Abstract
Objective:
Although preconditioning remains one of the most powerful maneuvers to reduce myocardial infarct size, it is not feasible in the clinical setting to pretreat patients prior to acute myocardial infarction (MI). The purpose of this study was to investigate the effect of more clinically relevant therapies of remote perconditioning, postconditioning, and the combined effect of remote perconditioning and postconditioning on myocardial infarct size in an anesthetized rat model.
Methods:
Anesthetized rats were subjected to 45 minutes of proximal left coronary artery occlusion followed by 2 hours of reperfusion. Remote perconditioning was performed 5 minutes after left coronary occlusion with 4 cycles of 5 minutes of occlusion and reperfusion of both the femoral arteries. Postconditioning was applied immediately prior to 2 hours of full reperfusion with 6 cycles of 10 seconds occlusion–reperfusion of the coronary artery. The combined effect was produced by preceding the postconditioning regimen with remote perconditioning, after 5 minutes of left coronary occlusion.
Results:
Remote perconditioning and postconditioning alone failed to reduce infarct size expressed as percentage of the risk zone (42.2% ± 3.9% and 45.0% ± 4.3%). The combination of remote perconditioning and postconditioning also failed to reduce infarct size (45.3% ± 4.1%) as compared to the untreated ischemia–reperfusion group (48.7% ± 3.4%). Hemodynamics including left ventricular end-systole and end-diastolic pressures, +dP/dt, −dP/dt, and heart rate did not show any improvement in the conditioning groups.
Conclusion:
This study shows that remote perconditioning and postconditioning alone or combined neither improve hemodynamics nor reduce infarct size in the rat model of MI.
Introduction
After ischemic injury, reperfusion salvages the myocardium but full-flow reperfusion immediately after ischemia has been postulated to cause a degree of reperfusion injury. Conditioning of the myocardium against ischemia–reperfusion increases cell survival. The most well studied and standard conditioning intervention is preconditioning, in which the brief periods of ischemia–reperfusion of the coronary artery makes the myocardium resistant to more prolonged ischemic injury and subsequent reperfusion. The applicability of preconditioning is limited to clinical trials involving elective procedures such as percutaneous coronary intervention (PCI) and coronary artery bypass grafting in which the timing of the onset of ischemic injury is known, as it is not possible to predict the time of an acute myocardial infarction (MI). Conditioning protocols that may be more relevant to clinical practice include postconditioning and perconditioning. With postconditioning, the culprit coronary artery is subjected to brief cycles of occlusion and reperfusion after the long period of ischemia. However, postconditioning has not been shown to reduce myocardial infarct size in every model studied. With remote perconditioning, the conditioning stimulus is applied to a more accessible organ such as the arm or lower limb during the MI. Brief episodes of ischemia and reperfusion to the remote organ protect the heart by a paracrine or neural-reflex mechanism and does not induce any additional stress on the heart itself. It is also a more feasible form of conditioning, which does not require medical facilities and expertise. For example, remote conditioning in humans has been achieved by simply inflating and deflating a blood pressure cuff on the arm.
Previously, we have shown that preconditioning reduced myocardial infarct size in our rat model of coronary occlusion and reperfusion; however, whether remote perconditioning or combination of remote perconditioning and postconditioning could reduce infarct size was unknown. Therefore, the 2 main objectives of conducting this study were (1) to determine whether remote perconditioning reduces infarct size in the rat. As previous experiments in our laboratory have shown that postconditioning alone does not reduce infarct size, our second objective was to determine whether (2) postconditioning combined with remote perconditioning would reduce infarct size and improve hemodynamics.
Methods
The study was reviewed and approved by the Institutional Animal Care and Use Committee of Good Samaritan Hospital, and the protocol conformed with the Guide for the Care and Use of Laboratory Animals published by the National Research Council (Eighth Edition, 2011).
