Abstract
The development of diabetic macrovascular complications is a multifactorial process, and melatonin may possess cardiovascular protective properties. This study was designed to evaluate whether melatonin attenuates arteriosclerosis and endothelial permeability by suppressing the myosin light-chain kinase (MLCK)/myosin light-chain phosphorylation (p-MLC) system via the mitogen-activated protein kinase (MAPK) signaling pathway or by suppressing the myosin phosphatase-targeting subunit phosphorylation (p-MYPT)/p-MLC system in diabetes mellitus (DM). Rats were randomly divided into 4 groups, including control, high-fat diet, DM, and DM + melatonin groups. Melatonin was administered (10 mg/kg/d) by gavage for 12 weeks. The DM significantly increased the serum fasting blood glucose and lipid levels, as well as insulin resistance and endothelial dysfunction, which were attenuated by melatonin therapy to various extents. Importantly, the aortic endothelial permeability was significantly increased in DM rats but was dramatically reversed following treatment with melatonin. Our findings further indicated that hyperglycemia and hyperlipidemia enhanced the expressions of MLCK, p-MYPT, and p-MLC, which were partly associated with decreased membrane type 1 expression, increased extracellular signal-regulated kinase (ERK) phosphorylation, and increased p38 expression. However, these changes in protein expression were also significantly reversed by melatonin. Thus, our results are the first to demonstrate that the endothelial hyperpermeability induced by DM is associated with increased expressions of MLCK, p-MYPT, and p-MLC, which can be attenuated by melatonin at least partly through the ERK/p38 signaling pathway.
Introduction
The serious human health consequences of diabetes mellitus (DM) result from chronic diabetic complications favoring the development of arteriosclerosis and microangiopathy. Given the severity of diabetic arteriosclerosis, 1,2 further investigations are required to understand the mechanisms of diabetes-accelerated atherosclerosis and to develop effective therapeutic strategies for the prevention of the clinical events associated with arteriosclerosis.
Endothelial barrier dysfunction causes vascular hyperpermeability and increases vascular leakage, resulting in the extravasation of fluids, solutes, and macromolecules. 3 –5 At the early stages of arteriosclerosis development, various detrimental factors 6 contribute to endothelial dysfunction, which is accompanied by enhanced vascular permeability that facilitates both the infiltration and the retention of oxidized low-density lipoprotein (ox-LDL) through endothelial gaps in the arterial intima. 7 As a result, reversal of hyperpermeability in diabetes has emerged as a promising therapeutic strategy to inhibit the progression of diabetic arteriosclerosis.
The paracellular permeability of the endothelial barrier structure is partially maintained by the contractile forces generated from the cytoskeleton of endothelial cells, in which actin and myosin serve as the major contractile components. 8 In vascular endothelial and smooth muscle cells, the actin–myosin interaction relies on the myosin light-chain phosphorylation (p-MLC) status, which is regulated by myosin light-chain kinase (MLCK) and myosin light-chain phosphatase (MLCP). 9,10 Our previous study along with studies from other groups demonstrated that the selective MLCK pharmacological inhibitors ML-7 and ML-9 clearly inhibited MLCK expression, prevented MLC phosphorylation, and led to a marked attenuation of vascular permeability. 11 –14 Compelling evidence also suggests that inhibiting phosphorylation of the myosin phosphatase-targeting subunit (MYPT), a large MLCP subunit, could further block MLC phosphorylation (p-MLC) in various cell types. 15,16 A large body of evidence also indicates that an increase in MLCK- and MLCP-dependent p-MLC Ser19/Thr18 levels and an enhanced cytoskeletal contraction function play pivotal roles in regulating the permeability of cultured endothelial cells as well as intact venular endothelium. 17 –19
Melatonin is produced in a rhythmic manner by the pineal gland and other organs. 20 A large number of studies have demonstrated the powerful capability of melatonin and its metabolites, both at physiological and at pharmacological concentrations, to oppose oxidative damage by scavenging free radicals. 21 –25 Furthermore, special emphasis has been given to the protective effect of melatonin against atherosclerosis progression, which involves the normalization of lipid profiles and improvements in endothelial function and anti-inflammatory effects. 26 –28 Notably, some physiological effects of melatonin have been identified as mediated through melatonin receptors, including membrane type 1 (MT1) present in mammalian arteries. 29,30 However, the mechanisms responsible for the protective effects induced by the interaction of melatonin with its specific receptor remain to be elucidated. The binding of Ca2+ to calmodulin (CaM), a necessary means to activate MLCK, induces actin–myosin contraction, and there is also evidence to suggest that melatonin stimulates CaM phosphorylation and inhibits Ca2+/CaM-dependent kinase II activity, thereby restricting the interaction of CaM with its target enzymes. 31 Based on previous evidence, Zhu et al 32 and Xiao et al 33 showed that melatonin prevents the ox-LDL-induced increase in MLCK activation and expression in human umbilical vein endothelial cells (HUVECs), possibly via the extracellular signal-regulated kinase (ERK)/mitogen-activated protein kinase (MAPK) and the p38/MAPK signal transduction pathways.
