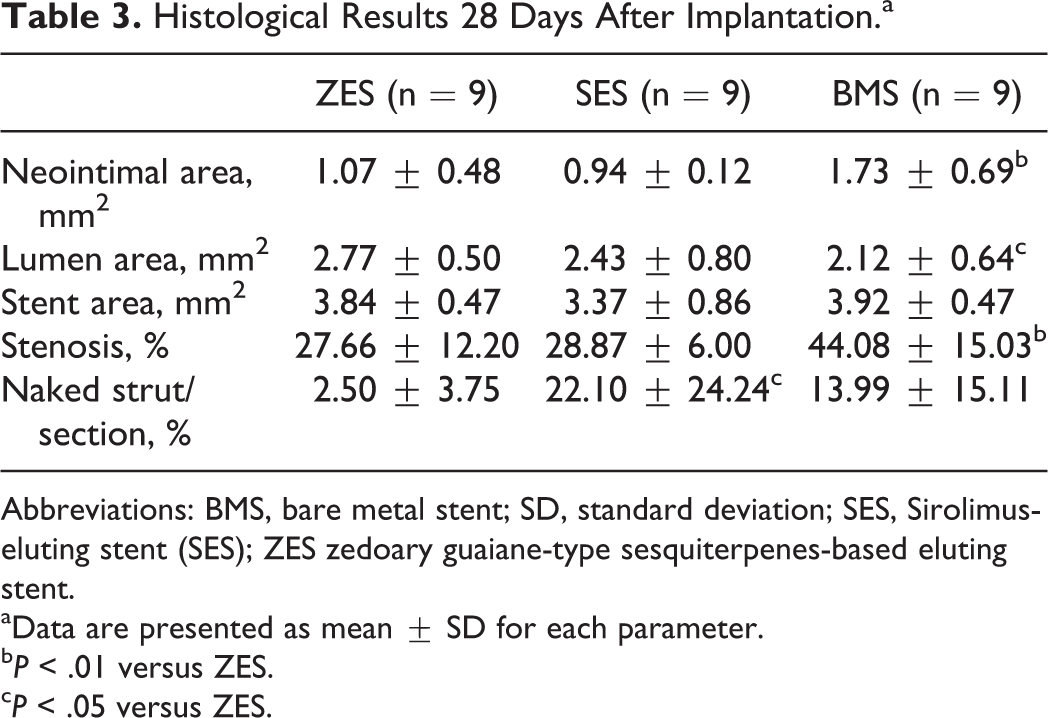
Abbreviations: BMS, bare metal stent; SD, standard deviation; SES, Sirolimus-eluting stent (SES); ZES zedoary guaiane-type sesquiterpenes-based eluting stent.
aData are presented as mean ± SD for each parameter.
b P < .01 versus ZES.
c P < .05 versus ZES.

Histomorphology of different stents after implantation (40×). (A), (D), and (G) Cross section of ZES showed complete endothelialium with thin neointimal thickness at 7, 14, and 28 days. (B), (E), and (H) uncovered struts were seen in SES at 7, 14, and 28 days. (C), (F), and (I) BMS showed nearly covered vessel surface, whereas neointimal hyperplasia was seen at 28 days. Black arrow indicated naked strut, arrowhead indicated stent-thrombus, and red arrow indicated neointimal hyperplasia.
Histopathological Analysis
The histopathological analyses were performed. At 28 days, reendothelialization score in ZES (2.83 ± 0.41) was higher than those in SES (1.43 ± 0.79, P < .01), while no difference was observed in BMS (2.17 ± 0.41, P > .05; Figure 3). Furthermore, the inflammation score was lower in ZES than either in SES or in BMS (both P < .01) at 28 days (Figure 4). And microthrombi were lower in ZES in compared with SES (P < .05) while was not different compared to BMS (P > .05). None of the stent thrombosis was observed in ZES during the experiment, whereas 2 cases in SES and 1 case in BMS occurred.

