Abstract
Effective treatment of high blood pressure (BP) represents a key strategy for reducing the burden of hypertension-related cardiovascular and renal diseases. In spite of these well-established concepts, hypertension remains poorly controlled worldwide. In order to improve BP control in patients with hypertension, several interventions have been proposed, among which (1) preferred use of more effective, sustained, and well-tolerated antihypertensive drug aimed to ensure adherence to prescribed medications and (2) extensive use of rational, integrated, and synergistic combination therapies, even as first-line strategy, aimed to achieve the recommended BP targets. Within the possible antihypertensive drug classes currently available for the clinical management of hypertension, both in monotherapy and in combination therapy, drugs inhibiting the renin–angiotensin system and calcium channel blockers (CCBs) have demonstrated to be effective and safe in lowering BP levels and achieving the recommended BP targets with a good tolerability profile. In particular, CCBs have been one of the most widely used classes of antihypertensive agents in the last 20 years, based on their effectiveness in reducing BP levels, good tolerability, and abundant evidence on reducing cardiovascular and renal consequences of hypertension. This article provides an updated overview of the evidence supporting the use of CCBs-based antihypertensive regimen, both in monotherapy and in combination therapies with different classes of antihypertensive drugs.
Keywords
Introduction
Hypertension is a major modifiable risk factor, which significantly and independently increases the risk of developing major cardiovascular, cerebrovascular, and renal complications. 1 On the other hand, an effective treatment of hypertension substantially reduces the risk of developing such complications and improves cardiovascular prognosis. 2 However, the control of blood pressure (BP) remains largely unsatisfactory worldwide. 3,4 In particular, analyses of data collected across different Western countries on BP control rates have consistently and independently confirmed that only 20% to 30% of treated patients with hypertension achieve the recommended BP targets. 5 -8 Inadequate BP control increases the risk of developing hypertension-related cardiovascular diseases, including myocardial infarction, ischemic stroke, end-stage renal disease, and congestive heart failure, and explains the persistently high burden of cardiovascular death related to hypertension. 9
Current recommendations from international guidelines stated that in all patients with hypertension, it is important to reduce BP until systolic and diastolic BP values below 140/90 mm Hg are achieved. 10,11 These BP goals are recommended for all adult patients with hypertension, regardless of gender, age, ethnicity, or other concomitant clinical conditions. 10,11 Results of large, randomized, controlled clinical trials demonstrated that it is possible to reach these BP targets in large proportions of treated patients with hypertension having different cardiovascular risk profiles. 12 -14 Indeed, the design of these clinical trials systematically included antihypertensive therapies based on drugs inhibiting the renin–angiotensin system (RAS) and calcium channel blockers (CCBs) compared to β-blockers and diuretics. 12 -14 On the basis of these findings, a preferred use of these antihypertensive drug classes have been pursued by recent hypertension guidelines, in order to bridge the gap between the attained and expected BP control rates, to ensure adequate adherence and persistence to prescribed medications and to improve cardiovascular outcomes in treated patients with hypertension. 10,11
Being the vast majority of these evidence based on the use of dihydropyridinic CCBs, these drugs are now recommended both as first-line therapy and as an ideal partner for dual or triple combination therapies for the clinical management of hypertension and hypertension-related comorbidities. 10,11
The aim of this article is to provide an updated overview of the evidence supporting the use of CCBs-based antihypertensive regimen, both in monotherapy and in combination therapies with different classes of antihypertensive drugs.
General Considerations on CCBs
Within the drug class, CCBs showed several important differences from the pharmacokinetic
15
and pharmacodynamic
16
point of views as well as for selectivity and duration of pharmacological action,
17
-19
although sharing the same ability to interact with

Schematic representation to explain the molecular mechanisms of actions of calcium channel blockers (CCBs).
Several classifications have been proposed to distinguish different compounds within this drug class. Among these, CCBs may be classified into 3 groups according to their selectivity for interactions with either cardiac or vascular (or both)
Classification of Different Compounds Within the CCB Drug Class, According to Their Selectivity for Interactions With Either Cardiac or Vascular (or Both)

Abbreviations: SR, slow release; GITS, gastrointestinal-transport system; ER, extended release.