Previous studies have emphasized the role of melatonin in the regulation of MLCK-dependent cytoskeletal contraction at the cellular level. However, there is a need to further establish the protective effects of melatonin and to elucidate the mechanisms involved in this role, especially in diabetic models. Therefore, we established a diabetic arteriosclerosis rat model to investigate the role of melatonin in endothelial hyperpermeability and in the progression of arteriosclerosis, in both hyperglycemia and hyperlipidemic conditions, and to examine whether aforementioned effects were mediated via MLCK- and MLCP-dependent MLC phosphorylation.
Materials and Methods
Animal Experimental Procedures
Thirty-two male Sprague-Dawley rats (weighing approximately 150-180 g) were obtained from the experimental animal center of Anhui Medical University. The rats were randomly divided into 4 groups, including control (n = 7), high-fat (HF) diet (n = 7), DM (n = 10), and DM + melatonin (n = 8) groups. All groups were subjected to a 12:12-hour light–dark cycle lights on at 06:00 under temperature (22°C ± 1°C) and humidity (50%-60%). With the exception of the control group, all rats received an HF diet (2% cholesterol, 10% grease, and 88% normal diet) for 8 weeks, after which time they were subjected to an intraperitoneal glucose tolerance test (IPGTT) and an intraperitoneal insulin tolerance test (IPITT). The rats in the DM and DM + melatonin groups were injected once intraperitoneally with a dose of streptozotocin (30 mg/kg) to induce diabetes, and the blood glucose levels were tested 1 week after the streptozotocin injection. Animals with glucose levels ≥11.1 mmol/L were considered diabetic. The rats in the control, HF, and DM groups were given normal saline, and the rats in the melatonin group were given 10 mg/kg/d of melatonin for 12 weeks. Melatonin was obtained from the Institute of Clinical Pharmacology of Anhui Medical University, dissolved in 1% alcohol, and given to the rats by gavage at 22:00. All of the rats were euthanized at the end of week 21. Following withdrawal of food overnight, the rats were deeply anesthetized using a 10% chloral hydrate solution at a dose of 0.3 mL/100 g. Blood samples were taken from the abdominal aorta. The aortas of all animals were removed and appropriately divided into 4 parts. One part of the thoracic aortas was fixed in 10% formalin and embedded in paraffin for immunohistochemical and morphological analysis and another part was embedded in optimum cutting temperature compound (OCT) and immediately frozen at −80°C for subsequent permeability analyses and oil red O staining. The third part of the thoracic aorta was placed in a dish containing ice-cold Krebs solution and continuously aerated with 95% O2 and 5% CO2. These samples were cut into rings (3 mm in length) to measure the isometric contractile tension. The fourth part of the thoracic aortas was immediately frozen at −80°C for Western blotting analysis. All experiments were approved by Ethics Committee of Anhui Medical University.
