Abstract
Introduction:
Recent clinical studies have reported the potential benefit of an early mineralocorticoid receptor (MR) blockade with potassium canrenoate (PC) on ventricular arrhythmias (VAs) occurrence in patients experiencing an ST-segment elevation myocardial infarction (STEMI). However, most of the electrophysiological properties of PC demonstrated to date have been investigated in normoxic conditions, and therefore, in vitro experiments during an acute myocardial ischemia–reperfusion were lacking.
Materials and Methods:
We used rabbit in vitro models and standard microelectrode technique to assess the electrophysiological impact of PC during myocardial ischemia–reperfusion, including right ventricle mimicking the “border zone” existing between normal and ischemic/reperfused areas (1 µmol/L, 10 and 100 nmol/L), isolated right ventricle, and sinoatrial node (SAN) experiments (1 µmol/L, respectively).
Results:
During ischemia–reperfusion, acute superfusion of PC 100 nmol/L prevented the increase in action potential (AP) duration at 90% of repolarization (APD90) dispersion between ischemic and nonischemic areas and in VAs occurrence induced by aldosterone 10 nmol/L (86 ± 3 vs 114 ± 4 milliseconds for aldosterone alone, P < .05). Potassium canrenoate also induced conduction blocks and significantly decreased Vmax during simulated ischemia (from 25 ± 5 to 12 ± 4, 14 ± 3, and 14 ± 5 V/s, respectively, for PC 1 µmol/L, 100, and 10 nmol/L, P < .05). Potassium canrenoate 1 µmol/L demonstrated cycle length (CL)-dependent effects on APD90 and on Vmax, and it also reduced SAN beating CL (from 446 ± 28 to 529 ± 24 millisecond, P < .05).
Conclusion:
Our experimental study highlights new evidence for an antiarrhythmic impact of PC during myocardial ischemia–reperfusion via multiple channels modulation. These results are in line with recent clinical trials suggesting that an early MR blockade in STEMI may be preventive of VAs.
Introduction
The deleterious impact of aldosterone in patients with heart failure and systolic left ventricular dysfunction is now well demonstrated.1 –3 Conversely, the deleterious role of high aldosterone levels, associated with sudden cardiac death (SCD) and ventricular arrhythmias (VAs) occurrence, at the acute phase of ST-segment elevation myocardial infarction (STEMI), has been more recently highlighted. 4 Therefore, the possible benefit of mineralocorticoid receptor (MR) blockade early after STEMI onset and even in preserved systolic left ventricular function has emerged. 5
Based on studies published during 1970s to 1980s, potassium canrenoate (PC; Soludactone), the principal active metabolite of spironolactone, is still approved in France (not United States) for the prevention of VAs.6 –9 Gradually, this indication had been forgotten in clinical daily practice. The use of PC as a preventive antiarrhythmic drug in STEMI was revisited recently5,10 and is actually evaluated in the large multicenter study Aldosterone Blockade Early After Acute Myocardial Infarction (ALBATROSS) 11 the results of which are eagerly awaited and may lead to changes in STEMI management. However, the underlying electrophysiological properties explaining such antiarrhythmic properties are to date poorly understood, and although some electrophysiological effects of PC and spironolactone were already described,12 –15 the exact impact of PC in an in vitro model of myocardial ischemia–reperfusion was lacking. In fact, compared to other MR antagonists, PC was little studied, and its antiarrhythmic properties were only investigated in patch-clamp experiments 12 or in normoxic in vitro 13 or in vivo 15 models. At last, the cycle length (CL) dependence of PC activity and its action on the sinoatrial node (SAN) were never investigated.
Therefore, the purpose of this study was to evaluate the electrophysiological impact and, therefore, the exact antiarrhythmic properties of PC during an acute myocardial ischemia–reperfusion. We used in vitro models and standard microelectrode techniques of high clinical relevance including rabbit right ventricle mimicking the “border zone” existing between normal and ischemic–reperfused areas, isolated rabbit right ventricle, and rabbit SAN experiments.
