Abstract
Background and Purpose:
Cardioprotection against ischemia–reperfusion (I/R) damages remains a major concern during prehospital management of acute myocardial infarction. Noble gases have shown beneficial effects in preconditioning studies. Because emergency proceedings in the context of myocardial infarction require postconditioning strategies, we evaluated the effects of argon in such protocols on mammalian cardiac tissue.
Experimental Approaches:
In rat, cardiac I/R was induced in vivo by transient coronary artery ligature and cardiac functions were evaluated by magnetic resonance imaging. Hypoxia–reoxygenation (H/R)-induced arrhythmias were evaluated in vitro using intracellular microelectrodes on both rat-isolated ventricle and a model of border zone in guinea pig ventricle. Hypoxia–reoxygenation loss of contractile force was assessed in human atrial appendages. In those models, postconditioning was induced by 5 minutes application of argon at the time of reperfusion.
Key Results:
In the in vivo model, I/R produced left ventricular ejection fraction decrease (24%) and wall motion score increase (36%) which was prevented when argon was applied in postconditioning. In vitro, argon postconditioning abolished H/R-induced arrhythmias such as early after depolarizations, conduction blocks, and reentries. Recovery of contractile force in human atrial appendages after H/R was enhanced in the argon group, increasing from 51% ± 2% in the nonconditioned group to 83% ± 7% in the argon-treated group (P < .001). This effect of argon was abolished in the presence of wortmannin and PD98059 which inhibit prosurvival phosphatidylinositol-3-kinase (PI3K)/protein kinase B (Akt) and MEK/extracellular receptor kinase 1/2 (ERK 1/2), respectively, or in the presence of the mitochondrial permeability transition pore opener atractyloside, suggesting the involvement of the reperfusion injury salvage kinase pathway.
Conclusion and Implications:
Argon has strong cardioprotective properties when applied in conditions of postconditioning and thus appears as a potential therapeutic tool in I/R situations.
Introduction
In patients with acute myocardial infarction, the restoration of coronary flow using a combination of percutaneous interventions and pharmacological approaches limits infarct size. However, reperfusion also contributes to myocyte cell death and to the occurrence of ventricular arrhythmias, contractile, and microvascular dysfunction subsequent to opening of the mitochondrial permeability transition pore (MPTP). 1 In this setting, ischemic and pharmacological postconditioning strategies were proposed to reduce infarct size. Anesthetic gases activate cardioprotective kinase pathways 2,3 but are only suitable in surgical situations.
Several noble gases have cardioprotective effects under experimental conditions. 4 -6 Helium exerts a cardioprotective effect not only in preconditioning in rabbit and rat 7,8 through an activation of the reperfusion injury salvage kinase (RISK) pathway 9 but also in in vivo postconditioning proceedings in rat. 10,11 However, helium conditioning is abolished in aged rats 7 as well as in diabetic Zucker obese rats. 12 Xenon applied in preconditioning reduces infarct size in rat and rabbit 9,13 but not in pig. 14 Cardioprotection also occurs when xenon is applied at the early stages of reperfusion in rabbit 15 or in postconditioning when it is combined with hypothermia in rat. 16 Nonetheless, xenon was shown to inhibit tissue plasminogen activator, leading to potentially deleterious effect in a rat model of thromboembolic stroke. 17 Moreover, its anesthetic properties under normobaric conditions prevent its use in the context of acute myocardial infarction. 18
Among noble gases, argon is much more abundant in the atmosphere than xenon, has no anesthetic effects at normal atmospheric pressure, 4 is well tolerated, and at high concentration (75%), does not inhibit thrombolytic efficiency of tissue plasminogen activator. 19 Previous results suggest a cardioprotective effect of argon applied in preconditioning conditions in rabbit and rat cardiomyocytes in culture. 9,20 In other experimental issues, argon exerts neuroprotective effects in cerebral ischemia as well. 5,21 -24
The aim of the present study was to measure the effect of postconditioning by argon in the context of ischemia–reperfusion (I/R) in mammalian heart, including human tissue, since data are lacking in this context. We have explored its effect on both contractile and electrical properties and have observed that postconditioning by argon is beneficial.
Methods
Study Approval and Animal Studies
All experimental procedures and protocols used in this investigation were reviewed and approved by our regional ethics committee for animal experiment and performed according to European Commission Directive 2010/63/EU. Studies involving animals are reported according to animal research: reporting of in vivo experiments (ARRIVE) guidelines. Animals were housed in conventional cages with a 12-hour/12-hour light/dark cycle with free access to food and water. A total of 73 rats and 13 guinea pigs were used.
Studies using human atrial appendages were made after the approval of local medical ethics committee (Comité de Protection des Personnes Nord Ouest III, Caen, France, ref: DC-2013-1967) and written informed consent.