The first generation of the compounds of these 3 groups included nifedipine, verapamil, and diltiazem, respectively. They were characterized by short-acting therapeutic actions, which have limited their clinical effectiveness due to potential risk of drug-related adverse reactions (eg, peripheral edema, reflex tachycardia, skin reactions). Following the availability of the first-generation molecules, over the years several other compounds have been developed for widespread use in treating hypertension and cardiovascular diseases. In particular, among dihydropyridinic agents, second-generation CCBs, including manidipine, 22,23 felodipine, 24,25 and nicardipine, 26 and third-generation CCBs, including lacidipine, 27 lercanidipine 28 barnidipine, 29 and amlodipine, 30 were characterized by high selection for vascular calcium channels and favorable pharmacokinetic and pharmacodynamic profile.
From a clinical point of view, dihydropyridinic CCBs are considered one of the first-line therapeutic options to treat hypertension and reduce hypertension-related cardiovascular morbidity and mortality. 10,11
According to the recommendations from the European guidelines, 10 all CCBs can be effectively and safely used for the treatment of hypertension, both in monotherapy and in combination therapies (Figure 2). In particular, according with the compelling indications (Table 2), they are now recommended for lowering BP levels in black individuals, elderly patients with isolated systolic hypertension as well as in patients with hypertension having metabolic syndrome, cardiac (left ventricular hypertrophy) or vascular (atherosclerosis) organ damage, previous stroke, or peripheral artery disease. In addition, nondihydropyridinic CCBs are also recommended for treating patients with hypertension having angina pectoris and for those with atrial fibrillation, with the aim of achieving ventricular rate control. 