Abstract
Diabetes mellitus is a major risk factor for cardiovascular events and patient death. Many animal and clinical studies are now being conducted exploring the potential of antidiabetic drugs such as glucagon-like peptide 1 (GLP-1) agonists and dipeptidyl peptidase IV (DPP-IV) inhibitors to improve cardiovascular outcomes. This review summarizes the effect of DPP-IV inhibitors on myocardial ischemia–reperfusion injury in animal models. The DPP-IV inhibitors prevent the rapid degradation and inactivation of incretins and lead to the accumulation of GLP-1 and other chemokines and cytokines, which appear to have both GLP-1 receptor-dependent and -independent cardioprotective, antiapoptotic, and anti-inflammatory effects. Conflicting results, however, have been reported regarding the effect of DPP-IV inhibitors on infarct size in nondiabetic and diabetic animal models. Some studies suggest that DPP-IV inhibitors given as part of preconditioning can decrease infarct size while others found no difference in infarct size compared to placebo. As postconditioning, one study suggested it does provide cardioprotection. No clinical trials have yet been conducted addressing the effect of DPP-IV inhibitors on infarct size. Thus far, clinical trials have not demonstrated improvement in cardiovascular events or mortality from any cause in high cardiovascular risk, type 2 diabetic patients with the use of DPP-IV inhibitors. Although further experiments and clinical trials will be warranted to confirm the results of these studies, the myocardial protection afforded by DPP-IV inhibitors in preclinical animal studies poses a potential breakthrough role for antidiabetic medications in attenuation of ischemia–reperfusion injury that occurs with cardiovascular disease.
Keywords
About 347 million people worldwide and 25.8 million Americans, who represent 8.3% of the US population, have diabetes mellitus (DM). 1,2 DM is the leading cause of lower limb amputations, blindness, and kidney failure in adults and the seventh leading cause of death in the United States. 2 Furthermore, the risk of stroke and death from cardiovascular disease is 2 to 4 times higher in people with DM compared to nondiabetic persons. 2 In fact, 50% to 80% of deaths in people with DM can be attributed to cardiovascular disease. 2 Although strict glycemic control decreases the microvascular complications of DM, 3 it is uncertain whether any treatment strategy decreases macrovascular complications. 4,5 Many animal and clinical studies are now being conducted exploring the potential of antidiabetic drugs such as glucagon-like peptide 1 (GLP-1) agonists and dipeptidyl peptidase IV (DPP-IV) inhibitors to improve cardiovascular outcomes.
Dipeptidyl peptidase IV inhibitors, a relatively new class of antidiabetic medication, also referred to as “gliptins,” inhibit the normally rapid degradation and inactivation of incretins and lead to the accumulation of endogenous GLP-1 (7-36), the active form of GLP-1, and other chemokines and cytokines, which affect the cardiovascular and vascular systems. 6,7 Incretins such as GLP-1 and glucose-dependent insulinotropic peptide (GIP) are secreted by the intestinal endocrine L cells following meals. Glucagon-like peptide 1 helps achieve glucose control by stimulating insulin secretion in a glucose-dependent manner and decreasing glucagon secretion and gastric motility. 8 –10
In this review, we will be summarizing the effect of DPP-IV inhibitors on myocardial ischemia–reperfusion injury in animal models.
Dipeptidyl Peptidase IV Inhibitors and Ischemia–Reperfusion Injury in Animal Studies
Several studies have shown that GLP-1 receptor agonists protect against ischemia–reperfusion injury and reduce myocardial infarct size in experimental models 11 as well as in the clinical setting. 12,13 Less information is available as to whether DPP-IV inhibitors can replicate the effect and protect against ischemia–reperfusion injury. Studies in in vivo models of ischemia–reperfusion injury have suggested that DPP-IV inhibitors limit infarct size. Nondiabetic mice preconditioned with sitagliptin 14,15 and nondiabetic pigs preconditioned with vildagliptin 16 had significantly smaller infarct size compared to control animals (Table 1). Furthermore, this cardioprotective effect was not attenuated even with longer treatment periods up to 14 days, 14 whereas it has been suggested that the protective effect of statins is lost after 1 week of treatment due to upregulation of phosphatase and tensin homolog (PTEN). 19 The DPP-IV inhibitors also limited infarct size after ischemia–reperfusion injury in diabetic animals (Table 2). Db/db mice with type 2 DM given MK0626, a DPP-IV inhibitor, had dose-dependent decrease in infarct size that was augmented by cilostazol, a phosphodiesterase 3 inhibitor that prevents the degradation of cyclic adenosine monophosphate (cAMP). 10 Similarly, administration of a DPP-IV inhibitor in prediabetic (diet-induced obesity [DIO]) rats reduced infarct size. 18 Interestingly, the addition of granulocyte-colony stimulating factor (G-CSF) to DPP-IV inhibition appears to enhance cardioprotection by increasing homing of circulating stem cells to damaged cardiac tissue. 15
Studies of Dipeptidyl Peptidase IV Inhibitors and Nondiabetic Animal Models of Ischemia–Reperfusion Injury.
