Abstract
Nitric oxide (NO) is an integral molecule especially important in the regulation of the cardiovascular system. Literature indicates that the number of studies continues to grow with regard to the effects of NO on cardiovascular disease and hypertension. “Prehypertension” is the clinical stage leading to hypertension. Diet and lifestyle modifications are the only treatment options for prehypertension. The objective of this study was to determine the effects of oral NO supplementation on blood pressure in patients with clinical prehypertension. This pilot phase study evaluated the effect of an orally disintegrating lozenge that generates NO in the oral cavity on blood pressure, functional capacity, and quality of life. Thirty patients with clinical prehypertension were recruited and enrolled in either the NO treatment or the placebo group over a 30-day period in an outpatient setting. Nitric oxide supplementation resulted in a significant decrease in resting blood pressure (138 ± 12 mm Hg in systole and 84 ± 5 mm Hg in diastole at baseline vs 126 ± 12 mm Hg in systole and 78 ± 4 mm Hg in diastole at follow-up, P < .001, vs baseline) and a significant increase in the achieved walking distance in the standard 6-minute walk test (596 ± 214 meters at baseline vs 650 ± 197 meters at follow-up, P < .005 vs baseline). Using a standardized questionnaire to assess quality of life, patients receiving NO supplementation showed improvement in the Physical Component Summary Score (PCS) and Mental Component Summary Score (MCS). Nitric oxide supplementation appears to lower blood pressure in patients with prehypertension and might be beneficial as a routine supplementation for cardiovascular protection.
Introduction
Nitric oxide (NO) is an integral molecule in regulating the cardiovascular system naturally produced and found in many different kinds of cells and organ systems. Nitric oxide has a short half-life of a few seconds in biological mixtures and is quickly oxidized to nitrite and subsequently to nitrate by oxygenated hemoglobin which is eventually excreted into urine
1
or concentrated in the salivary glands for recycling.
2
Continuous generation of NO is essential for the integrity of the cardiovascular system, and decreased production and/or bioavailability of NO is central to the development of many disorders. The production of NO from
Despite NO being recognized by the scientific and medical community as one of the most important molecules produced within the body and being named “Molecule of the Year” by Science in 1992 3 and a Nobel Prize in Physiology or Medicine awarded for its discovery, there are currently only 3 Food and Drug Administration (FDA)-approved products on the market directly related to NO: (1) organic nitrates, such as nitroglycerin for the treatment of acute angina (these have been used for centuries long before the discovery of NO); (2) inhaled NO therapy for neonates for treatment of pulmonary hypertension due to underdeveloped lungs and for adults, the main use of inhaled NO is for maintenance of light anesthesia and analgesia; and (3) phosphodiesterase inhibitors, such as sildenafil, which do not directly affect NO production but act through affecting the downstream second messenger of NO, cyclic guanosine monophosphate (cGMP). There are a number of NO-based therapies in development, including technologies designed to activate and promote NO synthesis from NOS, NO-donating compounds, therapies designed to modulate posttranslational protein modifications through S-nitrosation, and therapies designed to affect or prolong downstream signaling pathways from NO. The method of delivery of NO and cellular and molecular specificity is of utmost importance. Delivery of NO through controlled and enzymatic metabolism of organic nitrates is an effective acute treatment for angina but still causes some adverse effects when used chronically. The safe delivery of NO gas through inhalation therapy is also now in practice.
Utilizing intellectual property developed out of the University of Texas Health Science Center in Houston (US patents 8,298,589, 8,303,995, and 8,435,570), Neogenis Laboratories has licensed this technology and developed a GMP-certified, over-the-counter, all natural formulation that provides a system for generating NO in an endothelium-dependent and independent manner. This recently patented technology was developed by Nathan S. Bryan, PhD, and has been reviewed by the FDA as a safe, all natural formulation using generally recognized as safe (GRAS) ingredients. The NEO40 Daily product ingredients list and packaging was submitted to the FDA Office of Compliance by Neogenis Labs, Inc, for use as a dietary supplement (registration # 3008524085). Previously published data that incorporated in vitro biochemical assays, animal studies, and human clinical trials not only demonstrate a robust improvement in NO activity but also that the formula is effective at modifying biomarkers of cardiovascular risk and replenishing NO biochemistry in at-risk patients. 4 -6 The objective of the current clinical pilot phase study was to determine the effects of oral NO supplementation on blood pressure in patients with clinical “prehypertension.”
