Abstract
Effective treatment of chronic lower limb ischemia is one of the most challenging issues confronting vascular surgeons. There are a number of choices available to the vascular surgeon. Open or endovascular revascularization is the treatment of choice when applicable. Current pharmacological therapies play an auxiliary role and cannot prevent disease progression. Therefore, new methods of treatment are needed. We conducted a phase 2b/3 multicenter randomized controlled clinical trial of the intramuscular transfer of a plasmid DNA encoding vascular endothelial growth factor (VEGF) 165 with cytomegalovirus promotor (CMV) in patients with atherosclerotic lower limb ischemia. A total of 100 patients were enrolled in the study, that is, 75 patients were randomized into the test group and received 2 intramuscular injections of 1.2 mg of pCMV-vegf165, 14 days apart together with standard pharmacological treatment. In all, 25 patients were randomized into the control group and received standard treatment only. The following end points were evaluated within the first 6 months of the study and during a 1.5-year additional follow-up period: pain-free walking distance (PWD), ankle–brachial index (ABI), and blood flow velocity (BFV). The pCMV-vegf165 therapy appeared to be significantly more effective than standard treatment. The PWD increased in the test group by 110.4%, 167.2%, and 190.8% at 6 months, 1 year, and 2 years after treatment, respectively. The pCMV-vegf165 intramuscular transfer caused a statistically significant increase in ABI and BFV. There were no positive results in the control group. Thus, pCMV-vegf165 intramuscular gene transfer is an effective method of treatment of moderate to severe claudication due to chronic lower limb ischemia.
Introduction
According to the World Health Organization, cardiovascular diseases are the number 1 cause of death globally. 1 Cardiovascular disease includes both coronary heart disease, cerebrovascular disease, and peripheral arterial disease, which causes chronic lower limb ischemia. 2 Endovascular or open revascularization procedures are the main treatment methods for such patients, although many of them are not suitable for a revascularization due to severe distal or multifocal atherosclerotic lesions, failed grafts, or severe coexisting pathology. Thus, new methods to treat chronic lower limb ischemia should be used.
Along with open surgical, endovascular, and pharmacological treatment, gene therapy has been introduced to treat patients with chronic lower limb ischemia. Gene therapy is one of the most rapidly developing methods for treating ischemia. 3,4 The following different types of therapeutic genes that encode various growth factors have been used in clinical trials: vascular endothelial growth factor (VEGF) 165, 5 –7 basic fibroblast growth factor, 8,9 hypoxia-inducible factor, hepatocyte growth factor, 10,11 and others. Gene therapy is designed to induce angiogenesis via the expression of the aforementioned genes in skeletal muscles after intramuscular or intravascular delivery of gene products.
In 2010, we completed a phase 1 to 2a clinical trial of pCMV-vegf165 in patients with chronic lower limb ischemia (stage 2a to 3 according to Fontaine classification modified by A. V. Pokrovsky) who were not suitable for reconstructive surgery or endovascular treatment. This study demonstrated the safety, feasibility, and short-term (3 months) efficacy of pCMV-vegf165 gene transfer, 12,13 which lead to conducting a phase 2b to 3 multicenter clinical trial. The study was conducted under the control of the Russian Ministry of Health and was completed in 2011. Patients enrolled in the study were subjected to a 6-month follow-up period according to the study protocol and an additional 18-month follow-up period for a longer evaluation of study drug efficacy and safety. The results of the study are reported herein.
Materials and Methods
Rationale for the Clinical Study
Preclinical studies of general toxicity (acute, subacute, chronic, and local irritation) and specific toxicity (allergenicity, reproductive and immune toxicity, mutagenicity, and carcinogenicity) as well as the detection of specific drug activity were carried out at Russian State Federal Institution “Institute of Toxicology of Federal Medical Biological Agency of Russia,” Saint-Petersburg (2008). The safety, feasibility, and short-term efficacy of the study drug were then evaluated in a phase 1 to 2a multicenter randomized trial that was conducted in 2010 and enrolled 45 patients.