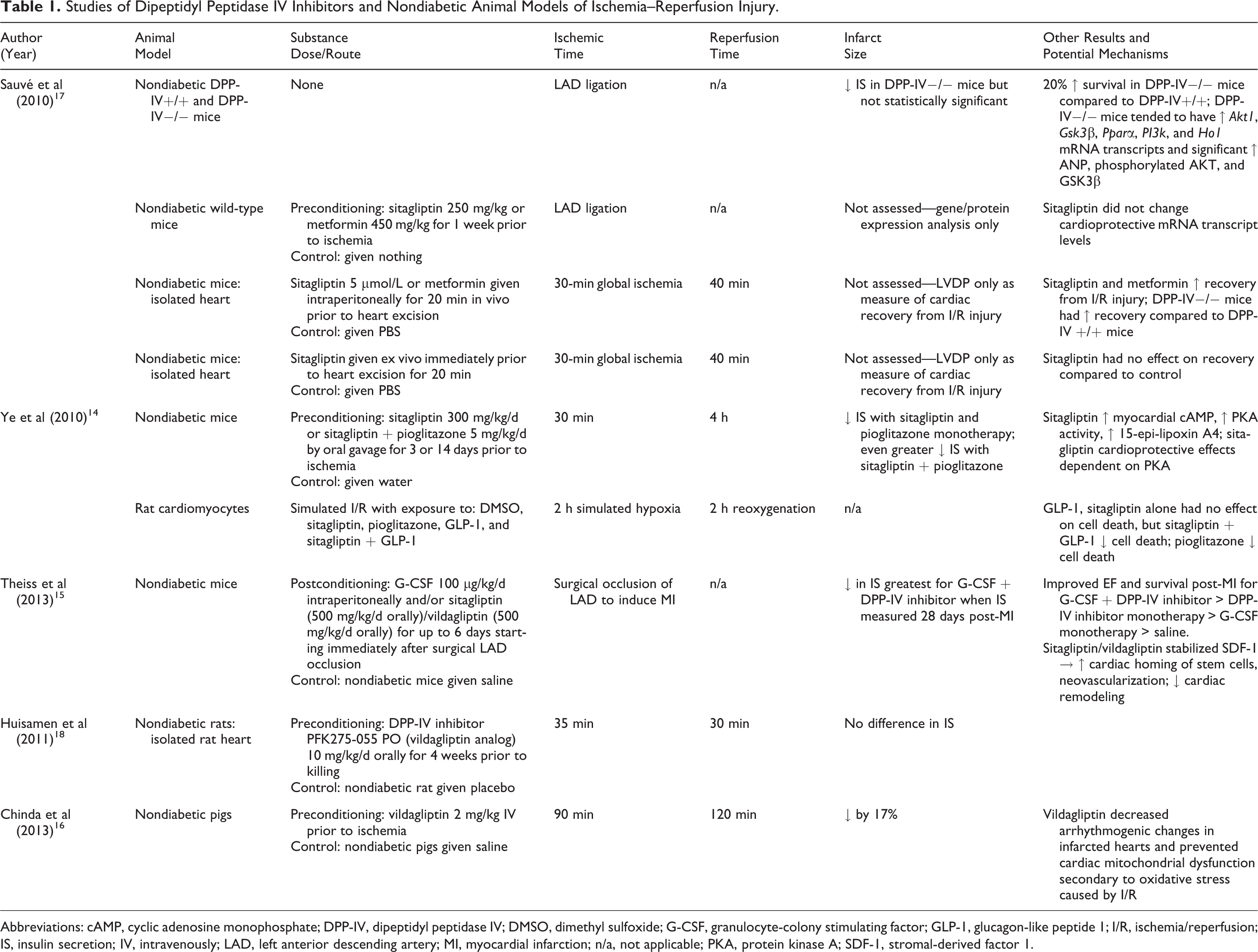
Abbreviations: cAMP, cyclic adenosine monophosphate; DPP-IV, dipeptidyl peptidase IV; DMSO, dimethyl sulfoxide; G-CSF, granulocyte-colony stimulating factor; GLP-1, glucagon-like peptide 1; I/R, ischemia/reperfusion; IS, insulin secretion; IV, intravenously; LAD, left anterior descending artery; MI, myocardial infarction; n/a, not applicable; PKA, protein kinase A; SDF-1, stromal-derived factor 1.
