Abstract
Background:
Pycnogenol, the standardized flavonoid-rich extract from the bark of French maritime pine (Pinus pinaster Ait), has been shown to modify a number of cardiovascular risk factors. However, that Pycnogenol modulates plasma lipid levels is unclear due to the inconsistent findings.
Objective:
To examine the impact of Pycnogenol on lipid profile through a meta-analysis of available controlled clinical trials.
Methods:
Controlled trials assessing the effects of Pycnogenol on lipid parameters (total cholesterol, low-density lipoprotein cholesterol [LDL-C], high-density lipoprotein cholesterol [HDL-C], and triglycerides) were identified by electronic search in Medline and Scopus. Weighed mean difference (WMD) and 95% confidence interval (CI) were calculated as effect size using random-effects meta-analysis. Sensitivity and meta-regression (for Pycnogenol dose and duration of supplementation) analyses were carried out using leave-one-out and unrestricted maximum likelihood methods, respectively.
Results:
A total of 7 trials comprising 442 patients (226 in the Pycnogenol and 216 in the control group) were identified. Meta-analysis did not reveal any significant effect of Pycnogenol on lipid parameters: total cholesterol: WMD: −0.03 mmol/L, 95% CI: −0.34 to 0.28, P = .83; LDL-C: WMD: −0.07 mmol/L, 95% CI: −0.29 to 0.15, P = .54; HDL-C: WMD: 0.00 mmol/L, 95% CI: −0.04 to 0.05, P = .86; and triglycerides: WMD: 0.05 mmol/L, 95% CI: −0.12 to 0.23, P = .55. All these effect sizes were robust in sensitivity analyses. Apart from a dose–effect association for the effect of Pycnogenol on LDL-C, there was no other association between Pycnogenol dose or its duration of supplementation with changes in any of the lipid parameters.
Conclusion:
Current clinical evidence does not suggest any significant effect of Pycnogenol on fasting lipid profile.
Keywords
Introduction
Dyslipidemia is a leading risk factor for atherosclerotic cardiovascular disease (ACVD) and stroke and is usually characterized as elevated levels of low-density lipoprotein cholesterol (LDL-C) and triglycerides but low levels of high-density lipoprotein cholesterol (HDL-C). It has been reported that more than a half of the patients with dyslipidemia fail to reach recommended lipid targets despite being treated with statins and other types of lipid-lowering drugs. 1 This is due to the limited efficacy of currently available medications on one hand and adverse events of intensive therapy with such medications on the other. 2 –4 Due to these limitations, there has been a surge of interest to find safe lipid-modulating agents of natural origin to be used as adjuncts to stains and other hypolipidemic agents. Such an adjunctive therapy would not only result in incremental reductions in plasma lipid levels but also would obviate the requirement for high-dose statin therapy and thereby limits the side effects observed with aggressive therapy including myopathies and hepatotoxicity. 3,4
Pycnogenol is a standardized hydroalcoholic extract obtained from the bark of French maritime pine (Pinus pinaster Ait). The extract is predominated by procyanidins (∼65%-75%) that are condensed biopolymers of flavonoids catechin and epicatechin, with varying chain length. Other constituents of Pycnogenol include monomeric flavonoids (catechin and taxifolin) and phenolic acids (derivatives of benzoic and cinnamic acids such as gallic acid, caffeic acid, and ferulic acid). 5 Pycnogenol is endowed with a multitude of pharmacological actions important for the improvement in cardiovascular health. Among these are enhancement of plasma antioxidant capacity through free radical scavenging and metal-chelating activities, stimulation of endogenous enzymatic antioxidants, and inhibition of lipid peroxidation. 6 –8 Along with mitigation of oxidative stress, Pycnogenol can counterbalance inflammation as another fundamental contributor of ACVD. It has been shown that Pycnogenol can downregulate nuclear factor κB pathway, thereby reducing the expression of proinflammatory cytokines such as tumor necrosis factor α and interleukin 1β. 9,10 Moreover, Pycnogenol improves endothelial function and vasodilation, 11 reduces vascular inflammatory markers (vascular cell adhesion molecule and intracellular cell adhesion molecule), 10 enhances microcirculation and endothelial nitric oxide generation, 12,13 reduces high blood pressure, 14 and inhibits platelet aggregation. 15 Owing to all these beneficial actions, Pycnogenol could be considered as a multifunctional supplement for patients at high risk of ACVD.