Federal Service on Surveillance of the Ministry Healthcare and Social Development of the Russian Federation has granted the approval to conduct a phase 2b to 3 study (approval notice No. 177, April 21, 2010). The study protocol was approved by the National Ethics Committee (protocol No. 62 from April 07, 2010); local ethics committees have also granted their approval to conduct the study.
All phases of clinical trials were conducted according to the Declaration of Helsinki of the World Medical Association “Recommendations guiding physicians in biomedical research involving human subjects” (1964, 2000), “Rules of Good Clinical Practice in the Russian Federation” OST 42-511-99, ICH GCP rules, and valid regulatory requirements.
Drug Characteristics and Administration Method
The study drug is an original gene construction which contains a supercoiled plasmid DNA (1.2 mg) encoding pCMV-vegf165 as the active substance and is now marketed as “Neovasculgen.” 12 The drug was supplied to the study centers as a sterile lyophilisate that was then dissolved in 2 mL of water for injections immediately prior to administration. The drug was administered intramuscularly (calf muscles) at 4 to 5 injection sites in the lower and middle third of the posterior part of the calf.
Patient Characteristics
The study included patients with chronic lower limb ischemia who were not suitable for an open or endovascular revascularization due to a severe distal or multifocal atherosclerotic lesion. The decision was made by a team of vascular surgeons and radiologists based on the angiographic and echographic findings, history of the disease, previous procedures, and concomitant pathology. Angiographic score was ≥7 points according to the Rutherford (1997) runoff classification.
The types of atherosclerotic lesions were defined as follows: proximal lesion—patency of proximal arterial segments (aortoiliac) with a diffuse atherosclerotic lesion (occlusion) of superficial femoral artery and a popliteal artery extending into the tibioperoneal trunk; multifocal lesion—patency of proximal arterial segments (aortoiliac) with a diffuse atherosclerotic lesion (occlusion) of the femoral, popliteal, and both tibial arteries; distal lesion—patency of proximal arterial segments (aortoiliac, femoral) with a diffuse atherosclerotic lesion (occlusion) of the popliteal artery with hemodynamically significant stenosis or occlusion of the tibial arteries.
All patients had a previous history of a long-term moderate to severe claudication. All patients received aspirin and statins on a daily basis to reduce the risk of adverse cardiovascular ischemic events and previously underwent treatment with pentoxifylline.
Inclusion criteria
age more than 40 years;
a history of stable claudication for at least 3 months;
stage 2 to 3 chronic ischemia according to Fontaine classification (modified by A. V. Pokrovsky);
absence of hemodynamically significant stenosis (>70%) of the aortoiliofemoral arterial segment or (if present) a patent proximal bypass graft (prosthesis) if revascularization surgery was performed no earlier than 3 months prior to the inclusion in the study; satisfactory patency of the deep femoral artery in the presence of hemodynamically significant femoropopliteal arterial lesions;
presence of hemodynamically significant (stenosis >70% and/or occlusion) diffuse lesions of the anterior and (or) posterior tibial arteries (distal lesion);
voluntary informed consent signed and dated by the patient.
Exclusion criteria
chronic lower limb ischemia of nonatherosclerotic genesis (autoimmune disorders, Buerger disease, congenital abnormalities, vascular injuries, etc);
stage 4 chronic ischemia according to Fontaine classification modified by A. V. Pokrovsky (ischemic ulcers and necrotic lesions);
severe concomitant pathology with life expectancy <1 year;
infectious diseases, history of cancer, or suspected malignancy;
decompensated diabetes mellitus (hemoglobin A1c > 8% and fasting plasma glucose > 11.1 mmol/L).
Study Design and End Points
The current pCMV-vegf165 gene transfer trial was as an open-label, prospective, randomized, controlled, and multicenter study. The patient distribution per study center and time period is presented in Figure 1.

