Abstract
Introduction:
Although hypothermia reduces myocardial infarct size, noninvasive and rapid cooling methods are lacking. This study tests the effectiveness of a novel cooling apparatus on myocardial infarct size and no-reflow area in 2 models of coronary artery occlusion (CAO).
Methods and Results:
Animals were randomized to normothermic (N) or hypothermic (H) groups after isolation of a proximal coronary artery. Animals were subjected to 30 minutes CAO and 3 hours reperfusion. In protocol 1 (rabbit, n = 8 per group), hypothermia was initiated, using the ThermoSuit apparatus (convective-immersion cooling), 5 minutes after the onset of CAO to a goal temperature of ∼32°C. In protocol 2 (rat, n = 5 per group), hypothermia was initiated 2 minutes after the onset of CAO to a goal temperature of ∼30°C. Goal temperature was reached in ∼20 minutes. In protocol 1, hypothermia caused an 82% reduction in infarct size as a percentage of the ischemic risk zone (N, 44% ± 5%; H; 8% ± 2%, P < 0.001) and an 89% reduction in the no-reflow area (N, 44% ± 4%; H, 5% ± 1%, P < 0.001). In protocol 2, hypothermia caused a 73% infarct size reduction (N, 51% ± 5%; H, 14% ± 6%, P < 0.01) and a 99% reduction in the no-reflow area (N, 33% ± 5%; H, 0.4% ± 0.3%, P < 0.01).
Conclusion:
The ThermoSuit device induced rapid hypothermia and limited infarct size and no reflow to the greatest extent ever observed in this laboratory with a single intervention.
Introduction
With over 900 000 myocardial infarctions (MIs) in the United States annually 1 and 30-day mortality for acute MI of approximately 10%, methods are still needed to decrease mortality. 2 Emphasis for improving outcome has focused on reducing door-to-balloon time in patients who require percutaneous coronary intervention to restore coronary flow, since door-to-balloon times of <90 minutes have shown reduced mortality compared to those of >90 minutes. 3 -5 Recently, however, a significant further decrease in door-to-balloon time showed no further decrease in mortality, suggesting a plateau effect. 2 Thus, new avenues must be examined to protect the myocardium until flow can be restored. One approach is to further reduce myocardial infarct size by adjunctive therapy in addition to reperfusion. Myocardial infarct size remains an important determinant of survival and clinical outcome. 6,7
Myocardial temperature is a major predictor of myocardial infarct size. 8 Previously, this laboratory has shown that mild regional hypothermia consistently decreases myocardial infarct size and anatomic zone of no reflow in the experimental rabbit model when hypothermia is achieved before reperfusion. This model utilized a bag of ice-cold saline placed directly on the ischemic risk area to achieve an intramyocardial temperature of 32°C to 33°C.9- 12 Other methods by which hypothermia has been achieved include heat-exchange catheters, 13 -16 infusion of cold fluid into the vasculature, 15 into the peritoneum, and into the intrapericardial space, 17 and use of surface cooling techniques. 18 Success of cooling and infarct-size reduction in clinical trials has been variable with these techniques; in some of the clinical trials, patients did not achieve adequate hypothermia prior to reperfusion. 19 One practical method of achieving mild hypothermia may be a new noninvasive cooling device, the ThermoSuit System, developed by Life Recovery Systems (Kinnelon, New Jersey). This device pumps ice-cold water over the surface of the patient (convective-immersion cooling, Figure 1). This water is constantly drained, rechilled, and returned until ideal patient temperature is reached, at which point the system is disengaged. Advantages of this device are that it is noninvasive, portable, reduces temperature to therapeutic levels within approximately 20 minutes, is already Food and Drug Administration approved for the treatment of hyperthermia, has been studied in neurologic preservation after cardiac arrest, 20 and has been shown to be safe in humans. 21 This therapy could be initiated in the emergency department, the ambulance, or even in the patient’s home prior to transfer to the hospital, as this device requires very little set-up time. It is not known whether the ThermoSuit device is cardioprotective in the setting of an acute MI. We hypothesized that infarct size and area of no reflow (ANR) would be reduced in our rabbit (protocol 1) and rat (protocol 2) models if this device reduced temperature to goal before reperfusion.
