Abstract
In the present study, we for the first time explored the protective effect of LOB (Chrysoeriol 7-O-[β-D-glucuronopyranosyl-(1→2)-O-β-D-glucuronopyranoside]) on myocardial ischemia–reperfusion (I/R) injury in a rat model and on hypoxia–reoxygenation (H/R) injury in a rat myocardium cell line. An I/R rat model and an H/R rat myocardium cell model were established. The animal and H9C2 (2-1) rat myocardium cells were pretreated with increasing doses of LOB before the I/R or H/R injury. Malondialdehyde (MDA), lactate dehydrogenase (LDH), interleukin 6 (IL-6), and the caspase 3 activity were measured. Pretreatment with LOB dose dependently reduced the elevated plasma levels of LDH and IL-6 and myocardium MDA level induced by the I/R injury in the rat model, and the elevated culture medium LDH, cell MDA levels, and caspase 3 activity induced by the H/R injury in the cell model were also reduced. Both in vivo and in vitro data showed that high-dose LOB (20 mg/kg or 10 μmol/L) had stronger protective effects than the positive control drug verapamil. In conclusion, our study for the first time provided both in vivo and in vitro evidence that LOB exerted significant cardioprotective effects on myocardial I/R injury in rats, suggesting that LOB could be a potential therapeutic agent for myocardial I/R injury.
Introduction
Each year in the United States, there are approximately 1 million myocardial infarctions (MIs) and 700 000 patients undergoing cardioplegic arrest for various cardiac surgeries. 1 Once the coronary flow is restored, however, the myocardium is susceptible to another form of insult stemming from reperfusion of the previously ischemic tissue. It has been reported that the histological changes seen following only 30 to 60 minutes of ischemia/reperfusion (I/R) were comparable to the degree of necrosis normally seen after 24 hours of permanent coronary occlusion. 2 Generation of free radicals through incomplete reduction in oxygen during I/R has been well described. 3 These oxygen species are highly reactive and can quickly overwhelm the cell’s endogenous free radical scavenging system. This, in turn, triggers cellular injury by reactions with lipids, proteins, and nucleic acids. 3 This kind of reactive oxygen species (ROS)-induced cardiac cell injury plays an important role in the pathogenesis of myocardial ischemia injury and myocardial infarction. 4 Accordingly, successful antioxidant interventions targeted to cleave ROS, which to date has attracted intensive interests from investigators, offer insights into treating or preventing I/R. 5,6
Chrysoeriol 7-O-[β-D-glucuronopyranosyl-(1→2)-O-β-D-glucuronopyranoside], called LOB in the present study, was first identified in the aerial parts of barrel medic (Medicago truncatula), with its structure established by spectrometric and spectroscopic electrospray ionization mass spectrometry/Mass spectrometry (ESI-MS/MS) and nuclear magnetic resonance (NMR) techniques. 7 LOB is a flavone (Supplementary Figure S1, available in the online version of this article at http://cpt.sagepub.com/supplemental/), that is of particular importance in the human and animal diet as there is evidence that they act as antioxidants. 8,9 Epidemiological studies have indicated that its consumption is associated with a reduced risk of cancer 8 and heart disease. 9 In the present study, we for the first time explored the protective effect of LOB on myocardial I/R injury in a rat model and on hypoxia–reoxygenation (H/R) injury in a rat myocardium cell line.