Design of clinical trial.
A total of 100 patients were enrolled in the study and randomized into 2 groups at a ratio of 3:1. Thus, 75 patients were included into the test group and received 2 injections of pCMV-vegf165 at a dose of 1.2 mg, 14 days apart (total dose—2.4 mg) into the calf muscles altogether with standard pharmacological treatment, and 25 patients were included in the control group and were given standard therapy alone. All patients signed and dated the informed consent documents.
In all, 13 patients had ischemic rest pain at baseline: 8 and 5 patients in test and control groups, respectively. In all, 11 patients underwent aortoiliac arterial reconstructive surgery within more than 6 months prior to the onset of the study (5 patients in the test group and 6 patients in the control group). In all, 18 patients had compensated diabetes mellitus: 12 in the test group and 6 in the control group.
In all, 5 patients in the test group and 1 patient in the control group had undergone limb amputations prior to study enrollment which did not allow to perform a treadmill test to evaluate pain-free walking distance (PWD). The analyzed population in the study included 94 patients: 70—in the test group and 24—in the control group. The AP value was variable depending on the technical equipment of each study site and the period of time following the study completion, which was considered when processing statistical data.
The initial time points were defined as follows: baseline, 14, 90, and 180 days. Follow-up period was extended to a total of 2 years, with additional time points at 1 and 2 years following the patients’ inclusion in the study.
Safety Criteria
Safety of pCMV-vegf165 gene transfer in terms of the trial protocol was initially evaluated within 6 months following the onset of the study (a 14-day in-patient hospital stay, with following out-patient office visits) with the registration of adverse event (AE) and serious adverse event (SAE) during both routine visits and unscheduled requests for medical care. Moreover, patients who gave their written consent for the extended follow-up procedures underwent blood and urine laboratory tests, chest X-rays, and abdominal echography in order to assess the oncological safety (Figure 1).
Efficacy End Points
Primary Efficacy End Point
Pain-free walking distance
The value of the PWD was defined as the primary (main) efficacy end point. According to the American College of Cardiology/American Heart Association Guidelines for the management of patients with peripheral arterial disease, this value is of the highest importance (class I recommendations). 14 The intragroup distribution of patients was based on the PWD value. The severity of the disease was determined according to the Fontaine classification modified by A. V. Pokrovsky, which is widely accepted in Russia: stage 2a—PWD more than 200 m; stage 2b—less than 200 m, but more than 50 m; and stage 3—less than 50 m or ischemic rest pain in absence of ischemic ulcers or necrotic lesions. The PWD was determined using a treadmill test with reduced initial speed (1 km/h), as the majority of elderly patients were unable to perform Gardner test or its equivalents. Information on patients with ischemic rest pain is also provided (Table 1).
Baseline Characteristics of Patients.

Abbreviations: ABI, ankle–brachial index; BFV, blood flow velocity; PWD, pain-free walking distance; SD, standard deviation.
Secondary End Points
Ankle–brachial index
Ankle–brachial index (ABI) was measured using a standard technique at each visit. Although ABI measurement is regarded as a first-line assessment tool, 14 it largely characterizes main arterial blood flow (macrohemodynamics), and its diagnostic value is limited in patients who are not suitable for arterial reconstructive surgery due to poor runoff.
Blood flow velocity
Doppler ultrasound techniques are useful in assessment of lower extremity atherosclerotic lesions and determining severity of the disease or progression of atherosclerosis. 14 Blood flow velocity (BFV) in the posterior tibial artery was evaluated (if patent).
Angiography
Thirty percent of patients enrolled into the study agreed to undergo a digital subtraction angiography using contrast enhancement at following time points: prior to the study, at 6 months, and 1 year after the onset of treatment. Primary and repeated angiography was performed using the same angiography system, by the same radiologist, and with the same time delay of images. Angiograms were assessed visually by the same experienced specialist.