Methods
Heart Sampling
Care of the animals complied with the European directive of 2010, published in the French Official Journal in February 2013. New Zealand white rabbits of either sex weighing 1.5 to 2.0 kg were euthanized under anesthesia with sodium pentobarbital 125 mg/kg within the marginal vein. The hearts were quickly removed after thoracotomy and placed in a cardioplegic solution at room temperature and bubbled with 95% O2 and 5% carbon dioxide (CO2).
Superfusion Solutions and Chemicals
Normal Tyrode solution was oxygenated with 95% O2 and 5% CO2 and kept at 37°C. The composition of this Tyrode solution was (in mmol/L) Na+ 135, K+ 4, Ca2+ 1.8, Mg2+ 1, H2PO4 − 1.8, HCO3 − 25, Cl− 117.8, and glucose 11. The pH was 7.4. The cardioplegic solution used for dissection differed from normal Tyrode by higher glucose (from 11 to 55 mmol/L) and hyperkalemia (from 4 to 30 mmol/L). The modified, ischemia-simulating Tyrode solution differed from normal by elevated K+ concentration (from 4 to 12 mmol/L), decreased HCO3 − concentration (from 25 to 9 mmol/L) leading to a decrease in pH (from 7.4 to 7.0), a decrease in PO2 induced by replacement of 95% O2 and 5% CO2 by 95% N2 and 5% CO2, and withdrawal of glucose. All chemicals were purchased from Sigma Aldrich (Saint Quentin Fallavier, France).
Data Acquisition and Analysis
Except when specified, the myocardial strips were stimulated at a basic CL of 1000 milliseconds (frequency = 1 Hz) via a bipolar silicon-coated steel wire electrode. Rectangular pulses of 2 milliseconds in duration and twice diastolic threshold intensity (around 2-2.5 V) were delivered by a programmable stimulator (SMP 310, Biologic, Grenoble, France). Transmembrane potentials were recorded by the use of intracellular glass microelectrodes pulled from borosilicate filament tubes (GC 200F-15; Phymep, France) on a single-barreled microelectrode puller (Narashige, distributed by OSI, France). Microelectrodes were filled with KCl 3 mol/L (tip resistance ranging from 10 to 30 MΩ) and coupled to Ag/AgCl microelectrode holders leading to a home-built high impedance capacitance-neutralizing amplifier. The following action potential (AP) characteristics were automatically stored and measured by a system of cardiac AP automatic acquisition and processing devices (DATAPAC, Biologic, Grenoble, France): resting membrane potential (RMP), AP duration at 90% of repolarization (APD90), and maximal upstroke velocity (Vmax). Whenever possible, the same impalement was maintained throughout the experiment; however, when it was lost, readjustment was attempted. If the readjusted parameters deviated ≤10% from the previous ones, experiments were continued; otherwise, they were discarded.
In vitro Model of Rabbit “Border Zone”
This in vitro model of “border zone” used for experiments as the data acquisition and analysis process and the experimental protocol used have been already described in detail.16 –18 Briefly, a custom-made dual perfusion chamber is bisected by a thin latex membrane, which is centrally perforated at its bottom to allow 1 single right ventricular strip to be passed carefully through and so to be divided into 2 zones, called the normal zone (NZ) and the altered zone (AZ), respectively. A simulated ischemia was induced in the AZ during 30 minutes by superfusion with the modified Tyrode solution, and then a 30-minute reperfusion period was induced by perfusion of normal Tyrode solution. The NZ remained constantly perfused with normal Tyrode solution. The drugs administration began concomitantly in the 2 zones with simulated ischemia and continued until the end of the reperfusion period. The preparations were randomly assigned to 6 groups, controls (n = 10), PC 1 µmol/L (n = 6), 100 nmol/L (n = 5), and 10 nmol/L (n = 6), aldosterone 10 nmol/L (n = 5), and PC 100 nmol/L (n = 5). Conduction blocks and spontaneous premature ventricular contractions (PVCs) were recorded and PVCs were classified in extrasystoles (1-3 continuous PVCs), salvos (4 to 9 continuous PVCs), and sustained activities (≥10 continuous PVCs). 17 To evaluate the ischemia-induced dispersion of repolarization, APD90 dispersion between NZ and AZ was calculated at the end of the simulated ischemia period as APD90 NZ − APD90 AZ.