Methods and Materials
Given that this study involves human participation, the institutional review board (IRB; RCRC IRB approval Neo 11-2012 prehtn) has approved the study to commence the clinical pilot phase of the effects of oral NO supplementation on functional capacity and blood pressure in “prehypertensive” healthy adults. According to The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure (JNC 7), prehypertension is the term used to identify patients who are at high risk for the development of hypertension. 7 The JNC 7 Complete Report classifies the following blood pressure range as prehypertensive: systolic and diastolic blood pressures of 120 to 139 mm Hg and 80 to 89 mm Hg, respectively. 7 This study is registered on clinicaltrials.gov as NCT01937754.
Thirty randomly selected patients without overt chronic cardiovascular history were recruited from outpatient cardiology clinics in a single center. The majority of the patients were referrals for preoperative clearance. Patients were considered eligible to the study if they met the following inclusion criteria: adults between the age of 18 and 80 years; two blood pressure readings of ≥ 135/80 mm Hg at rest, thus representing prehypertension as per the JNC 7 Complete Report definition
7
; and informed consent.
Exclusion criteria were: existence of any significant internal or cardiovascular disease; current use of any antihypertensive medication; and inability to give informed consent or fill out standard questionnaires or inability to follow-up clinically.
Only English-speaking patients were recruited.
The 30 study participants were then randomized to 2 groups: group 1 (n = 17) received NO and group 2 (n = 13) received placebo. The following parameters were analyzed: baseline and follow-up blood pressure (mm Hg); baseline and follow-up heart rate (bpm); baseline and follow-up distance from 6-minute walk test (meters); and baseline and follow-up quality-of-life assessment short form 36 (SF-36) questionnaire (Physical Component Summary Score [PCS] and Mental Component Summary Score [MCS]).
Patients were advised to take the NO lozenge or the placebo twice a day daily for a 30-day period. In the 2 groups of patients (on NO and placebo), the various parameters were measured at baseline and posttreatment, and the corresponding changes from baseline and posttreatment observed in the 2 groups were compared.
Using Stata Statistical Software, Release 10, we assumed an α of .05, power of 0.80, and a 2-tailed t test for the sample size calculation. This calculation was based on the steady state plasma nitrite levels. According to power analyses from previous clinical studies, 20 patients were sufficient for significant increase of 50% in plasma nitrite levels that have affected vasodilation. The placebo looked and tasted the same as the active NO supplement. Both the patients and the study coordinator were blinded to the treatment. After enrollment, baseline assessments were performed in which the patients had 2 Riva Rocci blood pressure readings at rest with 10 minutes apart from each reading. These readings were followed by an orthostasis testing with heart rate and blood pressure manometer readings. A full medical history and focused physical examination, which included collecting the said vitals, was performed and documented followed by a baseline 6-minute walk test at the initial office visit. The postwalk blood pressure and heart rate measurements were collected immediately after the walk and at 5 minutes subsequently. Each participant filled out a standard of quality-of-life questionnaire, SF-36, at the initial office visit in addition to a nonstandard individualized short questionnaire asking for overall well-being and activity levels.
Following the baseline measurements and examination, the participants were provided with a 30-day supply of the active NO product or the placebo. Via self-administration, patients were required to take a pleasant-tasting lozenge twice per day, 12 hours apart, for 30 days. This product’s formulation was designed to be a quick dissolve that melts in the mouth within 4 to 5 minutes. After 30 days, the participants returned to the outpatient clinic for a medical history, focused physical examination, and a follow-up assessment. We measured blood pressure and heart rate measurements performed at rest 10 minutes apart, orthostasis testing, and ability to walk 6 minutes as mentioned earlier at the baseline visit. Two questionnaires were provided to be completed again and returned before leaving the office.
Data from blood pressure, medical history, and physical examinations were collected in case report forms and reported in the patients’ medical files. The data were recorded electronically with unidentified labeling for each patient. As for statistical considerations, the baseline blood pressure was compared to the same measurements after 30 days of taking NO.