Transcutaneous Noninvasive Ultrasound Measurement of Vascular Diameter and Blood Flow Velocity
Transcutaneous noninvasive ultrasound evaluation of the vascular diameter and blood flow velocity were performed 2 days before the end of the experiment. Rats were anesthetized using a 10% chloral hydrate solution and placed in the dorsal decubitus position for the shaving of their abdomens. After 10 minutes of rest in the supine position, the ultrasonic examination of the abdominal aorta was performed using a 13-MHz ultrasound probe. The transducer was lubricated with ultrasound gel and placed at the level of the abdominal aorta with little pressure. Precisely, the probe was placed 1.0 cm below the renal artery to obtain a longitudinal axis view of the abdominal aorta. Images of the abdominal aorta were recorded continuously throughout the entire procedure. The systolic diameter (Ds), the diastolic diameter (Dd), the end diastolic velocity (EDV), and the peak systolic velocity (PSV) were recorded.
Measurement of Isometric Contractile Tension
Individual thoracic aorta rings were vertically suspended between 2 stainless steel wire hooks in a jacketed organ bath containing 5 mL of Krebs solution (pH 7.4), which contains 118 mmol/L NaCl, 4.7 mmol/L KCl, 1.1 mmol/L MgSO4, 1.2 mmol/L KH2PO4, 1.5 mmol/L CaCl2, 25 mmol/L NaHCO3, and 10 mmol/L glucose. The Krebs solution was replaced at 15-minute intervals. The bathing solution was bubbled continuously with a mixture of 95% O2 and 5% CO2 at 37°C. The isometric contractile tension was recorded continuously using a BL-420F biological function experimental system (Chengdu Tai Meng Science and Technology Co, Ltd, Chengdu, China). The resting tension applied to the aortic rings was increased stepwise to reach a final tension of 2 g. Then, the rings were equilibrated for 45 minutes. Following equilibration, the rings were precontracted with 1 µmol/L phenylephrine (Phe). Once a stable contraction plateau was obtained, 10−8 to 10−4 mol/L acetylcholine (Ach) or 10−8 to 10−4 mol/L sodium nitroprusside (SNP) was added to the organ bath cumulatively until a maximal vasodilator response was achieved. Cumulative vasodilator response data were expressed as the percentage of relaxation relative to the Phe-induced precontraction.
Serum Biochemical Analysis
Blood samples were taken from the abdominal aorta. Serum fasting blood glucose (FBG), total cholesterol (Tch), low-density lipoprotein cholesterol (LDL-c), triglyceride (TG), and insulin levels were measured with commercially available spectrophotometric assay kits (Beijing BHKT Clinical Reagent Co, Ltd, Beijing, China). The insulin sensitivity index was calculated using the [1/(FBG × fasting insulin)] formula.
Histological Examination
Thoracic aorta specimens were dehydrated, embedded in paraffin, sectioned (4 µm), and stained with hematoxylin and eosin (H&E) as previously reported. 34 Masson trichrome staining was used to detect extracellular matrix deposition by sequential addition of Bouin, Weigert, and Biebrich solutions on all of the aorta paraffin sections (4 µm), as described in the instruction manual. Lipid and fat content was evaluated by oil red O staining. Frozen sections were fixed with 10% formalin for 30 minutes. After rinsing with 60% isopropanol for 5 minutes, the aorta sections were stained with filtered oil red O working solution for 5 minutes, rinsed again with distilled water to reduce the background, and imaged. Photographs were taken, and the integral optical density values were measured using a JD-801 pathological image analysis system (Jiangsu JEDA Science-Technology Development Co., Ltd).
Immunohistochemistry Analysis
Briefly, the thoracic aorta tissues sections were deparaffinized, rehydrated, and fixed with a methanol–0.3% H2O2 solution for 20 minutes at room temperature. Antigens were unmasked by cooling for 30 minutes at room temperature after high compression heating. Primary antibodies against MLCK (Santa Cruz Biotechnology Inc, Santa Cruz, California) were incubated with the tissue sections overnight, followed by sequential incubation with a biotinylated antibody and horseradish streptavidin for 30 minutes at 37°C. Finally, the samples were incubated with diaminobenzidine for coloration and counterstained with hematoxylin for 2 minutes to stain the nuclei. Positively stained cells appeared brown in color. Photographs were taken, and the integral optical density values were measured using JD-801 pathological imaging analysis system.
