Abstract
Objective:
Postoperative atrial fibrillation (POAF) is the most common complication after cardiac surgery, leading to increased morbidity and mortality. The aim of this preliminary study was to evaluate a novel drug delivery system for local release of amiodarone.
Methods:
In the current prospective study, 9 goats underwent attachment of right atrial (RA) epicardial electrodes. Alginate-based glue with amiodarone was applied to the RA of the treatment groups. Rapid atrial response (RAR) to burst pacing was assessed before application and in the third postoperative day (POD3). Average RAR frequency was defined as the average percentage of inductions resulting in RAR per animal. Myocardial and extracardiac tissue amiodarone concentrations were analyzed.
Results:
Differences in RAR proportions between baseline and POD3 were greater in the treatment group versus the control group (P = .034). Average RAR frequency was reduced by 34% in the treatment group (baseline: 65%; POD3: 31%), while it was increased by 11.3% in the control (baseline:43.8%; POD3: 55%). The treatment group demonstrated a greater proportion of animals meeting the success criterion of net percentage reduction in RAR frequency greater than 25% (P = .047). The average amount of total amiodarone detected in the RA was 104.4 ± 28.9 µg; the transmural concentration was linearly distributed (P < .0001). Extracardiac tissue concentrations were below the detection level.
Conclusions:
Local alginate-based amiodarone delivery demonstrated an RAR frequency reduction of clinical importance in response to burst pacing. The electrophysiological response was achieved while maintaining below-detection systemic drug levels. Current findings may point to the system’s future applicability in reducing POAF risk in humans.
Objective
Atrial fibrillation (AF) is the most widespread complication encountered after cardiac surgery, 1 associated with prolonged hospitalization, morbidity, mortality, and an annual cost of more than US$1 billion to the American health care system. 2,3 Atrial fibrillation has been reported to predict postoperative stroke, congestive heart failure, and late AF among patients undergoing mitral valve surgery. 4 Postoperative AF (POAF) has been reported to occur in 60% to 65% of patients undergoing combined aortic valve and coronary artery bypass graft (CABG) surgery, in 45% to 50% of patients undergoing aortic valve surgery alone, in 40% to 45% of patients undergoing mitral valve surgery, and in 30% to 35% of patients undergoing CABG surgery alone. 5 The POAF peak incidence occurs on the second and third postoperative days (PODs). In all, 70% of patients develop this arrhythmia before the end of the fourth POD and 94% before the end of the sixth POD 6 ; notably, these observations address in-hospital events. Recent guidelines for the prevention and management of POAF were jointly published in 2006 by the American College of Cardiology, the American Heart Association, and the European Society of Cardiology. 7 These guidelines recommended (1) preoperative treatment with an oral β-blocker drug for prevention of POAF as a class IA indication and (2) preoperative administration of amiodarone for patients at a high risk of POAF as a class IIA indication.
Amiodarone is one of the most effective antiarrhythmic agents for the management of ventricular as well as supraventricular tachyarrhythmias. 8,9 In a group of patients undergoing complex cardiac surgery, amiodarone reduced the incidence of POAF from 53% in the placebo group to 25% in the treated group. 10 In keeping, the Amiodarone Reduction in Coronary Heart (ARCH) trial, including 300 patients with CABG, demonstrated that administration of postoperative intravenous amiodarone was associated with a 35% incidence of POAF compared with a 47% incidence in the placebo arm. 11 The trial of Prophylactic Oral Amiodarone for the Prevention of Arrhythmias that Begin Early After Revascularization, Valve Replacement, or Repair 12 (PAPABEAR) demonstrated that a 13-day perioperative course of oral amiodarone was well tolerated and effective for the prevention of postoperative atrial tachyarrhythmia. Amiodarone is associated with several complications, both cardiac and extracardiac. 13,14 ; among the cardiac complications, it is predominantly related to increased risk of bradycardia and hypotension, particularly when (1) administered intravenously, (2) using average daily doses exceeding 1 g, and (3) initiating prophylaxis during the postoperative period. Among the most severe extracardiac complications, amiodarone is associated with thyroid toxicity, lung fibrosis, and corneal damage, which may be aggravated by the drug’s delayed onset of action (2-3 days) and long elimination half-life time (up to 6 months). 7,14
A targeted, local delivery of amiodarone might uphold its advantages while resolving its aforementioned adverse effects, since (1) a relatively larger proportion of the drug will be absorbed by cardiac tissue, thus inducing the required effects at lower doses, and (2) extracardiac drug levels will be diminished and, consequently, peripheral side effects minimized. 15 –18 The current animal study examined the electrophysiological and drug release efficacy of a proprietary epicardial alginate-based amiodarone-delivering system in an acute goat model.