Quality of life
All patients completed the SF-36 questionnaire (“SF-36 Health Status Survey”) before enrollment and at 6 months after the onset of treatment (Figure 1). The following 7 scales were evaluated: physical functioning, physical role functioning, bodily pain, general health perceptions, vitality, social role functioning, emotional role functioning, and mental health. The values of each scale varied between 0 and 100, with 100 defined as complete health. All the scales were used to assess 2 parameters: psychological and physical well-being.
Statistical analysis
A sample size of 28 patients in each group was estimated to detect a 0.75 standardized difference (80% power, P = .05), assuming the target difference and SD for PWD to be 75 and 100 m, respectively. We decided to use a 3:1 test/control group ratio in order to make the test group sample more representative.
The absolute values of efficacy criteria (PWD, ABI, and BFV) were not normally distributed; therefore, nonparametric methods were used to test the hypothesis (Mann–Whitney U test and Wilcoxon test with Bonferroni correction to avoid a type I error). The SF-36 questionnaire scores were normally distributed, so the T test was used to compare the values of 2 groups.
Results
Baseline Characteristics of the Trial Participants
A total of 100 patients were enrolled in the clinical study: 75 were randomized into the test group and received 2 injections of pCMV-vegf165, 14 days apart (a total dose of 2.4 mg) into the calf muscles of the affected limb. The comparison of baseline characteristics between the groups showed that gender differences as well as differences in the primary and secondary end points were not statistically significant (Table 1). The values of PWD were similar between the 2 groups: 135.3 ± 12.2 and 114.3 ± 11.4 m in the test and control group, respectively. A more detailed analysis revealed that the severity of the disease and atherosclerotic lesion levels were comparable among the control and test groups. However, the control group did not include patients with stage 2a. Therefore, a precise comparison between the subgroups regarding the severity of ischemia was made only in patients with stages 2b to 3 disease.
Evaluation of Safety
No AE, SAE, or significant laboratory abnormalities were observed in either study group during both treatment and follow-up period. No peripheral edema was observed. Local pathological reactions, including allergic, anaphylactic, and neoplastic reactions, were absent immediately after study drug administration, at 6 months after the onset of treatment, and during the extended follow-up period.
During the first 6 months, 3 events precluded the continuation of the study: 2 acute ischemic strokes (test group) with a positive outcome and 1 acute myocardial infarction with a fatal outcome (test group). Apparently, these events were not related to pCMV-vegf165 gene transfer as the construction used in the study has a proven local action. The results of toxicological studies showed no relationship between the study drug and AEs. 15
Tumor growth, eyesight disorders, and other pathological conditions that could indirectly suggest complications of gene therapy were not observed in patients throughout the study and during the 1.5-year follow-up period.
Evaluation of Efficacy
Primary End Point
Pain-free walking distance
The first changes in clinical characteristics among the patients of pCMV-vegf165 group were noticed by the patients themselves within 2 weeks after the onset of treatment. More notable changes were observed at 45 to 60 days. The initial PWD level in the test group was 135.3 ± 12.2 m, increasing to 284.7 ± 29.8 m at 6 months (Tables 2 and 3). The differences between the baseline and subsequent PWD values within the test group and differences between the test and control group were statistically significant starting from day 90. During the first 6 months of the study, there was an increasing trend of PWD values in 62 (85%) patients of the test group. During the long-term follow-up period, the value of PWD continued to increase in the test group. The increase in the mean distance that a patient could walk without pain was 149.4 m in the study group after 6 months (110.4%), while its value decreased by 1.5 m in the control group compared to the baseline. The tendency remained positive throughout the 2 years of monitoring: PWD increased in pCMV-vegf165 patients by 167.2% and 190.8%, that is, by 226.3 and 258.1 m, at 1 and 2 years, respectively, while no statistically significant changes were observed in the control group.
