Abstract
In the adult heart, catalase (CAT) activity increases appropriately with increasing levels of hydrogen peroxide, conferring cardioprotection. This mechanism is absent in the newborn for unknown reasons. In the present study, we examined how the posttranslational modification of CAT contributes to its activation during hypoxia/ischemia and the role of c-Abl tyrosine kinase in this process. Hypoxia studies were carried out using primary cardiomyocytes from adult (>8 weeks) and newborn rats. Following hypoxia, the ratio of phosphorylated to total CAT and c-Abl in isolated newborn rat myocytes did not increase and were significantly lower (1.3- and 4.2-fold, respectively; P < .05) than their adult counterparts. Similarly, there was a significant association (P < .0005) between c-Abl and CAT in adult cells following hypoxia (30.9 ± 8.2 to 70.7 ± 13.1 au) that was absent in newborn myocytes. Although ubiquitination of CAT was higher in newborns compared to adults following hypoxia, inhibition of this did not improve CAT activity. When a c-Abl activator (5-(1,3-diaryl-1H-pyrazol-4-yl)hydantoin [DPH], 200 µmol/L) was administered prior to hypoxia, not only CAT activity was significantly increased (P < .05) but also phosphorylation levels were also significantly improved (P < .01) in these newborn myocytes. Additionally, ischemia–reperfusion (IR) studies were performed using newborn (4-5 days) rabbit hearts perfused in a Langendorff method. The DPH given as an intracardiac injection into the right ventricle of newborn rabbit resulted in a significant improvement (P < .002) in the recovery of developed pressure after IR, a key indicator of cardiac function (from 74.6% ± 6.6% to 118.7% ± 10.9%). In addition, CAT activity was increased 3.92-fold (P < .02) in the same DPH-treated hearts. Addition of DPH to adult rabbits in contrast had no significant effect (from 71.3% ± 10.7% to 59.4% ± 12.1%). Therefore, in the newborn, decreased phosphorylation of CAT by c-Abl potentially mediates IR-induced dysfunction, and activation of c-Abl may be a strategy to prevent ischemic injury associated with surgical procedures.
Introduction
In the pediatric population, congenital heart disease is the leading birth defect requiring corrective surgery for survival into adulthood. In order to repair these defects, newborns and infants are subject to corrective surgery involving cardiopulmonary bypass (CPB) that can lead to ischemia–reperfusion (IR) injury. Low-cardiac output syndrome (LCOS) is the most common complication encountered by postsurgical patients in the first 6 to 18 hours after CPB. 1 Left untreated, LCOS could lead to inadequate organ perfusion and untimely organ dysfunction. Aside from the evident myocardial damage, renal hypoperfusion, hypotension, and poor peripheral perfusion are other consequences of LCOS. 1 One potential mechanism for dysfunction due to IR injury is through formation of reactive oxygen species (ROS). During IR, there is a burst of ROS that leads to local cell death, inflammatory cell recruitment, and alterations in contractile protein regulation. It is therefore critical that steps are taken to limit further injury during CPB because, as we and others have previously reported, 2 the newborn heart is more susceptible to oxidative stress due to the developmental immaturity of newborn heart structures and lower antioxidant protection. Studies have shown decreased contractile reserve in newborn human heart that is consistent with immature calcium handling. 3 In addition, the immature sarcolemma contains high levels of polyunsaturated fatty acids that increase susceptibility to ROS injury. 4 Antioxidant levels also follow a developmental pattern, with low antioxidant levels being associated with younger age. Previous work has demonstrated that the major antioxidants (ie, catalase [CAT], superoxide dismutase, and glutathione) take 2 to 3 weeks following birth to reach maximum levels, progressively decrease to baseline levels thereafter, and then maintain plateau levels with further maturation. 5
Catalase is an essential scavenger of hydrogen peroxide (H2O2), and its regulation may be affected by various cofactors such as selenium and cysteine, cellular localization, and posttranslational modification. 6 –9 Mitochondrial localization of CAT has been demonstrated to play a significant role in dictating CAT activity; however, no studies have examined the differences in localization of CAT between adults and newborns. 10 In addition, posttranslational modification of CAT in the newborn may also be an important regulatory event of its scavenging activity. In the adult heart, increasing levels of H2O2 lead to the phosphorylation and activation of CAT by the tyrosine kinase c-Abl/Arg complex. However, when H2O2 levels become excessive, c-Abl/Arg dissociate, inactivating it and rendering it susceptible to ubiquitination by proteases. 8 We recently showed a significant increase in the production of H2O2 in the newborn right ventricle (RV; compared to adults) following IR without the corresponding increase in CAT activity required to scavenge H2O2. As oxidative injury occurs in both newborn and adult hearts, it is necessary in either case to determine mechanisms of regulation to limit myocardial injury. 2 This is vital during corrective surgery in newborns with congenital heart defects, as this is when their hearts are exposed to reactive oxygen intermediates that will lead to detrimental long-lasting sequelae. 11,12
In the present study, in order to elucidate the mechanism of CAT regulation in the newborn heart, we examined how phosphorylation of CAT contributes to its activation. Specifically, we demonstrate the role played by c-Abl tyrosine kinase in phosphorylating and activating CAT in the newborn heart, which may potentially provide the cardioprotection needed to limit injury during CPB.