Ischemia and Reperfusion In Vivo
In vivo myocardial I/R model in rat
Twenty-six male Wistar rats (240-380 g) were anesthetized by intraperitoneal administration of ketamine (100 mg kg−1) and xylazine (10 mg kg−1). Anesthesia depth was estimated by the loss of the podal reflex. After endotracheal intubation, controlled ventilation was performed using a rodent ventilator (SAR 830/P ventilator; CWE Inc, Colorado springs, CO, ASU), with room air (tidal volume 1 ml 100 g−1, respiratory rate 90 min−1, inspiratory time 50%). Atelectasis was prevented by maintaining a positive end-expiratory pressure of 8 to 12 mm H2O. Electrocardiographic monitoring was obtained using 3 subcutaneous electrodes and analyzed using dedicated software (LabScribe; Iworx). Body temperature was maintained within a physiologic range using a heating pad (ATC 1000, DC, temperature controller; World Precision Instruments).
When the depth of anesthesia was considered adequate, a left thoracotomy was performed in the fourth intercostal space and the pericardium was opened. A 6-0 nonabsorbable nylon suture was passed around the left descending coronary artery. The ends of the suture were threaded through a propylene tube to form a snare that was clamped to induce myocardial ischemia. Success of the ligation was verified by regional cyanosis and contraction loss. After 30 minutes of ischemia, reperfusion was allowed by releasing the snare. Pleural space was evacuated and the chest was closed in 3 layers (ribs, muscles, and skin). The rats were allowed to recover from anesthesia, with free access to water and food on a 12-hour/12-hour light/dark cycle. Subcutaneous buprenorphine (0.02 mg kg−1) was administrated for postoperative analgesia.
Animals were separated in 3 groups: I/R, sham operated, and argon postconditioning (APC). Ischemia–reperfusion group was submitted to a 30-minute ischemia by clamping of the snare, then reperfusion without additional proceedings (n = 11). In the sham-operated group (n = 7), the same surgical procedure was performed except that the snare was not clamped. In APC (n = 8), rats were submitted to ischemia, then received a mixture of argon (80%) and O2 (20%) for 20 minutes, started 5 minutes before reperfusion (instead of 80% N2 + 20% O2 in control). Gases were given through the rodent ventilator. Cardiac functions were thus evaluated by cardiac magnetic resonance (CMR) at 1 (W1) and 3 (W3) weeks after surgery.
Cardiac magnetic resonance
Cardiac magnetic resonance was performed using a dedicated small animal 7-Tesla MR system (Pharmacan 7 T; Bruker Biospin, Ettlingen, Germany). Anesthesia was induced and maintained with isoflurane (1.5%-3%), with animals breathing spontaneously an oxygen/nitrous oxide mix (50%-50%). Rats were positioned in the prone position. Electrocardiogram (ECG) and breath gating of the CMR sequences were achieved using a small animal MR ECG trigger unit (PC-Sam, SA Instruments). Physiological body temperature was maintained as described above. Left ventricular function was assessed using a retrospectively triggered FLASH sequence (IntraGate; Bruker Biospin). Six short axis slices were acquired from the basis to the apex (thickness: 1 mm, time to echo (TE): 2.107 ms/time to repetition (TR): 45 281 ms, flip angle 15°, field of view: 60 × 60 mm, matrix size: 256 × 256, spatial resolution: 0.273 mm pixel−1), with a temporal resolution set to 20 images per cardiac cycle.
Left ventricular function was measured using the Segment software after semiautomatic delineation of the endocardial and epicardial borders (Segment version 1, 8 R1675, Medviso AB, University of Lund, Sweden). Semiquantitative analysis of regional wall motion was performed using a 17-segment model and a 4-point grading system (normal = 0, hypokinetic = 1, akinetic = 2, and dyskinetic = 3). Wall motion score (WMS) was calculated by dividing the total WMS by the number of segments.
Electrical Properties In Vitro
Heart sampling
Experiments were conducted on 47 male Wistar rats (240-380 g) and 13 guinea pigs (300-400 g). After anesthesia with pentobarbital and euthanasia of the animal, the heart was quickly removed. A section of the right ventricle was isolated and placed in a superfusion chamber. In control condition, the preparation was continuously superfused using a peristaltic pump with a physiological solution at a rate of 7 mL mn−1 bubbled with 70% N2 + 30% O2 and maintained at 37°C. The physiological solution contained (in mmol L−1): NaCl 140, KCl 5.4, CaCl2 2, MgCl2 1, NaHPO4 0.3, glucose 10, and 2-[4-(2-hydroxyethyl)-1-piperazinyl]ethanesulfonic acid (HEPES) 10, adjusted at pH 7.4. To evaluate the impact of argon in normoxic conditions, the solution of superfusion was bubbled with 70% argon + 30% O2, using a specific calibrator vaporizer. Ischemic conditions were mimicked by bathing the preparation in a solution similar to the physiological solution but lacking glucose, with an acidic pH adjusted at 6.6 and bubbled with N2 instead of O2 (hypoxic solution).