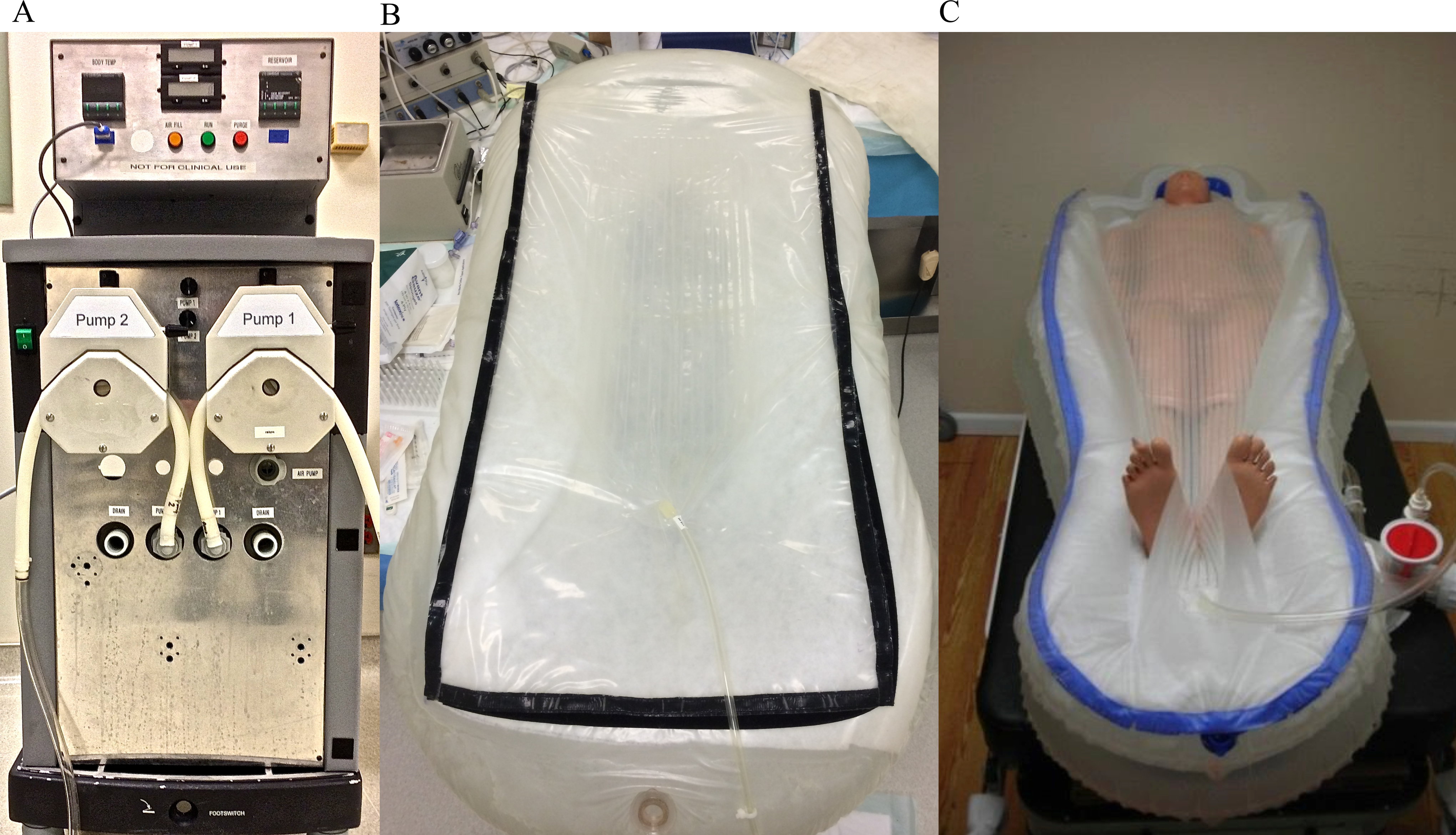
Images of the pump and ThermoSuit. A, The ThermoSuit pump. The pump has 2 output areas pumping water into the suit via the bottom and top of the suit. The suit also has 2 drains: one at each end of the suit. Water is returned to the pump, rechilled by the existing ice water, and then returned to the animal to continue cooling. B, The animal suit. This is the suit we used. The suit resembles an inflatable tub with a plastic cover in which the animal lays. Water is deposited directly onto the animal simultaneously from the top and bottom of the suit. C, The suit designed for human use.
Methods
The animals used in these studies were maintained in accordance with the policies and guidelines of the Position of the American Heart Association on research animal use (American Heart Association, 1985) and the Guide for Care and Use of Laboratory Animals (2010). The Good Samaritan Hospital Institutional Animal Care and Use Committee approved this protocol. Good Samaritan Hospital is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Protocol 1
Surgical preparation
Male New Zealand White rabbits (2.5-3.75 kg) were anesthetized with an intramuscular injection of ketamine (60 mg/kg; JHP Pharmaceuticals, Rochester, Michigan) and xylazine (5 mg/kg; Lloyd Labs, Shenandoah, Iowa). Intravenous pentobarbital (Lundbeck, Inc, Deerfield, Illinois) was given throughout the study to maintain deep anesthesia. All surgery was performed on the deflated ThermoSuit. The rabbit received a tracheostomy and was mechanically ventilated with room air enriched with 100% oxygen delivered at a flow rate of 0.1 L/min. Blood gasses were monitored at various time points throughout the study. Surgical procedures were carried out using clean instruments and gloved hands. Fluid-filled catheters were inserted into the jugular vein to deliver fluids and into the carotid artery to measure hemodynamics. A thermocouple probe was inserted into the esophagus. The chest was opened through the left fourth intercostal space, the pericardium incised, and the heart exposed. The first large anterolateral branch of the circumflex artery, or the circumflex artery itself, was encircled with a 4-O silk suture near the base of the heart to allow for coronary occlusion and reperfusion. If the circumflex was not readily visible or accessible, the left anterior descending artery was used. Coronary occlusion in this region normally results in ischemia of a large region of the anterolateral and apical left ventricular wall. The ends of the suture were threaded through a piece of tubing, creating a snare that was tightened to occlude the artery. The skin was closed with a purse string suture. After surgical preparation, rabbits were randomly assigned to the normothermia or hypothermia group.