Materials and Methods
Experimental Animals and Anesthesia
An established breeding colony for rabbits (Wagner Lab Facility, Emory University) was the source of newborn (2-5-day old) and adult (>8 weeks) rabbits for whole heart perfusion studies, while Sprague-Dawley rats (Charles River) were used for isolated cell experiments. Newborn rabbits and adult rats were heparinized 10 minutes before anesthetic induction. Xylazine (5-10 mg/kg) and ketamine (75-100 mg/kg) were administered subcutaneously for newborn and intravenously (5-10 mg/kg xylazine and 20-40 mg/kg ketamine) for adult rabbits. Adult rats were also given xylazine (10 mg/kg) and ketamine (75-100 mg/kg), but administration was via intraperitoneal injection. Anesthetic protocols adhered to the Guide for the Care and Use of Laboratory Animals. All experimental protocols were approved by Emory University Institutional Animal Care and Use Committee.
Langendorff Perfusion Method
In rabbits, following anesthesia, hearts were removed, the aorta cannulated, and perfused using the Langendorff method as described previously. 2 Right ventricular pressure was measured using a fluid-filled balloon and pressure transducer. Percentage of recovery of developed pressure (%RDP) was determined by calculating the ratio of developed pressure (difference between systolic and diastolic pressures) during the ischemia and reperfusion periods. For functional studies, Langendorff-perfused newborn rabbit hearts received 0.4 mL, while adult rabbits were given 2 mL, of indicated intervention via right ventricular (intramuscular) injection 2 minutes before the reperfusion period.
Cardiomyocyte Isolation and Hypoxia Treatment
Cardiomyocytes were isolated from newborn rat pups as reported previously and cultured in Dulbecco modified Eagle medium (Invitrogen) with 10% fetal bovine serum (Hyclone, Logan, Utah) and 1× penicillin–streptomycin–glutamine (P/S/G; Invitrogen, Grand Island, New York) in 6-well plates until reaching 80% confluency. 13 Flow cytometry was performed for α-sarcomeric actinin (Sigma, St. Louis, Missouri) to ensure purity. For isolation of adult rat cardiomyocyte, hearts were removed and perfused using Langendorff method with oxygenated calcium-free Tyrode solution for 5 minutes at 37°C followed by the same solution containing 0.25 mg/mL collagenase (Yakult Honshu Inc, Tokyo) and 0.05 mg/mL protease (type XIV; Sigma). After 10 to 15 minutes, perfusion with Kraft-Bruhe (KB) solution followed. Ventricular tissue was minced in KB solution, and the suspension was plated onto fibronectin-coated plates for subsequent hypoxia treatment. Isolated cardiomyocytes were incubated in a hypoxia chamber (5% CO2/95% N2) for 1 hour at 37°C. For drug treatment, the cells were incubated for 1 hour prior to hypoxia treatment with the indicated intervention.
Western Blot and Immunoprecipitation Studies
Proteins were extracted from cells by scraping with lysis buffer containing protease and phosphatase inhibitor cocktails (Sigma, St. Louis). Protein content was determined by microbicinchoninic acid assay (Fisher, Pittsburgh, Pennsylvania). Protein of 50 µg was loaded onto each lane and subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis followed by transfer to a polyvinylidene difluoride membrane. After blocking in 5% milk, the membrane was incubated overnight with the following primary antibodies: phosphotyrosine (Abcam, Cambridge, Massachusetts), c-Abl (Abcam), or CAT (Novus Biologicals, Littleton, Colorado), followed by the appropriate secondary antibody. For phosphorylated CAT (pCAT) and c-Abl measurements, immunoprecipitation against CAT and c-Abl was followed by immunoblotting with antiphosphotyrosine antibody. Membranes were then incubated with ECL prime-developing solution (Amersham, Pittsburgh, Pennsylvania), and bands were visualized using a Kodak camera system. Bands were quantified using ImageJ software (National Institute of Health, Bethesda, Maryland) and reported as arbitrary units (au).
Catalase Activity Assay
Levels of CAT in newborn and adult cardiomyocytes and whole heart homogenates were measured using decomposition of H2O2 at 240 nm utilizing a plate reader in the presence and absence of 3-amino-1,2,4-triazole (3-AT, a CAT inhibitor) as described previously. 2
Statistics
All statistics were performed using GraphPad Prism 5.0 software with the indicated test (unpaired t-test for 2 groups, analysis of variance for 3 or more groups). A P value of <.05 was considered significant.