Cycle Length Dependence of PC on Isolated Right Rabbit Ventricle
Cycle length-dependent effects of PC were tested on APD and Vmax in isolated right rabbit ventricle preparations using standard microelectrode. The ventricles were separated from the heart. The left ventricle was ablated, while the right ventricle was opened and pinned down, with the endocardial surface upward, in a superfusion chamber perfused with normal Tyrode solution. Each right ventricular preparation was initially stimulated at a basic CL of 1000 milliseconds during 15 minutes before determination of baseline AP characteristics. Then pacing CL was sequentially and instantly varied between 300 and 2000 milliseconds. After each CL changes, AP characteristics were stored after 25 APs. Experiments began with taking control records, and then the preparations were superfused with PC 1 µmol/L (n = 5) and the measurements were repeated at each pacing CL.
In Vitro Sinoatrial Rabbit Node Experiments
The right atrium and the superior vena cava were separated from the heart and pinned down, with the endocardial surface upward, in a superfusion chamber perfused with normal Tyrode’s solution. No electrical stimulation was applied, allowing recording of the free SAN activity. After 15 minutes of stabilization, PC 1 µmol/L (n = 6) was added in the superfusion solution during 30 minutes and then PC was washed out with normal Tyrode during 30 minutes. During the entire experiment, the spontaneous heart CL (milliseconds) was automatically stored.
Statistical Analysis
Results were expressed as means ± standard error of the mean (SEM). All data were tested for normality by Shapiro-Wilk test (normality accepted for P > .05). Fisher exact test paired or unpaired Student t test or nonparametric Wilcoxon test was used as appropriate. In all analyses, P < .05 was considered statistically significant.
Results
Electrophysiological Effects of PC on the “Border Zone”
Effects of PC on AP parameters
In normoxic conditions (NZ), after 1 hour of superfusion, PC alone at either concentration did not significantly affect any and not other APs parameters. Potassium canrenoate of 100 nmol/L prevented the APD90 lengthening (from 163 ± 2 to 155 ± 2 milliseconds, P < .05) and the RMP hyperpolarization (from –93 ± 4 to –86 ± 1 mV at 60 minutes, P < .05) induced by aldosterone 10 nmol/L alone.
During the simulated ischemia period (Table 1), PC significantly decreased Vmax (from 25 ± 5 to 12 ± 4, 14 ± 5, and 14 ± 3 V/s, respectively, for PC 1 µmol/L, 100, and 10 nmol/L, P < .05, Figure 1) without affecting RMP and APD90. Potassium canrenoate 100 nmol/L prevented the APD90 shortening induced by aldosterone 10 nmol/L alone.

Class I antiarrhythmic effects of potassium canrenoate. Effects of potassium canrenoate 1 µmol/L, 100, and 10 nmol/L on conductions blocks occurrence (panel A) and on Vmax (panel B) during the simulated ischemia period. Data are expressed as percentage of preparations with disturbances. Comparisons were made between control group and pharmacological groups. K+ Canr indicates potassium canrenoate; Vmax, maximal upstroke velocity.
Electrophysiological Effects of Potassium Canrenoate on Action Potential Parameters in Ischemic–Reperfused Conditions (AZ) in the “Border Zone” Model.a

Abbreviations: SEM, standard error of the mean; RMP, resting membrane potentiel; Vmax, maximal upstroke velocity; APD90, action potential duration at 90% of repolarization; AZ, altered zone; ms, millisecond.
aValues are mean ± SEM.
b P < .05 vs control values.