10 Finally, they are recommended for treating gestational hypertension or for preventing eclampsya. 10 On the other hand, dihydropyridinic CCBs are contraindicated for those patients with hypertension having tachycardia or congestive heart failure, while nondihydropyridinic CCBs are contraindicated for those patients with hypertension having grade 2 to 3 atrioventricular block, severe left ventricular dysfunction, or congestive heart failure.
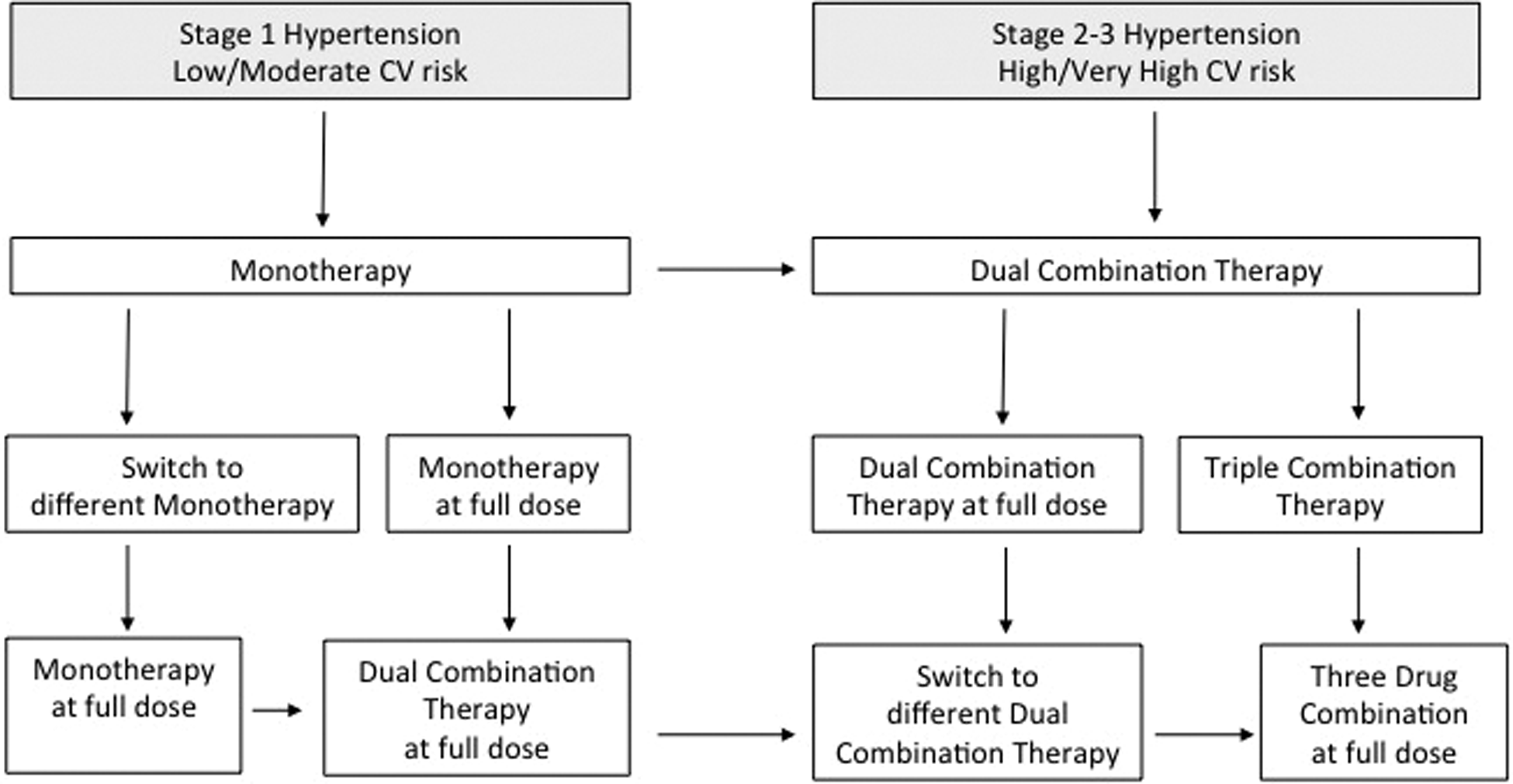
Schematic representation illustrating the different therapeutic options (monotherapy vs combination therapy) for the clinical management of hypertension, according to current European guidelines. 10
Compelling Indications and Contraindications for Using CCBs in the Clinical Management of Hypertension, According to Current European Guidelines. 10

Abbreviations: CCBs, calcium-channel blockers; ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blockers; BB, β-blockers, LVH, left ventricular hypertrophy; CV, cardiovascular, ISH, isolated systolic hypertension; AV, atrioventricular.
According to the recommendations from the evidence-based guidelines in the United States, 11 some selected dihydropyridinic CCBs (ie, amlodipine 2.5-10 mg and nitrendipine 10-20 mg) and only 1 nondihydropyridinic CCB (diltiazem extended release120-360 mg) may be used for lowering BP levels and reducing incidence of hypertension-related cardiovascular events. With the only exception of renal diseases, in which RAS-blocking agents should be preferred, CCBs