We expected a modest reduction in the systolic blood pressure as seen in the results published by Zand et al. 4 The data before and after were averaged, compared, and tested for statistical significance using a 2-tailed t test to determine whether NO supplementation leads to a significant change in systolic blood pressure in patients with prehypertension.
Results
The study was conducted between January 2013 and July 2013. All patients signed an informed consent and received NO lozenges or placebo in a blinded manner for a total duration of 30 days. All patients were advised to take 2 lozenges daily, the first one early in the morning after waking up and the second one in the late afternoon. Upon enrollment and randomization, all patients received a clinic appointment after 30 to 45 days for the study follow-up evaluation.
One patient in group 2 (placebo group) was lost to follow up, despite several attempts to reach him by mail or phone. Therefore, a total of 29 patients were analyzed. Baseline demographics are depicted in Table 1.
Baseline Demographics.a

Abbreviation: NO, nitric oxide.
aThe baseline demographics in group 1 (NO supplementation) and group 2 (placebo) are given.
Baseline Hemodynamics
The average baseline blood pressure in group 1 (NO supplementation, n = 17) was 138 ± 12 mm Hg in systole and 84 ± 5 mm Hg in diastole; the baseline heart rate was 75 ± 9 beats per minute (bpm). Baseline blood pressure in group 2 (placebo, n = 12) was 138 ± 21 mm Hg in systole and 80 ± 8 mm Hg in diastole; the baseline heart rate was 80 ± 10 bpm. There was no significant difference between the 2 groups except the diastolic blood pressure was slightly lower in group 2. At follow-up, blood pressure in group 1 at rest was 126 ± 12 mm Hg in systole and 78 ± 4 mm Hg in diastole (P < .001 vs baseline), indicating a 12 mm Hg reduction in systolic and 6 mm Hg reduction in diastolic pressures. Heart rate was 76 ± 8 bpm (nonsignificant vs baseline). In group 2, blood pressure at rest was 135 ± 17 mm Hg in systole and 82 ± 8 mm Hg in diastole (nonsignificant vs baseline), heart rate was 79 ± 8 bpm (nonsignificant vs baseline; Figure 1).

Blood pressure—graph shows individual blood pressure in group 1 (NO supplementation) and group 2 (placebo) at baseline and at 30-day follow-up. NO indicates nitric oxide.