Material and Methods
Animal Preparation
All animals received humane care in compliance with the European Convention on Animal Care; the study was approved by the Technion—Israel Institute of Technology animal committee.
The Local Drug-Delivery System
The local amiodarone delivery system consisted on SEAlantis, a patent-protected, alginate-based biomimetic glue, composed of a mixture of polyphenol, H2O2, KI, bromoperoxidase, alginate, and calcium ions in Milli-Q water (Millipore Corporation). 19 The glue contains the same ingredients as an alga-born glue (composed of components extracted from the brown alga Fucus serratus: alginate and polyphenol which was oxidized in the presence of bromoperoxidase, KI, H2O2, and calcium ions), yet the polyphenol was replaced by phloroglucinol. The adhesive strength of the biomimetic glue is comparable to that of the alga-born one. Moreover, both glues seem to adhere better to hydrophobic surfaces. Natural algal-born polyphenol can be successfully replaced by phloroglucinol, a low-molecular-weight synthetic analog, without considerable changes in the adhesion properties. Formulations composed of phloroglucinol, alginate, and calcium ions are capable of adhering to a variety of surfaces including porcine tissue. The glue is biodegraded in 2 weeks time. 20 It is to be emphasized that formulations containing native phloroglucinol were found to be safely applied to living cells without the risk of causing cell death. 21
In vitro tests were used to design a gel containing a high alginate concentration and amiodarone, solidified by adding calcium ions on top of the pregel. In vitro tests were performed to obtain a calibration curve for amiodarone concentration measurements using a spectrophotometer. Finally, a slow-releasing system for amiodarone based on SEAlantis prototypes was designed for testing the electrophysiological outcome and the drug delivery properties in a subchronic experiment.
Exploratory pilot in vivo experiments were performed on 4 common goats for establishing a proof of concept regarding the adhesion properties of both the alginate glue and the alginate–amiodarone medium in the cardiac atrial tissue. The adhesion properties were examined in 2 settings, namely, acute (3 hours postoperation; 2 goats) and subacute (3 days postoperation; 2 goats). In both settings, adhesion was qualitatively described either as a “positive” or as a “negative” finding according to the visual assessment of the surgeon. The subacute electrophysiological response was examined on the third POD (POD3); namely, a burst pacing test was performed and the rapid atrial response (RAR) was depicted. All 4 pilot experiments yielded “positive” outcomes and clear recordable responses, which attested for the potential capacity of the glue to be used as a drug delivery medium in the current prospective study. In both settings, the alginate glue and the alginate–amiodarone medium were applied to the right atrium; thereafter, the chest wall was approximated and closed.