Experimental protocol
After a 15-minute stabilization period, baseline hemodynamic parameters and temperature were obtained. Rabbits were then subjected to 30 minutes of coronary artery occlusion (CAO) followed by 3 hours of coronary artery reperfusion (CAR). Heart rate (HR), arterial systolic and diastolic pressure, and esophageal temperature were recorded throughout the study measured at baseline (before CAO), during occlusion, and during reperfusion.
Rabbits randomized to the normothermia group were maintained at a temperature of approximately 38.6°C throughout the protocol. Rabbits randomized to the hypothermia group had a target baseline temperature of 38.6°C. The target temperature for the hypothermia group was 32°C. The ThermoSuit (provided by Life Recovery Systems) was inflated prior to CAO. Hypothermia was initiated 5 minutes after CAO and was maintained for 1 hour after CAR, at which time active rewarming using heating pads was initiated. The ThermoSuit was disengaged once the hypothermic rabbit reached an esophageal temperature of 34°C. The animal’s temperature continued to drop after the ThermoSuit was disengaged, and the animal was then removed from the device when the esophageal temperature reached 32°C, approximately 20 minutes after starting the device. Of all animals, 88% reached a temperature below 32°C due to this phenomenon (minimum temperature = 30.7°C).
At the end of the protocol, a catheter was inserted into the left atrial appendage to inject blue dye and thioflavin S. A 4% solution of thioflavin S of 1 mL/kg (Sigma, St. Louis, Missouri) was injected into the heart to define the ANR. Thioflavin S, a fluorescent dye, stains endothelial cells, serves as a marker for perfusion, and is used as a standard marker for identifying areas of no reflow. The coronary artery was then reoccluded, and the ischemic risk area was delineated by injecting 4 mL of a 50% solution of Unisperse blue dye (Ciba Specialty Chemicals, Hawthorne, New York) into the left atrial appendage. The ischemic risk zone remained pink, while the nonischemic area that received blood flow stained blue. The deeply anesthetized rabbit was killed by an injection of 12 mEq potassium chloride into the left atrium, and then the heart was excised. No animals died prematurely during the protocol. Exclusion criteria were (1) death occurring before the end of the protocol or (2) an ischemic risk zone <10% of the left ventricle (LV).
Protocol 2
Surgical preparation
Female Sprague-Dawley rats (∼400 g) were anesthetized with an intraperitoneal injection of ketamine (75 mg/kg) and xylazine (5 mg/kg). Additional anesthesia was given intraperitoneally as needed throughout the protocol. The rats were intubated and mechanically ventilated with room air (60 cycles/min, tidal volume 1 mL/100 g body weight). The left jugular vein was cannulated for the injection of thioflavin S, blue dye, and KCl at the end of the experiment. The chest was opened through the fourth intercostal space. The left coronary artery was encircled with 4-O silk suture for occlusion and reperfusion of the artery as in protocol 1. The temperatures of the animals were monitored during the protocol by a thermocouple probe in the esophagus.
Experimental protocol
After preparatory surgery, rats were stabilized for 15 minutes. Rats randomized to the normothermia group were maintained at a temperature of 37°C throughout the protocol. Rats randomized to the hypothermia group had a baseline temperature of 37°C. The target temperature for the hypothermia group was 30°C. Hypothermia was initiated 2 minutes after CAO. Once temperature reached 32°C approximately 10 minutes later (12 minutes after the onset of CAO), the animal was removed from the ThermoSuit. The animal continued to cool after removal from the suit and was maintained at 30°C for the duration of the protocol using ice packs and heating blankets as needed. The temperature of all animals fell below 30°C during the study (minimum temperature = 27.4°C).
At the end of the protocol, thioflavin S was injected through the left jugular vein to demonstrate the no-reflow area. The heart was then reoccluded, and Unisperse blue dye was injected into the left jugular vein to delineate risk zone. Deeply anesthetized rats were then killed by 1 mL intravenous KCl. No animals died prematurely during the protocol. Exclusion criteria were (1) death occurring before the end of the protocol or (2) an ischemic risk zone <15% of the LV.
Analysis of no-reflow area, risk zone, and necrosis
The hearts were sliced transversely into 6 to 8 sections (protocol 1) or 4 sections (protocol 2) and photographed. The slices were photographed under ultraviolet light (254 nm) to identify ANRs (nonfluorescent perfusion defects) and under halogen lighting to identify the area at risk (AR; nonblue area). The slices were then incubated in a 1% solution of triphenyltetrazolium chloride (TTC), immersed in formalin, and rephotographed. Triphenyltetrazolium chloride stains viable myocardium brick red, and necrotic tissue appears white. 22 The ANRs, ischemic and normally perfused regions, and the areas of necrotic and nonnecrotic regions in each slice were determined by planimetry. These areas in each slice were multiplied by the weight of the slice, and the results summed to obtain the mass of the no-reflow, risk, and infarcted areas.
Statistical analysis
Data are calculated and tabulated using Excel work sheets. Student t tests were performed using StatPlus Software (AnalystSoft, Alexandria, Virginia). Repeated measures analysis of variance and analysis of covariance (ANCOVA) were performed with SAS (Version 9, Cary, North Carolina). Data are expressed as mean ± standard error of the mean. Statistical significance was accepted at P < 0.05.