During the reperfusion period (Table 1), PC did not significantly affect any AP parameters. Potassium canrenoate of 100 nmol/L prevented the APD90 lengthening and the RMP hyperpolarization induced by aldosterone 10 nmol/L alone.
Effects of PC on APD90 dispersion
At baseline, in normal conditions and before inducing simulated ischemia, there was no significant APD90 dispersion between the 2 regions in all the groups. Ischemic-like superfusion induced a significant increase in APD90 dispersion (P < .05) in all groups. Potassium canrenoate alone at either concentration did not modify APD90 dispersion. Potassium canrenoate 100 nmol/L prevented the increase in APD90 dispersion induced by aldosterone 10 nmol/L alone (86 ± 3 vs 114 ± 4 millisecond, P < .05, Figure 2).

Representative action potentials (APs; top), spontaneous arrhythmias, and conduction blocks (bottom) recordings obtained simultaneously in normal zone (NZ) and altered zone (AZ) in the presence of aldosterone (panel A, left) and aldosterone + potassium canrenoate (panel B, right), in same cell, during simulated ischemia. AP duration at 90% of repolarization (APD90) dispersion between NZ and AZ (ΔAPD90) was measured at the end of the simulated ischemia period as APD90 NZ − APD90 AZ and is expressed as mean ± standard error of the mean (SEM).
Effects of PC on of spontaneous PVCs and conduction blocks occurrence
Results are summarized in Table 2. At baseline, in normal conditions, spontaneous PVCs were observed in none of the groups. During both simulated ischemia and reperfusion periods, PC alone did not modify PVCs occurrence. During the reperfusion period, PC 100 nmol/L prevented increase in PVCs occurrence induced by aldosterone 10 nmol/L (Figure 2).
Effect of Potassium Canrenoate on APD90 Dispersion, Incidence of Spontaneous PVCs and Conduction Blocks in the “Border Zone” Model.a

Abbreviations: PVCs, premature ventricular contractions; APD90, action potential duration at 90% of repolarization; NZ, normal zone; AZ, altered zone; ms, millisecond; s, second.
aValues are percentage of preparations with disturbances. APD90 dispersion was calculation as APD90 NZ − APD90 AZ.
b P < .05 vs controls.
Potassium canrenoate at either concentration significantly increased the incidence of ischemia-induced conduction blocks (from 30% to 100%, 100%, 83%, and 80%, respectively, for PC 1 µmol/L, 100 nmol/L, 10 nmol/L, and PC 100 nmol/L + aldosterone 10 nmol/L; P < .05; Figure 1) and anticipated their occurrence time (from 1593 ± 165 to 775 ± 117 and 1167 ± 174 seconds for PC 1 µmol/L and 100 nmol/L; P < .05).
Cycle Length Dependence of PC
Potassium canrenoate demonstrated CL dependence effects on APD90 and Vmax. Potassium canrenoate of 1 µmol/L significantly increased APD90 proportional to the CL except for physiological heart CL (Figure 3). PC also significantly decreased Vmax for high heart CL.