are now recommended as first-line therapy in all stages of hypertension, independent of age, gender, race, and other comorbidities (Figure 3). They may be used both in monotherapy and in combination therapies with either angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) or thiazide diuretics. 11
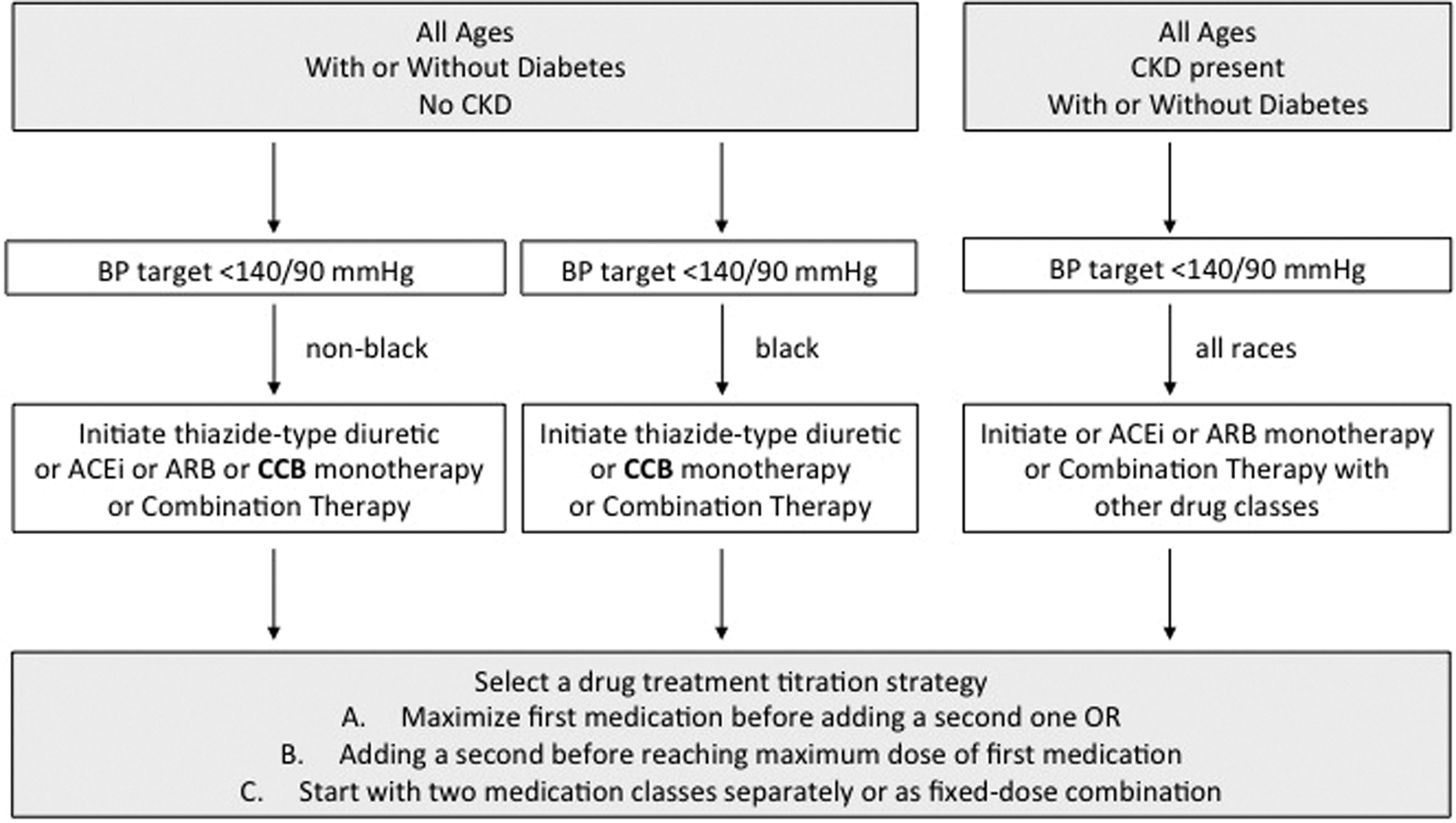
Schematic representation illustrating the different therapeutic options (monotherapy vs combination therapy) for the clinical management of hypertension, according to current United States guidelines. 11
Current Recommendations for Hypertension Treatment and Control
In view of the documented equivalence in terms of antihypertensive efficacy and in terms of reducing the risk of major cardiovascular events, it is now possible to choose among 5 antihypertensive drug classes, including ACE inhibitors, ARBs, β-blockers, CCBs, and diuretics, for starting and maintaining antihypertensive treatment in monotherapy. 10
In the selection of the first-choice antihypertensive drug class, however, it should be noted that, even in the presence of a substantial equivalence in terms of antihypertensive effectiveness, there are relevant differences in terms of compelling indications and contraindications among different antihypertensive drug classes (Table 2). 10 In addition, beyond evidence in favor of cardiovascular and renal protection, clinical trials have also demonstrated a better tolerability profile 31,32 and favorable metabolic properties 33 -35 by RAS-inhibiting drugs, especially ARBs, and by CCBs, compared to β-blockers and diuretics, thus promoting the use of these drug classes, both in monotherapy and in combination therapies, for hypertension management and control.