Another pharmacological effect of Pycnogenol that has been the subject of increasing research is its lipid-modulating properties. Although a number of clinical studies have reported interesting effects of Pycnogenol supplementation on lipid profile status, 7,16,17 others have failed to substantiate this effect. 11,17 –22 Such discrepancies are mainly due the interstudy variations regarding inclusion criteria, trial design, Pycnogenol dosage, and duration of supplementation. Therefore, the present study aimed to resolve the inconsistencies regarding the clinical relevance of Pycnogenol-induced changes in lipid levels by meta-analyzing published controlled trials.
Methods
Search Strategy
This study was designed according to the guidelines of the 2009 preferred reporting items for systematic reviews and meta-analysis (PRISMA) statement. 23 Medline (http://www.ncbi.nlm.nih.gov/pubmed) and Scopus (http://www.scopus.com) were searched using the combination of following key words in titles and abstracts: (hyperlipidemia OR hyperlipidaemia OR hyperlipidemic OR hyperlipidaemic OR hypolipidemic OR hypolipidaemic OR dyslipidemia OR dyslipidaemia OR dyslipidemic OR dyslipidaemic OR hypercholesterolemia OR hypercholesterolaemia OR hypercholesterolemic OR hypercholesterolaemic OR hypocholesterolemic OR hypocholesterolaemic OR “low-density lipoprotein” OR “high-density lipoprotein” OR cholesterol OR triglycerides OR hypertriglyceridemia OR hypertriglyceridaemia OR hypotriglyceridemic OR hypotriglyceridaemic OR LDL OR HDL) AND (Pycnogenol OR “French maritime”). The search was limited to studies in human. The literature was searched from inception to July 08, 2013. Selected articles were hand searched to identify further relevant studies.
Study Selection
Clinical trials with a case–control or case-crossover design were considered eligible for inclusion in this review provided that they fulfilled all the following criteria: (1) investigated the impact of Pycnogenol on at least one of the lipid profile parameters including total cholesterol, LDL-C, HDL-C, and triglycerides, (2) having an appropriate controlled design, and (3) presentation of sufficient information on plasma/serum lipid levels at baseline and at the end of study in both Pycnogenol and control groups. Clinical trials with any of the following characteristics were excluded from the review and analysis: (1) no or inappropriate controlling for Pycnogenol, (2) using nonstandardized French maritime pine bark extracts, and (3) lack of sufficient information about lipid levels at baseline and/or endpoint.
Data Extraction
Eligible studies were reviewed, and the following data were abstracted: (1) first author’s name; (2) year of publication; (3) study location; (4) number of participants in the case and control groups; (5) age, gender, and body mass index of study participants; (6) circulating concentrations of total cholesterol, LDL-C, HDL-C, triglycerides, high-sensitivity C-reactive protein, and glucose; and (7) systolic and diastolic blood pressures.
Quantitative Data Synthesis
Meta-analysis was conducted using the Cochrane Program Review Manager version 5.1 (Cochrane Collaboration, Oxford, United Kingdom). Blood lipid and glucose levels were collated in mmol/L using conversion factors 0.0259, 0.0113, and 0.0555 for cholesterol (total cholesterol, HDL-C, or LDL-C), triglyceride, and glucose, respectively. The effect size that was used in the present study was weighed mean difference (WMD) and 95% confidence interval (CI). Net changes were calculated for parallel and crossover trials as follows: (measure at end of follow-up in the treatment group − measure at baseline in the treatment group) − (measure at end of follow-up in the control group − measure at baseline in the control group). Standard deviations (SDs) of the mean difference were calculated using the following formula: SD = square root[(SDpretreatment)2 + (SDposttreatment)2 − (2R × SDpretreatment × SDposttreatment)], assuming a correlation coefficient (R) = .5. In case of reporting standard error of the mean (SEM), SD was estimated using the following formula: SD = SEM × square root (n), where n is the number of patients.
