Abstract
Macrophages are involved in every cardiovascular disease and are an attractive therapeutic target. Macrophage activation is complex and can be either beneficial or deleterious, depending upon its mode of action, its timing, and its duration. An important macrophage characteristic is its plasticity, which enables it to switch from one subset to another. Macrophages, which regulate healing and repair after myocardial infarction, have become a major target for both treatment and diagnosis (theranostic). The aim of the present review is to describe the recent discoveries related to targeting and modulating of macrophage function to improve infarct repair. We will briefly review macrophage polarization, plasticity, heterogeneity, their role in infarct repair, regeneration, and cross talk with mesenchymal cells. Particularly, we will focus on the potential of macrophage targeting in situ by liposomes. The ability to modulate macrophage function could delineate pathways to reactivate the endogenous programs of myocardial regeneration. This will eventually lead to development of small molecules or biologics to enhance the endogenous programs of regeneration and repair.
Introduction
The inability of the human heart to regenerate efficiently represents one of the greatest hurdles in cardiovascular medicine. Despite improvements in interventional and pharmacological therapy, cardiovascular disease is the leading cause of death in Europe, accounting for 4.1 million deaths annually, 46% of the total number of deaths per year. 1 Thus, there is a clear and immediate need for new effective therapies.
The adult heart responds to injury with inflammation, scar formation, and fibrosis, with minimal regeneration. 2 The inflammatory response to myocardial injury is an essential component of infarct healing. 3,4 However, inflammation may continue beyond the early period of healing and also spread into the remote myocardium, 5,6 resulting in adverse ventricular remodeling and heart failure. This inflammatory reaction has emerged as a therapeutic target. Although, encouraging results have been obtained in animal models, the results of clinical trials targeting the inflammatory response have been disappointing. 7,8
Regenerative cardiovascular medicine holds immense promise for treating damaged and diseased hearts. 9,10 The term regeneration defines the healing process that rebuilds damaged tissue to its normal structure and function. 11 The term repair defines the healing process that replaces the damaged tissue with a scar. 11 A potential therapeutic strategy is to augment endogenous regenerative mechanisms and to stimulate regeneration—a growth of new heart muscle from the milieu of cardiac progenitors and cardiomyocytes, spared by injury. 12
The most common cause of acute heart injury is acute myocardial infarction (MI). 13 Macrophages are the dominant cells in the infarcted myocardium, controlling the initiation, maintenance, and resolution of the inflammatory response (Figure 1). 14,15 They facilitate the healing process by removing dead cells, as well as by secreting trophic, angiogenic, and profibrotic cytokines, chemokines, and proteases. 15,17 These healing properties are attributed to distinct monocyte and macrophage subsets. 14,16,18–20 Thus, control of macrophage function could be a major therapeutic target in directing the process of infarct repair. 21,22

The aim of our review is to describe recent discoveries related to targeting and modulating macrophage function to improve infarct repair. The pivotal role of monocytes and macrophages in other cardiovascular diseases, such as atherosclerosis and heart failure, is beyond the scope of this review and has been extensively described elsewhere. 23,24 We will focus on macrophage polarization, plasticity and heterogeneity, their role in infarct repair, and bidirectional interaction with stem cells, particularly mesenchymal cells. Specifically, we will focus on the potential of macrophage targeting and modulation in situ by drug carriers, particularly liposomes. The ability to improve infarct healing and repair by macrophage modulation could advance the field of cardiovascular regenerative medicine, particularly in elderly and sick individuals who have an impaired and uncontrolled immune system and reparative capability.
Macrophages are Characterized by Plasticity and Heterogeneity
Macrophages are versatile cells that embrace diverse activation states in response to stimuli. Macrophages play an important role in many features of human health: from development and homeostasis to regulation of inflammation, angiogenesis, healing, regeneration, and repair. 16,25 –27 An important macrophage characteristic is its plasticity, which enables it to switch from one phenotype to another. 27 However, attempts to define and classify monocyte and macrophage subsets, particularly in vivo, are complex and have created confusion.
Monocytes, macrophage precursors, comprise around 10% of leukocytes in humans and 4% in mice. The old classical dogma dictated that tissue macrophages develop from hematopoietic stem cells (HSCs) in bone marrow via blood monocyte intermediates. However, new data show that some adult tissue-resident macrophages develop from embryonic progenitors independent of HSCs and can self-renew. 28 –31 The number of resident macrophages in normal adult tissue varies: they are abundant in the spleen, liver, and lung while barely detectable in the heart. However, following injury, monocytes migrate to the damaged heart and convert to macrophages. The ability of cardiac macrophages to proliferate in response to injury has been an enigma until lately.