Methods and Materials
Cell Line, Reagents, and Mice
The H9C2 (2-1) rat myocardium cell line was purchased from the American Tissue Culture Collection (ATCC, Manassas, Virginia). Male Sprague Dawley (SD) rats, aged 8 to 9 weeks and weighing 290 to 320 g, were purchased from Central South University (Changsha, China) and were housed at the Xiangya Hospital BioResources Centre. The animals were placed in a quiet, temperature (22°C ± 2°C)- and humidity (60% ± 6%)-controlled room with a 12-h:12-h light–dark cycle (light beginning at 8
Establishment of I/R Rat Model
The acute myocardial I/R model was established as previously described. 10 After an intraperitoneal injection of sodium pentobarbital (70 mg/kg, supplemented if necessary), the animal was tracheally intubated and mechanically ventilated with oxygen-enriched room air. The ventilation rate was 60 to 80 breaths/min; tidal volume, 2 to 3 mL/100 g body weight; and the inspiratory/expiratory ratio 1:1. By adjusting the ventilation rate and tidal volume, arterial blood gases were maintained at normal levels (pH, 7.35-7.45; PaCO2, 25-40 mm Hg; PaO2, 80-110 mm Hg). Throughout the experiment, the rat's body temperature was maintained at 36.5°C to 37.5°C by a heating pad. The right internal jugular vein was cannulated for fluid transfusion and blood sampling to assay the plasma levels of LDH and IL-6. The chest was opened by a left thoracotomy through the fourth intercostal space. After pericardiotomy, a 5-0 silk ligature was placed under the left anterior descending coronary artery (LAD), where it emerges from the left atrium, and the ends of the tie were threaded through a small plastic tube to form a snare for reversible LAD occlusion. After an equilibration period of 10 minutes, the ligature was tied to block the blood flow of LAD, producing local myocardial ischemia. Successful occlusion of the LAD was confirmed by the presence of ST segment elevation on electrocardiogram (ECG). Adequate reperfusion was verified by epicardial hyperemia and reversion of ECG changes, elevated ST segment descending more than 50% of the elevation level. 11 This study was conducted in accordance with our institutional guidelines on the use of live animals for research, and the experimental protocol was approved by the Laboratory Animal Users Committee at Xiangya Hospital, Central South University, Changsha, China.
Evaluation of Infarct Size
After a reperfusion period of 180 minutes, the LAD was reoccluded, and 1 mL of 2% Evans blue dye was injected into the right carotid artery to stain the normally perfused region of the heart and delineate the infarct area (white area). After the body was stained blue, the rat was killed by injection of 10% potassium chloride. The entire heart was excised and rinsed of excess blue dye, and the right ventricle and right and left atria were trimmed off. The remaining left ventricle was deep frozen at -20°C in a icebox. The frozen left ventricle was then cut into approximately 5 sections from apex to base; then, all tissues were incubated in a 1% solution of 2, 3, 5-triphenyl tetrazolium chloride for 15 minutes at 37°C. The infarcted tissue stains a characteristic white color, whereas the viable tissue stains red. After overnight fixation in 10% formaldehyde, the slices were digitally photographed. A blinded investigator measured the AAR and infarct size in the sections using Adobe Photoshop CS (Adobe Systems Inc, San Jose, California). The myocardial infarct area was expressed as percentage by the following formula: myocardial infarct area (%) = infarct area (white area)/whole sample area × 100%. 10,12
Experimental Groups of Animals
Sprague Dawley rats were randomly assigned to 7 groups (n = 8 per group): control group (mice without sugery, occlusion, and drug treatment), sham group (sham surgery-treated mice), I/R group (sublingual vein injection of 1 mL of LOB solvent 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours), LOB 5 mg/kg group (sublingual vein injection of 1 mL of LOB at 5 mg/kg 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours), LOB 10 mg/kg group, LOB 20 mg/kg group, and verapamil group (sublingual vein injection of 1 mL of verapamil at 2 mg/kg 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours). Plasma LDH and IL-6 and myocardium MDA levels were measured.