Action potential acquisition and analysis
Transmembrane potentials were recorded using glass microelectrodes filled with KCl (3 mol L−1) and with tip resistance around 10 MΩ. Microelectrodes were coupled to the input stages of an in-house impedance capacitance-neutralizing amplifier. The recordings were displayed and analyzed using cardiac action potential (AP) automatic acquisition software (iox 2; Emka Technologie, France, or DATAPAC; Biologic, Claix, France). In the model of border zone (see below), electrical activity was recorded by 2 intracellular microelectrodes impaled simultaneously in the normal zone (NZ) and the altered zone (AZ). When specified, the preparation was stimulated at a rate of 2 Hz by electrical square pulses, using a SMP-310 programmable stimulator (Biologic) connected to a bipolar electrode.
The effect of argon on early after depolarizations (EADs) was investigated using a model adapted from Simard et al. 25 To induce such arrhythmias, isolated rat ventricle was superfused by the hypoxic solution (see Section “Heart sampling”) for 30 minutes, then by an oxygenated solution for 30 minutes, to induce reoxygenation.
The effect of argon on conduction blocks and reentries was investigated using an in vitro model of the border zone between normal and ischemic myocardium known to reproduce such arrhythmias. 26 A ventricular strip was dissected and placed in a perfusion chamber bisected by a latex membrane which included a central hole allowing the myocardial strip to be passed through. The preparation is thus divided into 2 compartments called the “normal zone” and the “altered zone.” The 2 compartments can be independently bubbled and superfused by the appropriated gas and solution. 26 For these experiments, we used guinea pig ventricles since we have previously shown that this rodent is adapted for such experiments. 27
Arrhythmias were detected by visual screening of the recordings. In simple microelectrode recordings, in the absence of stimulation, the number of EADs was determined. An event was considered as an EAD when additional depolarization occurred during AP repolarization. To evaluate the variation of EAD occurrence, the number of EADs per 10 seconds was determined and normalized to the number of AP in the same period (EAD/AP). In the border zone model with 2 separated compartments and electrical stimulation in the NZ, conduction blocks between the 2 zones and spontaneous AP were looked for. Myocardial conduction blocks between the AZ and NZ were defined by the absence of AP detected in 1 zone after stimulation applied in the other zone. Blocks and spontaneous APs were coded as present or absent.
Human Atrial Trabeculae Contractility in an In Vitro Model of Hypoxia–Reoxygenation Injury
Human atrial trabeculae collection
Right atrial appendages were obtained during cannulation for cardiopulmonary bypass from patients scheduled for routine coronary artery bypass surgery or aortic valve replacement. All patients received total intravenous anesthesia with propofol, remifentanil, and pancuronium. Patients with chronic atrial arrhythmia or with diabetes mellitus were excluded from the study.
Experimental conditions
Right atrial trabeculae (1 or 2 per appendage) were dissected and suspended vertically between an isometric force transducer (MLT0202; ADInstruments, Sydney, Australia) and a stationary stainless clip in a 200 mL jacketed reservoir filled with a physiological solution containing (in mmol L− 1): NaCl 119, KCl 5, CaCl2 2, MgCl2 2, HEPES 25, and glucose 5.5 (pH = 7.4), maintained at 34°C and bubbled with O2. Isolated muscles were field stimulated at 1 Hz by 2 platinum electrodes with rectangular wave pulses of 5 ms duration 20% above threshold (CMS 95107; Bionic Instrument, Paris, France).
Trabeculae were equilibrated for 60 to 90 minutes to allow stabilization of their optimal mechanical performance at the apex of the length-active isometric tension curve. The force developed was measured continuously, digitized at a sampling rate of 400 Hz, and stored on a writable compact disc for analysis (PowerLab; ADInstruments).
At the end of experiment, the muscle cross-sectional area was calculated from its weight and length assuming a cylindrical shape and a density of 1. To avoid core hypoxia, trabeculae included in the study had a cross-sectional area less than 1.0 mm2, a force of contraction normalized per cross-sectional area (FoC) greater than 5.0 mN mm− 2, and a ratio of resting force/total force less than 0.50.