Studies of Dipeptidyl Peptidase IV Inhibitors and Diabetic Animal Models of Ischemia–Reperfusion Injury.

Abbreviations: cAMP, cyclic adenosine monophosphate; CREB, cAMP response element binding; DPP-IV, dipeptidyl peptidase IV; DIO, diet-induced obesity; eNOS, endothelial nitric oxide; GLP-1, glucagon-like peptide 1; HgbA1c, hemoglobin A1c; HFD-STZ, high-fat diet—streptozotocin; IS, insulin secretion; I/R, ischemia/reperfusion; LAD, left anterior descending artery; n/a, not applicable; PKA, protein kinase A; RISK, reperfusion-induced salvage kinase.
On the other hand, there have also been reports that DPP-IV inhibitors fail to limit infarct size in both nondiabetic and diabetic animal models. Preconditioning with a vildagliptin analog in isolated nondiabetic rat hearts failed to show a difference in infarct size compared to rats given placebo. 18 Infarct size after myocardial infarction (MI) induced by permanent left anterior descending artery (LAD) ligation in mice with streptozotocin-induced type 2 DM (high-fat diet—streptozotocin [HFD-STZ]) given sitagliptin was similar to that observed in HFD-STZ mice given placebo. 17
Investigations in animal models suggest that DM and/or hyperglycemia attenuate the cardioprotective effects of pre- and postconditioning against ischemia–reperfusion injury and lead to increased infarct size. 11 Therefore, one can hypothesize that improvement in glycemic control may lead to improved cardioprotection from ischemia–reperfusion injury. This raises the question whether the reduction in infarct size, reported by Huisamen et al 18 in prediabetic animals, but not in nondiabetic animals, is a nonspecific effect secondary to better glycemic control. In the study by Huisamen et al, 18 ischemia–reperfusion resulted in significantly larger infarct size in the DIO rats compared to the control nondiabetic rats. This could be secondary to improved glycemic control in the longer term with DPP-IV inhibitor administration. Nonfasting blood glucose levels between control and DIO rats were comparable at the time of killing regardless of whether a DPP-IV inhibitor was given. 18 However, measures of glycemic control and/or insulin sensitivity such as fasting glucose, hemoglobin A1c, or glucose-tolerance test were not reported. Others reported that DPP-IV inhibitors reduce infarct size using in vivo models in nondiabetic animals, which suggests a beneficial effect independent of improved glycemic control. 14,16 Huisamen et al 18 used an isolated heart model of ischemia–reperfusion. The isolated heart model could be problematic. Glucagon-like peptide 1 is produced only by the intestine. 11,14,20 Using an in vitro model, Ye et al 14 showed that cardiomyocytes were protected against simulated hypoxia–reoxygenation only when GLP-1 and DPP-IV inhibitor were combined. The GLP-1 alone was not protective, as it was rapidly degraded by the DPP-IV in the cardiomyocytes; whereas the DPP-IV inhibitor alone had no protective effect, as GLP-1 was not present in the cell culture. 14 Upon explantation, the heart is perfused with a solution lacking GLP-1 and/or DPP-IV inhibitor. With the short half-life of GLP-1 and lack of its supply from intestinal cells, it may be that any GLP-1 and/or the DPP-IV inhibitor were washed away with loss of any potential for cardioprotective effect. Thus, no infarct size reduction effect was seen in nondiabetic animals; whereas, the protective effect seen in DIO animals was likely nonspecific and related to improvement in glucose control/insulin resistance. Constant infusion of GLP-1 and the DPP-IV inhibitor would be needed for accurate clinical correlation between the isolated heart and the in vivo models. In addition, as DPP-IV inhibition alone in isolated hearts provided no protective effect, the nonincretin-related effects of DPP-IV inhibitors appear to play a less important role in ischemia–reperfusion injury.