Results
Baseline and Hypoxia-Regulated Levels of CAT
Western blot analysis of adult and newborn rat cardiomyocytes demonstrated a significant difference in total baseline CAT levels (98.6 ± 25.3 and 44.1 ± 1.1 au, respectively; P < .05; Figure 1A). To determine whether there was a difference in CAT activity between adult and newborn cardiomyocytes exposed to hypoxia, we measured CAT activity in the presence and absence of 3-AT, following 1-hour hypoxia treatment (Figure 1B). In the adult cells, CAT activity increased significantly from 15.2 ± 6.4 to 69.8 ± 20.1 U/mg protein (P < .01). In the newborn, there was no significant change in CAT activity (from 24.3 ± 7.7 to 6.1 ± 1.7 U/mg protein) demonstrating that the newborn, unlike the adult, is unable to increase CAT activity following hypoxia. To determine whether this difference in activity was regulated by phosphorylation of CAT, we measured the ratio of pCAT to total CAT (tot CAT) from newborn and adult rat cardiomyocytes subjected to 1 hour of hypoxia. We found in the adult, pCAT/tot CAT was increased almost 3-fold from 55.8 ± 12.3 to 146.5 ± 19.1 au (P < .005). No significant change was observed in the newborn (90.2 ± 7.8 to 112.5 ± 11.7 au), demonstrating that after hypoxia, levels of pCAT/tot CAT are unchanged (Figure 1C).

Age-related differences in catalase phosphorylation and activity in response to hypoxia. A, Western blot analysis demonstrating reduced baseline catalase (CAT) levels in newborn as compared to adult rat cardiomyocytes. Values are mean ± SEM; n = 4 hearts/group; *P < .05. B, Adult CAT activity was significantly increased following 1 hour of hypoxia, while there was no significant change in catalase activity in newborn myocytes. Values are mean ± SEM; n = 5 hearts/group; *P ≤ .01; ANOVA followed by Bonferroni posttest. C, Immunoprecipitation studies showed that while the ratio of phosphorylated to total CAT after hypoxia was significantly increased by hypoxia in adult cardiomyocytes, there was no significant change in newborn cardiomyocytes. Values are mean ± SEM; n = 4 to 6 hearts/group; *P < .005 ANOVA followed by Bonferroni posttest. Ad indicates adult; ANOVA, analysis of variance; Hx, hypoxia; NB, newborn; Nx, normoxia; SEM, standard error of the mean.
Basal and Phosphorylated Levels of c-Abl
Because c-Abl is known to modulate CAT, we examined basal protein levels of c-Abl in newborn and adult rat heart. Figure 2A shows that there was a significant decrease in c-Abl levels in newborn compared to adult (353.0 ± 179.1 vs 992.8 ± 159.4 au, P < .05). To determine whether hypoxia regulated phosphorylation of c-Abl, adult and newborn cardiomyocytes were exposed to hypoxia for 1 hour, and levels of phosphorylated c-Abl were measured. Similar to CAT, Western blotting following immunoprecipitation demonstrated an increase in the ratio of phosphorylated to total c-Abl in the adult (44.9 ± 13.9 to 116.9 ± 19.3 au; P ≤ .0005). This increase was not evident in newborn cardiomyocytes (43.9 ± 9.9 to 27.8 ± 4.1 au; Figure 2B). As interactions between c-Abl and CAT are known to regulate phosphorylation, we examined whether hypoxia regulated binding of these proteins. Coimmunoprecipitation and Western blot studies demonstrated an increased association between c-Abl and CAT in adult cardiomyocytes (30.9 ± 8.2 to 70.7 ± 13.1 au; P < .05), which was not present in newborn cardiomyocytes (50.7 ± 14.0 to 30.0 ± 3.1 au) after exposure to 1-hour hypoxia treatment (Figure 3).

Age-related differences in c-Abl phosphorylation following hypoxia. A, Western blot analysis demonstrating basal c-Abl levels was significantly lower in newborn cardiomyocytes compared to adult cells. Values are mean ± SEM; n = 4 to 5 hearts/group; *P < .05. B, Western studies demonstrating a significant increase in c-Abl phosphorylation in adult cardiomyocytes in response to hypoxia, with no increase in newborn cardiomyocytes. Values are mean ± SEM; n = 4 to 6 hearts/group; *P ≤ .0005 analysis of variance (ANOVA) followed by Bonferroni posttest. Ad indicates adult; Hx, hypoxia; NB, newborn; Nx, normoxia; SEM, standard error of the mean.