The experiment was conducted on 9 common goats (not the pilot goats) that were randomized such that they were alternately assigned either to the study group (5 goats) or to the control group (4 goats). Anesthesia was achieved by thiopental 20 mg/kg intravenously (IV) and maintained on isoflurane (2%-3%) with a 1:1 mixture of oxygen and air. Analgesia was achieved with buprenorphine (10 mg/kg IV). Limb-lead electrocardiogram (ECG), end-expiratory CO2, and oxygen saturation (pulse oximeter) were monitored throughout the procedure. An external heating pad was used to maintain a rectal temperature of 38°C to 39°C. Sodium chloride solution, 0.2 to 0.35 mL/kg/min, was given IV. The right carotid artery was cannulated with a catheter attached to a pressure transducer for measuring systolic and diastolic arterial pressures. Surgical approach was via a lateral right thoracotomy. A 6- to 8-cm long incision was made in the pericardium, and the drug delivery system was applied along the right atrium of the study group, namely, amiodarone concentration of 1 mg/kg of body weight in a constant glue volume of 4 mL. In the control group, 4 mL of glue without amiodarone was applied. Bipolar atrial temporary pacing wires (Mce, Medical Concepts Europe, VO60BB, Gemert, the Netherlands) were sutured to the right atrium (RA). The electrodes were placed in 2 areas of the RA: 1 just above the SA node area and the second 2 cm distally toward the right coronary artery. The applied glue covered, on average, an area of 4 × 4 cm2 of the RA, lateral to the electrodes’ location. Pericardial incision was approximated with sutures which were placed in 2- to 3-cm intervals, leaving the greater part of the pericardial sac open to the mediastinal space. The applied glue covered, on average, an area of 4 × 4 cm2. Due to anatomical variability, the chosen location was the most available smooth and flat area of the mentioned size in each atrium, allowing effective adherence. Much attention was directed at covering the same area at the same anatomical location in all animals. A complete hardening of the glue, which takes approximately 1 minute, was achieved before pericardium closure.
Drug Concentration Measurements
During the procedures, blood samples were drawn from a peripheral vein before medium placement and 60, 120, and 180 minutes thereafter. Additional blood samples were drawn in 12-hour intervals. Blood was centrifuged and the plasma stored at −80°C.
After euthanasia, 3 free-wall sections of 4 cm2 were dissected from the RA. Additionally, a single 4-cm2 section was dissected from the left atrium (LA), right ventricle (RV), and left ventricle (LV) respectively. Slices were frozen in liquid nitrogen and stored at −80°C. To assess transmural drug concentration gradients, frozen free-wall sections were sliced using a microtome, parallel to the epicardial surface, in a slice width of 500 mm.
Drug concentrations in plasma and tissue were determined by high-performance liquid chromatography (HPLC) coupled with fluorescence and ultraviolet (UV) detection for amiodarone. Additionally, samples from the following tissues were taken and examined for amiodarone drug concentration: (1) thyroid gland, (2) muscle (sternocleidomastoid muscle), and (3) liver. 16
The drug concentration analysis was performed in Maastricht, the Netherlands. The HPLC system consisted of a Shimadzu LC-10 AD liquid chromatograph, an SIL-10 AD autoinjector, and a SP8490 variable wavelength UV detector (Newport Spectra-Physics, Utrecht, the Netherlands) set at 254 nm. LabSolutions software (Shimadzu, Kyoto, Japan) was used for instrument control and data acquisition. 17
Electrophysiological Measurements
Electrocardiogram limb leads were connected to an electrophysiology monitoring system with integrated amplifier/stimulator; RR, PQ, QT, P, and QRS widths were measured during normal sinus rhythm to detect the occurrence of adverse cardiac events. All animals underwent electrophysiological testing at baseline and after placement of the drug delivery system. Additional electrophysiological tests were performed at the second POD and POD3. Atrial effective refractory periods (AERPs) were measured during bipolar pacing at 4 times the threshold. The AERP was assessed before drug application and on the POD3. Ten basic stimuli (S1) with 400-millisecond intervals in-between were followed by an extrastimulus (S2). The S1–S2 intervals were incremented by 2-millisecond steps and the longest S1–S2 interval failing to capture defined the effective refractory period. 17,18
To assess atrial vulnerability to arrhythmia, 10 basic stimuli (S1) with 400-millisecond intervals were applied, followed by a 1-second, 50-Hz burst pacing (S2). A RAR was considered inducible if a rapid irregular rhythm lasting 1 second occurred after S2. 17,18 The RAR was assessed before drug application and on the POD3. Thus, atrial vulnerability was examined twice (baseline and follow-up), while each test consisted of 20 burst pacing attempts and the tracing of the atrial response thereafter. This is according to the experimental protocol utilized by Bolderman et al. 16 –18
Statistical Analysis
All analyses were conducted using SPSS Version 19 (SAS Institute, Cary, North Carolina). The first study outcome measure was the epicardial average RAR frequency, which was defined as the average percentage of inductions resulting in RAR per animal. Average RAR frequency was quantified by the proportion of positive RAR inductions of 20 attempts. Differences in RAR frequency between baseline and POD3 were determined for each animal in each group (treatment vs control). Within groups, differences between baseline and POD3 were compared by 2-sided, nonparametric Wilcoxon rank-sum test. Between groups, differences per condition, that is, baseline and POD3, were tested using a 2-sided, nonparametric Mann-Whitney U test. To further assess the effectiveness of the amiodarone local delivery system, a success criterion of net percentage reduction in RAR frequency greater than 25% (on POD3 compared to baseline) was adopted. The difference in the success rate between the treatment and control groups was tested using 2-sided Fisher exact test.