It is documented that only a relatively low proportion of treated patients with hypertension (20% to 30%) can be maintained on a therapy based on a single class of antihypertensive medication (monotherapy). 36 On the contrary, larger proportions of treated patients with hypertension (70% to 80%) require a combination therapy based on at least 2 classes of drugs, in order to achieve the recommended BP goals. 36
In those patients with hypertension who have mild elevations in systolic/diastolic BP levels (ie stage 1 hypertension), CCB-based monotherapy can be effectively and safely used to control hypertension, especially in the presence of compelling indications. 10 Indeed, monotherapy based on CCBs has demonstrated to provide sustained antihypertensive efficacy and to reduce cardiovascular and renal complications compared to other antihypertensive drug classes. 37 -42
In those patients with hypertension who do not achieve satisfactory BP control under monotherapy, or in those with high or very high cardiovascular risk profile, a combination strategy based on the use of 2 classes of antihypertensive drugs, including either ARBs or ACE inhibitors plus CCBs or thiazide diuretics (dual combination therapy) should be used. Randomized clinical trials 43 -45 and large meta-analyses 46,47 confirmed that these combination strategies are characterized by an antihypertensive efficacy about 5 times greater than the doubling of the dose of each monotherapy. In particular, among different combination therapies, those based on RAS blockers and CCBs are now viewed as the most effective and better tolerated antihypertensive strategy compared to other drug classes in several clinical settings. 48
In those patients with hypertension who do not achieve satisfactory BP control under a combination therapy based on the use of 2 classes of antihypertensive drugs, it should be useful to use a combination strategy based on the use of 3 or more classes of antihypertensive drugs, including either ARBs or ACE inhibitors, CCBs, and thiazide diuretics (triple combination therapy). 48 Potential CCB-based combination therapies, both in dual or in triple combination therapies, are reported in Table 3.
Potential Combination Therapies Based on CCBs.

Abbreviations: ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker; CCBs, calcium-channel blocker.
a When available and tolerated, fixed combination therapies should be preferred to ensure adherence to prescribed medications.
In this latter regard, dual or triple combination therapies with CCBs have recently become available in fixed formulations (single pill). These preparations have been demonstrated to provide effective BP reductions, sustained BP control over 24 h, and improved adherence to prescribed medications compared to free combination therapies. Recent meta-analyses have consistently demonstrated that fixed combination therapy may provide effective systolic and diastolic BP control and better adherence to prescribed medications compared to free combination therapy. 49 -51 In addition, fixed combination therapies have demonstrated to provide sustained BP control over the 24-hour period, 52 thus reducing the potential risk of cardiovascular and cerebrovascular accidents due to partial or limited BP control during the nighttime period. 53,54 Finally, these formulations seem to have the advantage of an easier therapeutic regimen to be taken as a single pill, which of course has relevant consequence on pill burden and improves patient compliance and adherence to prescribed antihypertensive therapy as demonstrated even in a setting of real practice. 55,56
Calcium channel blocker-based therapy has also demonstrated to be very effective and safe for the treatment of patients with true resistant hypertension, particularly when combined with RAS-blocking agents (either ACE inhibitors or ARBs), thiazide diuretic, and antialdosterone agents. 57,58 In these cases, high-dose, integrated and synergistic antihypertensive strategies are required to achieve the recommended BP targets, in the presence of high adherence to prescribed medications and low risk of drug-related side effects. 48 Thus, CCBs represent a very attractive and useful partner for any antihypertensive regimen to be used in this clinical setting of high-risk patients with true resistant hypertension.
Calcium Channel Blockers and Hypertension: Lessons From Large Randomized Clinical Trials
Over the last years, several large, randomized, controlled clinical trials have convincingly assessed the ability of CCBs to reduce cardiovascular morbidity and mortality, and mostly cerebrovascular events, in patients with hypertension and high cardiovascular risk (Table 4).