Most recently, Epelman et al have shown by genetic fate mapping that most subsets of resident cardiac macrophages originate from yolk sac and fetal monocyte progenitors (Table 1). 28 During normal hemostasis, resident macrophages are maintained through local proliferation. However, after macrophage depletion, or during inflammation, circulating-Ly6Chi monocytes (M1 like) infiltrate the heart and dominant cardiac macrophages. In parallel, in response to injury or inflammation, resident macrophages also proliferate and expand. Expression of C-C chemokine receptor type 2 (CCR2) distinguished cardiac macrophages of adult monocytes from those of embryonic origin. These findings revealed the presence of multiple macrophage subsets in the heart, with different functions and origins (Table 1). 28
Abbreviations: IL, interleukin.
The heterogeneity of macrophages is dictated by different environmental signals from different tissues that undergo changes during injury, healing, and repair. A detailed discussion of the different classifications of monocytes and macrophages by their surface marker expression profiles is beyond the scope of this review and has been presented in Tables 2 and 3. In brief, monocyte/macrophage subsets are categorized by their chemokine receptors, surface markers, and function (Tables 2 and 3). Most of the studies on monocyte subsets have been performed in mice, which have a different distribution of mononuclear cells. For example, circulating “classic” monocytes in mice, such as Ly6Chi, account for 50% of blood monocytes compared with 90% cluster of differentiation (CD) 14hi CD16lo “classic” monocytes in humans. 33 Several monocyte/macrophage classifications using various markers have been suggested, without consensus, due to the fact that such classifications vary among mice, rats, and humans (Table 2). Probably, the most popular is the M1/M2 classification based on classic (interferon or lipopolysaccharide [LPS]) and alternative (interleukin [IL] 4 and IL-13) activation (Tables 2 and 3). Although this might represent an over simplification of a complex and heterogeneous condition, the M1 and M2 classification is used to simplify the wide spectrum of macrophage activation and polarization.