Permeability Assay
The permeability assay using the surface biotinylation technique was performed as described by Wang et al 35 with some modifications specifically for the aortic intima. The thoracic aorta canals were filled with a freshly made Sulfo-NHS-LC-Biotin solution in HBS at 1 mg/mL 30 minutes at room temperature. The aortas were rinsed with phosphate-buffered saline, embedded in OCT, and cryosectioned. Then, 10-µm frozen sections were incubated in blocking buffer for at least 5 hours. After washing 3 times with blocking buffer, the sections were incubated with Rhodamine600 Avidin D (XRITC-avidin). The samples were examined by fluorescence microscopy. Photographs were taken, and the integral optical density values were measured using a JD-801 pathological image analysis system (Jiangsu JEDA Science-Technology Development Co., Ltd).
Western Blotting
The MLCK, p-MLC, p-ERK, p-p38, and MT1 protein levels were measured by Western blotting. The p-MLC antibody was obtained from Cell Signaling (Danvers, Massachusetts). The β-actin, MLCK, p-MYPT, p-ERK, p-p38, and MT1 antibodies were purchased from Santa Cruz Biotechnology Inc. The primary antibody concentrations used were as follows: MLCK (1:500 dilution), p-MYPT (1:250 dilution), p-MLC (1:500 dilution), p-ERK (1:250 dilution), p-p38 (1:500 dilution), and MT1 (1:500 dilution). The protocol used was described in a previous study. 27 β-Actin levels were used to normalize the relative protein expression levels.
Statistical Analysis
All statistical analyses were undertaken using SPSS software, version 18.0. All data are presented as the means ± standard deviations. Comparisons between groups were carried out using 1-way analysis of variance and the Student-Newman-Keuls method. Statistical significance was confirmed at P < .05.
Results
Intraperitoneal Glucose Tolerance Test and IPITT
After 8 weeks of HF feeding, IPGTT and IPITT results showed that the blood glucose levels in HF rats were significantly higher than those in control rats at baseline and at 15, 30, and 60 minutes time points (P < .05). The area under the curve for the glucose levels increased significantly at week 8 in rats fed an HF diet (P < .05; Figure 1).