Results
Protocol 1
A total of 17 animals were used for this protocol. One animal was excluded due to a technical error. The remaining 16 animals were included in this protocol: hypothermia, n = 8 and normothermia, n = 8. Baseline temperature was 38.8°C ± 0.1°C in the hypothermia group and 38.6°C ± 0.1°C in the normothermia group. At 25 minutes of occlusion, temperature was 32.3°C ± 0.2°C in the hypothermia group and 38.6°C ± 0.1°C in the normothermia group (Figure 2A, P < 0.001 for group effect). There were no significant differences in HR and mean arterial pressure (MAP) at baseline between groups. At 25 minutes of occlusion, HR was 167 ± 4 beats per minute in the N group and 130 ± 8 in the H group and remained lower in the H group throughout reperfusion (P < 0.05 for group effect over time). At 25 minutes of occlusion, MAP was 67 ± 3 mm Hg in the N group and 54 ± 2 in the H group and remained lower in the H group throughout reperfusion (P < 0.05 for group effect over time).
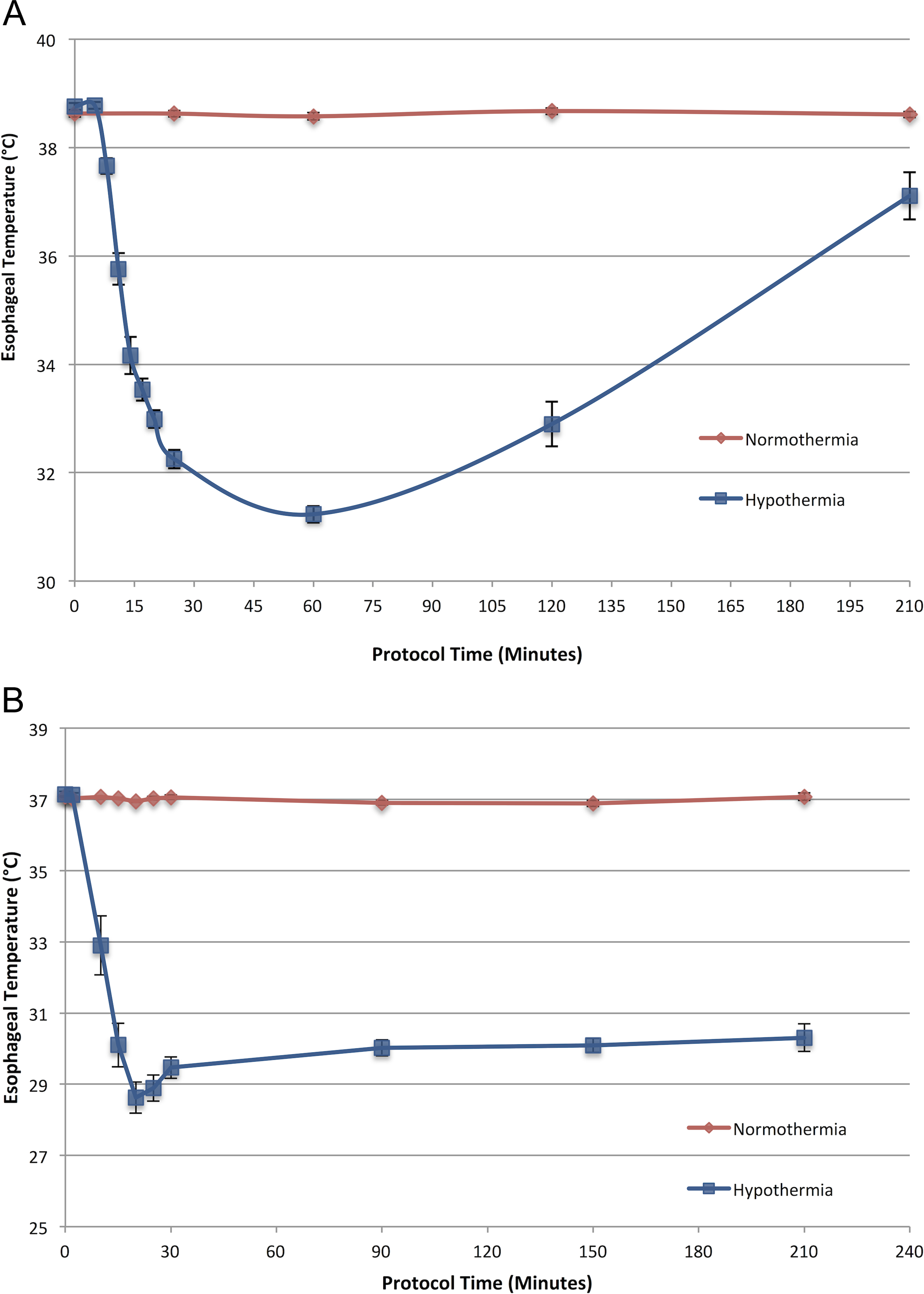
The temperature versus time comparison between groups. A, Protocol 1: The ThermoSuit was engaged at time (t) = 5 (minutes) and disengaged at approximately t = 15. The animal was removed from the ThermoSuit at approximately t = 25. Active rewarming began at t = 90 (P < 0.001 for group effect on temperature) Onset of CAO was t = 0, CAR began at t = 30. B, Protocol 2: The ThermoSuit was engaged at t = 2 and disengaged at approximately t = 12 (P < 0.001 for group effect on temperature). Onset of CAO was t = 0, CAR began at t = 30. CAO indicates coronary artery occlusion; CAR, coronary artery reperfusion.