A random-effects model and the generic inverse variance method were used to compensate for the heterogeneity of studies in terms of design (parallel or crossover), dose, duration of supplementation, and baseline characteristics of included populations. In order to evaluate the influence of each study on the overall effect size, sensitivity analysis was conducted using 1-study remove (leave-one-out) approach. 24 Interstudy heterogeneity was assessed using the Cochran Q test and I2 index. 25
Meta-Regression
Random-effects meta-regression was performed using unrestricted maximum likelihood method to evaluate the association between calculated net changes in plasma lipids and putative moderators, that is, Pycnogenol dose and duration of supplementation. Comprehensive Meta-Analysis V2 software (Biostat, New Jersey) 26 was used for performing meta-regression analysis.
Publication Bias
Potential publication bias was explored using visual inspection of Begg funnel plot asymmetry and Begg rank correlation and Egger weighted regression tests. Comprehensive Meta-Analysis V2 software 26 was used for performing publication bias analysis.
Results
Flow of Included Studies
Overall 118 articles were screened for eligibility after excluding duplicates (n = 11). Of these 118 articles, 15 were selected for full-text assessment. After careful evaluation, 7 studies met the inclusion criteria and selected for the meta-analysis. 11,17 –22 The other 8 studies were excluded for the following reasons: not assessing any of the lipid profile parameters, 14,27 –29 not having a controlled design, 7 reporting the same lipid profile data as another published study, 30 and insufficient information on the lipid levels. 31,32 A summary of the study selection process is illustrated in Figure 1.
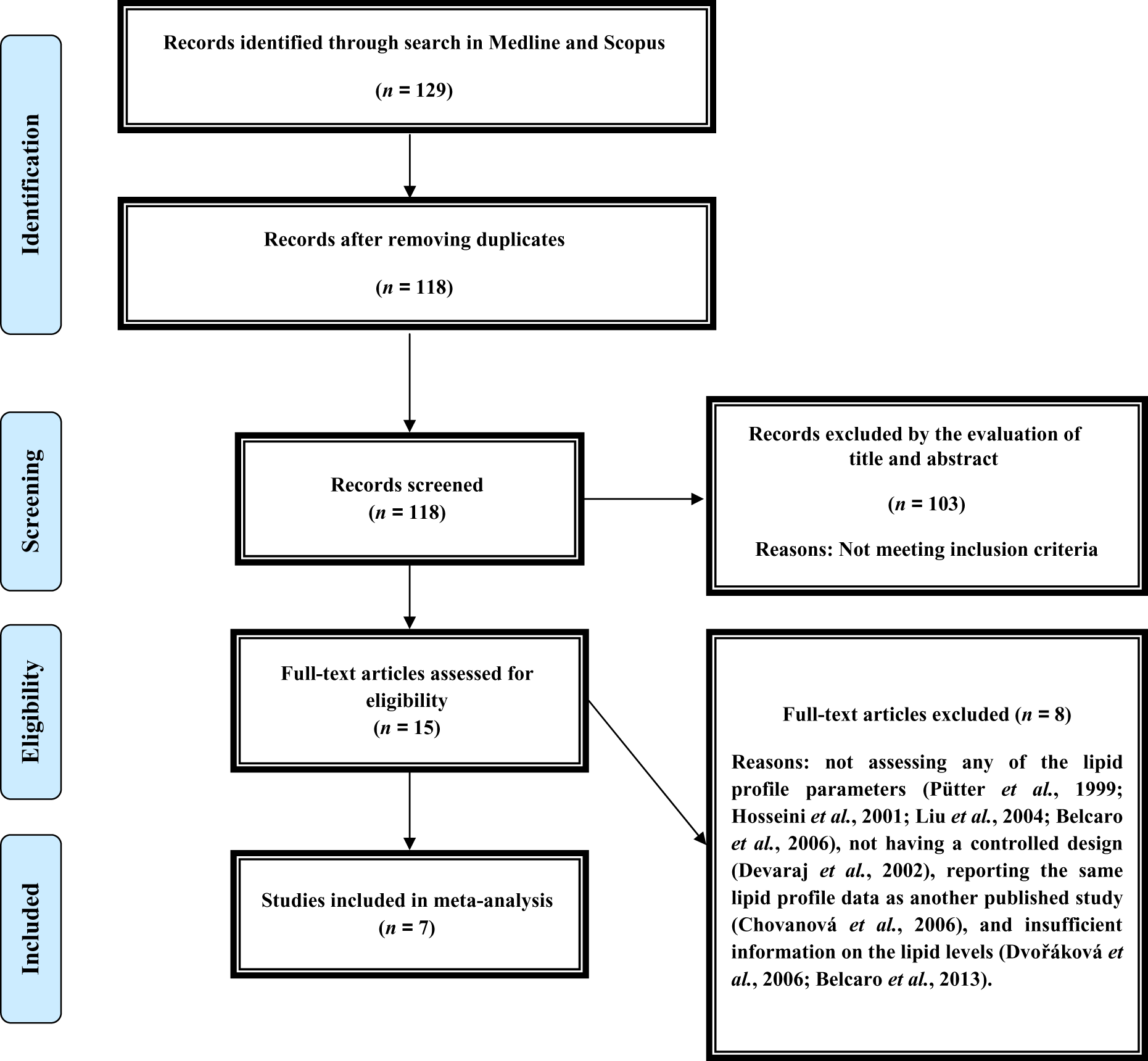
Flow diagram of the study selection procedure.