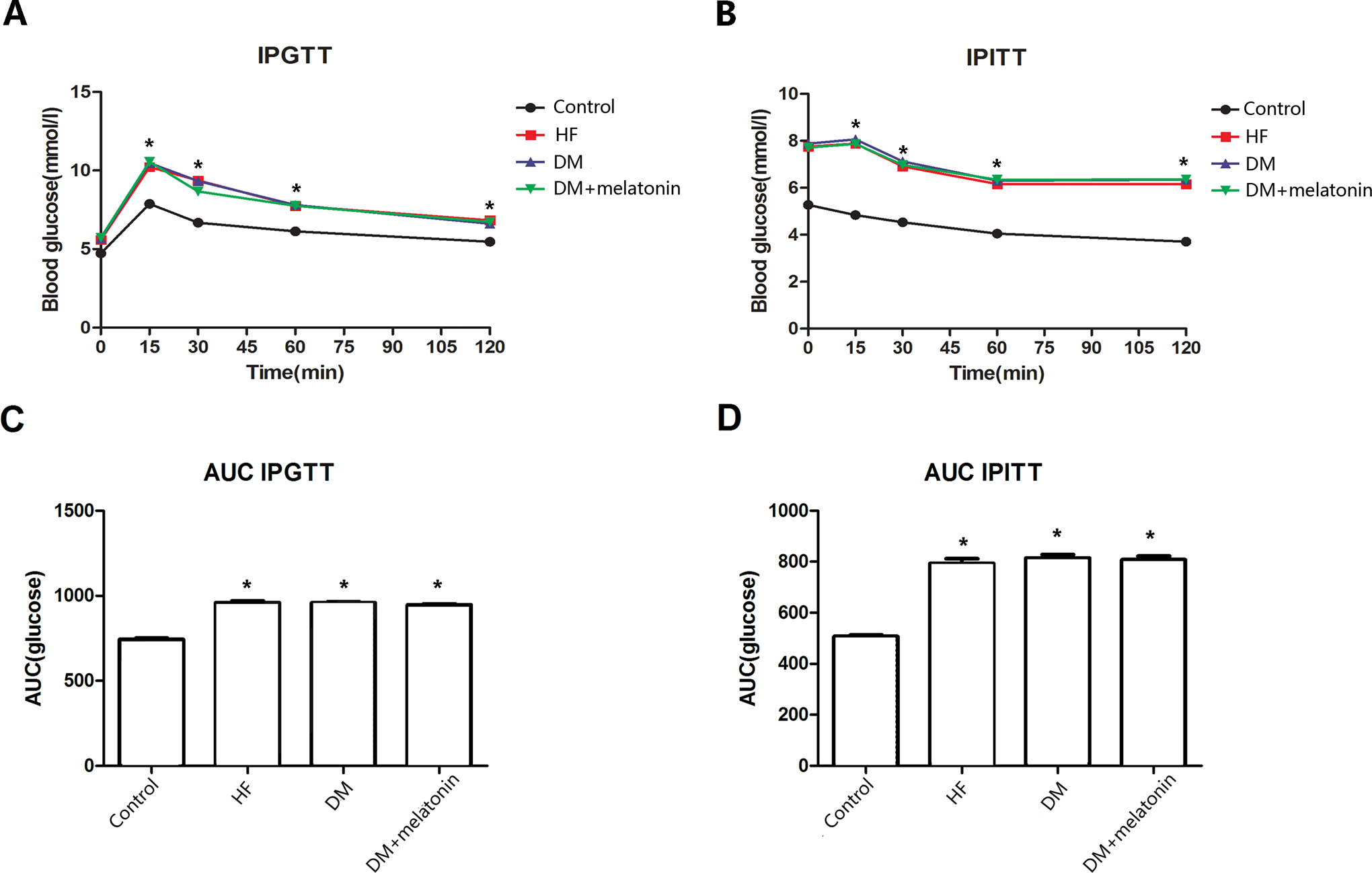
The IPGTT and IPITT after high-fat diet treatment for 8 weeks. A, Serum glucose levels during IPGTT. B, Serum glucose levels during IPITT. C, The AUC of glucose in IPGTT. D, The AUC of glucose in IPITT. Data are expressed as mean ± SD. *P < .05 versus controls. N > 6 per group. IPGTT indicates intraperitoneal glucose tolerance test; IPITT, intraperitoneal insulin tolerance test; AUC, area under the curve; SD, standard deviation.
Metabolic Parameters in Diabetic Models
After 12 week of treatment, FBG, Tch, TG, and LDL-c levels as well as insulin resistance were dramatically higher in the DM group compared with the control group (P < .05). However, melatonin therapy significantly reduced the serum Tch, TG, and LDL-c level (P < .05) in comparison to the DM group. Melatonin also reduced the extent of insulin resistance and levels of FBG, although these differences were not statistically significant (Table 1).
Basic Parameters of Rats in Different Experimental Groups.a

Abbreviations: FBG, fasting blood glucose; Tch, total cholesterol; LDL-c, low-density lipoprotein cholesterol; TG, triglyceride; ISI, insulin sensitivity index; HF, high-fat; DM, diabetes mellitus; MLT, melatonin; SD, standard deviation.
aData are mean ± SD values. ISI = 1/(FBG × fasting insulin).
b P < .05 versus control group.
c P < .05 DM + MLT group versus DM group.
Transcutaneous Noninvasive Ultrasound
At the end of the experiment, we measured the vascular diameter and blood flow velocity using transcutaneous noninvasive ultrasound. In the DM group, the EDV and PSV were significantly decreased (P < .05), while the Ds was dramatically increased compared with the control group (P < .05). Melatonin treatment significantly elevated the PSV (P < .05) and reduced both the Ds and the Dd values compared with the DM group, although not significantly (Table 2).
Transcutaneous Noninvasive Ultrasound Measurement.a

Abbreviations: HF, high fat; DM, diabetes mellitus; MLT, melatonin; SD, standard deviation.
aData are mean ± SD values.
b P < .05 versus control group.
c P < .05 DM + MLT group versus DM group.