Results of Measurements of Primary and Secondary End Points.a

Abbreviations: ABI, ankle–brachial index; BFV, blood flow velocity; PWD, pain-free walking distance.
aMean and standard error of the mean.
bSelection is too small to calculate statistical significance.
Results of Measurements of Primary and Secondary End Points.a

Abbreviations: ABI, ankle–brachial index; BFV, blood flow velocity; PWD, pain-free walking distance.
aMedian and interquartile range (IQR).
The largest increase in the PWD was observed in patients with advanced stages of ischemia (severe claudication or ischemic rest pain), that is, stage 3: a 96.4-m increase (231.2%) at 6 months, a 228.3-m increase (547.5%) at 1 year, and a 345.3-m increase (828%) at 2 years. The PWD increased by 129.4 m (108.3%) in patients with stage 2b disease (initial PWD increased by 50-200 m). Such positive results remained stable throughout a 2-year follow-up period. The PWD increased by 290.0 m (90.6%) in patients with stage 2a disease at 6 months and by 660.0 m (206.2%) and 517.5 m (161.7%) at 1 and 2 years, respectively.
Depending on the localization of atherosclerotic lesions, the results were as follows: the 6 months results showed that patients with multifocal arterial lesions of the lower limbs benefited from gene therapy. The average increase in PWD values in these patients was 259.0 m (180.7%); the PWD increased by 431.7 m (301.2%) at 1 year and by 363.7 m (253.8%) at 2 years compared to baseline. The opposite results were obtained in the control group: PWD increased by 34.0 m (35.4%) at 6 months; however, during the follow-up period, the PWD value decreased by 56.0 m (−58.3%) and 66.0 m (−68.7%) at 1 and 2 years, respectively, compared to the baseline.
The PWD in test group patients with predominantly distal vascular lesions increased by 179.7 m at 6 months (132.4%), by 230.3 m (169.7%) at 1 year, and by 342.9 m (252.7%) at 2 years.
Secondary End Points
Characteristics of macrohemodynamics: ABI
Six months following the onset of the study, there were statistically significant changes in ABI in the test group (a 0.05 increase, P = .009). The ABI did not change in the control group. The differences in absolute values among the test and control groups (patients with rest pain included) at each visit, as well as an increase in the absolute values between the groups, were statistically insignificant (Tables 2-4). The long-term follow-up (2 years) results demonstrated a slight but stable improvement in ABI in test group patients.
Mean Values and Standard Error of the Mean (M ± m) of ABI and BFV in Patients With Ischemic Rest Pain in Test (pCMV-vegf165) and Control Groups.

Abbreviations: ABI, ankle–brachial index; BFV, blood flow velocity.
Characteristics of macrohemodynamics: BFV
The BFV in the pCMV-vegf165 group patients increased by 8.4 m/s within 6 months (average growth by 59.1%). Within 1 year, the value of BFV slightly decreased but remained 5.5 m/s higher than the baseline. At 2 years, the tendency remained positive. There were no statistically significant changes in BFV in patients of the control group (Tables 2 and 3).
Angiography
Angiograms were performed and assessed visually by the same experienced radiologist. Improvement in the collateral vascular bed was observed in 75% of patients who agreed to undergo angiography. Enhanced contrast filling of the microcirculatory bloodstream due to an increased diameter of collateral vessels was recorded in 12.5% of patients who underwent angiography. A moderate increase in the number of the newly formed collaterals was recorded in 37.5% of patients. Significant growth of the collateral vessels was registered in 37.5% of patients who underwent angiography. Neoangiogenesis may be attributed to the growth of new collaterals and possibly to the opening of previously nonfunctioning vessels (Figure 2). There were no clinically important laboratory abnormalities throughout the entire period of treatment and follow-up in patients of both groups.

Angiographic images, patient of the test group: (A) before treatment and (B) 6 months after pCMV-vegf165 gene transfer.