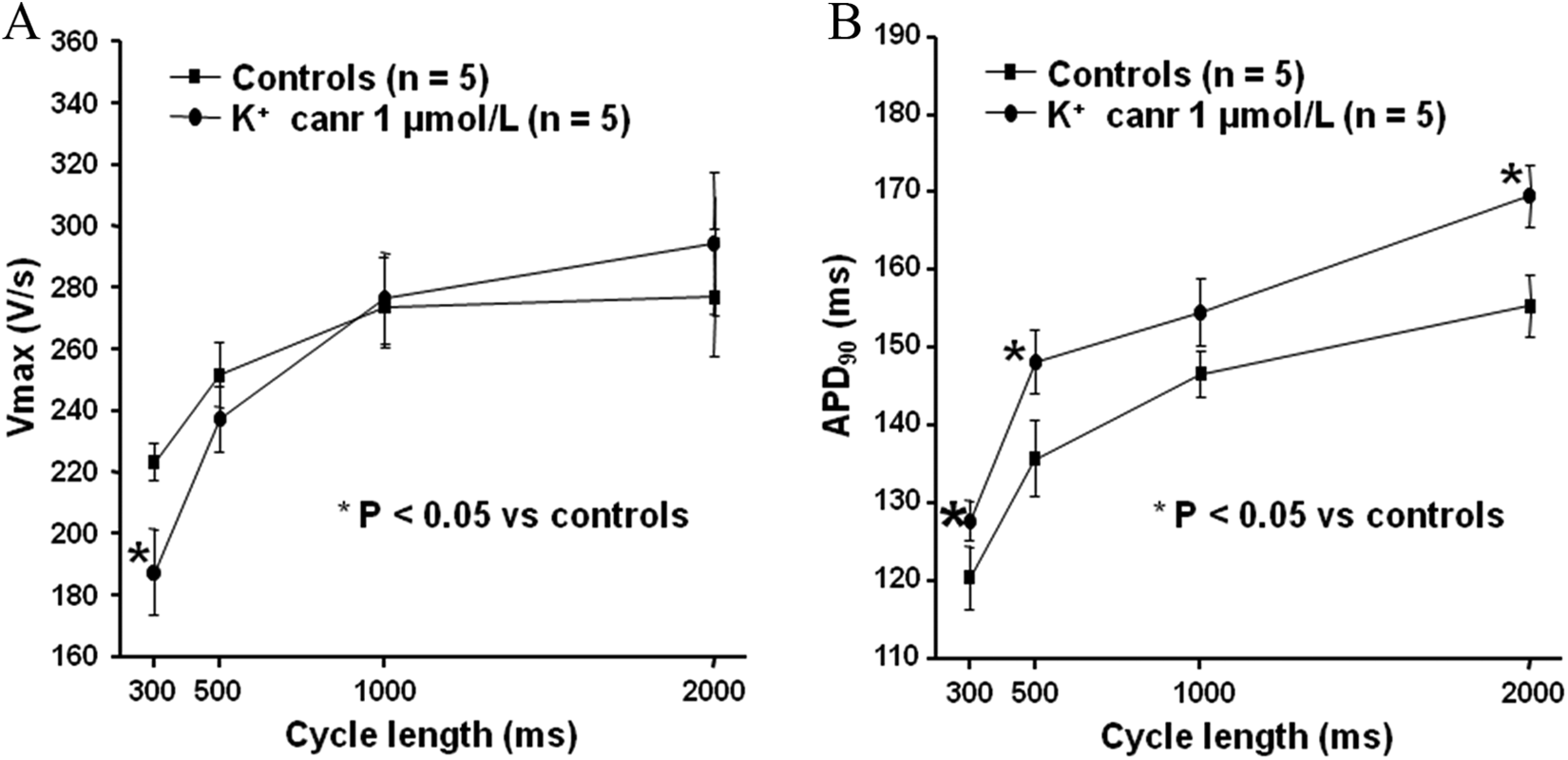
Cycle length dependence of potassium canrenoate 1 µmol/L on maximal upstroke velocity (Vmax; panel A) and on action potential duration at 90% of repolarization (APD90; panel B) in normoxic conditions. K+ Canr indicates potassium canrenoate.