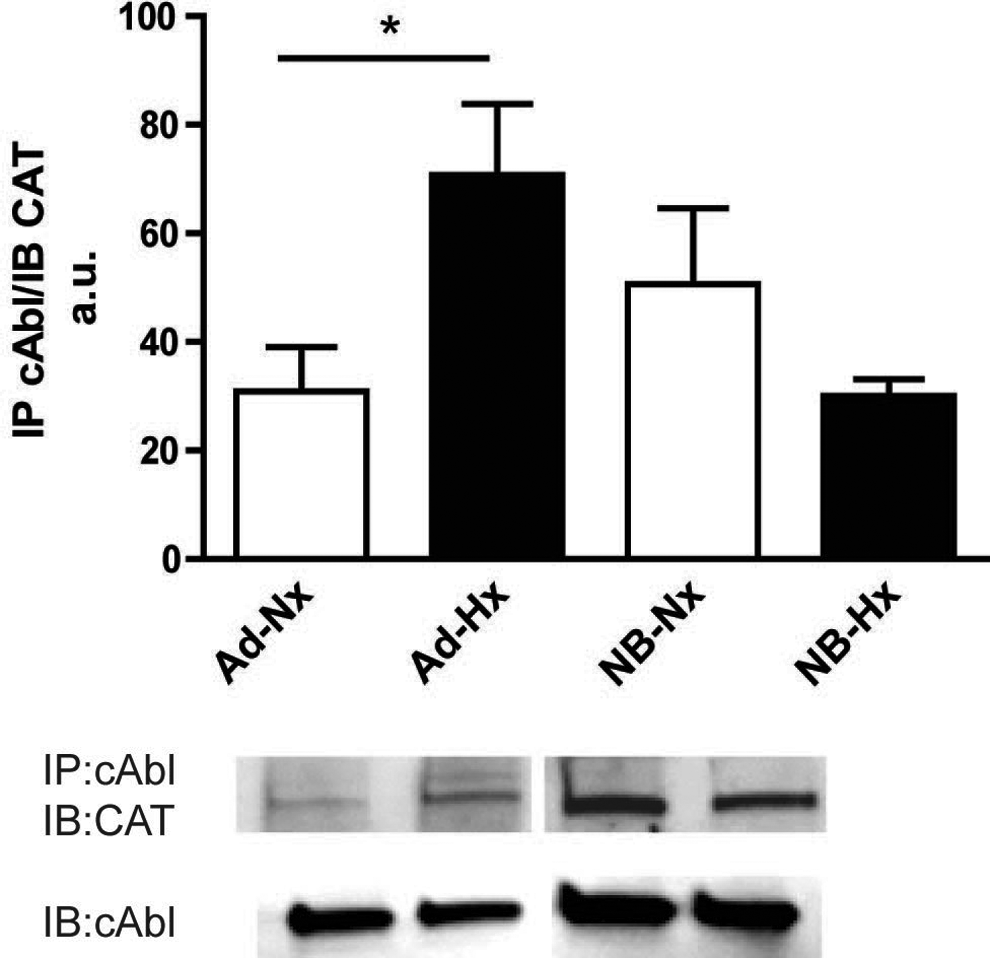
Association of c-Abl and catalase (CAT) in response to hypoxia. Immunoprecipitation against c-Abl followed by Western blot studies with c-Abl and CAT showed increased association between c-Abl and CAT after hypoxia treatment in adult cardiomyocytes. There was no increase in association between c-Abl and CAT after hypoxia in newborn. Values are mean ± standard error of the mean (SEM). n = 5 to 7 hearts/group; *P < .05; analysis of variance (ANOVA) followed by Bonferroni posttest. Ad indicates adult; Hx, hypoxia; NB, newborn; Nx, normoxia.
Activation of c-Abl in Newborn Cardiomyocytes Regulates CAT
To determine whether activation of c-Abl in newborn rat cardiomyocytes could enhance CAT activity and phosporylation, cells were incubated for 1 hour with 5-(1,3-diaryl-1H-pyrazol-4-yl) hydantoin (DPH, 200 µmol/L), a small molecule c-Abl activator, prior to hypoxia treatment. Treatment with DPH significantly increased the ratio of pCAT to tot CAT by 2.22-fold (from 146.1 ± 25.4 to 325.3 ± 57.2 au; P < .01) in the cardiomyocytes exposed to hypoxia alone versus those given DPH and hypoxia treatment, respectively (Figure 4A).