Re-endothelialization of different stents at 28 days. (A), (B), and (C) Different stents exhibited the degree of re-endothelialization after implantation (100×). (a), (b) and (c) represented ports of (A), (B) and (C), respectively(400×). Black arrow indicated endothelial cells, and red arrow indicated inflammatory cells. Data stated mean±SD. **P < .01, compared to ZMS.

Inflammation and micro-thrombi scores for stents at 28 days. ZES indicated ZGS-eluting stents; SES, sirolimus-eluting stents; BMS, bare metal stents. Data are stated mean±SD (A) or mean ± SE (B). *P < .05, **P < .01, compared to ZES.
The Cell Viability Analysis and the Cell Cycle Analysis
The ZGS showed no harmful effect on HUVECs viability in comparison with the control group at 24 hours (data not shown). At 72 hours, increased viability was seen in ZGS, whereas sirolimus significantly reduced HUVECs viability (P < .01) when compared to the control group (Figure 5).

Improvement the viability of HUVECs treated with ZGS. HUVECs were incubated with ZGS (10∼1000ug/ml) or sirolimus (1ug/ml) for 72h. Values were mean ± SE. Con indicated control group. Sir, sirolimus group. *P < .05, **P < .01 compared to Con; ## P < .01 compared to Con.
Compared to the control group, ZGS (800 and 1000 µg/mL) and sirolimus suppressed HUVECs at G0/G1 phage. However, the concentrations of ZGS (600, 800, and 1000 µg/mL) showed reduced inhibition of cell cycle at G0/G1 phase (60.33% ± 3.00%, 63.64% ± 1.59%, 65.47% ± 8.26%) when compared to sirolimus (73.88% ± 2.47%), as shown in Figure 6.