Spontaneous SAN Activity Modulation by PC
Typical SAN spontaneous APs with a diastolic depolarization (DD) were recorded (Figure 4). Baseline AP parameters (without drug) were RMP = –62 ± 3 mV, Vmax = 96 ± 24 V/s, APD90 = 89 ± 7 milliseconds, and DD slope = 22.3 ± 9.8 V/s (n = 6). Free beating CL was 446 ± 28 milliseconds (n = 6), in accordance with previous reports. 19 After 30 minutes of application of PC of 1 µmol/L, the spontaneous beating CL increased by 19% ± 2% (n = 6, P < .05) and the DD slope reduced by 21% ± 4% (n = 6, P < .05) without significant variation in the remaining AP parameters (Figure 4).
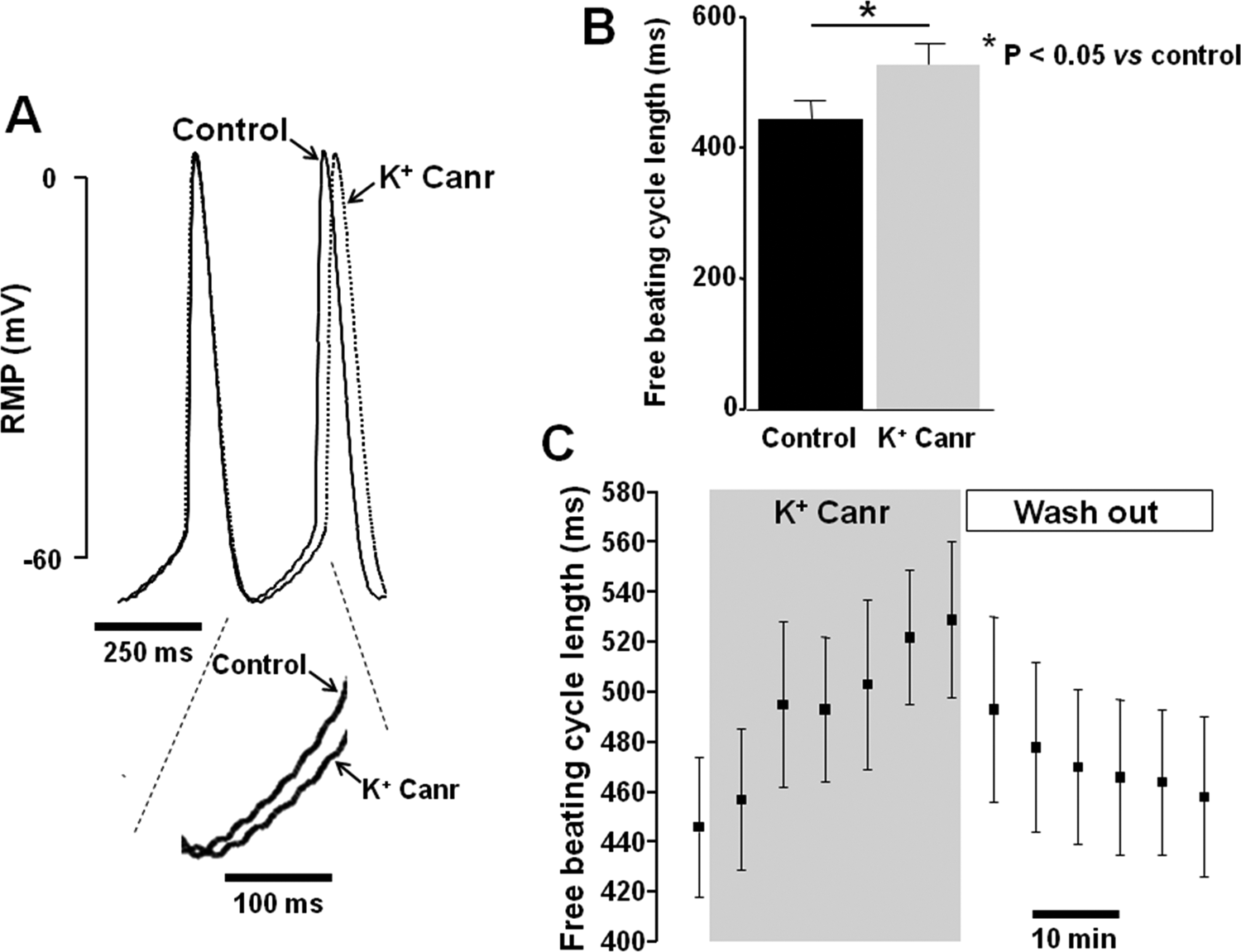
Effect of 1 µmol/L potassium canrenoate on rabbit sinoatrial node. A, Spontaneous action potentials (Aps) in the absence (control) or presence of potassium canrenoate 1 µmol/L in the same experiment. Spontaneous beatings in both conditions are overlaid. Note the reduction in diastolic depolarization (DD) slope in the bottom magnification. B, Average free beating cycle length measured in the presence of potassium canrenoate mol/L. C, Spontaneous beating cycle length for 5-minute periods plotted as a function of time. Values are expressed as mean ± standard error of the mean (SEM). Potassium canrenoate 1 µmol/L was superfused, as highlighted in gray. K+ Canr indicates potassium canrenoate; RMP, resting membrane potential.