In Vitro Model of Hypoxia–Reoxygenation (H/R) and LOB Treatment
The H/R cell model was established as described previously. 13 H9C2 (2-1) cells were exposed to hypoxic conditions (oxygen deprivation, 0.5% O2) for 24 hours. After hypoxia, the cells were reoxygenated under normoxic conditions (reoxygenation) for 12 hours. There were 6 experimental groups: control group, H/R group (add LOB solvent to the culture medium, culture for 1 hour, and then apply H/R injury), LOB 1 μmol/L group (add LOB to the culture medium to a final concentration of 1 μmol/L, culture for 1 hour, and then apply H/R injury), LOB 3 μmol/L group, LOB 10 μmol/L group, and verapamil group (add verapamil to the culture medium to a final concentration of 1 μmol/L, culture for 1 hour, and then apply H/R injury). Each group contains 6 replicates. Interleukin 6 and MDA levels in the culture medium and caspase 3 activity of the cells were measured.
In Vitro Cell Viability Assay
In vitro cell viability was determined with the MTT cell proliferation and viability assay kit as described by the manufacturer (R&D systems). Briefly, cells were cultured at 15 × 103 cells per well in 96-well tissue culture plates and treated as mentioned above. At the end of the culture period, cells were washed with PBS, the MTT reagents were added according to the manufacturer's recommendations, and the absorbance was measured at 570 nm using an enzyme-linked immunosorbent assay (ELISA) plate reader. The proliferation/viability of cells was shown as percentage changes in the control cells (designated as 100%). Each experiment was repeated 3 times in triplicates.
Statistical Analysis
Statistical analyses were performed with SPSS for Windows 10.0. Data were expressed as mean ± SD. Comparison of means among multiple groups were performed with 1-way analysis of variance followed by post hoc pairwise comparisons using the least significant difference method. The significance level of this study was set at a 2-tailed α = .05.
Results
Protective Effects of LOB in a Rat Myocardial I/R Model
As shown in Figure 1, I/R injury induced myocardial infarction (32.1% ± 3.5% infarct size) in the I/R group, while the control and the sham groups showed no infarction. Sublingual injection of LOB 10 minutes before ischemia dose dependently decreased the myocardial infarct size in the I/R injury. Compared with the I/R group, the LOB 10 mg/kg and 20 mg/kg groups significantly decreased the infarct size (P < .05). LOB pretreatment at 20 mg/kg showed significant stronger protective effects on the infarct size than verapamil. Typical images of myocardial slices in each experimental group are shown in Figure 2. Histological changes in the myocardium of all experimental groups are shown in Supplementary Figure S2.

Myocardial infarct size in experimental groups. Myocardial infarct size was evaluated in rats that had ischemia–reperfusion (I/R) injury. Ischemia–reperfusion group: sublingual vein injection of 1 mL of LOB solvent 10 minutes before occlusion, followed by left anterior descending coronary artery (LAD) occlusion for 1 hour, and then reperfusion for 3 hours. LOB 5, 10, and 20 mg/kg groups: sublingual vein injection of 1 mL of LOB at 5, 10 or 20 mg/kg 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours. Verapamil (VR) group: sublingual vein injection of 1 mL of verapamil at 2 mg/kg 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours. a P < .05 versus the I/R group; b P < .05 versus the LOB (5 mg/kg) group; c P < .05 versus the LOB (20 mg/kg) group.