Characteristics of Included Studies
A total of 442 patients were included in the 5 eligible studies, comprising 226 patients in the Pycnogenol group and 216 patients in the control group (patients in the crossover trial were counted in both groups). The smallest and largest trials had population sizes of 16 patients 18 and 155 patients 19 , respectively. Included studies were published during the period of 2003 to 2012 and were reported from Switzerland, 11 Slovakia, 17 Japan, 18 Taiwan, 19 Australia, 20 United States, 21 and Italy. 22 All studies were designed as parallel-group trials apart from the study by Enseleit et al, 11 which had a crossover design. All trials were double blind apart from the study by Cesarone et al, 22 which did not provide sufficient information as to the blinding status. The study by Cesarone et al 22 was the only one to evaluate the efficacy of Pycnogenol as adjunctive therapy to ramipiril, while other studies compared the efficacy of Pycnogenol versus placebo. Duration of supplementation with Pycnogenol ranged between 2 weeks 18 and 24 weeks. 19,22 Pycnogenol dosage varied between 120 and 200 mg/d. Case and control groups in most of the included studies were matched regarding age and gender. Selected trials were conducted among patients with concomitant type 2 diabetes and hypertension, 21 hypertension, 22 stable coronary artery disease, 11 erectile dysfunction, 17 elderly patients, 20 perimenopausal women, 19 and healthy participants. 18 Among the case groups that were included in the current review, baseline levels of total cholesterol, LDL-C, HDL-C, and triglycerides were above the normal range in 3 studies, 17,20,22 1 study, 20 and 1 study, respectively. 20 The HDL-C levels were within the normal range in all case groups. Demographic and baseline biochemical parameters of the included studies are illustrated in Table 1.