The second study outcome measure was the amiodarone transmural cardiac dispersion gradient and plasma as well as extracardiac drug concentrations. Drug concentrations were determined in corresponding slices of the each cardiac tissue free-wall sample, and an epicardium to endocardium transmural concentration gradient was established. The dependence of drug concentration (sum and average) on the joint RA slices was tested using a linear regression test. The average amount of detected amiodarone and the median number of extracted slices were calculated for the RA, RV, LA, and LV. Continuous variables are presented as average ± standard deviation (SD). Statistical significance was set at P < .05.
Results
Electrophysiological Findings
The Wilcoxon test indicated that differences in RAR proportions, between baseline and POD3 (Table 1), were significantly greater in the treatment group than in the control group (P = .034; Figure 1), suggesting a significantly larger reduction in RAR frequency associated with amiodarone application. Average RAR frequency was reduced by 34% in the treatment group (from 65% at baseline to 31% on POD3), while it was increased by 11.3% in the control group (from 43.8% at baseline to 55% on POD3). An exemplar electrode recording is brought in Figure 2 so as to allow a visual appreciation of the induced RAR.
Data of the Induction of Rapid Atrial Response in the Study and Control Animals.

Abbreviations: POD, postoperative day; AF, atrial fibrillation.
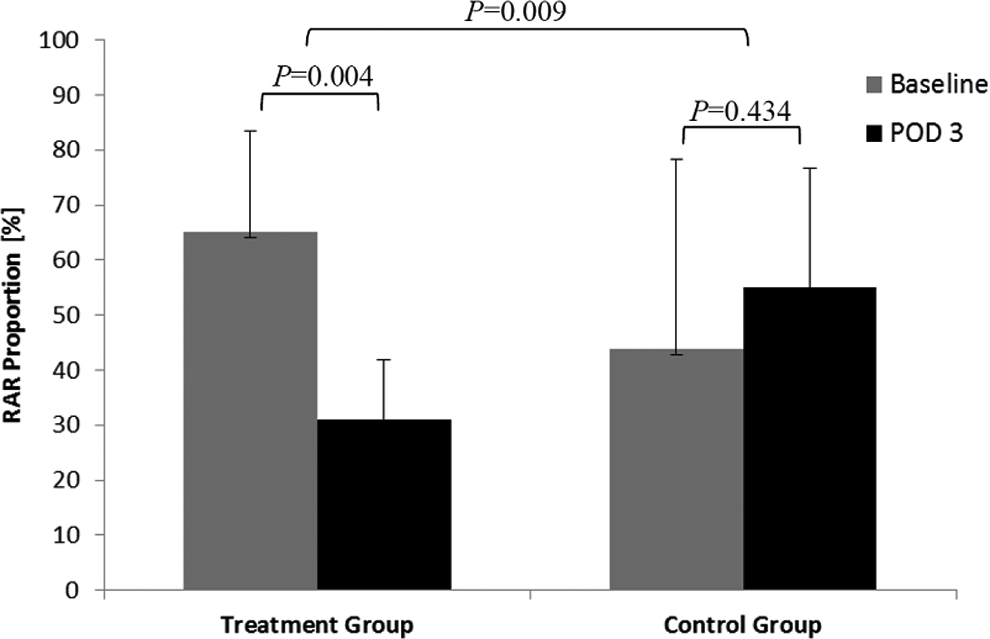
Differences in RAR proportions between the treatment and the control groups. RAR indicates rapid atrial response; POD, postoperative day.