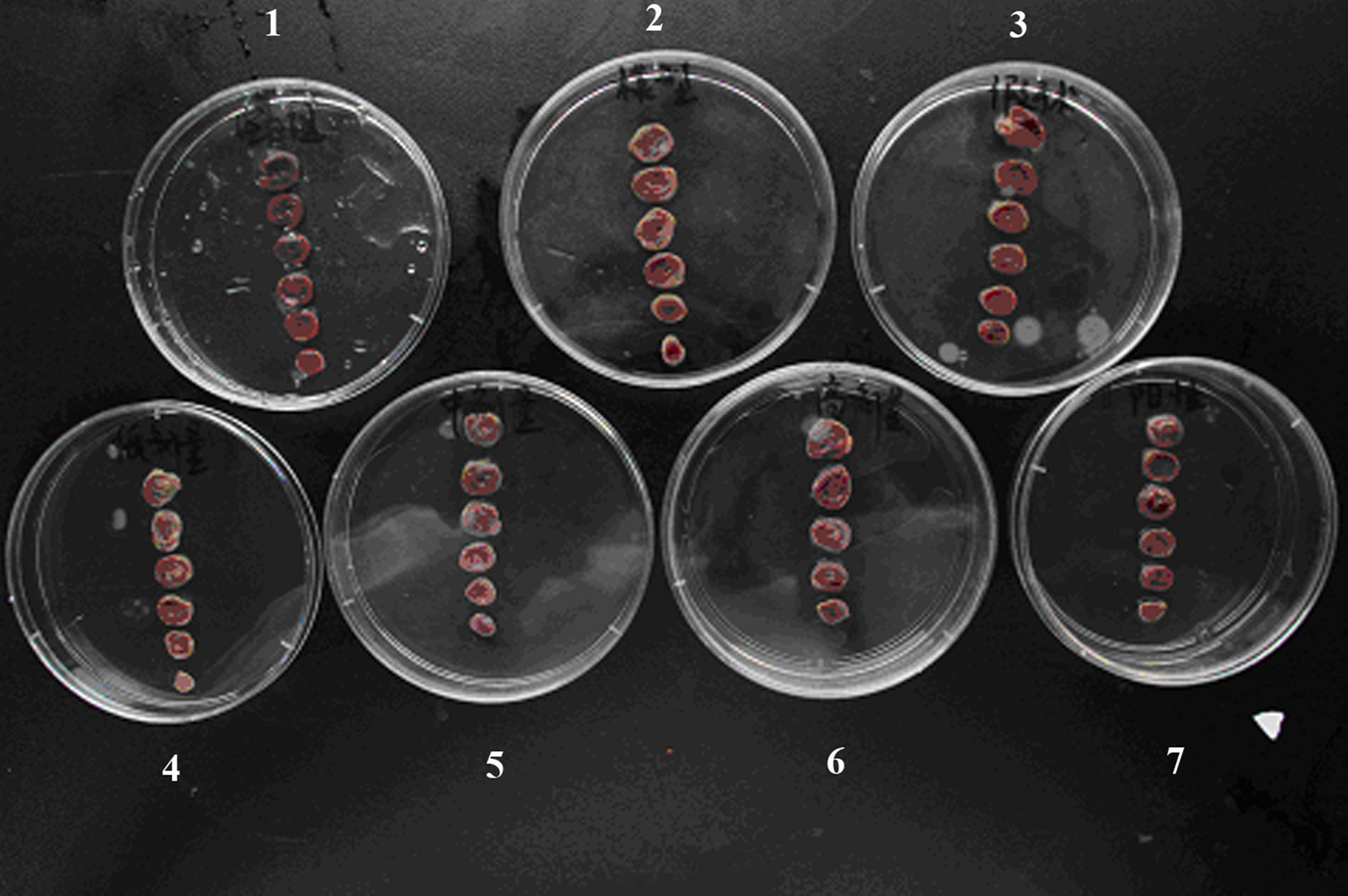
Typical myocardial slices in each experimental group. 1, control group (mice without sugery, occlusion, and drug treatment); 2, I/R group: sublingual vein injection of 1 mL of LOB solvent 10 minutes before occlusion, followed by left anterior descending coronary artery (LAD) occlusion for 1 hour and then reperfusion for 3 hours; 3, sham group (sham surgery-treated mice); 4-6, LOB 5, 10, and 20 mg/kg groups: sublingual vein injection of 1 mL of LOB at 5 mg/kg 10, or 20 mg/kg 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours; 7, verapamil (VR) group: sublingual vein injection of 1 mL of verapamil at 2 mg/kg 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours.