Endothelial Function
We next studied the effects of melatonin on the vascular endothelial function in diabetic rats. Isometric contractile tension experiments on isolated aortic rings were performed. As illustrated in Figure 2, in the DM group, we observed an inhibition of the cumulative contraction relaxation response of thoracic aorta rings to Ach compared with the control and HF groups (P < .05). However, there was no significant change in the accumulative contraction relaxation in response to SNP. Melatonin therapy reversed the diabetes-induced impairment of the cumulative concentration-dependent vasodilator responses to Ach on Phe-contracted thoracic aorta rings (P < .05) but did not affect the SNP-induced cumulative contraction relaxation responses.

Relaxation to Ach (A) and SNP (B) of thoracic aortic rings from rats with or without melatonin therapy. Aortic rings were exposed to cumulative doses of the indicated agent. Values are presented as percentage of relaxation relative to a Phe precontraction (1 µmol/L). Data are expressed as mean ± SD. *P < .05 DM + melatonin group versus DM group. N > 6 per group. Ach indicates acetylcholine; SNP, sodium nitroprusside; Phe, phenylephrine; SD, standard deviation; DM, diabetes mellitus.
Endothelial Permeability
To determine the effect of melatonin treatment on endothelial permeability, the transport of Biotinamidohexanoic acid 3-sulfo-N-hydroxysuccinimide ester sodium salt (NHSLC-biotin) across the aortic intima into the media was assessed. The NHSLC-biotin concentration profiles were obtained as a function of the radial distance through the aortic wall media layer. Only the endothelial surface of the aorta intima was biotinylated in rats fed a normal diet, indicating no paracellular leakage of the NHSLC-biotin. The HF rats’ aortic intima layers were incubated with NHSLC-biotin, and NHSLC-biotin leakage into the aortic intima layers was increased in diabetic rats. However, aortic endothelial permeability was clearly attenuated in the melatonin group compared with the DM group (Figure 3).

Effects of melatonin on permeability, fibrosis, and lipid deposition in diabetic rat aorta. A, Permeability assay of rat aorta intima by surface biotinylation technique (magnification ×400). B, Masson-stained sections of aorta (magnification ×50). C, Oil red O-stained sections of aorta (magnification ×200). 1: Control, 2: HF group, 3: DM group, and 4: DM + melatonin group. Data are expressed as mean ± SD. *P < .05 versus control, # P < .05 DM + melatonin group versus DM group. N > 6 per group. DM indicates diabetes mellitus; HF, high fat; SD, standard deviation.
Morphological Characteristics
We next examined the morphological characteristics of the arterial lesions on H&E, Masson trichrome, and oil red O-stained tissue sections. Compared with the control groups, endothelial cells adhering to inner elastic plates showed structural changes in the DM group. Indeed, the vascular smooth muscle cells (VSMCs) of the tunica media were arranged irregularly, as shown by irregular nuclei and uneven cytoplasm staining, and the inflammatory cells infiltration and the media layer thickness of the aorta were also clearly increased. However, melatonin treatment attenuated the structural impairments of the aortic intima, corrected the disarrangement of VSMCs, and inhibited inflammatory cell infiltration (Figure 4). Masson staining showed that in the DM group, the collagen content was significantly increased, and the thick collagen fibers were connected into irregularly arranged reticular or mass structures. However, melatonin treatment dramatically decreased the collagen content, as shown in Figure 3. Oil red O staining further revealed that diabetic rats fed HF diet showed a significantly increased lipid accumulation in endothelial cells compared with the control and HF groups, while melatonin treatment apparently attenuated this lipid accumulation, as shown in Figure 3.

Melatonin prevented the damage of diabetic rat aorta and decreased the expression of MLCK. A, The H&E-stained sections of aorta (magnification ×200). B, The MLCK protein expression was analyzed by immunohistochemistry staining (magnification ×400). C, Bar graph of protein quantification from 6 independent experiments. 1: Control, 2: HF group, 3: DM group, and 4: DM + melatonin group. Data are expressed as mean ± SD. *P < .05 versus control, # P < .05 DM + melatonin group versus DM group. MLCK indicates myosin light-chain kinase; H&E, hematoxylin and eosin; DM, diabetes mellitus; HF, high-fat; SD, standard deviation.