Risk Zone, Infarct Size, and ANR
Mean AR as a fraction of the LV was 0.28 ± 0.02 in the hypothermia group and 0.29 ± 0.04 in the normothermia group (Figure 3A, P = 0.80). The area of necrosis (AN) as a fraction of the AR was 0.08 ± 0.02 in the hypothermia group and 0.44 ± 0.05 in the normothermia group (Figure 3B, P < 0.001). Thus, hypothermia caused an 82% reduction in myocardial infarct size. Area of no reflow as a fraction of the AR was 0.05 ± 0.01 in the hypothermia group and 0.44 ± 0.04 in the normothermia group (Figure 3C, P < 0.001). Thus, hypothermia caused an 89% reduction in the size of the no-reflow zone. When AN/LV was expressed as a function of AR/LV, ANCOVA revealed a significant effect of group on the relationship (Figure 3D, P < 0.001). When ANR/LV was expressed as a function of AR/LV, ANCOVA revealed a significant effect of group on the relationship (Figure 3E, P < 0.001). Therefore, for any size risk zone, both AN and ANR were significantly smaller in the hypothermia group compared to normothermic values. An example of the heart slices in this protocol can be seen in Figure 4. Hypothermia did not cause arrhythmias, rewarming shock, or shivering. Figure 5 shows the second-from-apical, TTC-stained LV slice in all rabbits; large, confluent zones of necrosis are observed in normothermic but not in hypothermic rabbits.

Planimetry data from protocol 1. A, Area at risk comparison between groups: normothermia, 0.29 ± 0.04; hypothermia, 0.28 ± 0.02 (P = 0.80). B, Area of necrosis as a fraction of area at risk comparison between groups: normothermia, 0.44 ± 0.05; hypothermia, 0.08 ± 0.02 (P < 0.001). C, Area of no reflow as a fraction of area at risk comparison between groups: normothermia, 0.44 ± 0.04; hypothermia; 0.05 ± 0.01 (P < 0.001). D, AN/LV versus AR/LV (P < 0.001, ANCOVA). E, ANR/LV versus AR/LV (P < 0.001, ANCOVA). AN, area of necrosis; ANCOVA, analysis of covariance; ANR, area of no reflow; AR, area at risk; LV, left ventricle.
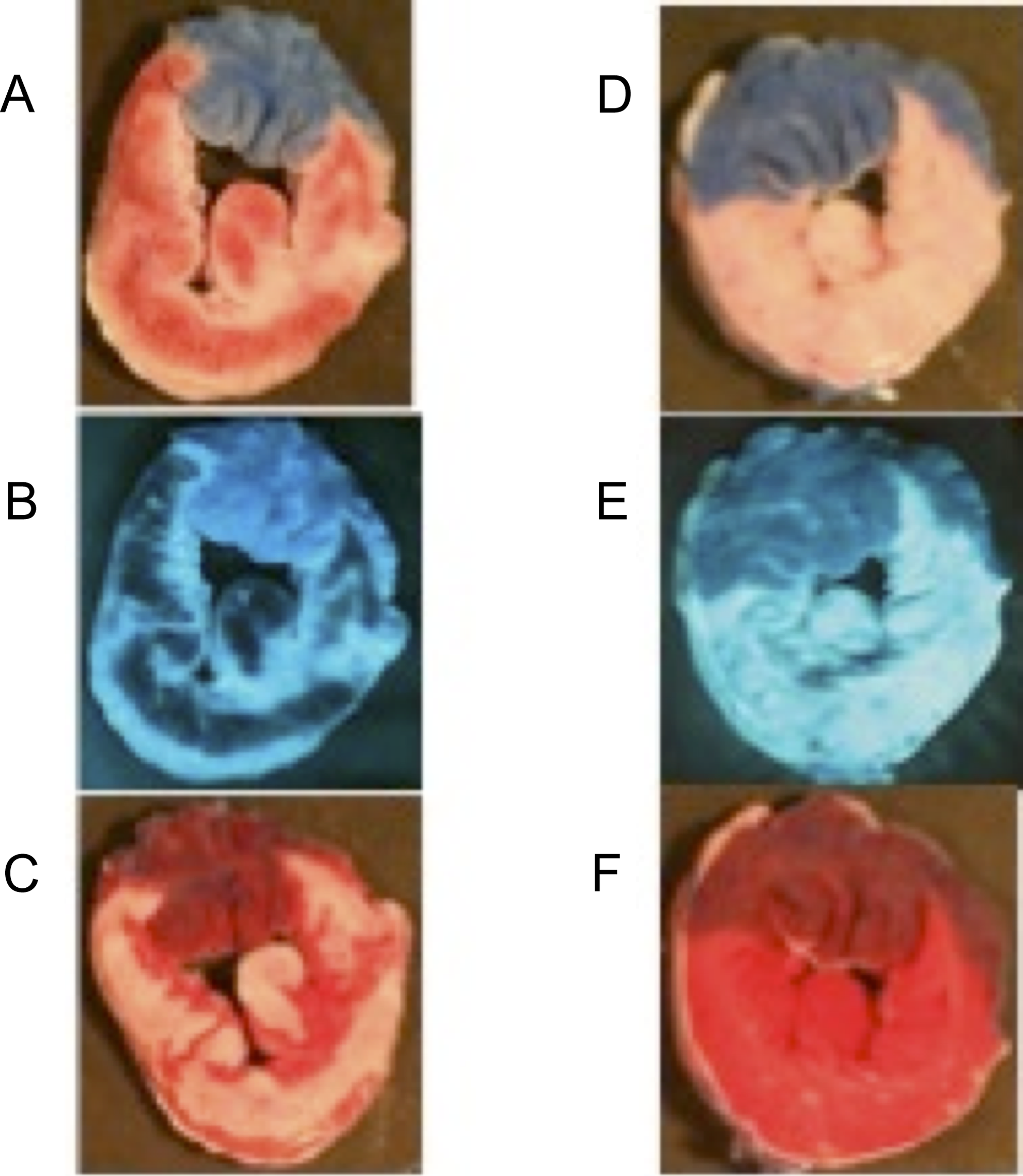
Examples of area at risk, area of no reflow, and area of necrosis in protocol 1. These are single transverse LV slices representative of our methods as well as differences between groups in the rabbit model. A, Area at risk in a normothermic heart (area at risk in pink is sizable involving >60% of this particular LV slice; area not at risk in blue). Note the areas of gross hemorrhage or vascular congestion in red. B, Area of no reflow in a normothermic heart (area of no reflow appears as dark and nonfluorescent crescent shapes within the risk zone). C, Area of necrosis in a normothermic heart (viable tissue is brick red, necrosis is white confluent area within the risk zone). D, Area at risk in a hypothermic heart (area at risk in pink is sizable involving ∼60% of this particular LV slice; area not at risk in blue). Note the lack of gross hemorrhage in the risk zone. E, Area of no reflow in a hypothermic heart (area of no reflow appears dark but is minimal). F, Area of necrosis in a hypothermic heart (viable tissue is brick red; necrotic area is not seen). LV indicates left ventricle.