Effect of ZGS on the cell cycle of HUVECs. Cells were incubated with either increasing concentrations of ZGS (600∼1000ug/ml) or sirolimus (1ug/ml) for 24h. A∼E indicated Con, ZGS (600ug/ml), ZGS (800ug/ml), ZGS (1000ug/ml), and siroliums (1ug/ml), respectively. Con indicated control group, Sir, sirolimus group. Values were means±SD, **P < .01, compared to control; # P < .05, ## P < .01, compared to the Sir.
Discussion
The present study demonstrated that ZGS-based eluting stents nearly accomplished endothelial coverage 7 days after implantation and maintained thin neointima as effective as SES in a porcine coronary injury model at 28 days. Moreover, less inflammatory response was observed in ZES when compared to either SES or BMS. Microthrombi in ZES were significantly less than SES. In addition, ZGS also improved cell viability and reduced suppression of cell cycle at G0/G1 phase when compared to sirolimus.
Despite suppressing neointimal hyperplasia, sirolimus delays endothelial regrowth and function, therefore contributing to late thrombotic events to some extent. Looking for antiproliferative drugs with less toxicity toward normal cells seems especially necessary. Previous studies demonstrated that Curcuma zedoaria, as an anticancer drug, has demonstrated to have less negative effects on normal cells, 8 –10 showing that fibroblasts of human mucus lineage maintained stable cell viability from 0 hours to 7 days, when treated with Curcuma zedoaria extract. In the present study, we found that ZGS, bioactive substances extracted from Curcuma zedoaria, increased the viability rates of HUVECs and significantly reduced the suppression of HUVECs cycle at the G0/G1 phase in comparison with sirolimus. Hence, ZGS had favorable endothelial viability and proliferation, indicating the advantages of its use in stents materials.
Using a stent implantation injury model, Rogers et al 22 demonstrated that despite a 60% loss in the endothelial cell monolayer within 1 hour after direct stent implantation, complete regeneration of the damaged endothelial cell layer occurred within 14 days. In the present study, ZES completed reendothelialization at 7 days. Although BMS completed reendothelialization at 14 days, neointimal hyperplasia was observed at 28 days. Part of SES struts were still naked during the experiment. Moreover, ZES suppressed neointimal hyperplasia as effective as SES. Histomorphology in our research showed that neointimal area and thickness, lumen area, and restenosis percentage were not different between ZES and SES, suggesting that ZES could promote reendothelialization effectively along with thin neointima.
Pathological studies demonstrated that Curcuma zedoaria inhibits growth of cancer cells at S/G(2) or induces cancer cell apoptosis by cleavage and activation of caspase-3, -8, and -9, polymerase cytochrome C translocation, or loss of mitochondrial membrane potential. 6,23 Although the detailed mechanisms of suppression of smooth muscle cell (SMC) proliferation after treatment with ZGS was not investigated, it was obvious that reestablishment of an intact endothelium can inhibit SMC proliferation in our study and others. 24
The layer of endothelial cells on the luminal surface of stents indicates restoration of an intact endothelium, serving as a natural “barrier” to regulate a variety of functions including inhibiting thrombosis and inflammation. 25,26 However, there was discrepancy among ZES, SES and BMS in neointimal components and the vascular responses. Hara et al 27 analyzed the neointimal tissue of stents in a rabbit study using intravascular near-infrared fluorescence molecular imaging, showing greater fibrin deposition and fibrin persistence in DES than in BMS at 7 and 28 days, respectively. Consistent with previous findings, 19 we also observed that less endothelial cells were adherent on neointimal surface of SES, hinting that neointima with less endothelial cells do not possess intimal functions, and endothelium of BMS may not be functional as reflected by inflammatory cells adhesion. In contrast, a layer of endothelial cells was adhesive onto the surface of ZES neointima, which could prevent against inflammation and thrombosis.
Persistent inflammatory cell infiltration largely induced by incomplete endothelium is a key factor to form late stent thrombosis. 28,29 In the present study, the relatively low level of inflammation in ZES was observed, whereas inflammation in both SES and BMS was increased, respectively, at 28 days. Accumulating researches have already identified the effects of sesquiterpenes of Curcuma zedoaria in preventing inflammatory responses. 12,30 –32 Cho et al 33 demonstrated that zedoarondiol, a compound of ZGS, dose dependently inhibited lipopolysaccharide-stimulated tumor necrosis factor α, interleukin (IL) 6, and IL-1β production in RAW 264.7 macrophage and mouse peritoneal macrophage cells by suppressing the phosphorylation of Nuclear factor-kappB (IkappB) kinase (IKK) and Mitogen-activated protein kinases (MAPKs) and subsequently by inactivating the nuclear factor-κB pathway. This demonstrates that ZES prevents inflammation. In addition, research also showed that inflammation is closely associated with thrombosis. 34 There was no in-stent thrombosis in ZES, while these events occurred in both SES and BMS. The mechanism of antithrombosis in ZES required further investigation.
Although the animal studies demonstrated overall favorable outcomes using the ZGS-based eluting stents, it may be possible that the results will have disparity when applied to an atherosclerotic state. However, certain contents of Curcuma zedoaria have protective role against hypercholesterolemic and lipidemic conditions via modulation of PPAR-α, LXR-α, and associated genes involved in lipid metabolism and transport. 35,36 The effects of ZGS on cholesterol metabolism merit exportation in detail.
Conclusion
The present study suggested that ZGS-based eluting stents enhanced reendothelialization with suppression of neointimal hyperplasia in a porcine coronary artery model.
Footnotes
Authors’ Note
Yuanyuan Cui and Fuhai Zhao are the cofirst authors and wrote the paper together.
Acknowledgments
The authors acknowledge the help of Lei Li, Yue Shi, Leiyan Ma, and Zhixin Li for their assistance in this study. The authors thank Prof. Xinmiao Liang and Prof. Yanfang Liu at the Dalian Institute of Chemical Physics, Chinese Academy of Sciences.
Author Contributions
C. Yuanyuan and Z. Fuhai contributed to acquisition, analysis, and interpretation, drafted the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. L. Jiangang, W. Xin, and D. Jianpeng contributed to acquisition, drafted the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. S. Dazhuo and C. Keji contributed to conception and design, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. D. Shi conceived the topic. K. Chen helped to draft the manuscript. J. Liu, J. Du, and X. Wang helped to finish this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (no. 81072931 and 81473530) and basic research programme of China academy of Chinese medical sciences (no. ZZ0708106).