Functional Capacity
The average distance of the 6-minute walk test in group 1 (NO supplementation) at baseline was 596 ± 214 meters versus 650 ± 197 meters at follow-up (P < .005 vs baseline) revealing a 55-meter improvement in the treatment group. The average distance of the 6-minute walk test in group 2 (placebo) at baseline was 590 ± 8 meters versus 606 ± 225 meters at follow-up (nonsignificant vs baseline). There was no significant difference in the 6-minute walk test at baseline and follow-up between the 2 groups (Figure 2).
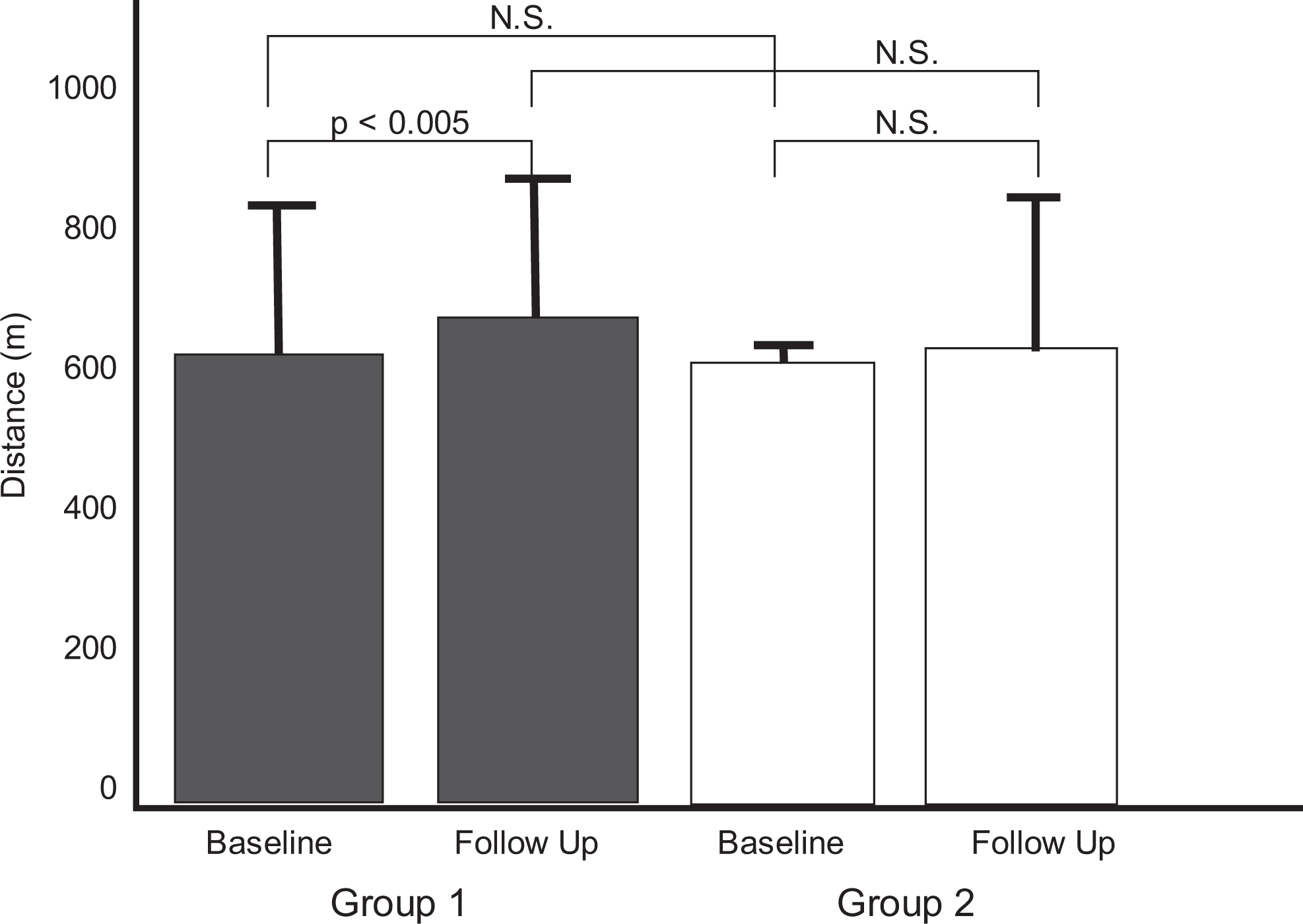
Six-minute walk test—graph shows the distance responses of the 6-minute walk test in group 1 (NO supplementation) and group 2 (placebo) at baseline and at 30-day follow-up. NS indicates nonsignificant difference; NO, nitric oxide.
Quality-of-Life Assessment
Quality of life was assessed using a standardized SF-36 questionnaire that computed 8 scaled scores. The presented study utilized the Physical Component Summary Score (PCS) and the Mental Component Summary Score (MCS) that were created based on the questionnaire responses. Many studies have shown that in the United States, 80% to 85% of the variance among the 8 scales are attributed to physical and mental health factors. 8 Therefore, the 2 summary measures used in the presented study, PCS and MCS, aggregate the 8 scores into their respective physical and mental health categories. The average PCS and MCS for group 1 (NO supplementation) at baseline were 48 ± 10 and 40 ± 9, respectively. At follow-up, PCS was 50 ± 8 and MCS was 45 ± 7, respectively (P < .05 vs baseline for both comparisons).
The PCS and MCS for group 2 at baseline were 43 ± 10 and 37 ± 9, respectively, versus 37 ± 11 and 37 ± 7 at follow-up. Of interest, there was a significant worsening in both scores at follow-up versus baseline (P < .05, Figure 3).

SF-36 questionnaire Scores—graph shows the Physical Component Summary Score (PCS) and Mental Component Summary Score (MCS) responses of the SF-36 questionnaire in group 1 (NO supplementation) and group 2 (placebo) at baseline and at 30-day follow-up. NS indicates nonsignificant difference; NO, nitric oxide; SF-36, short form 36.