Stroke Incidence in Randomized Clinical Trials in Hypertension, Performed With CCBS Versus Either Placebo or Other Antihypertensive Drugs.a,b

Abbreviations: nr, not reported; ACEI, angiotensin-converting enzyme; CCB, calcium channel blockers; BB, beta-blockers.
a In the Syst-Eur, 37 STOP Hypertension-2, 38 INSIGHT, 39 SHELL, 40 NORDIL, 41 CONVINCE, 42 FEVER, 59 ALLHAT, 44 ASCOT-PBPLA, 43 ACCOMPLISH, 45 SCOPE (49) and VALUE 60 trials, number of events included fatal and non-fatal stroke; in the INVEST trial, 61 number of events included non-fatal stroke; in the MOSES study, 62 number of events included fatal and nonfatal stroke and recurrent events.
b Adapted from Reference 14 .
A first evidence suggesting a beneficial effect of an antihypertensive regimen based on CCBs derived from the Systolic Hypertension in Europe (Syst-Eur) Trial, 37 which enrolled elderly patients aged 60 to 75 years with isolated systolic hypertension (as defined for systolic BP values more than 160 mmHg and diastolic BP lower than 95 mm Hg), randomly assigned to a CCB-based regimen or to placebo. As expected, this trial was prematurely interrupted after an average period of 2 years, due to the significant reduction in the incidence of the primary composite end point of both fatal and nonfatal stroke observed in the active compared to the placebo group. 37
The Swedish Trial in Old Patients with Hypertension-2 Study (STOP Hypertension-2) 38 was a prospective, randomized clinical trial, which enrolled elderly patients aged 70 to 84 years with severe hypertension (defined for systolic BP more than 180 mm Hg and diastolic BP more than 105 mmHg, or both). Patients were randomized to an antihypertensive strategy based on either β-blockers and diuretics or ACE inhibitors or CCBs. In the presence of comparable BP reductions among 3 groups during an average follow-up of 54 months, there was a trend toward reduction in the relative risk of fatal and nonfatal stroke in the overall comparison between either CCBs or ACE inhibitors and β-blockers plus diuretics. 38 However, when considering the comparison between CCBs or ACE inhibitors versus conventional drugs, no significant difference was observed for fatal and nonfatal stroke nor in the comparison between CCBs and ACE inhibitors. 38
The International Nifedipine GITS Study: Intervention as a Goal in Hypertension Treatment (INSIGHT) Study 39 was a prospective, double-blind, randomized clinical trial that enrolled adult patients aged 55 to 80 years with stage 1 to 2 hypertension (as defined for BP levels more than 150/95 mmHg) or isolated systolic hypertension (systolic BP more than 160 mmHg) and at least 1 additional cardiovascular risk factor. Patients were randomly assigned to either a CCB or a diuretic. 39 Even in this trial, in the presence of comparable BP reductions during an average follow-up of 4 years, no significant differences were observed in the 2 treatment groups in terms of primary composite end point, all-cause mortality, nonfatal events, both fatal and nonfatal stroke. 39
Even in the Systolic Hypertension in the Elderly (SHELL) trial, 40 which enrolled patients aged older than 60 years with stage 1 to 2 hypertension (as defined for systolic BP more than 160 mmHg and diastolic BP more than 95 mm Hg) randomly assigned to receive an antihypertensive treatment based on the CCB or the diuretic, no difference in any cardiovascular or cerebrovascular event and in total mortality was observed between the 2 treatment groups. Substantial similar results were also observed in other clinical trials, performed with nondihydropiridinic CCBs, such as in the Nordic Diltiazem Study (NORDIL) 41 and in the Controlled Onset Verapamil Investigation of Cardiovascular End-Points (CONVINCE) trial. 42
Calcium Channel Blockers, Hypertension, and High Cardiovascular Risk: Lessons From Large Randomized Clinical Trials
Several international, randomized, controlled clinical trials have tested the antihypertensive efficacy, safety and tolerability of CCB-based therapy in patients with hypertension at high or very high cardiovascular risk. The results of these trials have consistently and independently confirmed the greater efficacy of CCBs in lowering BP levels and reducing incidence of major cardiovascular events compared to either diuretics or β-blockers and substantial equivalence to RAS-blocking agents (Table 4).