Sinus rhythm prior to rapid atrial pacing attempt of 1-second duration (A) and RAR in response to a rapid atrial pacing attempt (B).
The Mann-Whitney U test indicated no difference between the 2 groups in the RAR frequency at the baseline condition (P > .1), while a significant difference was observed on POD3 between the 2 groups (P = .02).
Based on the Fisher exact test, the treatment group demonstrated a significantly greater proportion of animals meeting the success criterion of net percentage reduction in RAR frequency greater than 25% (on POD3 compared to baseline; P = .047), attesting to the superior effectiveness of the local drug delivery system in RAR reduction, as compared with the control group. Differences in AERP remained unaffected in the treatment group (P > .2); namely, the pretreatment AERPs in the 5 animals comprising the treatment group were 190, 200, 210, 200, and 230 milliseconds, while the posttreatment AERPs were 190, 210, 210, 210, and 220 milliseconds, respectively. No bradyarrhythmias were detected during the study period in either group.
Pharmacodynamic Findings
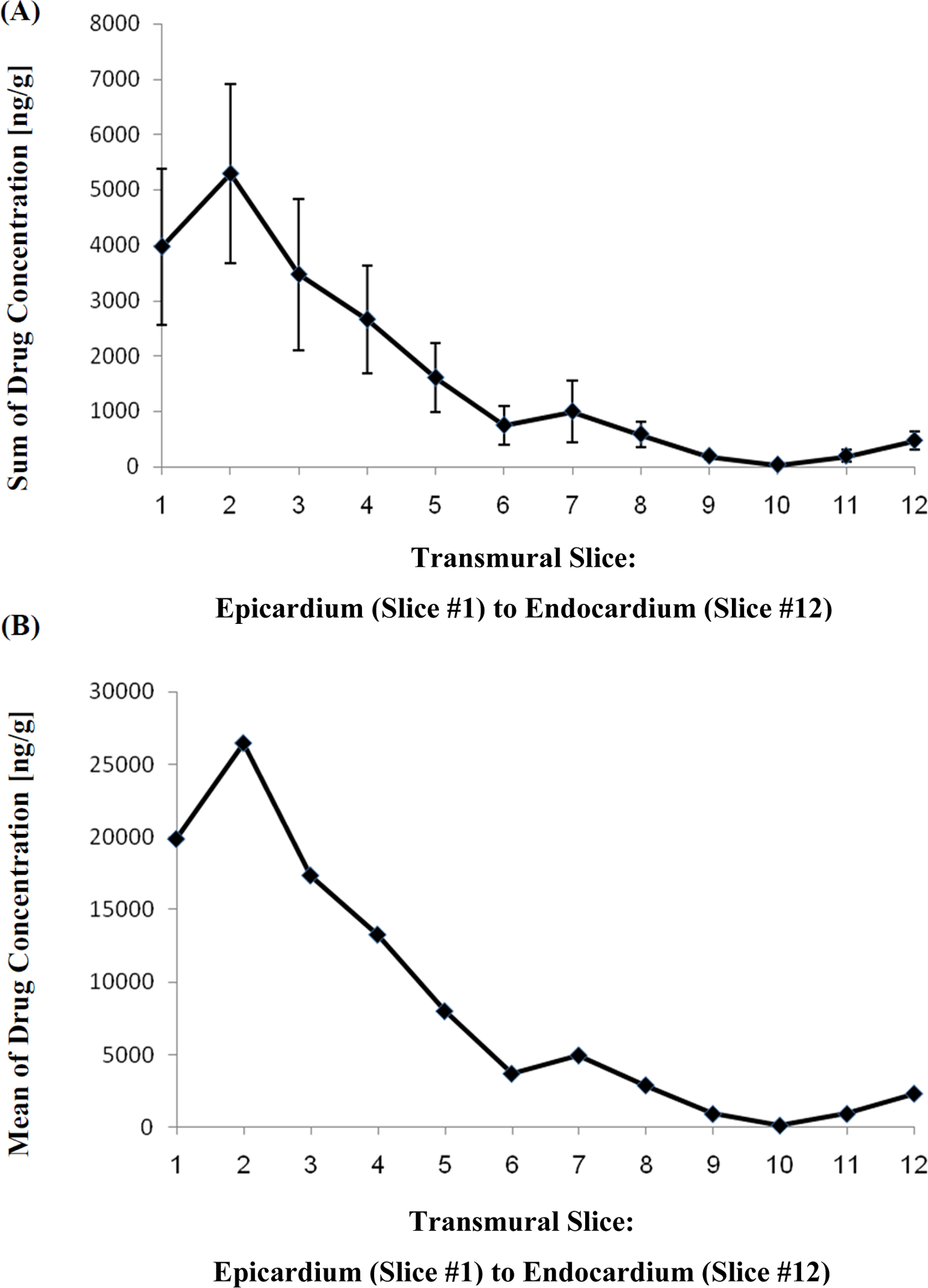
At the RA, where the amiodarone was locally applied, both the average and the sum of amiodarone transmural concentration, collectively measured in all 3 free-wall sections, were linearly distributed along the examined slices of the cardiac tissue (R 2 = 0.90, P < .0001, Figure 3A; R 2 = 0.883, P < .0001, Figure 3B, respectively). The average amount of total amiodarone detected in the RA was 104.4 ± 28.9 µg. The median number of slices extracted from the RA tissue samples was 11 (range: 8-15).

Average (A) and sum (B) of amiodarone transmural dispersion in the right atrium.
The average amount of total amiodarone detected in the LA was 1.9 ± 1.7 µg. The median number of slices extracted from LA tissue samples was 10 (range: 8-12). Both the average and the sum of amiodarone transmural concentration were linearly distributed along the examined slices of the cardiac tissue (R 2 = 0.795, P < .0001, Figure 4A; R 2 = 0.774, P < .0001, Figure 4B, respectively).