As mentioned earlier, Sauvé et al 17 used a permanent LAD ligation in their HFD-STZ diabetic mice. However, it is important to note that preconditioning is protective against ischemia–reperfusion injury only if there is a limited period of ischemia followed by reperfusion. The original study by Murry et al 21 describing the protective effect of ischemic preconditioning in dogs showed a significant reduction in infarct size after 90 minutes of ischemia. However, the protective effect of ischemic preconditioning was lost when the ischemic period was prolonged to 3 hours. 21 Thus, the study done by Sauvé et al 17 suggests that DPP-IV inhibitors may not alter remodeling postinfarction but do not address the issue of protection against ischemia–reperfusion injury.
There is one study reporting the effect of DPP-IV inhibition as postconditioning therapy. Theiss et al 15 induced MI in nondiabetic mice by surgical occlusion of the LAD and immediately began treatment with G-CSF and/or oral sitagliptin. At 28 days post-MI, infarct size was significantly reduced in mice that received sitagliptin or sitagliptin with G-CSF. Sitagliptin therapy led to enhanced cardiac homing of stem cells and neovascularization, decreased cardiac remodeling, and reduced overall mortality. However, it is unclear how drugs can reach the infarct zone when the artery is occluded.
Mechanism of Action
Dipeptidyl peptidase IV is expressed on the surface of most cell types. The DPP-IV converts the endogenous active GLP-1 [GLP-1 (7-36)] form to an inactive form [GLP-1 (9-36)]. It also deactivates glucagon-like peptide 2 (GLP-2) and GIP. 22 The DPP-IV inhibitors, thus, augment the effects of endogenous incretins by preventing their degradation (Figure 1). Activation of the GLP-1 receptor leads to generation of cAMP with downstream activation of protein kinase A (PKA). Protein kinase A leads to the downstream activation of cAMP response element binding (CREB) protein, Akt, extracellular signal-related kinase 1/2, 5-lipoxygenase, and endothelial nitric oxide synthase (eNOS). 14,23,24 The CREB is a nuclear transcription factor that acts as a positive regulator of Bcl-2, which reduces apoptosis. 14,25 The GLP-1 (7-36) also has anti-inflammatory effects that may contribute to its anti-ischemic properties. 26 The phosphorylation of 5-lipoxygenase by PKA leads to the production of the anti-inflammatory eicosanoid 15-epi-lipoxin A4. 10 PKA also mediates normalization of PTEN and reduction in structural protein degradation via deactivation of cytoskeletal actin, Rho kinase, and caplain. 