Animals
A total of 129 animals were randomized into 4 different groups. Animals (n = 40) with failed occlusion of the left coronary artery, inadequate infarct size, poor demarcation of the risk area, and inadvertent preconditioning were excluded from the study. The data of 89 rats divided into the following groups: 22 in the ischemia–reperfusion group, 21 in the postconditioning group, 23 in the remote perconditioning group, and 23 in the combined remote perconditioning + postconditioning group were analyzed.
Surgical Preparation
Female Sprague-Dawley rats weighing 200 to 250 kg were anesthetized with intraperitoneal injection of ketamine (75 mg/kg) and xylazine (5 mg/kg), intubated, and ventilated with room air at 60 strokes/min. The animal ventilator was set to 1.5 mL/100 g tidal volume. The body temperature was maintained at 37°C with a heating pad. The neck, left side of the chest, and hind limbs were shaved and cleaned with ethanol. A midline neck incision was made, and the left jugular vein was cannulated for administration of Unisperse blue dye and KCl. The carotid artery was isolated, and a catheter was advanced to the left ventricle for recording the left ventricular (LV) pressure during the experimental protocol. The LV end-systolic pressure (ESP) and end-diastolic pressure (EDP), +dP/dt, −dP/dt, and heart rate (HR) were monitored and recorded at baseline, 45 minutes of ischemia, and 2 hours of reperfusion in all the experimental groups. A left thoracotomy was performed through the fourth intercostal space after injecting bupivacaine hydrochloride (0.1 mg/kg) for regional analgesia. The ribs were retracted to expose the heart, and the pericardium was removed. A 4-0 suture was passed beneath the left coronary artery, 3 to 4 mm from its origin and threaded through a small tube to form a snare. The proximal left coronary artery was occluded by tightening and clamping the snare and reperfused by releasing it. In the control ischemia–reperfusion group, the left coronary artery was occluded for 45 minutes and subsequently reperfused for 2 hours. In this study, 3 different conditioning interventions were evaluated in comparison to the ischemia–reperfusion group. For remote perconditioning, a cut down was performed below the inguinal ligament to expose the femoral artery on both sides. The femoral artery was occluded and reperfused by clamping and releasing the bulldog clamp placed on the artery.
Conditioning Protocols
In the postconditioning group, prior to the beginning of the sustained 2 hours reperfusion, the proximal left coronary artery was subjected to 6 cycles of 10 seconds reperfusion followed by 10 seconds of occlusion. In the remote perconditioning group, conditioning was commenced after 5 minutes of coronary occlusion. In addition to the index ischemia of the left coronary artery, both femoral arteries were subjected to 4 cycles of 5 minutes occlusion and 5 minutes of reperfusion. In the combined remote perconditioning plus postconditioning group, both femoral arteries were subjected to 4 cycles of 5 minutes occlusion–reperfusion after 5 minutes of left coronary artery occlusion, followed by 6 cycles of 10 seconds reperfusion-occlusion of the coronary artery just before the 2 hours sustained reperfusion.
Analysis of Risk Zone and Infarct Size
After the completion of 2 hours of reperfusion, the proximal left coronary artery was retied and blue dye was injected via the jugular vein to demarcate the area at risk (area not perfused with blue dye). Animals were euthanized under anesthesia with KCl to arrest the heart in the diastolic state. The heart was excised and cut into 4 transverse sections. The heart slices were incubated into 1% 2,3,5-triphenyltetrazolium at 37°C for 15 minutes and then dipped in 10% formalin to delineate the area of necrosis (pale area). The area at risk and area of necrosis were photographed and quantified using Image J software. The area at risk and area of necrosis for each heart were calculated by summing the product of percentage of risk or necrotic zone and weight of the slice and then dividing it by the sum of weight of all the slices.
Statistical Analyses
Data were expressed as mean ± standard error of the mean. Statistical analysis was performed using SPSS Software version 20. Hemodynamics, area at risk, and area of necrosis were analyzed using 1-way analysis of variance with Tukey post hoc test. The P value <.05 was considered as significant.