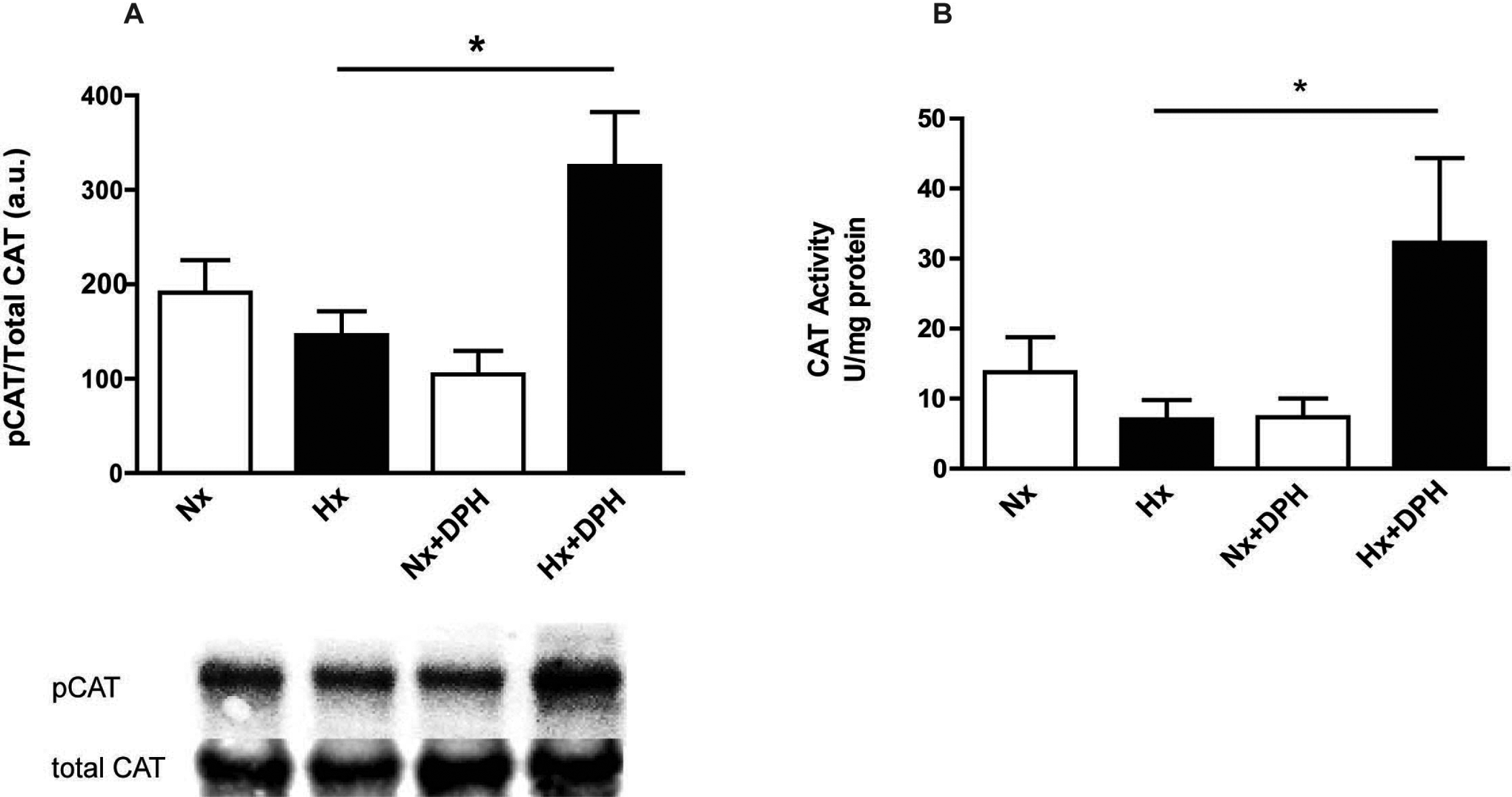
Activation of c-Abl increases catalase activity in hypoxic newborn cardiomyocytes. A, Ratio of phosphorylated to total catalase (CAT) was increased in newborn cardiomyocytes following 1 hour of hypoxia treatment in cardiomyocytes pretreated with DPH, a small molecule c-Abl activator. Values are mean ± SEM; n = 4 to 5 hearts/group; *P < .01 ANOVA followed by Bonferroni posttest. B, Activity levels of CAT were significantly increased following 1 hour of hypoxia treatment in newborn cardiomyocytes pretreated with DPH. Values are mean ± SEM; n = 4 to 6 hearts/group; *P < .05 ANOVA followed by Bonferroni posttest. ANOVA indicates analysis of variance; DPH, 5-(1,3-diaryl-1H-pyrazol-4-yl)hydantoin; Hx, hypoxia; Nx, normoxia; SEM, standard error of the mean.
To determine whether this activation of c-Abl led to an increase in CAT activity, we treated newborn myocytes with either 10 or 200 µmol/L DPH followed by hypoxia exposure for 1 hour. A small but not significant increase in CAT activity was seen with the lower dose of DPH (data not shown). Increasing DPH further resulted in a significant increase in hypoxia-induced CAT activity (6.1 ± 1.4 to 32.3 ± 12.1 U/mg protein, P < .05; hypoxia without DPH vs hypoxia with 200 μmol/L DPH; Figure 4B).
Hypoxia Alters Ubiquitination of CAT, But Inhibition of This Does Not Improve Activity
To determine the difference in CAT ubiquitination levels in adult versus newborn cardiomyocytes, we treated adult and newborn myocytes with hypoxia and immunoblotted for ubiquitinated CAT. Western blot studies demonstrated a trend toward decreased levels of ubiquitinated CAT following hypoxia in adult myocytes (139.8 ± 25.50 to 53.92 ± 10.37 au; P = not significant [ns]), while newborn myocytes showed an opposite trend (224.6 ± 62.6 to 684.3 ± 351.7 au; P = ns; Figure 5A). Furthermore, when newborn myocytes were incubated with either 200 or 400 µmol/L MG132 (an ubiquitin inhibitor), CAT activity did not improve and was even decreased significantly from normoxic levels following the administration of the drug at the 400 µmol/L dose (28.6 ± 6.2 to 0.8 ± 1.1 au; P < .05; Figure 5B).