Comparison of the second from apical rabbit LV slices after incubation in triphenyltetrazolium chloride (TTC). Each heart from protocol 1 is included in this figure. Notice the white, confluent infarcts in the normothermic hearts (top). The hypothermic hearts (bottom) are largely devoid of these areas.
Protocol 2
A total of 10 animals were used for this protocol. No animals were excluded: hypothermia, n = 5 and normothermia, n = 5. Baseline temperature was 37.1°C ± 0.09°C in the hypothermia group and 37.1°C ± 0.03°C in the normothermia group. At 25 minutes of occlusion, temperature was 28.9°C ± 0.4°C in the hypothermia group and 37.0°C ± 0.1°C in the normothermia group (Figure 2B, P < 0.001 for group effect).
Risk Zone, Infarct Size, and ANR
Mean AR/LV was 0.38 ± 0.08 in the hypothermia group and 0.41 ± 0.03 in the normothermia group (Figure 6A, P = 0.73). Mean AN/AR was 0.14 ± 0.06 in the hypothermia group and 0.51 ± 0.05 in the normothermia group (Figure 6B, P < 0.01). Thus, hypothermia caused a 73% reduction in infarct size. Mean ANR/AR was 0.004 ± 0.003 in the hypothermia group and 0.33 ± 0.05 in the normothermia group (Figure 6C, P < 0.01). Thus, hypothermia caused a 99% reduction in the size of the zone of no reflow. When AN/LV was expressed as a function of AR/LV, ANCOVA revealed a significant effect of group on the relationship (Figure 6D, P < 0.01). When ANR/LV was expressed as a function of AR/LV, ANCOVA revealed a significant effect of group on the relationship (Figure 6E, P < 0.001). Therefore, for any size risk zone, both AN and ANR were significantly smaller in the hypothermia group compared to normothermic values. An example of the heart slices in this protocol can be seen in Figure 7.

Planimetry data from protocol 2. A, Area at risk comparison between groups: hypothermia, 0.38 ± 0.08; normothermia, 0.41 ± 0.03 (P = 0.73). B, Area of necrosis as a fraction of area at risk comparison between groups: hypothermia, 0.14 ± 0.06; normothermia, 0.51 ± 0.05 (P < 0.01). C, Area of no reflow as a fraction of area at risk comparison between groups: hypothermia, 0.004 ± 0.003; normothermia, 0.33 ± 0.05 (P < 0.01). D, AN/LV versus AR/LV (P < 0.01, ANCOVA). E, ANR/LV versus AR/LV (P < 0.001, ANCOVA). ANCOVA, analysis of covariance; ANR, area of no reflow; AN, area of necrosis; AR, area at risk; LV, left ventricle.

Examples of area at risk, area of no reflow, and area of necrosis in each group of protocol 2. These are single transverse left ventricle (LV) slices representatives of our methods as well as differences between groups in the rat model. A, Area at risk in a normothermic heart (area at risk in pink; area not at risk in blue). B, Area of no reflow in a normothermic heart (area of no reflow appears dark and nonfluorescent). C, Area of necrosis in a normothermic heart (viable tissue is brick red, necrotic area is white and appears as a large homogeneous infarct). D, Area at risk in a hypothermic heart (area at risk in pink; area not at risk in blue). E, Area of no reflow in a hypothermic heart (area of no reflow appears minimal). F, Area of necrosis in a hypothermic heart (viable tissue is brick red; necrotic area is white). Nearly no necrosis is observed with the hypothermic treatment.