Results
The numbers of animals in each group with successful experiments used to analyze the results were 22 in the untreated ischemia–reperfusion group, 21 in the postconditioning group, 23 in the remote perconditioning group, and 23 in the combined remote perconditioning plus postconditioning group. In the remote perconditioning group, 1 heart was excluded due to poor perfusion of blue dye as a result of low arterial pressure at the end of reperfusion. In this group, 22 animals were considered for the quantification of area at risk and area of necrosis.
Area of Risk and Infarct Size
The area at risk was 46.6% ± 2.4% of the left ventricle in the ischemia–reperfusion group, 46.3% ± 2.4% in the postconditioning, 48.8% ± 2.3% in the remote perconditioning, and 48.6% ± 2.3% in the combined remote perconditioning + postconditioning group (P = not significant among the groups; Figures 1 and 2). Similar area at risk in all the groups confirmed that the anatomic extent of ischemia was similar among the 4 groups. The infarct size expressed as percentage of area of necrosis/area at risk was 48.7% ± 3.4% in the ischemia–reperfusion group, 45.0% ± 4.3% in the postconditioning, 42.2% ± 3.9% in the remote perconditioning, and 45.3% ± 4.1% in the combined remote perconditioning + postconditioning group (Figures 1 and 3). There was no significant reduction in the infarct size in any of the 3 conditioning groups as compared to the untreated ischemia–reperfusion group, indicating failure of these conditioning interventions to salvage the myocardium from ischemic injury, in this anesthetized rat model.

Transversely cut sections of heart showing area at risk and area of necrosis of a representative rat from the (A and B) ischemia–reperfusion group, (C and D) postconditioning group, (E and F) remote perconditioning group, and (G and H) combined remote perconditioning + postconditioning group. All the animals were subjected to 45 minutes ischemia followed by 2 hours of reperfusion along with the respective conditioning intervention. After 2 hours of reperfusion, the left coronary artery was retied and Unisperse blue dye was injected through the jugular vein. Hearts were excised and sliced from apex to base, photographed and incubated in triphenyl tetrazolium chloride (TTC), and rephotographed. The viable myocardial cells absorb TTC stain and turn brick-red in color, whereas nonviable cells appear white. (A, C, E, and G) represent area at risk (ischemia zone or nonblue which was not perfused with blue dye) and (B, D, F, and H) represent infarct area (white) in the same heart slices after incubation in TTC. Both area at risk and area of necrosis were similar in all the groups.
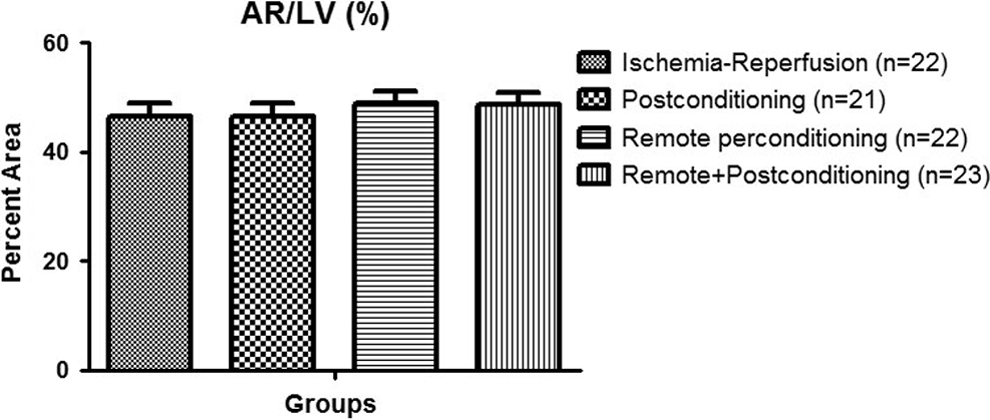
Bar graph showing the area at risk as the percentage of the left ventricular area in all the groups.