Ubiquitination of catalase in adult and newborn cardiomyocytes and effect on catalase activity. A, In adult cells, ubiquitination of catalase tended to decrease following hypoxia treatment. In the newborn, an increased trend in ubiquitination of catalase was observed. Values are mean ± SEM; n = 3 to 5 hearts/group. B, Newborn cardiomyocytes preincubated with MG132 (ubiquitin inhibitor) prior to hypoxia treatment, displayed no significant improvement in catalase activity compared with cardiomyocytes treated with hypoxia alone. Values are mean ± SEM; n = 3 hearts/group. Ad indicates adult; Hx, hypoxia; M200, MG132 (200 µmol/L); M400, MG132 (400 µmol/L); Nx, normoxia; NB, newborn; SEM, standard error of the mean
Activation of c-Abl Improves Cardiac Function in Newborn But Not Adult Rabbit Hearts
Our prior studies show that IR-induced damage to the newborn RV is regulated by H2O2. To determine whether RV function was improved by administration of a c-Abl activator, 200 µmol/L DPH (in 0.4 mL) was given as an intracardiac injection into the RV of a newborn rabbit heart immediately before reperfusion in a Langendorff (isolated heart preparation) model. Recovery of developed pressure was just 74.6% ± 6.6% following IR, indicating impaired function (100% indicates the baseline). Although there was no significant change in vehicle treatment, DPH administration resulted in a significant increase in RDP to 118.7% ± 10.9% (P < .002; Figure 6A), higher than original contractility. Furthermore, measurement of CAT activity in these hearts demonstrated a 3.9-fold increase from 19.1 ± 8.9 to 75.3 ± 12.0 U/mg tissue (P < .02; Figure 6B) with no effect of vehicle treatment. In contrast, when DPH was injected into the RV of adult rabbits, there was no significant change in RDP (71.3% ± 10.7% to 59.4% ± 12.1%). These data demonstrate that activation of c-Abl enhances CAT activity and RV function following IR injury in the newborn but not in adult hearts.

Activation of c-Abl enhances newborn not adult right ventricular function. A, Ischemia–reperfusion (IR) injury resulted in reduced right ventricular function in newborn rabbit heart as measured by recovery of developed pressure (RDP) where 100% signifies a return to baseline contractility. Although treatment with vehicle (DMSO) had no effect, administration of DPH improved function above that of baseline levels. Values are mean ± SEM; n = 5 to 6 animals/group; *P < .002 ANOVA followed by Bonferroni posttest. B, A significant increase in catalase (CAT) activity was observed following intracardiac DPH injection into the newborn right ventricle with no effect of vehicle. Values are mean ± SEM; n = 5 animals/group; *P < .02 ANOVA followed by Bonferroni posttest. C, No change in RDP was observed in the adult right ventricle (RV) after IR following administration of DPH. Values are mean ± SEM; n = 5 to 6 animals/group. ANOVA indicates analysis of variance; DPH, 5-(1,3-diaryl-1H-pyrazol-4-yl)hydantoin; SEM, standard error of the mean.
Discussion
The newborn heart’s susceptibility to oxidative stress has been demonstrated by our previously published work as well as by others. 2,14 Lower baseline levels of antioxidants (ie, CAT) or reduced activity could account for this inability to defend against oxidants such as H2O2 and superoxide. Many proteins are developmentally regulated and based on prior reports antioxidants are in this category. 5 Although it has become well established that oxidative stress is regulated differently in newborns versus adults, there are few studies examining mechanistic differences in this regulatory pathway. In this article, we sought to examine the regulation of CAT based on our prior studies, demonstrating that the newborn RV is more sensitive to changes in H2O2 without corresponding upregulation of CAT.
During conditions of oxidative stress, CAT undergoes phosphorylation at tyrosine-231 and tyrosine-386 by c-Abl and Arg, 2 tyrosine kinases involved in regulating responses to oxidative stress as well as DNA damage and cytoskeletal reorganization. 15 Phosphorylation of CAT confers activation and allows it to function normally as a scavenger of H2O2. However, when the levels of H2O2 become excessive, c-Abl and Arg dissociate from CAT, exposing it to ubiquitination and the activity of phosphatases, thus rendering it inactive. 16 Our findings agree with this observation. We show that CAT is dysfunctional in the newborn heart and that c-Abl is a regulator of CAT activity. During exposure to hypoxia, the ratio of pCAT to total CAT as well as CAT activity in isolated newborn rat cardiomyocytes was unchanged from normoxic levels. In contrast, in the adult heart, phosphorylation and activity of CAT increased significantly following hypoxia signifying an adequate antioxidant response. These data agree with studies done by Cao et al, which demonstrated that phosphorylation of CAT indeed occurs following exposure to oxidative stress. 17,18 Specifically, Michigan Cancer Foundation 7 cells from mouse embryo fibroblasts were treated with 0.25, 1.0, and 2.0 mmol/L H2O2, and pCAT levels were measured via Western blot. Their results demonstrated that phosphorylation was increased starting at 1.0 mmol/L H2O2. In addition, ST1571, a c-Abl/Arg kinase inhibitor, reversed the increase in CAT phosphorylation previously observed, thus supporting an essential association of c-Abl/Arg in the phosphorylation and activation of CAT. Furthermore, in our present study, we show reduced basal levels of CAT and c-Abl in the newborn versus the adult cardiomyocytes. This further demonstrates how diminished tot CAT and pCAT levels are possibly tied to c-Abl/Arg levels and needs to be further investigated.