Experimental protocol
At the end of the stabilization period, the trabeculae were randomly assigned to 1 of the experimental groups. In all groups, hypoxia–reoxygenation (H/R) was performed by replacing O2 with N2 in the buffer for 30 minutes, followed by a 60 minutes oxygenated recovery period. In the nonconditioned group (n = 6), trabeculae were exposed to the H/R protocol alone. In the argon treatment groups (n = 6), 70% argon–30% N2 was administered during the last 2 minutes of hypoxia followed by 70% argon–30% O2 during the first 3 minutes of reoxygenation. In an additional group, the effect of 2 sequences of argon administration was examined: 5 minutes after the first exposition, argon was readministered during 5 minutes (argon 2 cycles group; n = 6).
Mechanisms involved in argon-induced postconditioning were studied during a 5-minute exposure to argon 70%, and in the presence of 100 nmol L− 1 wortmannin (wort), a PI3K inhibitor (argon + wort; n = 6), 20 µmol L− 1 PD98059, an ERK 1/2 inhibitor (argon + PD98; n = 6), and 50 µmol L− 1 atractyloside (atract), an MPTP opener (argon + atract; n = 6). Pharmacological agents were administered 2 minutes before, throughout, and 12 minutes after argon exposure. In 3 additional groups, muscles were exposed to 100 nmol L− 1 wort (n = 6), 20 µmol L− 1 PD98059 (PD98; n = 6), and 50 µmol L− 1 atract (n = 6) 5 minutes before and in the first 15 minutes of reoxygenation.
These concentrations of wort, PD98059, and atract have been shown to inhibit PI3K, ERK1/2 pathways, and to open MPTP, respectively, in the present model. 2,3,28 Wortmannin and PD98059 were dissolved in dimethyl sulfoxide (DMSO), and atract was dissolved in ethanol. It has been previously shown that DMSO and ethanol are without effect in the present model. 29 Wortmannin and PD98059 were purchased from Calbiochem (VWR International, Fontenay-sous-Bois, France), and atract was obtained from Sigma Aldrich (Saint-Quentin-Fallavier, France).
Statistical Analysis
For the in vivo experiments, no data from previous studies on a similar model were available for appropriate sample size calculation. However, our preliminary data revealed that a group of 6 experiments are sufficient to reveal a 20% variation at P < .05. For in vitro experiments using intracellular microelectrodes recordings, group’s size was based on similar protocols reported in the literature. 25,26 According to these, a group of 7 experiments are sufficient to detect significant variations in arrhythmias occurrence at P < .05. For in vitro experiments using human atrial appendages, the power analysis demonstrated that a group’s size of n = 5 was necessary to detect a difference of 20% in the FoC, as described previously. 30
For the analysis of left ventricular function, a linear time series model was used (least square method, JMP version 11; SAS Institute Inc). When a significant effect was found at the P < .05 level, post hoc t tests were computed for each parameter (treatment group and time) and their interactions.
For the in vitro electrophysiological experiments, data were tested for normality by a Shapiro-Wilk test (normality accepted for P > .05), then compared using a paired Student t test. An unpaired Student t test was used to compare data from different series of experiments. A Fisher exact test was used to compare the percentage of appendages exhibiting arrhythmias in different conditions.
In the contraction experiments, the primary end point of the study was the recovery of FoC at 60 minutes of reoxygenation (FoC60, expressed as percent of baseline). Data are expressed as mean (SD). Baseline values of main mechanical parameters, age, left ventricular ejection fraction (LVEF), and FoC were compared by univariate analysis of variance with group factor as the independent variable. Statistical analysis has been performed with Statview version 5.0 software (Deltasoft, Meylan, France). All P values were 2 tailed. For P < .05, a Bonferroni post hoc analysis was performed. Within-group data were analyzed over time using a 2-way analysis of variance for repeated measures and Bonferroni post hoc analysis with group factor and time (baseline, hypoxia 5, 10, 20, 30 minutes, and reoxygenation 5, 10, 20, 30, 40, 50, and 60 minutes) as independent variables.
In all experiments, differences were considered statistically significant when P < .05 and are indicated in the graphs by asterisks. The term “n” refers to the number of animals or human atrial appendages that were used.
Results
Effect of Postconditioning by Argon on Rat Cardiac Properties In Vivo
Cardiac I/R was induced in rats by transient (30 minutes) ligature of the left descending coronary artery. Compared to sham-operated animals, rats submitted to I/R showed a significant impairment of global and regional left ventricular function on CMR. In this group, CMR demonstrated ventricular enlargement, LVEF decrease, and WMS increase that were observed at W1 and W3 (see Table 1). In animals exposed to argon therapy at the time of reperfusion (20 minutes starting 5 minutes before reperfusion), left ventricular function was preserved. Compared to sham-operated animals, the number of normokinetic segments was significantly decreased in the I/R group but not in the argon group, demonstrating myocardial protection. Note that argon did not prevent I/R-induced left ventricular diastolic volume (LVEDV) increase.