10,11 The GLP-1 (9-36) may also protect against ischemia in a GLP-1 receptor-independent way. 27 However, as DPP-IV inhibitors prevent the formation of GLP-1 (9-36), these effects may be irrelevant. The cardioprotective kinases protein kinase C, adenosine monophosphate-activated protein kinase, and phosphoinositide 3-kinase are activated by the GLP-1 receptor as well. 6,23 However, DPP-IV inhibitors appear to have potential GLP-1 receptor-independent cardioprotective, antiapoptotic, and anti-inflammatory effects (Figure 1). 10,11,14,18 Aside from GLP-1, DPP-IV also degrades several other cytokines and chemokines, 6,7 and the significance of increasing their levels by DPP-IV inhibition is unknown. The effects of GIP and GLP-2 on ischemia–reperfusion injury have not been reported. The GLP-1 receptor-independent effects may occur via prevention of degradation of other non-GLP-1 substrates such as B-type natriuretic peptide, substance P, neuropeptide Y, peptide YY, bradykinin, and stromal-derived factor 1α (SDF-1α). 22 The SDF-1α increases circulating levels of endothelial progenitor cells and may contributes to recovery of function after infarction. 22,28 The role of these substances in protecting against ischemia–reperfusion injury and mediating the effects of DPP-IV inhibitors has not been clarified. It has been suggested that protection against ischemia–reperfusion injury occurs via progenitor cell mobilization, neovascularization and vasodilation, and increased levels of cardioactive peptides. 6,7 Studies have also suggested that DPP-IV inhibitors protect cardiac mitochondria against oxidative stress induced by reducing reactive oxygen species production and mitochondrial membrane potential changes that occur during ischemia–reperfusion. 16
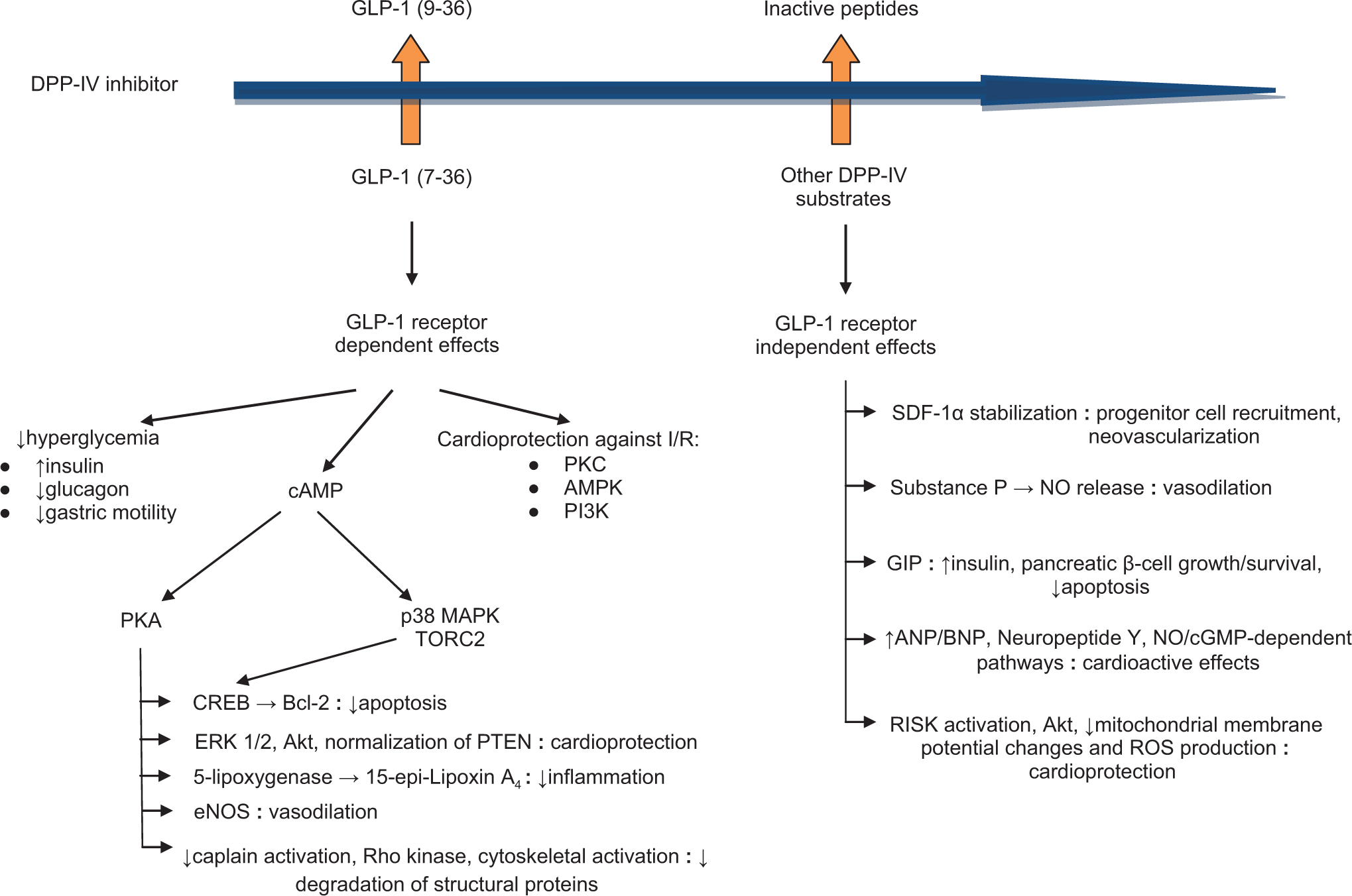
Protective effects and mechanisms of signaling action of glucagon-like peptide 1 (GLP-1) and dipeptidyl peptidase IV (DPP-IV) inhibitors on ischemia/reperfusion (I/R) injury based on the literature. PKC indicates protein kinase C; ERK1/2, extracellular signal-related kinase 1/2; AMPK, adenosine monophosphate-activated protein kinase; PI3K, phosphoinositide 3-kinase; cAMP, cyclic adenosine monophosphate; PKA, protein kinase A; p38 MAPK, p38 mitogen-activated protein kinase; TORC2, transducer of regulated CREB activity 2; CREB, cAMP response element binding protein; eNOS, endothelial nitric oxide synthase; SDF-1α, stromal-derived factor 1α; NO, nitric oxide; GIP, glucose-dependent insulinotropic peptide; ANP/BNP, atrial natriuretic peptide and brain natriuretic peptide; cGMP, cyclic guanosine monophosphate; RISK, reperfusion-induced salvage kinase; PTEN, phosphatase and tensin homolog; ROS, reactive oxygen species.