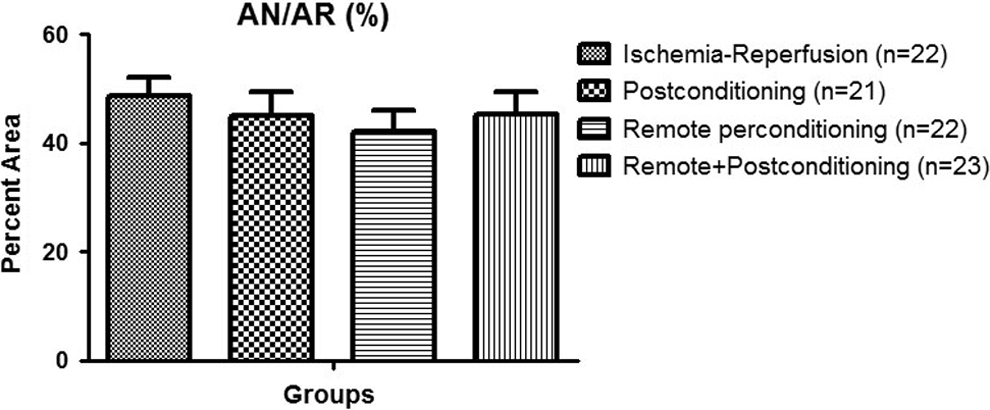
Bar graph showing the area of necrosis as the percentage of the risk area in all the groups.
There was a correlation between area of necrosis and area at risk in all the groups (Figure 4). The coefficient of determination, that is, the r 2 value explains the proportion of variability in area of necrosis accounted for or described by area at risk. The “r 2” value was .48 (P < .0003) in the ischemia–reperfusion group, .14 (P < .09) in the postconditioning, .26 (P < .01) in the remote perconditioning, and .19 (P < .03) in the combined remote perconditioning + postconditioning group. The group per se had no effect on the relationship between area of necrosis and area at risk by ANCOVA (Figure 5).
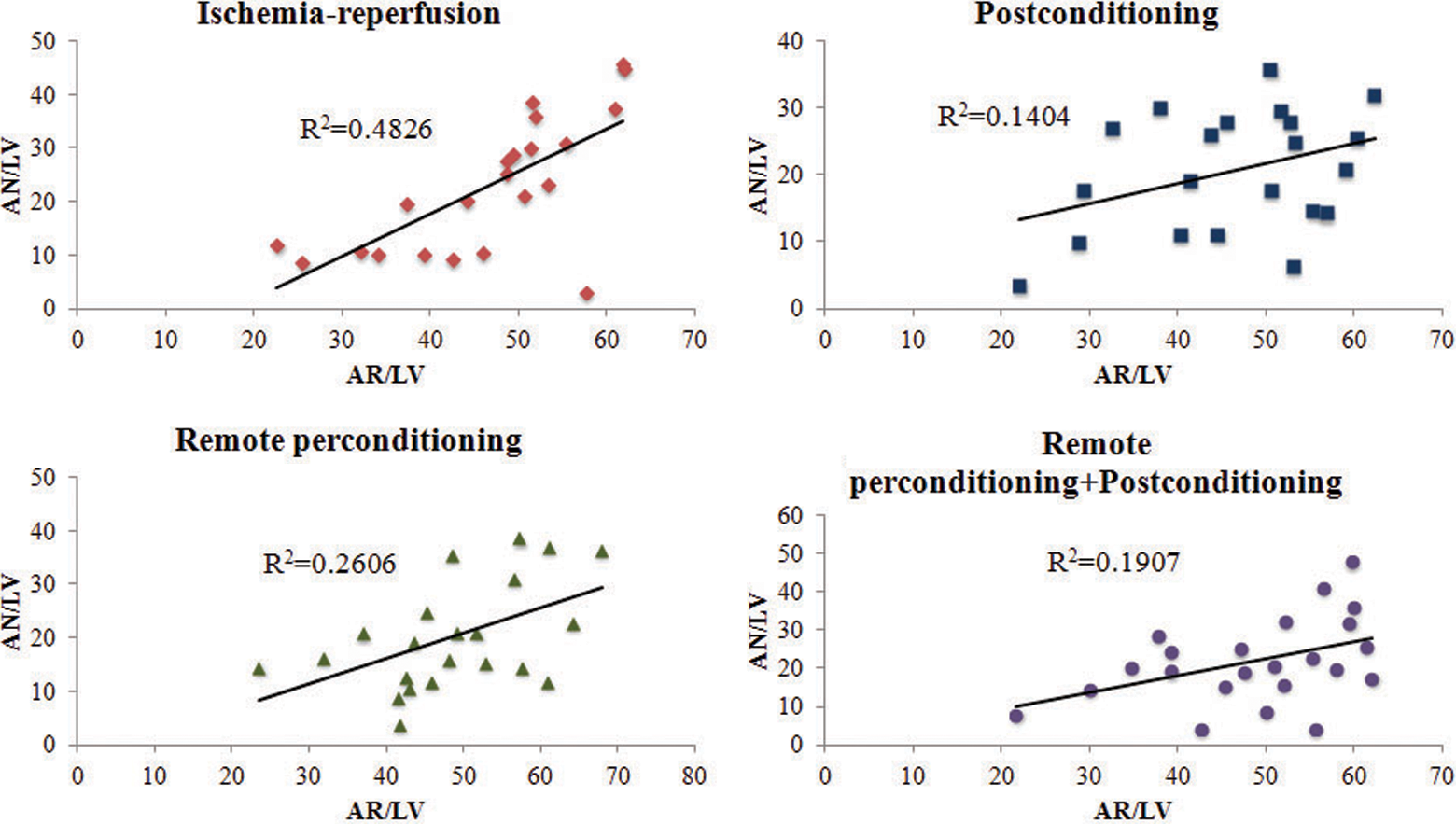
Scatterplot showing the relationship between area of necrosis and area at risk in all the groups.

Scatterplot showing the effect of conditioning intervention on the relationship between area of necrosis and area at risk in all the groups.
Hemodynamics
There was a slight decrease in HR after 45 minutes of ischemia but it significantly increased by the end of 2 hours reperfusion as compared to baseline and end of ischemia in all the groups (Table 1). There was no significant difference in HR among the groups. We did not observe any change in LVESP at different time points in any of the groups. The LVEDP increased after 45 minutes of ischemia but it failed to reach significance except in the combined remote perconditioning + postconditioning group. Slight decrease in LVEDP after 2 hours