Quality of life
A statistically insignificant improvement in physical health was observed in patients of the test group. Mental health also slightly improved in patients of the pCMV-vegf165 group (Table 5). Control patients had a higher quality of life regarding mental health compared to patients who were treated with gene transfer.
Values of Physical and Mental Health in Patients of Test (pCMV-vegf165) and Control Groups According to SF-36 Questionnaire.

Discussion
The concept of gene therapy for paracrine vascular growth regulation, that is, therapeutic angiogenesis, began evolving after the pioneering works of Isner. 5,16,17 Gene therapy evolved due to the accomplishment of experimental and clinical trials which investigated different therapeutic genes. 3 A number of delivery vectors were used: viral (mainly adenoviruses) 3,7 and nonviral (mainly naked plasmids). 3,5,6,18 Majority of clinical trials demonstrated safety of both approaches of local administration of gene products at different dosage levels in terms of systemic allergic or anaphylactic reactions and the absence of neoplastic reactions, for example, proliferative retinopathy, vascular tumors, induction of dormant tumors, and so on. 3,18 However, data regarding the efficacy of gene therapy were more variable. Certain studies were considered a failure due to the chosen requirements regarding efficacy end points, such as the number of amputations or the survival curve, 19 heterogeneity of patients enrolled into the study, and selection of a therapeutic gene, for example, not the most promising candidate genes for angiogenesis.
Present study aimed to determine the safety and efficacy of the pCMV-vegf165 gene product in patients who were not suitable for surgical or endovascular revascularization. The absence of ischemic ulcers and necrotic lesions in patients (stage 4 according to Fontaine classification modified by A. V. Pokrovsky) allowed to study the effect of gene transfer in patients with viable limbs. Majority of previous studies enrolled patients with ulcers or gangrenes, which had a negative impact on further investigations of gene products and their effects or use in patients with moderate to severe claudication. 19 –21
Within the study (180 days) and follow-up period (another 1.5 years) neither of 3 study centers reported any adverse effects (AEs and SAEs) or other complications. The selected mode of pCMV-vegf165 administration at the selected dosage regimen was safe during the therapy and at least 2 years thereafter.
All lethal outcomes (5 in the test group and 2 in the control group) were attributed to acute myocardial infarction (Table 6). Peripheral arterial disease is an independent predictor of worse outcomes in patients with ischemic heart disease. We believe that there is no relationship between lethal outcomes and gene transfer in terms of this study. These findings correspond with the results of other studies of plasmid VEGFf165 gene products. 3,17,22
Number of Amputations and Patients Who Died During the Observation Period in Test (pCMV-vegf165) and Control Groups.

aFour amputations were performed in patients with ischemic rest pain at baseline.
bOne amputation was performed in patients with ischemic rest pain at baseline.
The evaluation of efficacy appeared more difficult. The use of gene therapy in patients who were not suitable for an open or endovascular revascularization allowed significant increases in PWD. This positive tendency was stable both during the first 6 months and the following 1.5 years. The PWD continued to increase at the end of the 2-year monitoring period. Improvement in PWD in the test group was compared to the changes in PWD in the control group: while the conventional therapy alone did not have major successful results, gene transfer had statistically significant positive effects (Tables 2 and 3). Unfortunately, our data cannot be compared to the results of similar studies, as most of the previous studies enrolled patients with critical lower limb ischemia who could not undergo the treadmill exercise testing. 7,8,11,23,24 Although the study design (not blinded, not placebo-controlled) contributed to comparison difficulties, we were able to notice that the number of test group patients with less severe stage of the disease increased, while a negative tendency was observed in the control group due to an increased number of patients with more advanced stages of the disease, including those which resulted in amputation (Figure 3). At 1 year following the onset of treatment, 4 of the total 5 amputations in the test group were performed in patients with pain at rest at baseline (Table 6). Of a total of 2 amputations in the control group, 1 was performed in a patient with ischemic rest pain at baseline. Limb loss was attributed to the disease progression leading to irreversible ischemia. Patients enrolled in the study were not suitable for revascularization, consequently performing an amputation was the only option left.