Monocyte/Macrophage Characteristics.
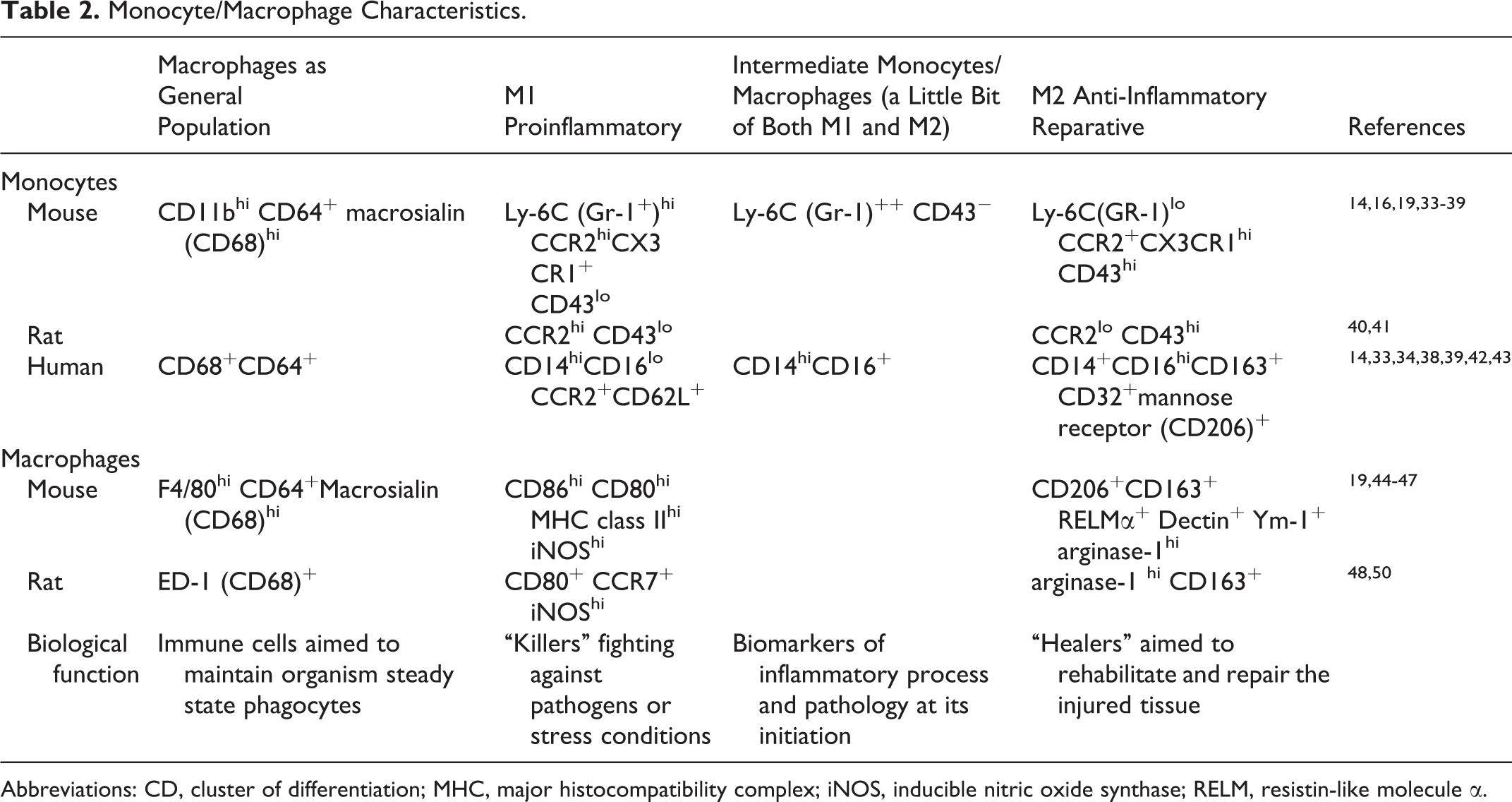
Abbreviations: CD, cluster of differentiation; MHC, major histocompatibility complex; iNOS, inducible nitric oxide synthase; RELM, resistin-like molecule α.
Mouse Macrophage Subsets.

Abbreviations: IFNγ, interferon γ; LPS, lipopolysaccharide; TLR, toll-like receptor; CD, cluster of differentiation; MHC, major histocompatibility complex; IL, interleukin; TNF, tumor necrosis factor; ROS, reactive oxygen species; iNOS, inducible nitric oxide synthase; PAMPs, pathogen-associated molecular patterns; HMGB, high-mobility group box; ATP, adenosine triphosphate; TGF, transforming growth factor.
In response to local triggers, macrophages are “reeducated” and polarize into either an M1 (proinflammatory) or an M2 (anti-inflammatory and reparative) phenotype. 19,62,66,67 M1 macrophages are proinflammatory 65,68 and have been linked to the development of atherosclerosis 69,70 as well as to acute inflammation after skeletal muscle injury and MI. 14,16 On the other hand, M2 macrophages are anti-inflammatory and reparative 65 and are implicated in the advanced stages of atherosclerosis, 34 MI healing, 14,16 fibrosis, and the development of pulmonary hypertension. 71 Thus, modulating macrophage polarization could lead to a therapeutic strategy to prevent and treat these disorders.
There is evidence that the ability to modulate macrophage function relies on local signals, cell contact-dependent mechanisms, paracrine effects through the release of soluble factors, and epigenetic changes (reviewed by Krampera, Doorn, Delaroasa, and Galli). 57,72 –74 It has been suggested that a broad panel of molecular pathways could affect macrophage polarization.