Effect of Postconditioning by Argon on Cardiac Properties in Rat In Vivo.

Abbreviations: I/R, ischemia–reperfusion; LVEDV, left ventricular diastolic volume; LVEF, left ventricular ejection fraction; LVESV, left ventricular endsystolic volume; WMS, wall motion score; NS, not significant.
a P < .001 versus I/R week 1.
b P < .01 versus I/R week 3.
c P < .05 versus I/R week 3.
d P < .05 versus argon week 3.
e P < .01 versus sham week 3.
f P < .05 versus argon week 1.
g P < .0001 versus I/R week 1.
h P < .0001 versus I/R week 3.
i P < .01 versus sham week 3.
Effect of Postconditioning by Argon on Cardiac Electrical Activity In Vitro
Hypoxia–reoxygenation-triggered arrhythmias
Hypoxia–reoxygenation is known to induce triggered arrhythmias, such as EADs, 31,32 which result from changes in AP duration. To induce such arrhythmias, isolated rat ventricles were superfused by a hypoxic solution for 30 minutes, then by an oxygenated solution for 30 minutes. Only 1 ventricular sample was used from each animal. In all samples (n = 22), the spontaneous activity recorded in control conditions was abolished by hypoxic conditions but reappeared under reoxygenation. In 12 samples (54.6%), EADs appeared under reoxygenation (Figure 1A) while spontaneous AP without EADs appeared in others. In those exhibiting EADs, bubbling with argon (10 minutes), 30 minutes after reoxygenation, reduced EADs occurrence by 90.1% (n = 6; Figure 1B). In a postconditioning protocol, argon was superfused for 5 minutes starting 2 minutes before the end of the hypoxic episode. Early after depolarizations were detected in only 11% of experiments (n = 9; Figure 1C), indicating a significant effect of argon against triggered arrhythmias. Such experiments were also conducted on ventricles stimulated at 2 Hz to simulate triggering by the sinus node. While additional ectopic AP were detected under reoxygenation (n = 6), none of the ventricles postconditioned by argon exhibited such abnormal activities (n = 6).

Effect of argon on hypoxia–reoxygenation induced triggered arrhythmias from isolated rat ventricles. Spontaneous APs were recorded from isolated rat right ventricle superfused with an oxygenated physiological solution using the intracellular microelectrode technique. A, Representative spontaneous APs in an hypoxia–reoxygenation protocol where the preparation was submitted to 30-minute normoxic conditions, 30-minute hypoxic conditions, then 30-minute normoxic conditions corresponding to the reoxygenation (reoxy). Spontaneous beating disappeared in the hypoxic condition and triggered activities (EADs) appeared during reoxygenation. B, Left: representative experiment showing the effect of argon on EADs, when applied after reoxygenation. Right: Mean number of EAD normalized to that of APs (EAD/AP) in reoxygenation and with application of argon during reoxygenation (mean of 6 experiments). C, Number of isolated ventricles exhibiting EADs (in % of experiments), when the preparation was postconditioned (white) or not (black) by argon, according to the protocol schematized at the top of the histogram. Number of ventricles is indicated in each column. AP indicates action potential; EADs, early after depolarizations.
Hypoxia-reoxygenation induced conduction blocks and reentry arrhythmias
The effect of argon on conduction blocks and reentries was investigated using the in vitro model of the border zone between normal and ischemic myocardium (see section “Methods”) in which a guinea pig ventricle is placed in a perfusion chamber bisected by a latex membrane into 2 compartment called the NZ and the AZ. The AZ was superfused for 30 minutes by the hypoxic solution followed by 30 minutes reoxygenation while the NZ was maintained in the normoxic conditions during all proceeding. Hypoxic conditions induced conduction blocks in 54% of experiments and spontaneous APs in 85% (n = 13). Reoxygenation induced spontaneous APs in all experiments but no conduction block was detected (n = 6; Figure 2A and B). One group (n = 7) was postconditioned 5 minutes with argon (from 2 minutes before to 3 minutes after reoxygenation) as schematized in Figure 2C. In these conditions, no arrhythmias were detected, indicating that APC protects against reentry arrhythmias (Figure 2D).