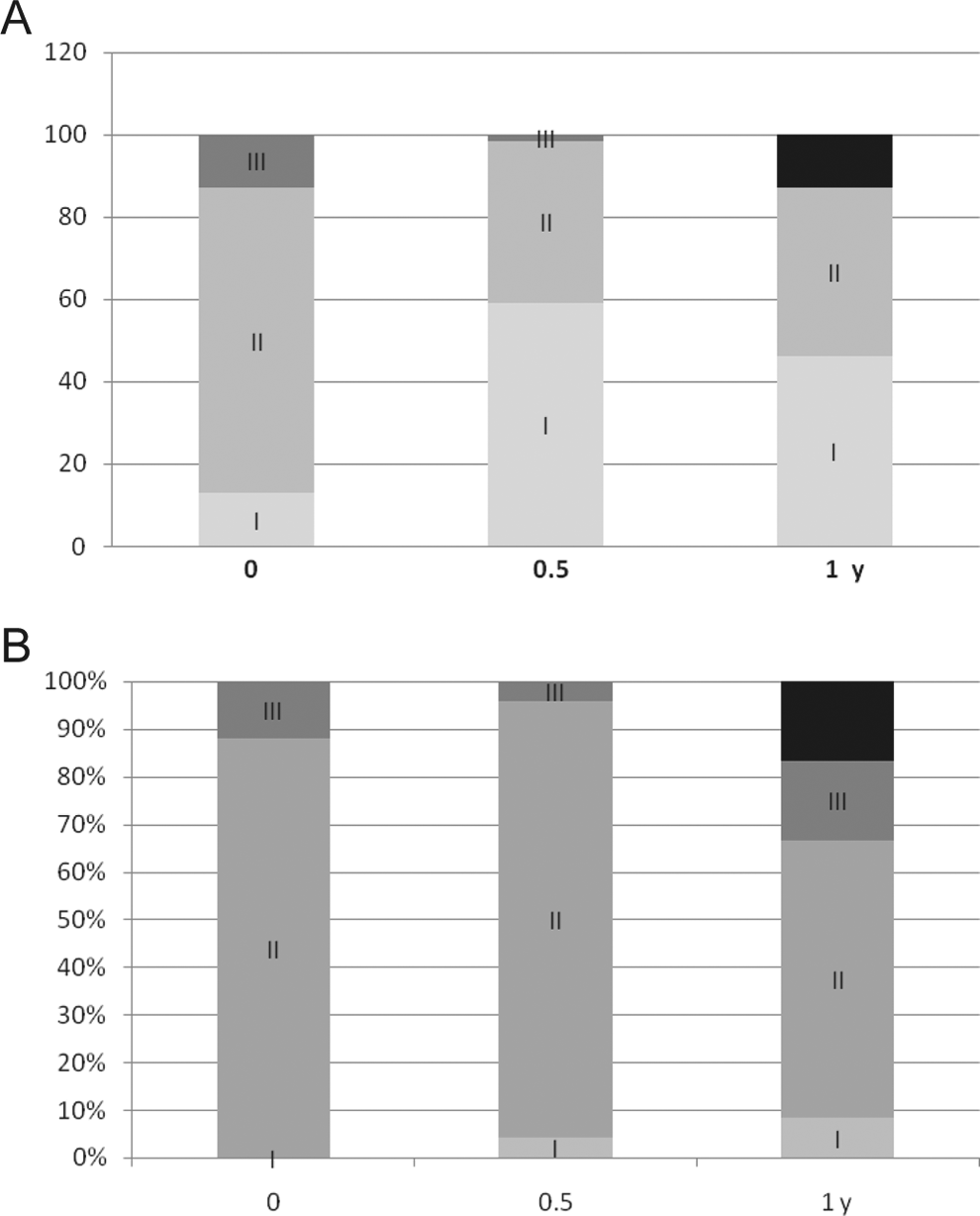
Patient intergroup ratio according to the stage of disease at baseline, at 0.5, and 1 year after administration: (A) test group (pCMV-vegf165) and (B) control group. I—stage 2a; II—2b; III—stage 3; and black—amputations.