The International Verapamil-Trandolapril Study (INVEST) 61 was a randomized, open-label, clinical trial, which enrolled patients aged 50 years or older, with hypertension and coronary artery disease. Patients were randomly assigned to receive a nondihydropiridinic CCB or a β-blocker. After a 2-year follow-up and in the presence of similar BP reductions, no significant difference was observed between the 2 treatment regimens in terms of main cardiovascular outcomes, including fatal and nonfatal stroke. 42
The Felodipine Event Reduction (FEVER) trial 59 was a prospective, double-blind, parallel group, placebo-controlled, randomized clinical trial, which enrolled patients aged 50 to 79 years, with 1 or 2 additional cardiovascular risk factors or disease. Patients, whose BP levels remained uncontrolled after 6-week therapy based on thiazide diuretic, were randomly assigned either to CCB or to placebo. In the presence of a small difference in BP levels, the primary end point (fatal and nonfatal stroke) was significantly reduced in the felodipine compared to the placebo group. Among secondary end points, all cardiovascular events were reduced by 27% (P < .001), all cardiac events by 35% (P = .012), death by any cause by 31% (P = .006), coronary events by 32% (P = .024), heart failure by 30% (P = .239), and cardiovascular death by 33% (P = .019) were reduced in active compared to placebo group.
The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), 44 was a double-blind, active-controlled, randomized clinical trial, which enrolled patients, aged 55 years or older, with a history of hypertension and at least 1 additional vascular risk factor. Patients were randomly assigned to receive thiazide diuretic, CCB, ACE inhibitor, or α-blocker in a setting of general practice for planned follow-up of approximately 4 to 8 years. At the end of the follow-up period, systolic BP levels were significantly higher in the amlodipine (+0.8 mm Hg, P = .03) and lisinopril (+2 mm Hg, P < .001) groups compared with chlorthalidone, whereas diastolic BP was significantly lower with amlodipine (−0.8 mmHg, P < .001 for both comparisons). These BP differences, however, were paralleled with no differences between treatment groups for the incidence of the primary composite end point as well as for that of all-cause mortality.
The Anglo-Scandinavian Cardiac Outcomes Trial – Blood Pressure Lowering Arm (ASCOT-BPLA) was a prospective, randomized, clinical trial, which enrolled patients with hypertension and at least 3 cardiovascular risk factors. 43 Patients were randomized to either a CCB-based regimen or a β-blocker-based regimen. 43 Compared with the atenolol-based regimen, BP values were lower throughout the follow-up in those patients allocated to the amlodipine-based regimen. In particular, these differences were largest (5.9/2.4 mm Hg) at 3 months, and the average difference throughout the study was 2.7/1.9 mm Hg. By the end of the trial, about 53% patients had reached both the systolic and the diastolic BP targets, and about 78% were taking at least 2 antihypertensive agents. This trial (originally powered for an estimated 1150 primary end points) was prematurely stopped when only 903 primary end points had occurred. Thus, the primary end point of fatal cardiovascular disease and nonfatal myocardial infarction was not achieved (HR 0.90 [95% CI: 0.79-1.02]; P = .1052). However, when the components of the primary and secondary end points were considered, the amlodipine-based regimen was consistently better than the comparator, by significantly reducing all-cause mortality by 11% (P = .0247), cardiovascular mortality by 24% (P = .0010), total cardiovascular events and procedures by 16% (P < .0001), total coronary end points by 13% (P = .0070), fatal and nonfatal stroke by 23% (P = .003) as compared to the atenolol/thiazide regimen. Also, amlodipine-based regimen significantly reduced incidence of new-onset diabetes by 30% (P < .0001) compared to the atenolol-based regimen.
Two additional trials have tested the clinical efficacy and safety of CCB-based compared to ARB-based regimen in patients with hypertension at high cardiovascular risk.