Compared to the control group, the plasma LDH (Figure 3A) and IL-6 (Figure 3B) levels and the MDA level in myocardium homogenate (Figure 3C) in the I/R group increased significantly (P < .05). Pretreatment with LOB at 5, 10, and 20 mg/kg dose dependently decreased the elevated plasma LDH (Figure 3A) and IL-6 (Figure 3B) levels and the myocardium MDA level (Figure 3C) induced by the I/R injury. LOB pretreatment at 20 mg/kg showed significant stronger protective effects on LDH and MDA levels than verapamil.

Plasma lactate dehydrogenase (LDH) (A) and interleukin 6 (IL-6) (B) levels and the myocardium malondialdehyde (MDA) (C) level in experimental groups. Ischemia–reperfusion (I/R) group: sublingual vein injection of 1 mL of LOB solvent 10 minutes before occlusion, followed by left anterior descending coronary artery (LAD) occlusion for 1 hour and then reperfusion for 3 hours. LOB 5, 10, and 20 mg/kg groups: sublingual vein injection of 1 mL of LOB at 5, 10, or 20 mg/kg 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours. Verapamil (VR) group: sublingual vein injection of 1 mL of verapamil at 2 mg/kg 10 minutes before occlusion, followed by LAD occlusion for 1 hour and then reperfusion for 3 hours. a P < .05 versus the control or the Sham group; b P < .05 versus the I/R group; c P < .05 versus the LOB (5 mg/kg) group; d P < .05 versus the LOB (20 mg/kg) group.
Protective Effects of LOB in a Rat Myocardium Cell H/R Model
As shown in Figure 4, LOB at 10 μmol/L showed no significant effects on the viability of normal H9C2 (2-1) rat myocardium cells. Compared to the control group, the cell viability was decreased to 79.5% ± 6.5% in the H/R group (P < .05). Pretreatment with LOB at 1 μmol/L, 3 μmol/L, and 10 μmol/L 1 hour before hypoxia dose dependently restored the cell viability, showing similar effects as Verapamil.

Cell viability of H9C2 (2-1) myocardium cells in experimental groups. LOB 10 μmol/L group: treat normal H9C2 (2-1) myocardium cells with 10 μmol/L of LOB; Verapamil (VR) group: treat normal H9C2 (2-1) myocardium cells with 1 μmol/L of VR; hypoxia–reoxygenation (H/R) group: add LOB solvent to the culture medium, culture for 1 hour, then apply H/R injury; +LOB 1, 3, and 10 μmol/L groups: add LOB to the culture medium to a final concentration of 1, 3, or 10 μmol/L, culture for 1 hour, then apply H/R injury; +verapamil (VR) group: add verapamil to the culture medium to a final concentration of 1 μmol/L, culture for 1 hour and then apply H/R injury. Cell viability of the control group was defined as 100%. a P < .05 versus the control group; b P < .05 versus the H/R group; c P < .05 versus the +LOB (1 μmol/L) group; d P < .05 versus the +LOB (10 μmol/L) group.
Compared to the control group, the LDH (Figure 5A) level in the cell culture medium and the MDA level in the cells (Figure 5B) of the H/R group increased significantly (P < .05). Pretreatment with LOB at 1 μmol/L, 3 μmol/L, and 10 μmol/L dose dependently decreased the elevated LDH (Figure 5A) level in the cell culture medium and the MDA level in the cells (Figure 5B) induced by the H/R injury. LOB pretreatment at 10 μmol/L showed significant stronger protective effects on LDH and MDA levels than verapamil.

Lactate dehydrogenase (LDH) (A) level in culture medium and the malondialdehyde (MDA) (B) level in H9C2 (2-1) myocardium cells in experimental groups. Hypoxia–reoxygenation (H/R) group: add LOB solvent to the culture medium, culture for 1 hour, then apply H/R injury; +LOB 1, 3, and 10 μmol/L groups: add LOB to the culture medium to a final concentration of 1, 3, or 10 μmol/L, culture for 1 hour, then apply H/R injury; +verapamil (VR) group: add verapamil to the culture medium to a final concentration of 1 μmol/L, culture for 1 hour, then apply H/R injury. a P < .05 versus the control group; b P < .05 versus the H/R group; c P < .05 versus the +LOB (1 μmol/L) group; d P < .05 versus the +LOB (10 μmol/L) group.