Dipeptidyl Peptidase IV Inhibitors in Clinical Trials
In December 2008, the Food and Drug Administration and European Medicines Agency mandated that all new antidiabetic medications must demonstrate long-term cardiovascular safety. Two clinical trials have been published evaluating the effect of exenatide, a GLP-1 analog, on infarct size. Woo et al 13 found that exenatide given subcutaneously at currently approved doses decreased infarct size by approximately 40% in patients with ST-segment elevation MI (STEMI) when measured by cardiac magnetic resonance imaging 1 month after percutaneous coronary intervention (PCI). Intravenous exenatide infusion reduced final infarct size by about 30% in patients with STEMI who underwent primary PCI and had short systemic delay (defined as ≤132 minutes). 12 No clinical trials have been published assessing the effects of DPP-IV inhibitors on infarct size, however. In the clinical setting, oral administration of a drug before reperfusion therapy for STEMI may not reach maximal protective effect and parenteral administration of GLP-1 receptor agonists may have an advantage. Yet oral DPP-IV inhibitors, alone or in combination with other drugs that have been shown to protect against ischemia–reperfusion injury (ie, statins), 29 –31 can potentially be used for preconditioning of the heart and other organs before elective procedures, such as PCIs or surgery.
It should be noted, however, that numerous agents and interventions that have been shown to effectively reduce myocardial infarct size, especially in rodents, failed to do so in the clinical setting. The limitations of translating results from small animal models to the clinical setting have been discussed in detail at the UCL-Hatter Cardiovascular Institute 6th International Cardioprotection Workshop 32 and by the Working Group of Cellular Biology of the Heart of the European Society of Cardiology. 33 The response of the heart to ischemia–reperfusion insult varies among species and with age. Most patients with coronary artery disease are older and have coronary artery disease. Moreover, in the clinical setting, patients receive several drugs and the final effects of the combination of these agents may differ from each drug alone. For example, statins reduce myocardial infarct size and are commonly used in patients with coronary artery disease. It is plausible that the protective effect of DPP-4 inhibitors could be masked by concomitant use of statins.
The Examination of Cardiovascular Outcomes with Alogliptin versus Standard of Care (EXAMINE) 34 and Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus—Thrombolysis in Myocardial Infarction 53 (SAVOR-TIMI 53) 35 trials published in 2013 address the safety and efficacy of DPP-IV inhibitors in reduction in cardiovascular events (Table 3). The EXAMINE 34 and SAVOR-TIMI 5335 trials were large, prospective, randomized, and placebo-controlled trials including 5380 patients with a median follow-up period of 18 months and 16 492 patients with a median follow-up period of 2.1 years, respectively. They enrolled type 2 diabetic patients with high cardiovascular risk. The DPP-IV inhibitors were given in addition to standard-of-care treatment for diabetes and cardiovascular risk factors and/or disease. In fact, glycemic control was statistically better in the DPP-IV groups. For both studies, no differences were observed between the DPP-IV inhibitor and placebo groups in the primary outcome, which was defined as composite of death from cardiovascular causes, nonfatal MI, nonfatal stroke, or overall mortality from any cause. The number of adverse events due to alogliptin was similar compared to placebo in the EXAMINE 34 trial. However, more hypoglycemic events (P < .001) and hospitalizations for heart failure (P = .07) were noted with saxagliptin in the SAVOR-TIMI 53 trial. 35 The Cardiovascular Outcome Study of the DPP-IV Inhibitor Linagliptin (CAROLINA) 36 and Trial Evaluating Cardiovascular Outcomes with Sitagliptin (TECOS) 37 are currently ongoing and will help confirm the results of the EXAMINE 34 and SAVOR-TIMI 5335 trials. No studies have been published on the long-term effects of direct GLP-1 agonists on cardiovascular outcomes.