Discussion and Conclusion
In these 2 separate animal models, we have shown a profound reduction in both myocardial infarct size and ANR size (82% and 89%, respectively, in protocol 1 and 73% and 99%, respectively, in protocol 2). These reductions in infarct size are greater than any other single intervention previously observed in our rabbit and rat models (Table 1). In the rabbit, the only treatment that rivaled our results was the combination of regional hypothermia, ischemic preconditioning, and treatment with cariporide. 23
Interventions That Have Reduced MI Size in the Rabbit and Rat Models of 30 to 60 Minutes of Occlusion Followed by 3 to 4 Hours of Reperfusion in the Kloner Laboratory.
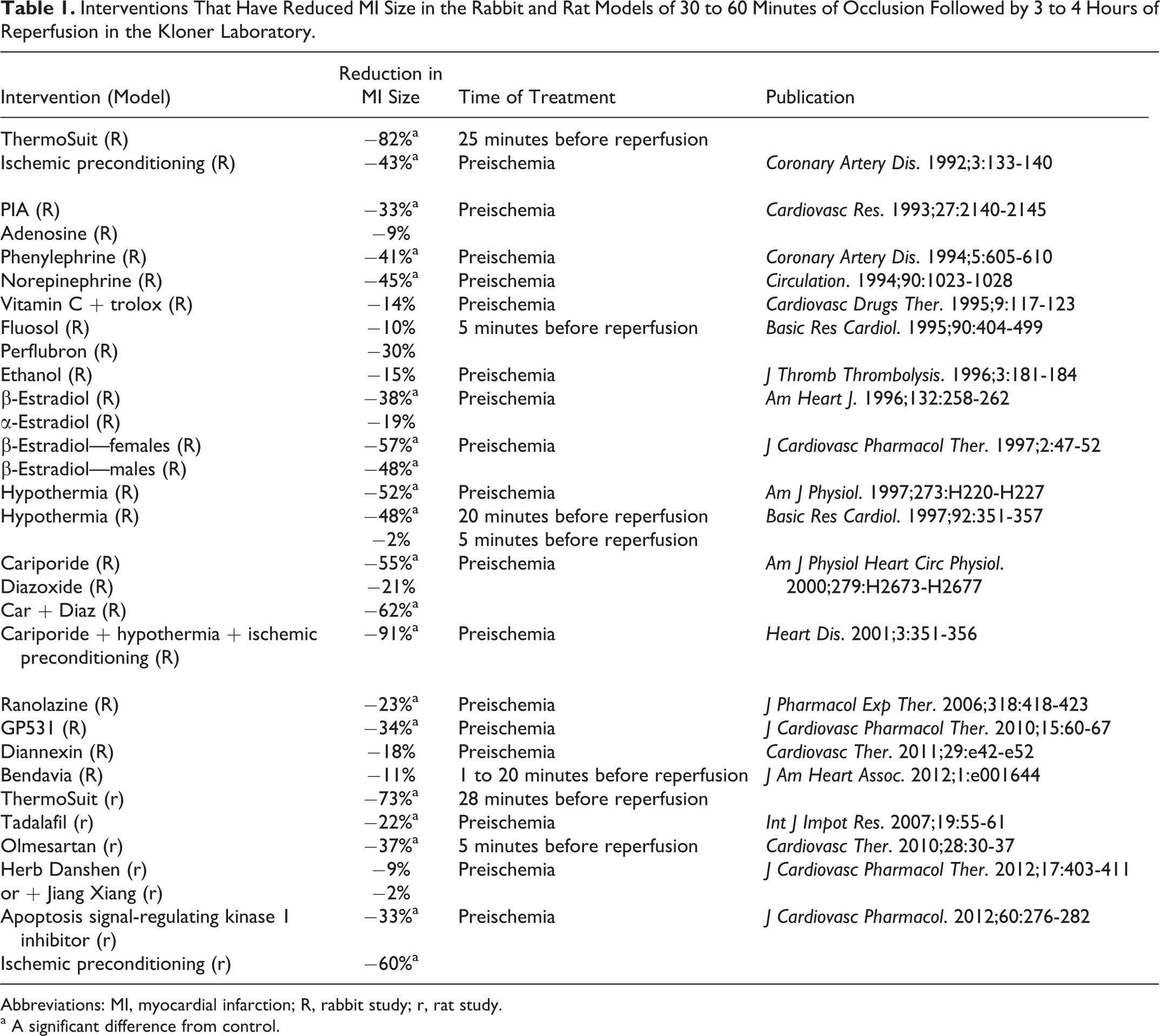
Abbreviations: MI, myocardial infarction; R, rabbit study; r, rat study.
a A significant difference from control.
Unlike other techniques used to induce hypothermia such as the bag of iced saline,9- 12 heat-exchange catheters, 13 -15 infusion of cold fluid into the vasculature, 15 into the peritoneum, 16 and into the intrapericardial space, 17 and use of other surface cooling techniques, 18 the ThermoSuit is effective, noninvasive, rapid in its induction of hypothermia and portable. This technique would theoretically allow for cooling to begin in either the home of the patient or the ambulance en route to a catheterization laboratory. Although being portable, the ThermoSuit cools the animal or patient very rapidly (in approximately 20 minutes) when compared to several hours of cooling needed by equally noninvasive cooling blankets. 18 As seen in our studies, once the subject is cooled, temperature remains reduced for an extended period of time, even after removal from the suit. In theory, a patient could be treated, removed from the suit, and then transported to an invasive catheterization laboratory for PCI while remaining hypothermic.