Average (A) and sum (B) of amiodarone transmural dispersion in the left atrium.
The average amount of total amiodarone detected in the RV was 3.1 ± 1.7 µg. The median number of slices extracted from RV tissue samples was 10 (range: 8-12). Both the average and the sum of amiodarone transmural concentration were linearly distributed along the examined slices of the cardiac tissue (R 2 = 0.585, P = .003, Figure 5A; R 2 = 0.454, P = .016, Figure 5B, respectively).

Average (A) and sum (B) of amiodarone transmural dispersion in the right ventricle.
The average amount of total amiodarone detected in the LV was 0.7 ± 0.7 µg. The median number of slices extracted from LV tissue samples was 10 (range: 8-12). Both the average and the sum of amiodarone transmural concentration were linearly distributed along the examined slices of the cardiac tissue (R 2 = 0.790, P = .0001, Figure 6A; R 2 = 0.768, P = .0002, Figure 6B, respectively).

Average (A) and sum (B) of amiodarone transmural dispersion in the left ventricle.
Amiodarone concentrations in the blood and extracardiac tissues, namely, the thyroid gland, sternocleidomastoid skeletal muscle, and liver were below the detection level (10 μg). No ECG measures fell outside the normal value range.
Discussion
The main findings of this preliminary study are that epicardial alginate-based application of amiodarone results in (1) persistently high drug concentrations in the RA on the POD3 and drug diffusion indicated a gradual, efficient transmural dispersion of amiodarone; (2) negligible drug levels in the LA, RV, and LV; (3) undetected drug levels in the blood and extracardiac tissues; (4) decreased vulnerability to burst pacing-induced atrial tachyarrhythmias; and (5) no bradyarrhythmias. Based on these observations, local amiodarone administration may be efficacious in reducing atrial vulnerability to arrhythmias.