Clinical Trials Involving DPP-IV Inhibitors.

Abbreviations: ACS, acute coronary syndrome; CAROLINA, Cardiovascular Outcome Study of the DPP-IV Inhibitor Linagliptin; CV, cardiovascular; DPP-IV, dipeptidyl peptidase IV; EXAMINE, Examination of Cardiovascular Outcomes with Alogliptin versus Standard of Care; HgbA1c, hemoglobin A1c; MI, myocardial infarction; SAVOR-TIMI 53, Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus—Thrombolysis in Myocardial Infarction 53; TECOS, Trial Evaluating Cardiovascular Outcomes with Sitagliptin; T2DM, type 2 diabetes mellitus; UA, unstable angina.
One must keep in mind, however, that the end point of infarct size used in animal studies is not equivalent to the end point of long-term cardiovascular outcomes used in the clinical trials studying DPP-IV inhibitors. Aside from GLP-1, DPP-IV also degrades several other cytokines and chemokines. 6,7 Taken together, these substances can affect a variety of body systems and inhibition of their degradation may lead to results that are ultimately different or potentially deleterious compared to that found with GLP-1 receptor activation alone with GLP-1 receptor agonists. Furthermore, the levels of GLP-1 achieved with DPP-IV inhibitors may not be enough to produce a clinically significant effect on protection against a robust insult of ischemia–reperfusion injury. There is also the possibility of tolerance after prolonged treatment in humans as has been suggested for statins 19,38 although the protective effects were not lost after 14 days of pretreatment in the nondiabetic mice. 14 In addition, in the clinical setting, patients often take a combination of other drugs such as statins, angiotensin-converting enzyme (ACE) inhibitors, or angiotensin receptor blockers, which may also provide protection against ischemia–reperfusion injury. 29 –31,39,40 It is possible that any beneficial effect of DPP-IV inhibitors is obscured by the effects of these other drugs. However, the potential remains to use drug combinations involving statins, ACE inhibitors, cilostazol, among others, to augment the cardioprotection in high-risk patients. The EXAMINE 34 and SAVOR-TIMI 5335 trials also had a relatively short duration of follow-up. A longer duration of follow-up may be required to demonstrate any benefit in terms of macrovascular complications. Further studies will need to be done to confirm the results of these trials and to determine whether there is any effects on myocardial infarct size.
Conclusion
Diabetes mellitus is a major risk factor for cardiovascular events and patient death. The myocardial protection afforded by DPP-IV inhibitors in preclinical animal studies poses a potential breakthrough role for antidiabetic medications in attenuation of ischemia–reperfusion injury that occurs with cardiovascular disease. Although some studies in animal models suggest DPP-IV inhibitors can decrease infarct size, several clinical trials are currently underway to ascertain whether they reduce macrovascular outcomes in high cardiovascular risk, type 2 diabetic patients. Thus far, no differences have been observed between the DPP-IV inhibitor and placebo groups in cardiovascular events or overall mortality from any cause. No studies have yet been conducted addressing the effect of DPP-IV inhibitors on infarct size. Further experiments and clinical trials will need to be performed to confirm the results of the studies reviewed here.
Footnotes
Authors’ Note
Alyssa H. Yoon collected the data and wrote the manuscript. Yumei Ye reviewed and edited the manuscript and contributed to original data. Yochai Birnbaum mentored Dr Yoon in writing the manuscript and reviewed and edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Research grants from Merck, Amylin, and BMS.