Hypothermia is a concept being tested not only in our laboratory9- 12, 17 but also in many other laboratories with varying degrees of success, as myocardial temperature has been shown to correlate well with myocardial infarct size. 8 Dae and coworkers have shown that endovascular cooling in pigs caused a large reduction in infarct size. 13 These results were not always replicated in a human population, 14 perhaps because many patients were not adequately cooled prior to reperfusion. However, analysis of some clinical trials that were negative overall showed subgroups of patients who had smaller infarct sizes when patient temperatures were reduced to <35°C prior to reperfusion of an anterior infarction. 24,25 Issues with endovascular cooling devices are that they cool at a much slower rate than the ThermoSuit device and have not consistently resulted in temperatures <35°C prior to reperfusion.
The ThermoSuit device has already been approved for use during episodes of hyperthermia, thus showing that the ThermoSuit is safe for other human applications. The ThermoSuit is also being used off label after cardiopulmonary resuscitation to preserve brain function, as hypothermia has been researched extensively and may be of benefit in neurological models. 20,26
Although our study was not meant to look directly at the mechanism of cardioprotection, the effect of hypothermia on myocardial biochemistry has been previously investigated. One hypothesis suggests that hypothermia could slow cardiac energy metabolism, thus protecting cardiomyocytes from ischemic death. Simkhovich and coworkers from our group found elevated levels of adenosine triphosphate (ATP) and glycogen stores in hypothermic myocardium compared to normothermic myocardium in rabbits, suggesting that ATP and glycogen are preserved during periods of hypothermia. 27 Adenosine triphosphate preservation, as well as the preservation of myocardial function in hypothermic hearts, was also observed by Ning and coworkers. 28 Although hypothermia lowered HR and MAP in the rabbit, a reduction in oxygen demand due to lower HR and MAP alone is unlikely to explain the reduction in infarct size, since we have previously shown that pacing the cold heart back to baseline HR and MAP did not abolish hypothermic cardioprotection. 10 An additional and not exclusionary hypothesis is that extracellular signal-regulated kinase (ERK) activity may be preserved during hypothermia. Yang and coworkers demonstrated that during the period of ischemia, phosphorylated ERK and mitogen-activated protein kinase (extracellular signal-regulating kinases) levels were significantly higher in hypothermic hearts when compared to normothermic hearts. 29
Although rabbits and rats are very small animals, it is thought that the cooling times for these animals would be much shorter than larger animals or in humans in a clinical environment. Janata et al examined this phenomenon in swine (29-35 kg) and achieved a 5.5°C temperature drop in 9 minutes using the ThermoSuit device. 20 Howes and coworkers used the ThermoSuit device in patients to protect brain function after cardiopulmonary resuscitation. Patients who were sedated with propofol prior to the hypothermic therapy achieved goal body temperature (<34°C) in 27 minutes. 21 Other clinical studies examining hypothermia to improve outcome in the setting of MI ( (Intravascular Cooling in Myocardial Infarction as Adjunctive to Percutaneous Coronary Intervention [ICE-IT] and Rapid Intravascular Cooling in Myocardial Infarction as Adjunctive to Percutaneous Coronary Intervention, [RAPID-MI-ICE]) have used a combination of buspirone and meperidine for patient tolerance and to limit shivering, although these trials used endovascular cooling plus a cold saline infusion to cool the patients. 30
The present study is the first to investigate the use of the ThermoSuit in experimental MI models. We have shown that hypothermia was induced very rapidly using this technique, and that the ThermoSuit profoundly reduced infarct size compared to normothermic controls (82% in the rabbit and 73% in the rat). Furthermore, the ThermoSuit markedly reduced the ANR by 89% in the rabbit and 99% in the rat. These reductions in infarct size and no-reflow area are the largest percentage of reductions with a single agent that this laboratory has observed with more than 35 years of experience. This method of cardioprotection is eminently practical with regard to clinical applications. More studies are required to examine the effect of this method of hypothermia induced at different times during the CAO and different CAO durations.
Limitations
Although myocardial necrosis and no reflow were limited markedly in this study, this study was not without limitations. First, the time of reperfusion was short (3 hours). This did not allow for animals to reach baseline temperature at the end of the protocol and may not have allowed for sufficient time to see the development of necrosis. We are planning on investigating this occlusion and cooling protocol in a 6-week, chronic study. This will allow for the study of long-term damage as well as cardiac function post-MI.
Additionally, the n value (5) in protocol 2 was quite small. This arm of the study served as an acute pilot study before undertaking the aforementioned chronic study, in which the n values will be much greater. Despite this low n value, we showed highly significant differences between groups. Such would not be possible with a low n value examining a therapy with no biological impact.
Footnotes
Author's Contribution
Michael J. Herring, Wangde Dai, Sharon L. Hale, and Robert A. Kloner contributed to conception and design of the study; Michael J. Herring, Wangde Dai, and Sharon L. Hale substantially contributed to data acquisition; Michael J. Herring, Wangde Dai, Sharon L. Hale, and Robert A. Kloner contributed to analysis and interpretation of the data; Michael J. Herring involved in drafting of the manuscript; Michael J. Herring, Wangde Dai, Sharon L. Hale, and Robert A. Kloner contributed to critical revision of the manuscript for intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