Additionally, M2 macrophages contribute to the pathogenesis of cancer. Solid tumors induce M2 macrophage activation, the so-called tumor-associated macrophages, which then stimulate angiogenesis and tumor growth. 75 In summary, caution is recommended when classifying infarct macrophages. Due to the complexity that arises from the mixed phenotypes in vivo, some investigators classify macrophages based on their function: host defense, wound healing, or immune regulation (Table 2). 51,68
Macrophages in Acute MI
Because the adult mammalian heart lacks significant regenerative power, repair of the infarcted myocardium is dependent upon the coordinated process of inflammation and scar formation. 4 During MI, cardiomyocyte death triggers inflammation that is essential to clean the debris and subsequently induce scar formation. 76 However, disproportionate activation of inflammatory monocytes and M1 macrophages could propagate tissue damage and worsen post-MI remodeling. 4,77,78
Macrophages are the central immune cells in the infarcted myocardium, controlling the progression and resolution of inflammation. 16 Early depletion of monocytes or macrophages, 16,79 –82 as well as inhibition of macrophage migration, 83 worsen healing and provoke adverse left ventricular (LV) remodeling after MI. On the other hand, controlled activation of macrophages 21,84 improves myocardial healing and repair, thus subsequently improving remodeling and function. These findings were reproduced in a mouse model of kidney disease when ex vivo-activated macrophages reduced the severity of chronic renal disease and fibrosis. 85 Additionally, selective mineralocorticoid receptor blockade with eplerenone, immediately after MI, accelerates macrophage infiltration and switches to M2 activation during infarct healing, leading to improved tissue repair, angiogenesis, and remodeling. 86
Nahrendorf et al 16 were the first to show that 2 distinct subsets of monocytes, precursors of tissue macrophages, participate in healing after MI in a sequential manner. CD11bhigh/Ly-6Chigh cells, exhibiting phagocytic and proinflammatory functions, accumulate during phase I (1-3 days post-MI), while CD11bhigh/Ly-6Clow cells, attenuating the inflammatory response and expressing vascular endothelial growth factors (VEGFs), are present during phase II (4-7 days post-MI). 16 Rapid monocyte turnover in the infarcted myocardium has been reported. Although the Ly-6Chigh monocytes infiltrate the infarct in large numbers, 16,87 many of them depart or die within an average of 20 hours. 87 Macrophages derived from infiltrating monocytes acquire M1-proinflammatory characteristics. This rapid monocyte turnover in the infarcted heart could be significant for future monocyte/macrophage targeting. Nevertheless, as inflammation gives way to resolution, other Ly-6Cint/low monocytes, equivalent to M2, emerge and contribute to infarct healing and repair. 16,87 These pioneering studies on sequential monocyte and macrophage subset dynamics in the infarcted myocardium have been confirmed by us and others. 26,81,82 Thus, control of macrophage function could be a major therapeutic target in optimizing the process of infarct repair.
Macrophages and Myocardial Regeneration
Due to their plasticity, macrophages coordinate and influence tissue remodeling, regeneration, and repair. 88 –93 Macrophages are abundant in the embryonic heart and are essential in cardiac morphogenesis. Subsequently, macrophage depletion leads to cardiac malformation. 94 Macrophages secrete a variety of trophic mediators including platelet-derived growth factors, transforming growth factors, insulin-like growth factor 1, Wnts ligands, and thymosin β4 (Table 4). Macrophage-derived factors dictate cell fate decisions and promote regeneration of neurons, 95 nerves, 96 skeletal myocytes, 88,92 and hepatocytes. 97 Thus, macrophage activation is a critical process contributing to healing and repair.
Cytokines Secreted by Macrophages.

Abbreviations: IFNγ, interferon γ; TGF, transforming growth factor; VEGF, Vascular endothelial growth factor; bFGF, basic fibroblast growth factor; TNF, tumor necrosis factor; MI, myocardial infarction; Th1, T-helper 1; Th2, T-helper 2.
The neonatal heart of rat and mouse maintains certain regenerative mechanisms, but the window of regeneration is limited and disappears soon after birth. 2,125,126 We noticed a massive transient accumulation of monocytes and macrophages at the site of myocardial resection and subsequent regeneration in the neonatal heart of mouse (Konfino T, MSc, unpublished data). 127
Indeed, most recently, Aurora et al have identified macrophages as mediators of neonatal heart regeneration. 128 Following MI and monocyte and macrophage depletion in newborn (day 1) mice, they found that monocytes and macrophages are essential for neonatal heart regeneration, particularly myocardial angiogenesis. Comparison between the immune reaction after MI between mice at day 1 after birth (regenerative phase) and at day 14 (nonregenerative phase) revealed differences in phenotype and function of cardiac monocytes and macrophages. Macrophages derived from newborn mouse represent a unique population that does not match the classical characteristics of M1 or M2 macrophages. Thus, the monocytes/macrophages from newborn mice might have regenerative functions different from those of the profibrotic activities of adult macrophages. 128 Understanding the role of macrophages in cardiac regeneration may lead to the development of novel therapies that could improve cardiac function after MI. 129
Macrophages have been suggested to interact with stem cells, progenitor cells, and myocytes and play a critical role during skeletal muscle regeneration. 88,92,130,131 Furthermore, a most recent study has suggested that macrophages are essential for limb regeneration in salamander. 132 Systemic macrophage depletion during limb amputation resulted in failure of limb regeneration and fibrosis. Significantly, full regenerative capacity was restored by reamputation once endogenous macrophage populations had been replenished. Together, these studies suggest that identification of macrophage-derived therapeutic molecules may promote myocardial regeneration. 132
Although myocardial regeneration in the adult heart is scarce, its mechanism is unclear. It may originate from resident cardiac stem cells, 133,134 cardiomyocyte dedifferentiation and proliferation, 125,135 or both. 136 Macrophages might play a role in myocardial regeneration. For example, zebra fish have an amazing ability to regenerate their hearts. 137 However, treatment with an anti-inflammatory glucocorticoid inhibits myocardial regeneration in the zebra fish. 137 Thus, manipulating its immune response could regulate zebra fish cardiac repair.