Limb salvage rates at 2 years were 93.3% in the test group and 88% in the control group (Table 6). However, the differences were not statistically significant. So, we did not observe the amputation reduction in the test group. More observations are needed.
Clinical signs majorly improved in patients with distal or multifocal atherosclerotic lesion.
Changes in ABI and BFV were not significant which may be explained by the fact that the study drug is designed to induce angiogenesis at the microcirculatory level and does not affect macrohemodynamic. Nevertheless, slight improvement in ABI and BFW may be attributed to the general improvements in the collateral arterial flow and decrease in the peripheral arterial resistance.
Safety and efficacy of the studied pCMV-vegf165 gene product marketed as “Neovasculgen” were demonstrated in selected patients throughout a 2-year follow-up period. 15 Within this period, we were able to track both limb salvage and patient survival (Table 6). Gene therapy with pCMV-vegf165 did not affect mortality. Limb salvage largely depended on the presence of rest pain at baseline. However, these parameters should be analyzed in larger cohorts of patients.
Despite the marked improvement in claudication symptoms in the test group patients, gene therapy did not significantly affect the quality of life. Mental health score was higher in the control group as compared to those of the test group. Apparently, these findings were attributed to the initial differences in baseline SF-36 scores among the patients of both groups (Table 5) and presence of concomitant pathology which decreased the positive impact of PWD increase in the overall quality of life. Such findings may indicate the presence of an underlying depressive disorder in patients with chronic lower limb ischemia.
Conclusion
The use of the plasmid DNA gene product encoding VEGF165 (pCMV-vegf165) in combination with standard pharmacological therapy significantly improves clinical signs of claudication in patients with chronic lower limb ischemia. A 2-year follow-up demonstrated a stable PWD improvement. The results of the study were sufficient for the registration of “Neovasculgen” as a drug which is used in the treatment of patients with moderate to severe claudication due to stage 2a to 3 atherosclerotic chronic lower limb ischemia. However, further studies enrolling larger groups of patients are needed to completely evaluate the effects of pCMV-vegf165 gene transfer in patients with pain at rest due to peripheral atherosclerosis, ischemia caused by diabetes mellitus or autoimmune disorders, and those who undergo peripheral arterial revascularization.
Footnotes
Author Contributions
R. Deev contributed to design, contributed to analysis and interpretation, drafted the article, critically revised the article, and gave final approval. I. Bozo contributed to analysis and interpretation, drafted the article, critically revised the article, and gave final approval. N. Mzhavanadze contributed to acquisition and analysis, drafted the article, critically revised the article, and gave final approval. D. Voronov contributed to acquisition and analysis, critically revised the article, and gave final approval. A. Gavrilenko critically revised the article and gave final approval. Y. Chervyakov contributed to acquisition, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. I. Staroverov contributed to acquisition, critically revised the article, and gave final approval. R. Kalinin contributed to acquisition, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. P. Shvalb contributed to conception and design, contributed to acquisition, critically revised the article, and gave final approval. A. Isaev contributed to conception and design, contributed to analysis and interpretation, critically revised the article, and gave final approval.
Acknowledgments
Authors would like to thank Prof S. L. Kiselev for his contribution in developing the gene construction and participating in the studies.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A. A. Isaev, I. Ya. Bozo, and R. V. Deev are employees of the OJSC “Human Stem Cells Institute.” A. A. Isaev is shareholder of the OJSC “Human Stem Cells Institute.”
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: OJSC “Human Stem Cells Institute” (Moscow, Russia) sponsored the clinical trial.