As shown in Figure 6, the caspase 3 activity was significantly increased in the H/R group compared to the control group (P < .05). Pretreatment with LOB at 1 μmol/L, 3 μmol/L, and 10 μmol/L dose dependently decreased the elevated caspase 3 activity induced by the H/R injury. LOB pretreatment at 10 μmol/L showed significant stronger protective effects against caspase 3 activity than verapamil.
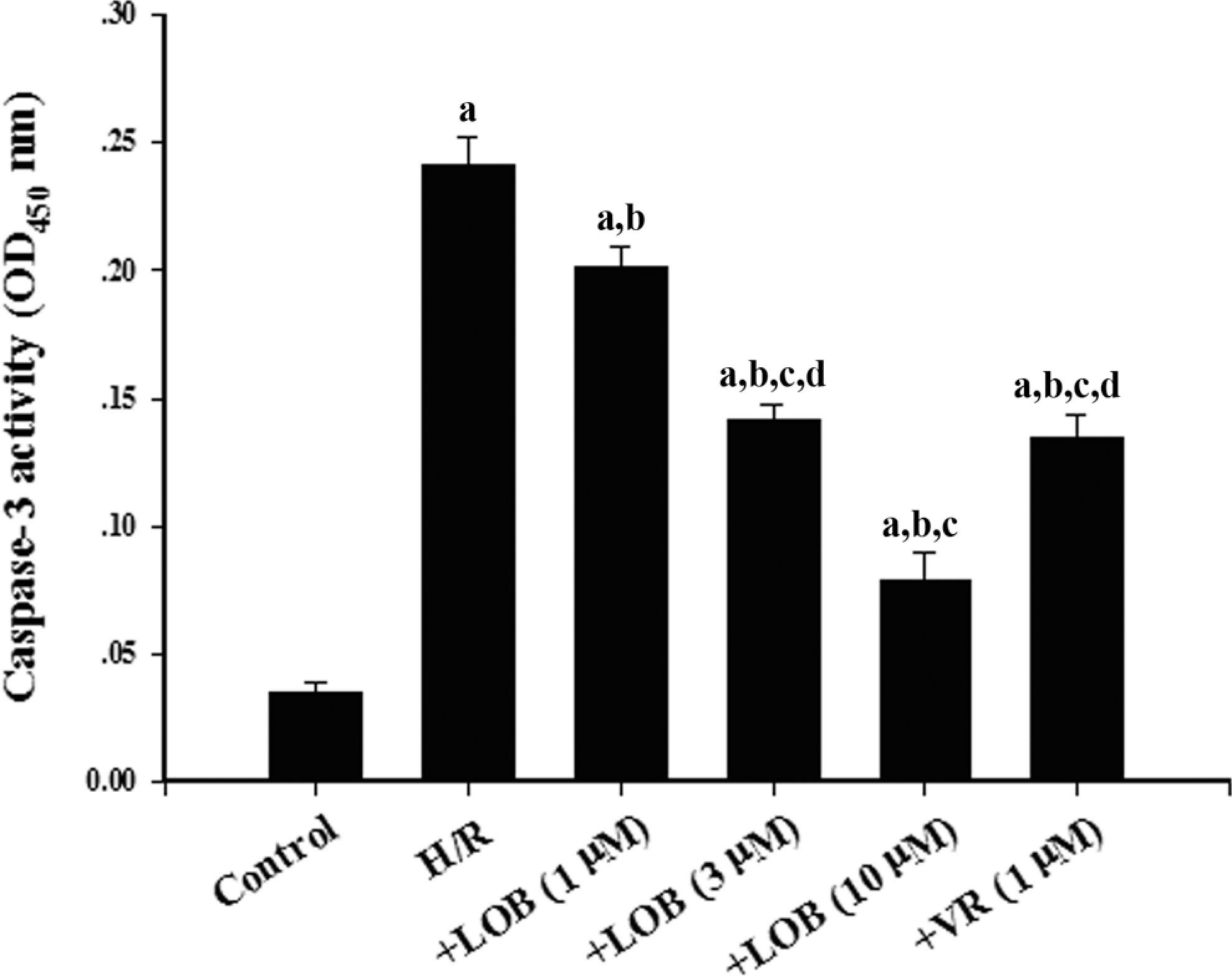
Caspase 3 activity in H9C2 (2-1) myocardium cells in experimental groups. Hypoxia–reoxygenation (H/R) group: add LOB solvent to the culture medium, culture for 1 hour, then apply H/R injury; +LOB 1, 3, and 10 μmol/L groups: add LOB to the culture medium to a final concentration of 1, 3, or 10 μmol/L, culture for 1 hour, then apply H/R injury; +verapamil (VR) group: add verapamil to the culture medium to a final concentration of 1 μmol/L, culture for 1 hour, then apply H/R injury. a P < .05 versus the control group; b P < .05 versus the H/R group; c P < .05 versus the +LOB (1 μmol/L) group; d P < .05 versus the +LOB (10 μmol/L) group.
Discussion
In the present study, we evaluated the protective effects of LOB, a natural flavone, on myocardial I/R injury in a rat model and on H/R injury in a rat myocardium cell line. Our findings for the first time provided evidence that LOB has strong protective effects against I/R injury in vivo and H/R injury in vitro.
Usually, cytosolic enzymes such as LDH and MDA serve as diagnostic markers of myocardial ischemia injury, leak out from the damaged myocardial tissues to the blood stream when the cell membrane becomes permeable or ruptures. The released enzymes are the main indicators of the extent of myocardial ischemia injury and also an important indicator to determine whether the cell is reversibly or irreversibly damaged. 