During acute MI in mouse, macrophages in the infarcted tissue and border zone express high levels of oncostatin M (OSM). 138 Oncostatin M is a member of the IL-6 family. It exerts several unique effects, such as stimulation of cardiomyocyte dedifferentiation. Dedifferentiation of adult cardiomyocytes has been suggested to be the initial step before cardiomyocyte proliferation. 125,135 Thus, it is tempting to speculate that macrophage OSM can promote cardiomyocyte proliferation and myocardial regeneration. However, it should be noted that sustained dedifferentiation, cardiac remodeling, and heart failure characterize continuous uncontrolled stimulation with OSM. 138 Together, these studies on the potential role of macrophages in myocardial repair and regeneration suggest that future research is necessary to understand how modulation of macrophage response to cardiac injury can influence heart regeneration and functional outcomes. 129
Interaction of Macrophages with Mesenchymal Stem/Stromal Cells
A relatively simple and feasible approach to target and modulate macrophage function in situ is to use mesenchymal stromal cell (MSC) therapy. Mesenchymal stromal cells are nonhematopoietic, multipotent, progenitor cells found in various adult tissues, particularly bone marrow and adipose tissue. 139 They are characterized by their paracrine reparative and immunomodulatory properties. Mesenchymal stromal cells ameliorate infarct healing and repair, 140 but their mechanism of action is not entirely clear.
In the last few years, it has been recognized that MSCs are modulators of immune response 141 and can attenuate a systemic inflammatory response in animal models of lung injury and sepsis. 142 These protective effects could be mediated by macrophages. 22 Recent reports have shown that MSCs act via macrophages, switching them from M1 to M2 (reparative) phenotype, in vitro 143 –146 and in vivo (Table 5). 81,82
Mesenchymal Stromal Cells Switch Macrophages From M1 to M2 Subset.

Abbreviations: MΦ, macrophages; MI, myocardial infarction; BM-MSCs, bone marrow-derived mesenchymal stromal cells; CD, cluster of differentiation; IL, interleukin; TNF, tumor necrosis factor; IFNγ, interferon γ; GMSC, gingiva-derived mesenchymal stem cells.
Mesenchymal stromal cell-based macrophage reprogramming is associated with increased secretion of M2 cytokines such as IL-10 and VEGFs. 146 Concomitantly, MSCs decrease secretion of inflammatory cytokines such as IL-1α, tumor necrosis factor α (TNF-α), IL-12, and IL-17 from macrophages. 146 Significantly, this interaction is bidirectional, and macrophages also stimulate MSCs to secrete cytokines such as IL-4 and IL-13, which are M2 inducers. 146
We have recently shown that MSC therapy significantly increased the percentage of reparative M2 macrophages (F4/80+CD206+) in the infarcted myocardium of mice, 3 and 4 days after MI. 82 Macrophage cytokine secretion, relevant to infarct healing and repair, was significantly increased after MSC therapy or incubation with MSCs or an MSC supernatant. Notably, with or without MSC therapy, transient, early macrophage depletion increased mortality after MI and produced the greatest negative effect on infarct size, LV remodeling, and function as well as a significant incidence of LV thrombus formation. These adverse effects were attenuated with macrophage restoration and MSC therapy. 82
Dayan et al 81 investigated the effect of human MSCs in an MI model of immune-deficient mice. Human bone marrow-derived MSCs and human perivascular cells from umbilical cord were infused 48 hours after MI. Mesenchymal stromal cells reduced the number of macrophage/monocytes, while the proportion of M2 macrophages was increased in the circulation and the heart, 24 hours after treatment. Moreover, LV contractility was improved 2 weeks after cell infusion but was similar to controls 16 weeks after MI.