There was no significant difference at baseline between the 2 groups for both scores (nonsignificant vs baseline for both comparisons). There was no significant difference at follow-up between the 2 groups for the PCS. The MCS in group 1 at follow-up (45 ± 7) was significantly higher than that of group 2 (37 ± 7, P < .05, Figure 3), indicating that well-being improves in patients exposed to NO supplementation versus the placebo.
The parameter values (mean ± standard deviation [SD]), at baseline and posttreatment, and mean ± SD of individual differences between the 2 measurements (ie, changes) are shown in the form of a table together with P values for any differences (Table 2).
Summary of Parameter Values.a
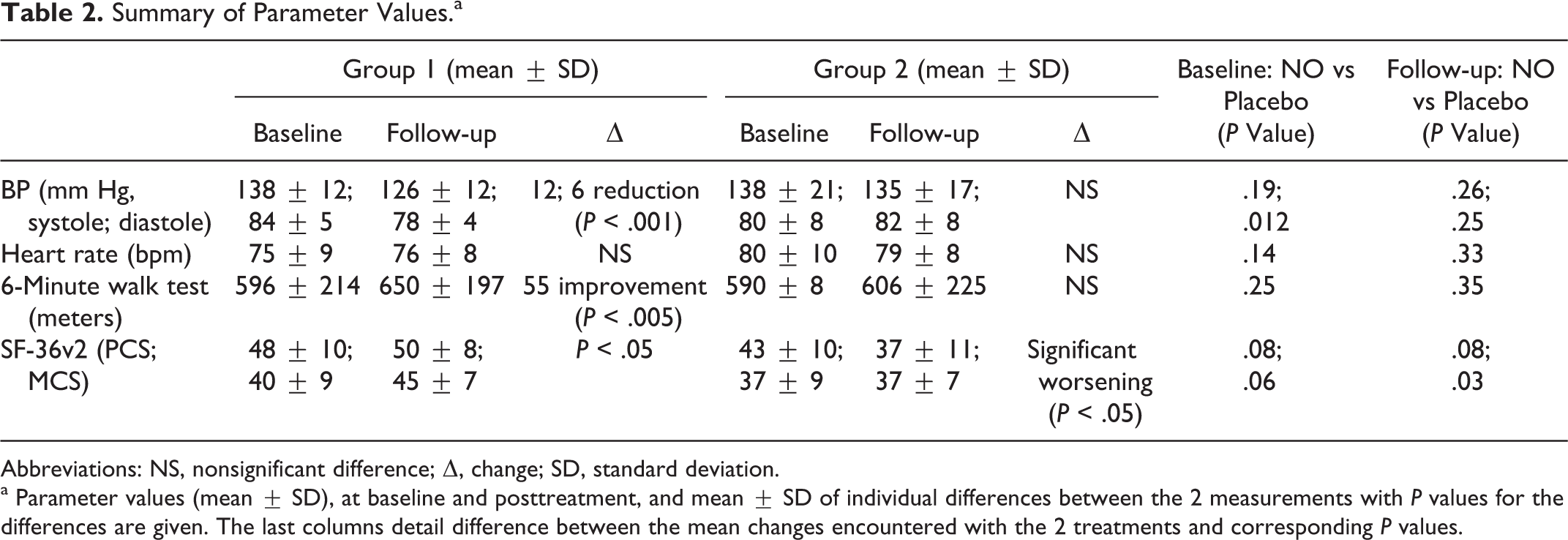
Abbreviations: NS, nonsignificant difference; Δ, change; SD, standard deviation.
a Parameter values (mean ± SD), at baseline and posttreatment, and mean ± SD of individual differences between the 2 measurements with P values for the differences are given. The last columns detail difference between the mean changes encountered with the 2 treatments and corresponding P values.
Discussion
This pilot phase study evaluated the effect of NO supplementation using a commercially available NO lozenge on blood pressure, functional capacity, and quality of life in a small number of patients with clinical prehypertension placed in the NO treatment group versus the placebo group over a 30-day period in an outpatient setting. Nitric oxide supplementation resulted in a significant reduction in resting blood pressure as well as a significant increase in the achieved walking distance in the standard 6-minute walk test. Although there was no significant difference at baseline between placebo versus NO-treated patients with regard to the PCS and MCS or at follow-up with only regard to the PCS as assessed in a standardized quality-of-life questionnaire, patients receiving NO supplementation did appear to show improved PCS and MCS. The NO-treated patients significantly improved in both the PCS and the MCS (P < .05 and P < .05, respectively).