reperfusion was observed compared to 45 minutes ischemia, except it remained significantly elevated in the remote perconditioning group. There was a decline in + dP/dt after 45 minutes ischemia, which continued even after 2 hours reperfusion in all the groups; statistical significance was observed in postconditioning and remote perconditioning groups. There was a marked fall in −dP/dt after 45 minutes of ischemia as compared to baseline in all the groups. The values slightly increased after 2 hours reperfusion but failed to reach significance in all the groups. No significant difference in any of the hemodynamic values at any time point among the groups was observed (Table 1).
Hemodynamic Parameters at Baseline, 45 minutes After Ischemia, and 2 Hours After Reperfusion in All the Groups.

Abbreviations: EDP, end-diastolic pressure; ESP, end-systolic pressure. Data are expressed as mean ± SEM. a p < 0.05, b p < 0.01, c p < 0.001 vs baseline; d p = 0.06 vs postconditiong; e p < 0.01, f p < 0.001 vs 45 min after Ischemia.
Discussion
In this study, we have demonstrated that remote perconditioning performed during the 45 minutes index ischemia did not reduce infarct size or improve hemodynamics in an anesthesized rat myocardial infarct model. In accordance with the previous studies done in our laboratory, postconditioning also failed to show any improvement in either of these endpoints. Moreover, combining remote perconditioning with postconditioning also did not show any synergetic effect in terms of infarct size reduction or improvement in cardiac hemodynamics.