4,14 In addition, IL-6 has been shown to be a key mediator in the pathophysiology of myocardial I/R. 15 In the present study, we found that LOB dose dependently reduced the elevated plasma levels of LDH and IL-6 and myocardium MDA level induced by the I/R injury, suggesting that LOB could reduce the extent of cell membrane damage and protein release in acute myocardial ischemia injury, and depress the systemic inflammatory response of the myocardial I/R injury.
Treatment with LOB at 1 μmol/L in vitro was equivalent to 0.04 mg/kg injection in vivo based on the rat’s average weight and blood volume, about 100-fold lower than the minimum concentration (5 mg/kg) used in vivo. The in vitro data in H9C2 (2-1) rat myocardium cells were in agreement with the in vivo data, in that LOB dose dependently reduced the elevated culture medium LDH level and cell MDA level induced by the H/R injury. In line with the findings, LOB dose dependently restored cell viability reduced by the H/R injury, probably due to its antiapoptotic effect by inhibiting the caspase 3 activity. Interestingly, both in vivo and in vitro data showed that high-dose LOB (20 mg/kg or 10 μmol/L) had stronger protective effects than the positive control drug verapamil (2 mg/kg or 1 μmol/L), suggesting that LOB could be a potential therapeutic agent for myocardial I/R injury. Further studies are needed to uncover molecular mechanisms underlying the myocardial protective effects of LOB.
Conclusions
Our study for the first time provided both in vivo and in vitro evidence that LOB exerted significant cardioprotective effects on myocardial I/R injury rats by reducing the extent of cell membrane damage, depressing the systemic inflammatory response to the myocardial I/R injury, and possibly inhibiting the caspase 3 activity in cardiomyocytes, thereby alleviating myocardial I/R injury and improving cardiac cell viability. Thus, LOB may have therapeutic potential as a cardioprotective agent.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