Effect of postconditioning by argon on hypoxia–reoxygenation induced reentry arrhythmias from isolated guinea pig ventricles in the border zone model. A guinea pig ventricle was placed in a double superfusion chamber. The normal zone (NZ) is continuously superfused by an oxygenated solution while the altered zone (AZ) is successively submitted to hypoxia, then reoxygenation. APs are induced by electrical stimulation (indicated by arrows) and recorded simultaneously in both chambers by intracellular microelectrodes. A, Representative activity during reoxygenation in the NZ and AZ following the protocol schematized at the top. B, Percentage of ventricles with conduction blocks and spontaneous APs (spontan AP) in hypoxia or reoxygenation. Numbers in columns are the numbers of experiments. The values 3 to 10 and >10 refer to the number of spontaneous AP detected in 1 experiment (see section “Methods”). C, Representative experiment similar to that shown in (A), after postconditioning with argon. D, Percentage of ventricles with arrhythmias (conduction blocks + spontaneous AP) in reoxygenation, without (black) or with (white) postconditioning by argon.
Effect of Postconditioning by Argon on Human Atrium Contractility In Vitro
The effect of postconditioning by argon was evaluated on cardiac contractile properties in vitro using human trabeculae from right atria. Hypoxia and reoxygenation protocols are described in the section “Methods” and schematized in Figure 3. Fifty-four human right atrial trabeculae were studied. There was no significant difference between groups for trabecular length at the apex of the length-active isometric tension curve, cross-sectional area, and ratio of resting-to-total force. The main characteristics of patients and LVEF were also not different between groups (Table 2).

Effect of postconditioning by argon on contractile force from human right atrial trabeculae. Human right atrial trabeculae were placed in a superfusion chamber and force of contraction was measured after electrical stimulation. A, Left: force of contraction time course (in percentage of the initial force) in trabeculae submitted to hypoxia then reoxygenation with (open squares) or without (black circles) postconditioning by argon. Symbols are mean (SD) of 6 experiments. Right: representative contractions in the initial normoxic conditions and after reoxygenation in the 2 groups. B, Mean (SD) recovery of force of contraction at the end of the 60-minute reoxygenation period following 30 minutes hypoxia in the nonconditioned group and groups submitted to 1 or 2 postconditioning cycles by argon (see section “Methods”). The control group was not submitted to hypoxia nor application of argon. n = 6 for each group. C, Mean (SD) recovery of force of contraction at the end of the 60-minute reoxygenation period in several conditions as indicated under columns. Dotted line indicates the reference level of the nonconditioned group (nc), as shown in (B). Asterisks indicate statistically significant difference with the argon 1 cycle group. Atract indicates atractyloside; PD98, PD98059; wort, wortmannin.
Control Values of Main Mechanical Parameters of Human Right Atrial Trabeculae and Patients Demographic Data.a
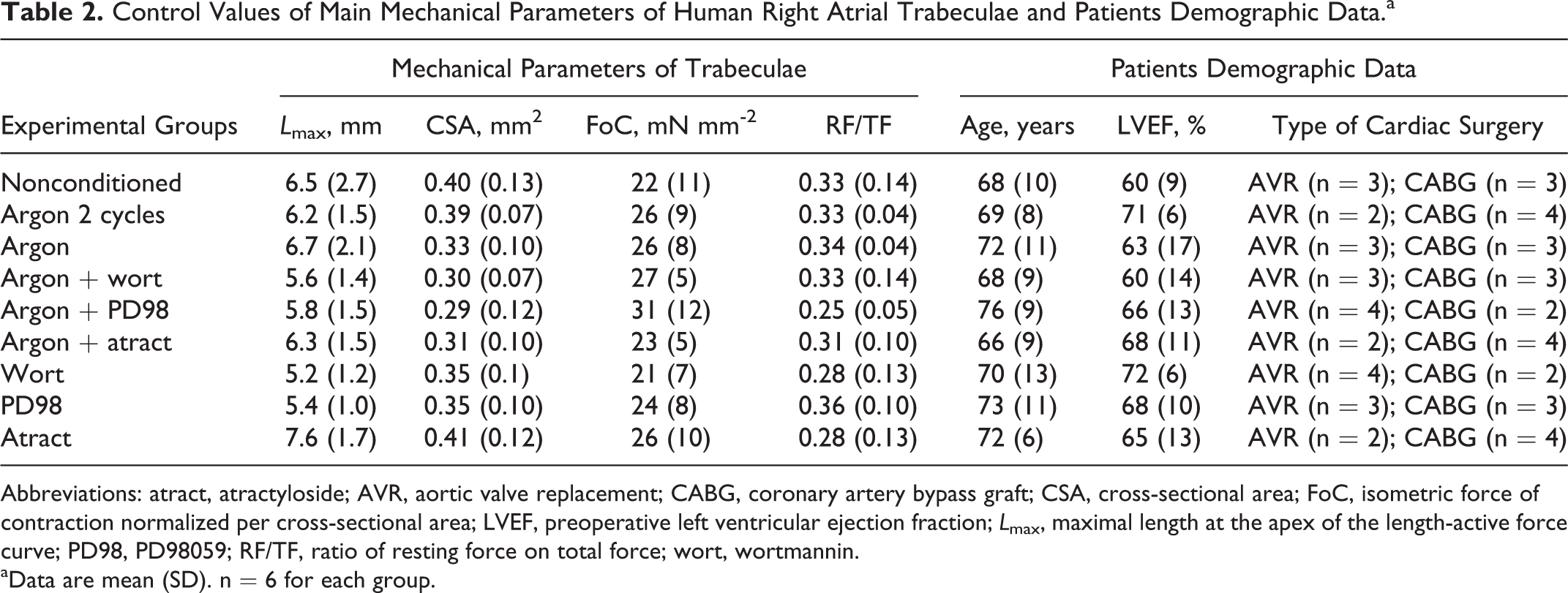
Abbreviations: atract, atractyloside; AVR, aortic valve replacement; CABG, coronary artery bypass graft; CSA, cross-sectional area; FoC, isometric force of contraction normalized per cross-sectional area; LVEF, preoperative left ventricular ejection fraction; L max, maximal length at the apex of the length-active force curve; PD98, PD98059; RF/TF, ratio of resting force on total force; wort, wortmannin.