Discussion
The main findings of this study may be summarized as follows: (1) during ischemia–reperfusion, PC prevented PVCs induced by aldosterone via multiple antiarrhythmic properties; (2) PC prevented the increase in APD90 dispersion induced by aldosterone; (3) PC induced conduction block and decreased Vmax during simulated ischemia; and (4) acute superfusion of PC reduced SAN beating rate.
Because of its aqueous solubility, PC is used as an injectable nonselective MR antagonist with similar therapeutic activity and metabolites to spironolactone. 20 The PC as an antiarrhythmic drug enjoyed its heydays during the 1970s with the studies of Yeh and colleagues in the context of digitalis-induced VAs. 21 Thereafter, just a few investigations have continued to study antiarrhythmic properties of PC.7 –9 Due to its easy administration at the acute phase of STEMI and the potential benefit of early MR blockade in STEMI in order to reduce the occurrence of SCD and VAs,5,10,11 PC’s use as an antiarrhythmic drug was revisited recently.5,11 However, the underlying electrophysiological properties occurring during acute myocardial ischemia–reperfusion were poorly studied. The PC concentrations used in clinical practice are around 1 to 10 µmol/L. 20 Thereby, we initially choose to use a PC concentration of 1 µmol/L in our 3 models and because we observed electrophysiological effects at this concentration, we also decided to test lower concentrations (10 and 100 nmol/L) in our “border zone” model.
In our “border zone” experiments, PC successfully prevented aldosterone-induced VAs during the reperfusion period. These results are in accordance with recent clinical trials. 5 Interestingly, PC alone did not decrease occurrence of PVCs compared with controls in our “border zone” model. This may be explained by the low incidence of PVC in our control group. The preventive antiarrhythmic effect of PC may be related to classes I and III antiarrhythmic properties observed in our experiments. In fact, PC-induced conduction blocks anticipated their occurrence time and decreased Vmax during simulated ischemia, thus evoking class I properties. A modulation of AP propagation via an impact of PC on cardiac connexin seems to be not involved in our experiments regarding the effect of PC on Vmax (Table 1). This inhibition of ventricular Na+ channels was already demonstrated in previous in vitro and normoxic observations.13,15,22 Regarding our results (moderate Vmax depression indicating a moderate phase 0 depression and absence of AP prolongation, Table 2), PC seems to be likely a class Ib agent, but further experiments are required to confirm this hypothesis. Interestingly, PC decreased Vmax only in AZ during the simulated ischemia period. The potentiating effect of our strong and prolonged simulated ischemia and our experimental model which was reported to be less sensitive to highlight Vmax variations may explain such findings. Moreover, the physiological stimulation CL (1000 milliseconds) imposed in our experiments may not allow to observe class I antiarrhythmic effects as they are typically expressed at high heart rate. 23
In our experiments, PC prevented the increase in APD90 dispersion between ischemic and nonischemic areas induced by aldosterone. The dispersion of repolarization is a well-known arrhythmia substrate, leading to the development of reentrant VAs. 24 This class III antiarrhythmic property of PC may be related to the previously demonstrated effect on rapid (IKr ) and slow (IKs ) components of the delayed rectifier K+ currents in guinea pig ventricular myocytes. 12 Interestingly, PC did not decrease APD90 in our “border zone” experiments as we expected. The physiological stimulation CL (1000 milliseconds) imposed in our experiments may be not compatible with the class III antiarrhythmic properties of PC, which seem to be less or not effective at physiological heart rate.