The Valsartan Antihypertensive Long-term Use Evaluation (VALUE) trial 60 was a prospective, multinational, double-blind, randomized, active-controlled, parallel group trial, which enrolled high-risk patients aged 50 years or older, with a history of hypertension and predefined combinations of cardiovascular risk factors or cardiovascular diseases. Patients were randomly assigned to valsartan-based regimen or to amlodipine-based regimen during a mean follow-up period of 4.2 years. 60 As observed for the ASCOT-BPLA, 63 even in this trial the amlodipine-based regimen induced greater BP reductions than valsartan-based regimen throughout the study, and especially during the first 6 months of the follow-up, during which the highest frequency of cardiovascular events, including stroke, was recorded. Authors interpreted the slightly reduced incidence of fatal and nonfatal stroke observed in the amlodipine group compared to the valsartan group as a consequence of these BP differences. 60 Subsequent analysis of the study population, stratified according to the same degree of BP reductions in both treatment groups, seems to confirm similar beneficial effects provided by both ARB-based and CCB-based therapy in terms of cardiovascular protection and reduced incidence of cerebrovascular events, mostly stroke. 64
The MOrbidity and mortality after Stroke—Eprosartan compared with nitrendipine in Secondary prevention (MOSES) trial 62 enrolled patients with hypertension having a history of cerebrovascular events. Patients were randomized to either ARB or CCB. 62 In the presence of comparable BP reductions, the eprosartan-based regimen reduced the incidence of the primary composite end point, including cardiovascular and cerebrovascular events and noncardiovascular death, significantly more than the nitrendipine group. 62 However, no significant differences were observed between 2 treatment groups in terms of incidence of cerebrovascular events. 62
More recently, the Avoiding Cardiovascular Events through Combination Therapy in Patients Living with Systolic Hypertension (ACCOMPLISH) trial 45 was a double-blind, randomized clinical trial, which enrolled patients with hypertension at high cardiovascular risk. Patients were randomized to receive a first-line combination therapy based on the association of the ACE inhibitor plus either the CCB amlodipine or the thiazide diuretic hydrochlorothiazide once daily. 45 Even in this case, the trial course was terminated early after a mean follow-up of 36 months because of the documented superior efficacy of benazepril plus amlodipine compared with benazepril plus hydrochlorothiazide. 45 At the time of trial interruption, combination therapy with benazepril-amlodipine significantly reduced the incidence of both primary (hazard ratio [HR] 0.80, [95% confidence interval, CI: 0.72 to 0.90]; P < .001) and secondary (HR 0.79, [95% CI: 0.67 to 0.92]; P = .002) end points compared to combination therapy with benazepril-hydrochlorothiazide. 45
Conclusions
In conclusion, the benefits obtained by achieving effective and persistent BP control in patients with hypertension having different cardiovascular risk profile in terms of reduction in cardiovascular morbidity and mortality have been repeatedly demonstrated. Despite such solid evidence, large international surveys still document persistently low rates of BP control in the general population with hypertension. The relatively low use of combination therapy and the lack of drug dosage optimization during chronic antihypertensive treatment represents 2 of the plausible reasons for this paradox.
Antihypertensive strategy based on the use of dihyropyridinic CCBs, both in monotherapy and in combination therapy with drugs inhibiting the RAS and/or thiazide diuretics, have demonstrated that this approach may significantly contribute to improvement in BP control in the presence of a good tolerability profile. In particular, fixed combination therapies based on RAS-blocking agents and CCBs have proven to be effective and safe in different clinical settings over the cardiovascular and renal continuum. Hence, this strategy could be viewed as a viable way to improve BP control rates in general outpatients with hypertension and to ensure effective and sustained BP control over the 24 h.
On the basis of the currently available clinical evidence, this strategy is particularly indicated for elderly patients with isolated systolic hypertension, in patients with hypertension having metabolic disorders (dyslipidemia, metabolic syndrome, hyperuricemia),in those with diabetes mellitus, and in those with renal impairment or diabetic nephropathy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