aData are mean (SD). n = 6 for each group.
At the end of 60-minute reoxygenation, 2 sequences as well as 1 sequence of 5 minutes of argon significantly increased the FoC as compared to the nonconditioned group (argon 2 cycles: 80% ± 7% of baseline; argon 1 cycle: 83% ± 7% of baseline; P < .001 vs 51% ± 2% of baseline control group). There was no significant difference between the FoC60 of argon 2 cycles and argon 1 cycle groups (P = .53; Figure 3A and B).
We further investigated the mechanisms involved in APC using RISK pathway modulators and an MPTP opener. Administration of wort, a PI3K inhibitor (FoC60: 59% ± 10% of baseline), PD98059, an ERK1/2 inhibitor (FoC60: 60% ± 10% of baseline), and atract, an MPTP opener (FoC60: 57% ± 14% of baseline), during the argon exposure, inhibited the argon-induced postconditioning (P < .001 vs argon group; Figure 3C).
Administration of wort (FoC60: 57% ± 5% of baseline; P = .68 vs control group), PD98059 (FoC60: 51% ± 9% of baseline; P = .99 vs control group), and atract (FoC60: 53% ± 6% of baseline; P = .73 vs control group) alone during the reoxygenation period had no significant effect (Figure 3C).
Discussion
Our data show that application of argon with a postconditioning protocol in an in vivo model of I/R in rat produces a protective effect on cardiac properties. In addition, argon prevents H/R-induced arrhythmias in in vitro cardiac preparations from the rat and guinea pig and further restores contractile properties in human atrial appendages. By demonstrating functional improvement against H/R-induced myocardial insults, these data taken together extend previous findings which only showed that argon preconditioning reduces infarct size or protects isolated cardiomyocytes. 9,20
A central element of I/R injuries, targeted in cardioprotection, is the MPTP. This non-selective channel of the inner mitochondrial membrane opens at reperfusion, following mitochondrial calcium and phosphate overload. 1,33 Mitochondrial permeability transition pore opening produces depolarization of the mitochondrial membrane and uncouples oxidative phosphorylation, resulting in adenosine triphosphate (ATP) depletion and cell death. 1 In our experiments on human appendages, application of atract, an MPTP opener, prevents the cardioprotective effect of argon. This suggests that argon acts directly or indirectly by inhibiting MPTP opening, thus protecting the mitochondria. However, MPTP is also known to be controlled by the RISK pathway which activation prevents MPTP opening. Here, we show that inhibiting the PI3K-Akt and MEK/ERK1/2 prosurvival signaling kinases of the RISK pathway, respectively, by wort and PD98059, suppresses the cardioprotective effect of argon, thereby suggesting that the RISK pathway is involved in the inhibiting effect of argon on MPTP opening. In line with our findings, previous data have reported that preconditioning with argon leads to an activation of the RISK pathway prosurvival signaling kinases. 9 This condition has been shown to induce beneficial effects in cardiac postconditioning studies with volatile anesthetics. 2,28,29
In the in vivo experiments, it is surprising that LVEDV was not preserved by argon despite its protective effect on LVEF. Although additional experiments are needed to understand this discrepancy, data obtained on human atrial samples confirmed the beneficial effect of argon on myocardium against I/R-induced contraction force decrease.