CL dependence is a characteristic property of antiarrhythmic drugs belonging to the Vaughan William classes I and III. 25 Interestingly, as typical class I, PC’s effect on Vmax is most marked at fast heart rates and nearly nonexistent at slow or physiological heart rates. PC also exerts a reverse CL dependence regard to the AP lengthening (Figure 3). Typical class III drugs exhibit reverse rate dependence regarding APD lengthening. 25 This reverse rate dependence of the APD lengthening is undesirable because it minimizes drug effects on repolarization during VAs and enhances their proarrhythmic potential at normal rates. 25 Unlike typical class III drugs, PC presents atypical class III properties with the prolongation of the APD most marked at both high and slow heart rates that could be explained by the associated class I effects. Finally, via multichannel properties, PC exerts classes III and I effects at high heart rates, class III effects at slow heart rates, and neutral effect at physiological heart rates.
In our experiments, PC also modulated SAN beating rate. The APs in SAN cells are triggered by a slow DD driving the membrane potential over the threshold of Ca2+ currents (I
Ca, T, I
Ca, L) activation, responsible for the AP upstroke.
26
The DD is due to the activation of the funny current (I
f)
27
associated with the activation of the inward Ca2+-dependent current with the major contribution of Na+/Ca2+ exchange.
28
The effect of PC on SAN is not surprising because canrenone (the main metabolite of both PC and spironolactone) is known to reduce
Study Limitations
The study was performed in rabbit myocardium, which differs from human myocardium while rabbits have repolarizing ion current characteristics similar to humans. 31 We did not use left ventricular myocardium because of its excessive thickness which might be associated with the risk of poor perfusion and subsequent ischemia in the center of the tissue. Finally, we did not test concentration higher than 1 µmol/L of PC in our models. However, it would be interesting to test higher concentration (the concentrations used in clinical practice are between 1 and 10 µmol/L20) in a forthcoming work to clarify precisely the electrophysiological effects of PC regarding its plasma concentrations, particularly in case of a favorable issue of the ALBATROSS trial.
Conclusions
During ischemia–reperfusion and at clinical concentrations, PC exhibited preventive antiarrhythmic properties via multiple channel modulation, including classes I and III antiarrhythmic effects and the modulation of SAN beating rate. Our experimental study highlights new evidence for an antiarrhythmic impact of PC during myocardial ischemia–reperfusion, which was hypothesized in previous normoxic experimental models.12 –15 These results are also in line with recent clinical trials, 5 suggesting that an early MR blockade in STEMI may be preventive of VAs. The ALBATROSS trial should soon provide a definitive answer to this hypothesis. 11
Our data also suggest that the timing of PC administration could be critically important as it would be necessary to administer the drug early enough during acute ischemia in order to benefit from lower SAN beating rates over the ischemic period and thus reduce the reperfusion VAs occurrence.
Footnotes
Author Contribution
J. Alexandre contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. F. Beygui contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. P. E. Puddu contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy; A. Manrique contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. R. Rouet contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. P. Milliez contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. All the authors wrote the manuscript, designed and performed the research, and analyzed the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Alexandre has received a research grant from the Fédération Française de Cardiologie.