As ubiquitination of CAT is another contributing factor of diminished CAT activity, we also examined levels of ubiquitinated CAT following hypoxia in isolated newborn rat cardiomyocytes and compared these to adult levels. A decreased trend in ubiquitination of CAT was observed in the adult but not in the newborn heart (Figure 5A). Based on the above published study, 16 this would seem to be an appropriate response to hypoxia, which was absent in the newborn. 18 Despite these changes in ubiquitination, treatment with an ubiquitin inhibitor (MG132) did not significantly improve CAT activity in newborn cardiomyocytes (Figure 5B). Previous studies on inhibition of the ubiquitin-proteasome pathway have demonstrated a cardioprotective role of MG132 via the induction of heat shock proteins. 18 Based on our current data, it would appear that ubiquitination of CAT may be secondary to inactivation by other mechanisms such as phosphatase activity. Phosphatases have been found to be more abundant in the newborn (compared to the adult) heart, specifically protein phosphatase (PP) 1 and 2A, suggesting that the newborn heart is more dependent on phosphatase inhibitors for the regulation of certain proteins. 19 Therefore, it is possible that the increased levels of phosphatases may be involved in the dephosphorylation and inactivation of CAT observed in the newborn heart, and inhibition of these phosphatases may in part restore the functional activity of CAT. It has been reported that calcium influx leads to the inhibition of phosphatase 1B in pancreatic acinar cells. 20 Since calcium handling in the newborn heart is also dysregulated, it may be possible that phosphatase inhibition does not occur, leading to aberrant phosphatase activity in the newborn heart. It is also possible that the well-balanced combination of phosphatase inhibition with kinase activation is instrumental in regulating CAT activity. In human erythrocytes, incubation with 0.4 or 2 mU of PP2A resulted in a decrease in CAT activity to 18% of control levels. Protein kinase A and C on the other hand resulted in an increase in CAT activity to greater than 150% of control levels. 21 Thus, the precise regulation of phosphatases and kinases may be critical for regulating CAT activity in the newborn heart.
In addition to posttranslational modification, CAT localization and cofactors of CAT, namely, selenium and cysteine, may also be instrumental in regulating CAT activity. Previous studies by Chiba et al have demonstrated that serum selenium levels, serum glutathione peroxidase activity, and myocardial glutathione peroxidase activity in infant and selenium-deficient adult rats were significantly lower than that of adult rats fed with a normal diet. 14 Thus, differences in the levels of these cofactors may also contribute to the diminished CAT activity observed in newborn hearts. Although there are no specific studies outlining the differences in localization of CAT between adult and newborns, various studies have demonstrated a significant role of mitochondrial CAT in H2O2 scavenging. One such study described the binding of cyanide to ferric hemeproteins (cytochrome c oxidase and CAT) located in the mitochondria inhibiting the binding of H2O2 to these hemeproteins and thus indicating mitochondrial localization. 10 Another study showed significant inhibition of peroxidase activity by aminotriazole in mitochondrial samples, suggesting mitochondrial CAT activity. 22 Thus, CAT localized to the mitochondria may play a significant role in regulating CAT activity following oxidative stress, and developmental changes in this are an interesting area for future studies.
As the phosphorylation state of CAT plays a significant role in regulating its scavenging activity, we incubated our newborn cardiomyocytes with DPH, a c-Abl activator in an attempt to restore CAT phosphorylation. DPH is described as the first cell-permeable molecule that binds to the myristoyl binding site of the kinase domain of c-Abl causing it to be phosphorylated and activated. 23 In the presence of DPH, there was a 2.2- and 5.3-fold increase in the levels of phosphorylation and activity of CAT, respectively, in response to hypoxia validating the role c-Abl plays in regulating CAT activity. The addition of DPH did not increase levels of phosphorylation or activity of CAT in normoxic conditions, indicating that perhaps this is not the only step in the pathway and other signals from stressors may be needed. In fact, prior studies show that c-Arg may form a complex with c-Abl to activate CAT, 17 ; thus, this step may not be active under basal conditions but stimulated by hypoxia. Other studies have demonstrated that c-Abl undergoes continuous shuttling within the compartments of the cell but resides predominantly in the cytoplasm where it controls cell survival and proliferation. When DNA damage occurs, c-Abl translocates to the nucleus where it initiates an apoptotic pathway. 24 In the newborn heart, it is unclear how subcellular localization of c-Abl affects the regulation of CAT but remains an intriguing concept nonetheless. In addition, 1 study describes how activated (phosphorylated at tyrosine-245 and tyrosine-412) wild-type c-Abl is more unstable than the dephosphorylated form. However, when c-Abl was structurally altered, phosphorylation at these residues did not play a major role in stabilizing c-Abl. 16 Although we did not examine the specific residues on c-Abl that regulate association with CAT, it remains an area for future mechanistic studies.