The NO lozenges used in the presented study utilizes physiological nitrate and nitrite concentrations that are provided via diet to produce more NO molecules to overcome the body’s inability to generate NO. 4,9 Nitric oxide has a very short half-life. A reduction in nitrate to nitrite occurs through oral commensal bacteria 10 then nitrite occurs by providing the substrates and machinery needed for such stepwise reduction. The NO lozenges allow the body to restore or replete NO availability by restoring NO production without endothelial dependency while lowering the burden of disease. Our data are consistent with a recent case report using the NO lozenge to reduce blood pressure in a pediatric patient with Argininosuccinic aciduria with resistant hypertension. 11
The current literature shows the significance of NO in human physiology. 12 However, there is no effective therapeutic drug treatment that has been developed to reestablish the NO homeostasis in individuals at risk of cardiovascular disease. Prehypertension leads to hypertension, a risk factor for atherosclerosis and coronary heart disease. Patients with coronary artery disorders cannot produce enough NO to regulate normal vascular integrity due to endothelial dysfunction. Early detection and proper treatment are vital to prevent negative effects of hypertension and its clinical sequelae. Using oral supplements of NO or nitrite-derived NO supports vasodilation and may help normalize blood pressure in individuals with prehypertension along with a delay in the need for antihypertensive medications as indicated by the presented study’s findings of a significant decrease in blood pressure at follow-up.
Another potential effect of NO supplementation on the cardiovascular system is the process in which NO inhibits
The presented study has several limitations. First, this is a pilot study with a limited number of participants. Therefore, the findings might not be easily extrapolated to larger populations. Second, the quality-of-life assessment is of subjective nature. The medical attention and treatment of any kind theoretically can affect a follow-up questionnaire. However, the data were obtained in the frame of a double-blind, randomized controlled study. Third, the NO lozenges also contain vitamin C (100 mg) and vitamin B12 (1000 μg). Vitamin B12 especially, when supplemented, has been shown to be a safe way to decrease elevated blood levels of total homocysteine, a risk factor for coronary and atherosclerotic vascular diseases. 15,16 Therefore, the described effects cannot be solely attributed to NO but to the combination of NO and vitamin C and B12. Fourth, the majority of the enrolled patients were referrals for preoperative clearance. This means that they may have had anxiety or a white coat effect on blood pressure and therefore may not represent genuine prehypertension. Fifth, no quantitative NO measurements were performed in the patients, therefore, it is unknown whether or not some of the individuals were actually NO depleted or not. Similarly, no quantitative NO measurements were performed at study follow-up. Therefore, it is unknown whether the NO supplementation in fact increased body concentration of NO. However, based on previously published data using this lozenge, 5,12 it appears likely that it increases NO production and availability. A quantitative assessment of absolute NO concentration was not within the frame of the current study design.
This small pilot study only looked at short-term effects, had no systematic strategy, and was not designed to examine adverse (long or short term) or compliance/concordance with the treatment. Moreover, twice daily dosing imposes a compliance/concordance burden, particularly for a long-term intervention.
Conclusion
This is the first study demonstrating a significant reduction in systolic and diastolic blood pressure levels after a 30-day period of NO supplementation in adults with clinical prehypertension. In addition, functional capacity improved. Quality of life was assessed by the physical and mental health summary scores of the standardized SF-36 questionnaire improved after NO supplementation. These preliminary results need to be confirmed by large-scale controlled studies.
Footnotes
Authors’ contribution
O. Biswas substantially contributed to data acquisition, analysis, and interpretation of the data, drafting of the manuscript, and critical revision of the manuscript for intellectual content; V. Gonzalez contributed to analysis and interpretation of the data and drafting of the manuscript; E. Schwarz contributed to conception and design of the study, with substantial contribution to data acquisition, analysis and interpretation of the data, drafting of the manuscript, and critical revision of the manuscript for intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: The study was supported by an educational grant from Neogenis Labs.