Another aspect of cardioprotection by argon is its antiarrhythmic effect that was observed on isolated cardiac preparations. Argon reduces both triggered and reentry arrhythmias, which mainly result from a variation in AP shape 34 and from an increase in AP parameter dispersion between the normoxic and ischemic zones, 26 respectively. Because both types of arrhythmias vary from each other even if basically caused by I/R episodes, which mainly results from Ca2+ overload and modulation of Ca2+-dependent currents, 31,35 the beneficial effects of argon on arrhythmias could occur either through a common mechanism or through independent convergent processes. This could occur through a direct action of argon on channel proteins or through modulation of their regulators. Action of noble gases on ion channels is poorly documented but realistic. As an example, a KATP channel was shown to be opened by xenon. This gas directly targets the Kir6.2 subunit of the channel without affecting the sulfonylurea receptor-1 subunit. 36 This is particularly interesting since KATP channel activation is known to exert antiarrhythmic effects. In that way, helium-induced cardiac preconditioning in vivo in rabbit has also been shown to be mediated by the mitochondrial KATP channel. 8 In addition, although data on argon are not so strength than those with xenon or helium at demonstrating interactions with ion channels, it has to be noted that argon has been shown using a pharmacological approach to be an agonist of the type A γ aminobutyric acid ion channel receptor. 37 Although there is now a main consensus that the inert gases, like the volatile anesthetics, act by disrupting directly protein functions, 37 -41 an alternative or additional way to modulate ion channels activity might be that the inert gases, including argon, act by disrupting the structure and dynamics of the lipid membranes and thereby indirectly alter the protein functions. As such, the Meyer–Overton rule of a high correlation between activity and solubility in lipids of the inert gases (with xenon > krypton > argon > oxygen > nitrogen > helium) 42 has for a long time promoted this hypothesis, and in that way, interestingly, xenon has been recently shown to affect lipid raft arrangement within an artificial bilayer membrane. 43
Alternatively, it should be examined whether argon could induce side effects such as alveolar or cellular hypoxia when given at high concentration of 75% or 80% with the remaining being oxygen at 25% or 20%. Although this possibility is not trivial, it seems unlikely to be true given that no adverse effect has been reported in human individuals breathing 80% argon at atmospheric pressure and in divers exposed to hyperbaric pressure equivalent to 300% to 600% argon. 44 -46 In addition, argon possesses oxygen-like properties as first demonstrated by Semenov, the Nobel Prize Laureate of chemistry in 1956 with Sir Cyril Norman Hinshelwood, whose work about branched chain reactions showed that argon, which is incapable of chemical reactions, behaves however as a sort of catalyst for some of them by producing a kind of oxygen synergy. 47 In that way, argon at such concentrations has been shown, like oxygen but unlike xenon and helium, to favor the catalytic activity of tissue plasminogen activator. 17,19,48,49 Alternatively and interestingly, argon has been shown to allow rats exposed to hypoxic conditions incompatible with life to adapt their oxygen demand and to restore, in part, mitochondrial respiratory enzyme activity in rats exposed to nonlethal hypoxic conditions. 50
Since it is difficult to obtain human ventricular samples, particularly for ethical reasons, our experiments on contraction were conducted on atrial samples. This might be a limitation of our work since differences exist between the atrial and ventricular myocardium. However, a significant number of studies evaluating pharmacological or ischemic preconditioning or postconditioning reported similar effects on contractile force restoration both on atrial and ventricular muscle. This was, for example, shown for sevoflurane-induced cardioprotection on rat ventricle, 51 which is similar on human atrial samples. 3
Despite the fact that our results are attractive for postconditioning cardioprotection, the translatability for treatment of humans remains to be evaluated since discrepancies between in vivo and in vitro results might appear. As example, meta-analysis from randomized clinical trials of cardioprotection with anesthetic gases provides encouraging but not yet definitely convincing results. 52 For noble gases, a recent study conducted in humans undergoing coronary artery bypass graft surgery reported that postconditioning with helium does not produce significant change in the activity of enzymes from the RISK pathway, including ERK1/2, 53 while it was efficient in animal models. 10,11 Argon might provide another opportunity to achieve this cardioprotection. One of its advantages for human application comes from the fact that it is already used in human as a gas carrier in diagnostic procedures, including the scintigraphic diagnosis of pulmonary embolism, without side effects. 54
Footnotes
Author Contributions
SL and JLH designed and performed the contraction experiments. MS, SL, RR, RG, CS, and LS designed and performed the electrophysiological experiments. KB, AL, DL, and AM designed and performed the in vivo experiments. AM, RG, LC, SA, JA, and LS designed the study. RG, SL, AM, and JA wrote the manuscript.
Authors’ Note
Sandrine Lemoine, Katrien Blanchart, and Mathieu Souplis contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a grant from the Fondation de France (Paris, France) and a grant from Conseil Régional Basse-Normandie (Caen, France). Sandrine Lemoine received a fellowship from the Fondation de France (Paris, France).