The utility of the small molecule c-Abl activator in affording cardioprotection to the newborn heart via its positive regulation of CAT is further evidenced by the improvement in right ventricular function observed in our model of IR injury using a Langendorff perfusion method. In the adult heart, however, this phenomenon was not replicated. There was no significant change in ventricular function following administration of DPH, suggesting further activation of c-Abl in the adult heart confers no additional protection beyond its inherent protective mechanism. Our previous study also demonstrated a similar effect with the administration of encapsulated ebselen to the adult heart. 2 No further improvement in RDP was afforded by ebselen in addition to the adult heart’s initial response to the ischemic insult. The IR injury results in poor right ventricular function in newborns as measured by RDP, and we found a significant increase in function when DPH was administered via intracardiac injection directly into the RV of the newborn heart 2 minutes before reperfusion. We have found that direct intramuscular injection is a more effective method of drug delivery as administering the drug in the perfusate in our prior studies had no significant effect on RDP likely because it wasn’t targeted directly to the area of damage or was diluted too greatly. In our present studies, not only was the RDP improved in the newborn heart but also the force and quality of contraction exceeded baseline levels, indicating the potential for other beneficial effects afforded by DPH following IR injury. It is possible that the mechanism of action of DPH involves regulation of contractile proteins, as Arg/Abl binding proteins have been shown to associate with stress fibers and Z-discs of cardiomyocytes and influence contractile properties of these cells. 25
It is well known that c-Abl is an oncogene, and kinase activation has been implicated in the development of various cancers. Therefore, although research has been underway in understanding the mechanisms involved in kinase inhibition, little progress has been made in the area involving kinase activation. Recently, reports have emerged regarding the role of c-Abl activation in the prevention of breast cancer aggressiveness 26,27 as well as in the treatment of neutropenia. 28,29 Thus, future studies on the effects of c-Abl activation in newborn cardiac disease should focus not just on the cardioprotection but also on the secondary effects as well. To further reduce the possibility of off-target effects, our laboratory has developed targeted nanoparticles that have been used to deliver site-specific therapeutics to the myocardium, and this may be a potential area for further investigation. 30
Conclusion
We have determined that the newborn heart is unable to increase CAT activity in spite of increasing levels of H2O2 during IR due to the dysregulation of CAT. The tyrosine kinase c-Abl, which is responsible for the phosphorylation of CAT, is in the dephosphorylated state and is unable to activate CAT and allow it to function normally as a scavenger of H2O2. Figure 7 depicts a schematic summarizing these studies. In the adult, increasing H2O2 levels during IR triggers the phosphorylation of c-Abl and subsequent association with and phosphorylation of CAT, leading to increased activity. This may explain our prior studies showing increasing levels of H2O2 due to decreased CAT activity only in newborn cardiomyocytes. 2 The administration of DPH, an activator of c-Abl, into the RV of the Langendorff-perfused newborn heart increased the activity of CAT, leading to an increase in functional recovery of ventricular pressure suggesting a vital role for c-Abl in the regulation of CAT and a potential therapeutic intervention in protecting the newborn heart against oxidative injury.

Proposed mechanistic differences in the regulation of catalase between newborn and adult cardiomyocytes. In the adult, increasing hydrogen peroxide (H2O2) levels during ischemia–reperfusion (IR) triggers the phosphorylation of c-Abl and subsequent association with and phosphorylation of catalase, leading to increased activity. Conversely, the newborn phosphorylation of both catalase and c-Abl is impaired, leading to a failure to increase activity. Ubiquitination of catalase may also have age-related differences, although inhibiting this process had no effect on activity. Administration of 5-(1,3-diaryl-1H-pyrazol-4-yl)hydantoin (DPH), a c-Abl activator, to the newborn heart results in phosphorylation and activity of catalase similar to that found in adult cardiomyocytes, leading to improvements in function.
Footnotes
Author’s Contribution
E. Cabigas: Conception and design of the study, Substantial contribution to data acquisition, Analysis and interpretation of the data, Drafting of the manuscript, Critical revision of the manuscript for intellectual content; J. Liu: Substantial contribution to data acquisition; A. Boopathy: Substantial contribution to data acquisition, Analysis and interpretation of the data, Critical revision of the manuscript for intellectual content; P. Che: Substantial contribution to data acquisition; B. Crawford: Substantial contribution to data acquisition; G. Baroi: Substantial contribution to data acquisition, Analysis and interpretation of the data; S. Bhutani: Substantial contribution to data acquisition, Critical revision of the manuscript for intellectual content; M. Shen: Substantial contribution to data acquisition, Analysis and interpretation of the data; M. Wagner: Conception and design of the study, Analysis and interpretation of the data, Drafting of the manuscript; M. Davis: Conception and design of the study, Analysis and interpretation of the data, Drafting of the manuscript, Critical revision of the manuscript for intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by an Emory-Children’s Center for Cardiovascular Biology Pilot Grant from Children’s Healthcare of Atlanta, as well as grant HL094527 from the National Heart, Lung, and Blood Institute to MED.