Remote preconditioning was demonstrated for the first time by Przyklenk et al in 1993 with 4 cycles of 5 minutes occlusion and 5 minutes of reperfusion of the left circumflex artery that reduced myocardial infarct size in the left anterior descending (LAD) bed from 16% ± 5% to 6% ± 2% of the risk region in dogs. 1 Subsequent studies have demonstrated that this infarct size reducing effect occurs even when other organs such as brain, kidney, intestine, and skeletal muscle were used for remote preconditioning. Birnbaum et al showed a 65% reduction in infarct size when the preconditioning stimulus was applied by partial stenosis of the femoral artery with concomitant electrical stimulation of the gastrocnemius muscle for 30 minutes followed by 30 minutes ligation of the left coronary artery and 4 hours reperfusion in rabbits. 2 But like ischemic preconditioning, remote preconditioning is also limited to preclinical studies and elective surgical interventions such as cardiac surgery and PCI where time and duration of ischemia–reperfusion injury are known. On the other hand, MI is an unpredictable event; therefore, there is a need to explore modifications in timing and duration of conditioning interventions. Schmidt et al reported in the closed chest porcine model that 4 cycles of 5 minutes of occlusion and 5 minutes of reflow when applied to 1 hindlimb during 40 minutes of myocardial ischemia and before the onset of 120 minutes reperfusion reduced the infarct size from 60% ± 5% to 38% ± 5%, preserved LV function, and reduced malignant rhythm disturbances. As pigs have negligible coronary collateral blood flow, they suggested that a circulating effector exerts its protective effect during reperfusion. They also speculated that perconditioning modifies the reperfusion-induced calcium overload. 3 In another study, the same perconditioning regimen of four 5 minutes cycles of lower limb ischemia–reperfusion applied during 60 minutes index ischemia and 1 minute before 180 minutes myocardial reperfusion showed reduction in infarct size to 18% ± 3% from 44% ± 3% in pigs. 4 In a proof-of-concept clinical trial, Bøtker et al demonstrated that 4 cycles of 5 minutes inflation and deflation to the arm using a blood pressure cuff during the ongoing ischemia administered in the ambulance reduces the infarct size in patients with ST-segment elevation MI receiving primary PCI. 5 Subsequent studies confirmed the cardioprotective potential of remote perconditioning on the basis of different cardiac injury markers. Reduction in troponin levels was observed when a conditioning stimulus was applied by inflation and deflation of a cuff at 600 mm Hg placed at the leg for 3 × 4 minutes. 6 Rentoukas et al also showed that inflation of a blood pressure cuff at 200 mm Hg placed around the arm for 3 cycles of 5 minutes each significantly resolved the ST-segment elevation in patients undergoing primary PCI. 7,8 Therefore, remote perconditioning seems to be a more clinically relevant, safe, and feasible intervention during the MI. We also used the same protocol of 4 cycles of 5 minutes occlusion and 5 minutes reperfusion of both femoral arteries 5 minutes after the onset of index ischemia. However, it failed to reduce infarct size between control and remote perconditioning rats in our experimental model.