Taken together, some of the protective effects of MSCs on infarct repair are mediated by macrophages, which are essential for early healing and repair. Thus, targeting macrophages by MSCs could be a novel strategy to improve infarct healing and repair.
Targeting Macrophages with Liposomes
Carrier-mediated drug targeting provides several advantages that improve the pharmacokinetics and bioavailability of therapeutic and imaging agents. Drug targeting by a carrier requires recognition and high-affinity binding between 2 partners: (1) a receptor (in its broadest definition) sufficiently unique to, and/or overexpressed at, the target cell membrane and (2) a targeting agent that constitutes an integral part of the carrier (with 1 macrophage-linked exception that will be discussed later), thus assuring that it will not dissociate in vivo causing carrier-targeting ability loss. Recognition and high-affinity binding operate at short range, and conditions have to be created or met in a living system in order to bring the targeted carrier close enough to its target for it to produce the desired effect. The in vivo conditions required to bring the 2 targeting partners close enough have been discussed elsewhere. 147 In the present section, we will focus on macrophages.
Targeting particular drug carriers to macrophages that are part of the reticuloendothelial system (RES) warrants special attention. One of the physiological roles of macrophages, that is, its ability to bind and internalize foreign microparticles (whether made from natural or synthetic materials) was recognized in the world of liposomes and has led to the terms active and passive targeting. 148 –151 Passive targeting is when the RES is the therapeutic target, and active targeting is when the therapeutic target is outside the RES avoiding both the RES and transferring the carrier to its target. We wish to point out that the term passive targeting (for drug carriers) is also used in another context—the enhanced retention and permeability in tumor therapy. In this review, dedicated to macrophages and infarct repair, the use of the term passive targeting will be applied only to the RES as the therapeutic target.
The case of passive liposome targeting to macrophages is the exception to the need for both partners. Macrophages possess unique scavenger receptors that recognize and bind liposomes, negating the need for a second partner (ie, the targeting agent) since the macrophages recognize and bind regular (conventional) liposomes as they are. 148 –151 Using liposomes as macrophage-targeted drug carriers has been applied for several different therapeutic goals, usually utilizing passive targeting. 152 Two veteran objectives were in the fields of cancer and Leishmania. For cancer therapy, the goal was to render macrophages tumoricidal by liposome-formulated muramil tripeptide. 148,151,153 The parasite causing Leishmania resides within the macrophages and was eradicated using antimonial drugs formulated in liposomes. 154 In the field of cancer therapy, the need for active tumor targeting was recognized early on, and extensive efforts have been successfully invested for more than 2 decades in identifying and establishing liposome requirements for RES avoidance or uptake. 155 Consequently, it is to the older literature one frequently has to go for the fundamental work done on liposome-mediated drug targeting to the RES.
We recently studied macrophage interactions with 4 liposome types. Two were regular (conventional) nano- and microsized liposomes denoted regular unilamellar (RL-ULV) and regular multilamellar (RL-MLV), respectively, and 2 were veteran developments by our team, nano- and microsized liposomes, surface-modified by covalent anchoring of hyaluronan, denoted HA-ULV and HA-MLV, respectively. 155 The macrophages bound all liposome types, with the affinity sequence of HA-MLV > HA-ULV > RL-MLV > RL-ULV, but only the RL-MLVs were internalized. 155 The HA-MLVs and RL-MLVs emerged as the favored candidates for drug delivery to macrophages. The finding that, despite being microparticles, HA-MLVs are not internalized by macrophages, yet bind to them with high affinity (the highest), is a manifestation of their binding to hyaluronan-specific receptors (the CD44 family) harbored by macrophage membranes rather than to scavenger receptors. 155 Although CD44 receptors are expressed in the majority of tissues, the one expressed on normal tissues is usually CD44s (also named CD44H), whereas in cancer and in inflammatory diseases those expressed are usually the CD44 splice variants (CD44v). 156 It is well known that the affinity of hyaluronan to the latter is significantly higher than to CD44s. 156
Testing HA-MLV targeting to infarct macrophages in mouse (intravenous [IV] tail vein injection, 3 days after MI), we found a high accumulation of these liposomes in the infarcted heart, but none in the liver, spleen, or lungs. There was also no HA-MLV accumulation in the healthy hearts of the control mice. Taken together, these results are in vivo proof of the concept that HA-MLVs actively target macrophages, which are pivotal cells in the inflammatory process.