Based on the fact that POAF reportedly occurs mostly during the second POD to POD3, the current study examined the efficacy of the local amiodarone delivery system 3 days after its application. The observation that at that time point local amiodarone administration reduced RAR frequency and resulted in no electrophysiological complications, for example, bradyarrhythmias and heart blocks may attest to its prospective physiological effectiveness and advantageous timing in POAF treatment. Importantly, the electrophysiological assessments reported in the current study delineate the achievable effects of amiodarone; they do not constitute a clinical surrogate for POAF but rather for amiodarone efficacy.
Amiodarone concentrations were found to be below the detection level in blood as well as in extracardiac tissues, namely, thyroid gland, sternocleidomastoid muscle, and liver. This may indicate a slow diffusion of amiodarone to the plasma and relatively rapid redistribution from the plasma to extracardiac tissues. This drug delivery profile stands to reason as amiodarone is characterized with a large volume of distribution due to high tissue affinity. 21,22
Previous methods of sustained local drug delivery to the myocardium relied on intrapericardial catheterization or the use of nonbiodegradable polymeric drug delivery systems. 23 –27 However, these methods were found to be associated with increased risks of complications, including infections and fibrosis. On the other hand, Bartoli et al 24 found over a 1-year period that silicone intrapericardial catheters remained bidirectionally patent and mobile with minimum inflammation and no infection. Furthermore, in nonbiodegradable systems, such complications may necessitate surgical removal of the drug delivery matrix. Notably, the resulting drug delivery profile produced by these systems was not found to be localized to the cardiac atrial tissue but rather to diffuse freely within the pericardial space to all of the cardiac chambers. This distribution profile was associated with conduction disturbances and extracardiac drug distribution.
Other potential clinical applications of amiodarone-releasing, alginate-based glue include treatment of paroxysmal AF and focus-induced ventricular arrhythmias. Such treatment may be enabled by alginate-based drug release of different diffusion profiles, providing longer periods of sustained drug release and advanced minimally invasive percutaneous techniques for local application. Regarding the latter, new approaches, such as subxiphoid and transatrial access, have already been evaluated. 28,29
Several limitations within the present study are to be acknowledged. First, a small number of animals was tested, which limits the ability to generalize current observations. Second, depiction of the drug absorption properties should be delineated for longer follow-up periods. Third, the site of local amiodarone application was the RA; since the aim of the current animal experiment was to examine the future applicability of the drug release system in humans, the RA was chosen due to its surgical accessibility during open heart surgery. In patients with paroxysmal AF, the pulmonary veins are important in arrhythmogenesis 30 ; therefore, it would be informative to study the effects of such a system by its application along the LA, around the pulmonary veins. Fourth, the surgeon was not blind to the patch type (with/without drug) as the 2 patches differed in color due to a differential reaction of the glue with the amiodarone. Finally, the control group demonstrated a lower baseline RAR incidence than that of the treated group; this may be accounted, at least partially, to physiological variability and a relatively small sample. The statistical analysis was adjusted to this variation by exploring the differences in RAR frequency between baseline and POD3 within each group.
Conclusion
Epicardial application of amiodarone-eluting, alginate-based glue yielded a significant RAR frequency reduction of clinical importance in response to burst pacing. This may point to the system’s future applicability in reducing the risk of POAF in humans. Additionally, such electrophysiological response was achieved while maintaining below detection systemic drug levels, which minimizes the risk of extracardiac adverse effects. Further chronic studies should be performed to substantiate these findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Beckerman worked for SEAlantis from January 2010 to December 2011. Dr Bolotin served as a consultant for SEAlantis from July 2009 to October 2010. Dr Maessen served as a consultant for SEAlantis from October 2009 to September 2010. Mrs Azran has been working for SEAlantis since January 2010. Dr Bianco-Peled serves as a Director, CSO, PI, and consultant for SEAlantis since January 2010 and is a patent-holder of the described technology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by SEAlantis.