Postconditioning is another conditioning intervention that is clinically feasible in patients with MI. Zhao et al reported for the first time that before the sustained reperfusion, brief repetitive occlusion of the LAD coronary artery significantly attenuated the infarct size in dogs. They demonstrated that 3 cycles of 30 seconds reperfusion followed by 30 seconds occlusion before sustained 3 hours reperfusion attenuated infarct size, tissue edema, and neutrophil accumulation. 9 Furthermore, it has been shown that the time window when the postconditioning stimulus is applied at the onset of reperfusion is critical for cardioprotection. Kin et al reported that the infarct limiting effect was comparable when 2 postconditioning regimens, that is, 3 cycles or 6 cycles of 10 seconds reperfusion and 10 seconds occlusion of the left coronary artery were applied before 3 hours reperfusion in rats. In contrast, a delay of 1 minute before commencing the 3-cycle regimen completely abrogated the protective response. In the same study, the postconditioning stimulus could not limit infarct size comparable to preconditioning but it was significantly reduced as compared to control. 10 Staat et al reported on the basis of proof of concept trial that postconditioning regimen of 4 cycles of 1 minute inflation each separated by 1 minute deflation applied using angioplasty balloon reduced infarct size by 36% and improved myocardial perfusion. 11
Several studies have shown that postconditioning reduces ventricular arrhythmias in the rat model of proximal left coronary artery occlusion and reperfusion without significant reduction in infarct size and recovery of cardiac function. 12 –14 In our previous study, a postconditioning regimen of 4 cycles of 20 seconds reperfusion/20 seconds occlusion, which ablated the arrhythmias, failed to reduce the infarct size when rats were subjected to 45 minutes ischemia and 2 hours reperfusion. In the same study, postconditioning applied by 4 cycles of 10 seconds occlusion and 10 seconds reperfusion or 20 seconds occlusion and 20 seconds reperfusion after both 30 and 45 minutes of ischemia also did not show any reduction in infarct size. 15 Therefore, we tested 6 cycles of 10 seconds occlusion and 10 seconds reperfusion regimen after 45 minutes ischemia and before 2 hours reperfusion. In line with our previous studies, we have found that postconditioning did not show any reduction in infarct size in the rat model. A recent study has shown that remote perconditioning when combined with postconditioning enhanced the cardioprotective effect of postconditioning applied after 45 minutes ischemia and before 2 hours reperfusion in the rat model. However, appropriate number and duration of cycles are required to observe the desired effect. In this study, they applied the perconditioning in a dose-dependent fashion; the optimum effect was observed when 4 cycles of 5 minutes occlusion and intermittent reperfusion were applied using a femoral artery cutdown approach during the late phase of ischemia followed by 6 cycles of 10 seconds occlusion and reperfusion of the left coronary artery (2 minutes intervention without any delay). 16 We investigated the effect of combined postconditioning and remote perconditioning on infarct size using the same bilateral femoral artery cutdown approach and number and duration of cycles. In contrast, in our study, there was no significant reduction in infarct size. In addition, it has also been reported that combining remote perconditioning with postconditioning improves LV pressure and volume. 3,16 In contrast, we did not observe any stabilizing effect of conditioning stimulus alone or in combination on cardiac hemodynamics. In fact, we have observed the progressive increase in HR during the ischemia–reperfusion duration in all the groups as compared to baseline. This increased HR could also contribute to worsening ischemia as it increases the myocardial oxygen consumption per minute and shortens the duration of diastole reducing coronary blood flow per single cycle. 17 In addition, decrease in −dP/dt represents delay in isovolumic ventricular relaxation and also contributes to reduced coronary blood flow.
Why were the results of our study negative while other reports in the literature have suggested that postconditioning and perconditioning may be effective? Many previous conditioning studies have shown positive effects in the rat model, in which the sample size has been relatively small (n value of approximately or less than 5). In our study, we utilized >20 rats in each group. Problems with groups of small numbers of animals are that 1 or 2 outliers can give false positive or negative results; 1 or 2 missed coronary occlusions can result in a smaller infarct size mean for the group; 1 or 2 rats with coronary anatomy leading to small infarcts or higher collateral flow in the risk zone can lead to a smaller infarct size mean for the group. A larger number of rats per group lessen the chances of such spurious results. In addition, there may be species difference in the effectiveness of postconditioning and remote perconditioning. It is entirely possible that large animal species may be more amenable to these conditioning procedures resulting in positive effects on infarct size. For example, while postconditioning was shown to reduce infarct size in the dog, it was not effective in our laboratory in smaller animals such as rat or rabbit. 9,15,18 Even in 1 study in which postconditioning was effective in the rat, its absolute reduction in infarct size was small. 14 Perhaps the higher metabolic rate of small rodents such as the rats makes this species harder to postcondition.
Conclusion
In this study, we investigated both remote perconditioning and postconditioning alone or in combination and observed that they failed to attenuate infarct size in an anesthetized rat model of MI. In addition, both conditioning interventions have neither shown attenuation nor recovery of LV dysfunction induced by ischemia–reperfusion injury.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