Measurement of Relative Protein Expression Levels and Signaling Pathways Activity
As shown in Figure 4, high MLCK expression levels were observed in the arterial wall of the HF and DM group rats, in comparison with controls. However, melatonin treatment led to significant downregulation of MLCK expression in the arterial walls of diabetic rats. Western blot analysis further revealed that the conditions of hyperglycemia and hyperlipidemia in the DM group dramatically enhanced expression levels of MLCK, p-MYPT, p-MLC, p-ERK, and p-p38 and suppressed expression of MT1 (P < .05), whereas melatonin therapy reversed these effects on protein expression (P < .05; Figure 5).

Effect of melatonin on the expression of membrane type 1 (MT1), MLCK, phosphorylation of MLC, ERK, and p38. A, Representative Western blot image. B, Bar graph of protein quantification plotted from 3 independent experiments. 1: Control, 2: HF group, 3: DM group, and 4: DM + melatonin group. Data are expressed as mean ± SD. *P < .05 versus control, # P < .05 DM + melatonin group versus DM group. N > 6 per group. MLCK indicates myosin light-chain kinase; MLC, myosin light chain; ERK, extracellular signal-regulated kinase; DM, diabetes mellitus; HF, high fat; SD, standard deviation.
Discussion
In the present study, we utilized streptozotocin injection to induce an insulin secretion defect and HF feeding to induce an insulin resistant state in a rat model of diabetes. The dose of melatonin used (10 mg/kg/d) was selected based on previous studies. 36,37 Our results demonstrated that melatonin ameliorated lipid metabolism, hemodynamic and endothelial dysfunctions, and vascular hyperpermeability to a various extent in diabetic rats. Most importantly, our study is the first to show that the suppression of MLCK- and p-MYPT-mediated p-MLC expression, which was partly associated with the ERK/p38 signal transduction pathway for the alleviation of hyperpermeability in both hyperglycemic and hypercholesterolemic states, played a crucial role in the protective effects of melatonin.
The endothelial barrier functions via intercellular junctions that restrict the passage of plasma components into the interstitial space. At the intracellular level, junction proteins are linked to adaptor proteins that are connected to the actin cytoskeleton. 8 The cytoskeletal tension is controlled by actomyosin contractility and is regulated mainly by the MLC phosphorylation status, which is controlled by the balance between phosphorylation by MLCK and dephosphorylation by MLCP. 38 –40 Phosphorylation of MYPT, a subunit of MLCP, reduces the levels of MLCP and MLC phosphorylation. Indeed, the endothelial hyperpermeability associated with increased actomyosin contractility is MLCK and MLCP dependent. 8 Our previous data 41 suggested that endothelial permeability was increased under a hypercholesterolemic status and associated with elevated MLCK expression and activity. Moreover, increased endothelial permeability is a hallmark of the early stages of diabetic macrovascular complications. Guo et al demonstrated that advanced glycation end products increased the permeability of endothelial monolayers and led to actin cytoskeleton reorganization. 42 Animal experiments have further indicated that diabetes-associated abnormal vascular reactivity and the diabetes-induced altered regulation of endothelial and smooth muscle cells contraction are specifically controlled by the opposing roles of MLCK and MLCP. 43 Our study further demonstrated that endothelial permeability in diabetic rats aortas was dramatically increased and, more importantly, that MLCK expression and MYPT and MLC phosphorylation status, which are associated with contractile forces, were significantly augmented in comparison with control and HF diet-fed rats. This finding indicates that the combination of a hyperglycemic and hyperlipidemic condition aggravates endothelial barrier dysfunction to a greater extent than hyperlipidemia alone.