Bearing in mind that the goal is to target the drug to its site of action, the targeted carrier being a means to this end, it is not enough to have the carrier preferentially accumulate at the target. Getting the drug to its molecular target also requires consideration of the pathways for drug supply from the carrier to the cell. Contrary to a prominently held assumption, carrier internalization is not a critical requirement for small-molecular-weight drugs that operate inside the cell, such as steroids and nonsteroidal anti-inflammatory drugs (NSAIDs) applied for the treatment of inflammations and infections. One of the advantages of carrier-mediated drug delivery is the potential to perform as slow-release drug depots for the many cases that such release profiles benefit the therapy. For example, half-lives of drug release for anti-inflammatory liposomal formulations of diclofenac, dexamethasone, or their combination are in the range of 1 to 3 days. 157 The following scenario can be envisioned for liposome-mediated drug supply to the macrophage cytosol: the RL-MLVs undergo internalization—a fairly fast process that can take under an hour and is considered complete within a few hours. 151,155,158 Once the liposome is disrupted in the course of release from the endosome, the cytosol is exposed at once to the whole drug load. Whether this is beneficial or detrimental may be drug species dependent, but clearly drug supply to the cytosol will be dominated by the endocytotic process rather than by the slow-release property of the liposomal formulation. For the HA-MLVs that stay as high-affinity bound drug depots at the macrophage membrane, it is reasonable to assume that their slow-release property will dominate drug supply to the cytosol, the drug diffusing out of the liposome across the cell membrane and into the cell. Thus, it may well be that in such cases carrier internalization is not necessary and may actually be less effective than a membrane-adhering drug depot.
In conclusion, 2 types of microsized liposomes are strong candidates for drug targeting to macrophages: regular liposomes that are bound and internalized and surface-modified liposomes, bearing a ligand directed against a receptor on the macrophage membrane, that remain bound without internalization, as exemplified earlier for hyaluronan and CD44. In vitro studies on liposomes and macrophages yield data on binding thermodynamics and cellular localizations and also shed light on whether internalization does or does not take place. With a given drug, in vitro studies in established or primary macrophages can also provide insight into pathways of drug supply to the cells. However, in vivo studies with animal models of the designated pathology are required to obtain proof of concept for liposome-mediated drug targeting to macrophages for infarct repair.
An original approach to improve infarct repair is based on the delivery of apoptotic-mimicking particles, such as phosphatidylserine-presenting liposomes. 159 The rational for this approach is based on the observation that macrophages recognize apoptotic cells by exposed phosphatidylserine. 160 Phagocytosis of apoptotic cells by macrophages inhibit secretion of proinflammatory cytokines, stimulate secretion of anti-inflammatory cytokines, and thereby resolve inflammation. 160 Indeed, reprogramming of infarct macrophages by IV delivery of phosphatidylserine-presenting liposomes improved angiogenesis and prevented adverse cardiac remodeling in rat after MI. 159
Modulating Macrophage Function by Other Approaches
Short interfering RNAs (siRNAs) are an emerging technology aimed at silencing specific gene targets. 161 Unlike small molecule drugs, siRNAs are highly specific toward their target sequence and have little off-target effects, making them a useful tool in basic research and a potential new approach to treat different gene-related diseases. Several siRNAs are already in clinical trials for different diseases 162,163 including age-related macular degeneration (ClinicalTrials.gov Identifier: NCT00259753), solid tumors (ClinicalTrials.gov Identifier: 00689065), acute kidney injury after cardiac bypass surgery (ClinicalTrials.gov Identifier: NCT00554359), and more. 164 However, a major impediment for siRNA treatment still remains: that of safe and efficacious delivery to target cells. Short interfering RNA delivery faces many obstacles when administered in vivo, such as degradation by serum nucleases, immune cell phagocytosis, and renal excretion. Lately, scientists have developed new strategies to improve stability and specificity of siRNA by use of nanoparticle-mediated delivery (ie, liposomes and polymers) and conjugation to ligands. 163,165 These modifications have made siRNAs both target specific and less toxic, making them much more relevant for clinical use.