Low levels of nocturnal melatonin secretion have been reported as a risk factor for the metabolic syndrome, DM, and coronary diseases. 44,45 Experimental evidence also indicates that melatonin produces beneficial effects on glucose metabolism. 46 For instance, melatonin protected diabetes-prone rats that fed a high-calorie diet from developing hyperlipidemia, hyperglycemia, and hyperleptinemia, and melatonin administration to insulin-resistant mice reversed the insulin resistance and improved glucose metabolism. 47,48 Our data further showed that melatonin reduced FBG levels and insulin resistance although not significantly. In addition, hyperlipidemia was markedly attenuated in the melatonin-treated group in comparison with the DM group. The metabolic disorder-induced hemodynamic dysfunction was also improved in DM rats under melatonin therapy. Furthermore, a growing body of evidence supports the notion that melatonin exerts beneficial effects by inhibiting the development of atherosclerosis via various pathways. Our histological analyses showed that aortic wall stiffness was significantly alleviated under melatonin therapy, in parallel with decreased collagen deposition and lipid accumulation in the vascular wall. Moreover, the disarrangement of the aortic intima structure, including the large number of infiltrated inflammatory cells, observed in diabetic rats was markedly attenuated by melatonin treatment. Melatonin has also been reported to ameliorate vascular endothelial dysfunction in HF diet-fed rabbits by suppressing the Toll-like receptor 4/nuclear factor κB pathway. 28 Our findings indicate that melatonin improved the endothelium-dependent vasodilation in diabetic rats, which may explain the aforementioned results.
It is noteworthy that many protective effects of melatonin are reportedly mediated through melatonin membrane receptors, including MT1. 49,50 Zhu et al demonstrated that microRNA-29b promotes HF diet-stimulated endothelial permeability in apolipoprotein E knockout mice by downregulating expression of MT1. This result indicates the crucial role of MT1 in endothelial permeability. 51 Therefore, it is highly possible that the protective effect of melatonin that we observed on endothelial permeability may also be mediated by MT1. Cui et al reported that the melatonin membrane receptor antagonist luzindole partially inhibited the action of melatonin on HUVEC proliferation and blocked the action of melatonin on intracellular signaling pathways. Thus, these results linked the melatonin membrane receptors with intracellular signaling pathways in HUVECs. 52 As expected, we demonstrated that melatonin treatment significantly upregulated expression of MT1 and inhibited the ERK/p38 signaling pathway in aortas of diabetic rats. Based on previous studies, we postulate that melatonin therapy inhibits the ERK/p38 signaling pathway partly via the elevation in expression of MT1. In addition, our laboratory recently reported that melatonin prevents the ox-LDL-induced increase in MLCK activation and expression in HUVEC via the MAPK signal transduction pathway. 32,33 In the present study, our data further verified that melatonin therapy could significantly attenuate aortic hyperpermeability in DM rats, possibly by suppressing MLCK expression and MLC phosphorylation and at least partly by regulating the ERK/p38 signal pathway as discussed previously. Considering that MLC phosphorylation is also regulated by MLCP activity, we further demonstrated that melatonin reduced expression levels of p-MYPT, thereby increasing MYPT activity and inhibiting MLC phosphorylation in diabetic rat aortas.
Taken together, our observations confirmed that melatonin exerts beneficial effects on the regulation of insulin resistance, lipid metabolism disorder, arterial interstitial fibrosis, and hemodynamic and endothelial dysfunctions in diabetic rats. Based on this in vivo study as well as our previous studies in HUVEC, the inhibitory effect of melatonin on hyperpermeability in the setting of DM may represent a novel mechanism contributing to the improvement in aortic endothelial permeability and arteriosclerosis associated with the MAPK/MLCK/p-MLC and MLCP/p-MLC systems. Our findings may also provide important implications for diabetic patients with arteriosclerosis, for whom melatonin could exert specific effects for the prevention and treatment of hyperpermeability in large blood vessels.
Footnotes
Authors’ Note
Song-tao Tang and Huan Su contributed equally to this work.
Author Contribution
Song-tao Tang contributed to conception and design and acquisition, analysis, and interpretation. Huan Su drafted the article and critically revised the article. Qiu Zhang and Hai-qin Tang critically revised the article. Chang-jiang Wang contributed to analysis. Qing Zhou contributed to interpretation. Wei Wei gave final approval and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Hua-qin Zhu and Yuan Wang critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (No. 81470568, 81270372 and 81070232).