Macrophages are an appealing candidate for siRNA delivery, not only because they play roles in various diseases 166,167 but also because they are easy to target due to their phagocytic capability. As previously mentioned, macrophages are essential for the healing of infarcted myocardium. 16,82 Hence, modulation of macrophage function with siRNAs could be a novel method to treat cardiovascular disease. Several studies have begun investigating the effects of different siRNAs targeted to macrophages. In a study by Leuschner et al, silencing monocyte chemokine receptor CCR2 by nanoparticle-mediated siRNA delivery has shown to diminish the migration of inflammatory monocytes to the heart after MI and shift the balance toward an M2 anti-inflammatory subset. 168 This anti-inflammatory environment helped attenuate infarct size as well as reduce lesion size in a mouse model of atherosclerosis. Inhibition of macrophage-induced inflammation has also proven useful in a model of murine arthritis, where TNF-α siRNA nanoparticles targeted to peritoneal macrophages, lowered systemic inflammation, and joint swelling. 169 Another example of the beneficial use of macrophage-targeted siRNAs is the work of Aouadi et al, where macrophage administration of Map4k4-siRNA reduced cytokine toxicity in a mouse model of LPS-induced systemic inflammation by inhibiting TNF-α and IL-1β production. 170 In summary, it seems that the application of siRNAs for macrophage gene silencing could be used for these and many more targets involved in macrophage migration, cytokine secretion, and even polarization.
Another approach that can both reduce the number of inflammatory macrophages and switch their function to reparative phenotype after MI is administration of thymosin β4 sulfoxide. 108 Administration of thymosin β4 sulfoxide in mouse after MI improves healing and limits fibrosis. 108 Finally, another potential method to modulate macrophage function is by targeting heme oxygenase 1 (HO-1). Heme oxygenase 1, also known as heat shock protein 32, has long been known for its anti-inflammatory properties. Heme oxygenase 1 exerts its protective effects through the degradation of heme and the subsequent production of anti-inflammatory, antiapoptotic, and antioxidative molecules. 171 Hemin is an iron-containing porphyrin that potently upregulates the expression of HO-1 in macrophages. 172,173 Heme oxygenase 1 decreases macrophage inflammatory cytokine levels, such as TNF-α, through an IL-10-mediated pathway, 173,174 making macrophage HO-1 a potential therapeutic target, especially in diseases associated with prolonged inflammation. Indeed, several studies have shown that administration of hemin alleviates various pathologies in murine models of cardiovascular disease, NSAID-induced intestinal injury, graft transplantation, and others. For example, in a rat model of ischemia reperfusion injury, hemin preconditioned hearts had improved postischemic cardiac function compared with control rats. 175 Jadhav et al 176 recently showed that the anti-inflammatory and antioxidative influence of hemin-ameliorated cardiac function restored normoglycemia and reduced blood pressure in a rat model of diabetic cardiomypoathy. Furthermore, they also showed that hemin administration decreased macrophage infiltration and enhanced the anti-inflammatory type 2 macrophage phenotype, while decreasing levels of inflammatory M1 macrophages. 176
This finding is in agreement with a previous study describing the role of HO-1 in macrophage polarization 177 and the study of Yoriki et al, which showed that upregulation of HO-1 in macrophages induces M2 characteristics. 178 These findings shed more light on the many mechanisms through which hemin exerts its beneficial effects and demonstrates new ways to polarize macrophages toward a cytoprotective, reparative phenotype. Hemin-based macrophage polarization may be of particular interest due to its translational possibilities, since it is already an administered drug against porphyria. 179
Summary and Future Directions
Macrophages play a critical role in the pathobiology of MI and are attractive therapeutic targets. Macrophage function can be stimulated or inhibited to improve healing, repair, and regeneration. Potential therapeutic approaches include reducing proinflammatory macrophage infiltration or the induction of a reparative and antifibrotic phenotype to guide repair and regeneration. The principles and strategies described here could be applicable to other cardiovascular diseases associated with macrophages, such as atherosclerosis, myocarditis, and pulmonary hypertension.
As this review highlights the actions of macrophages are complex and can be either beneficial or deleterious. Further research needs to be carried out to increase our understanding of the processes that control human macrophage activation during the various stages of acute and chronic myocardial disease. Better understanding of macrophage regulatory action could lead to the development of specific macrophage-based theranostic, which could improve infarct repair and recovery of patients after MI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grants from the Israel Ministry of Science, Culture, and Sport (JL and RM) and the Israeli National Nanotechnology Initiative and Helmsley Charitable Trust for a focal technology area on Nanomedicines for Personalized Theranostics (JL and RM).