Demographic Characteristics of the Included Studies.a
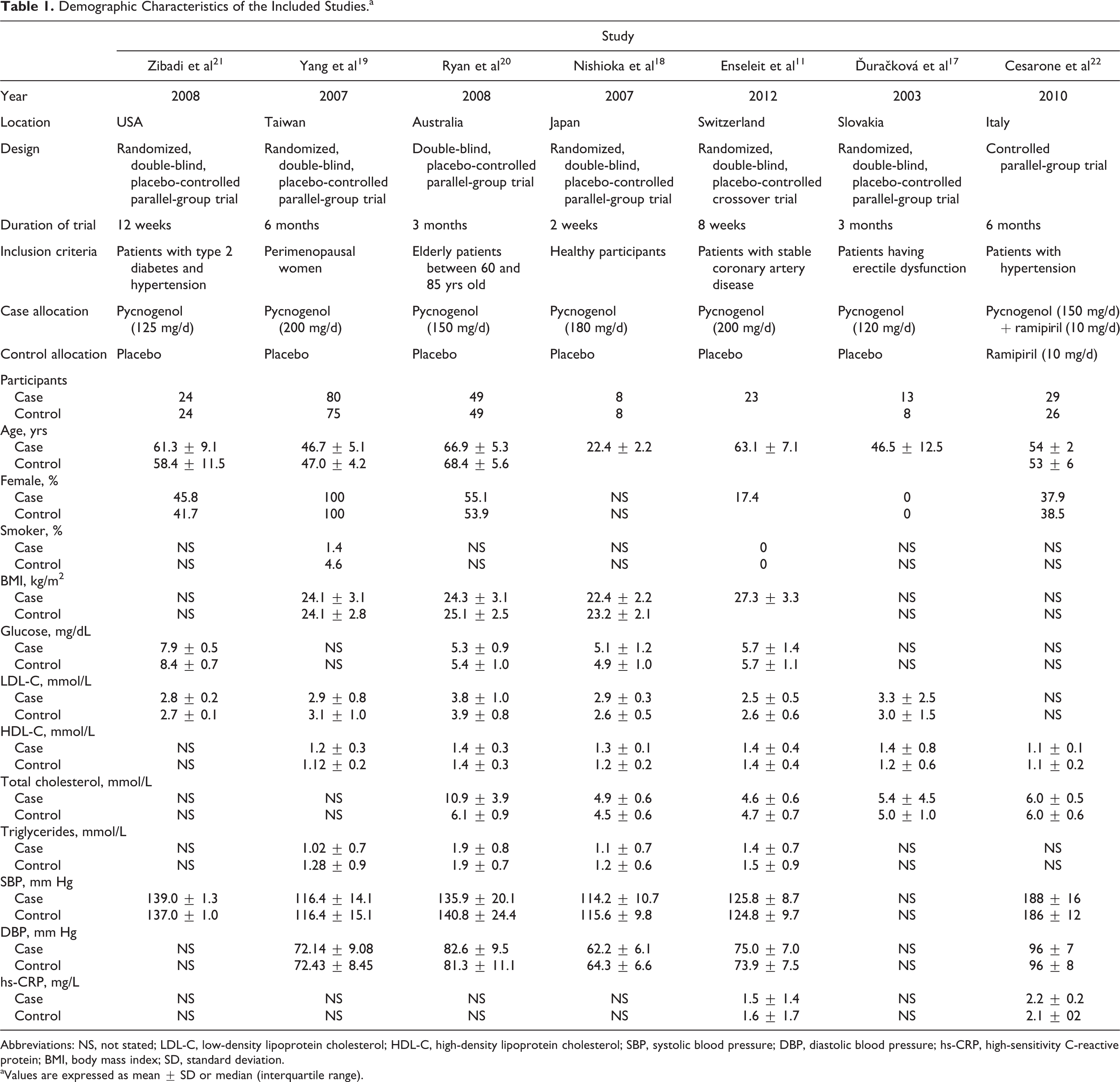
Abbreviations: NS, not stated; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; SBP, systolic blood pressure; DBP, diastolic blood pressure; hs-CRP, high-sensitivity C-reactive protein; BMI, body mass index; SD, standard deviation.
aValues are expressed as mean ± SD or median (interquartile range).
Quantitative Data Synthesis
Among the included studies, total cholesterol, LDL-C, HDL-C, and triglycerides were assessed in 5, 6, 6, and 4 studies, respectively. The net effect of Pycnogenol on circulating concentrations of total cholesterol (WMD: −0.03, 95% CI: −0.34 to 0.28, P = .83), LDL-C (WMD: −0.07, 95% CI: −0.29 to 0.15, P = .54), HDL-C (WMD: 0.00, 95% CI: −0.04 to 0.05, P = .86), and triglycerides (WMD: 0.05, 95% CI: −0.12 to 0.23, P = .55) was not found to be statistically significant. Forest plots summarizing the meta-analysis of trials on each lipid parameter are demonstrated in Figure 2.
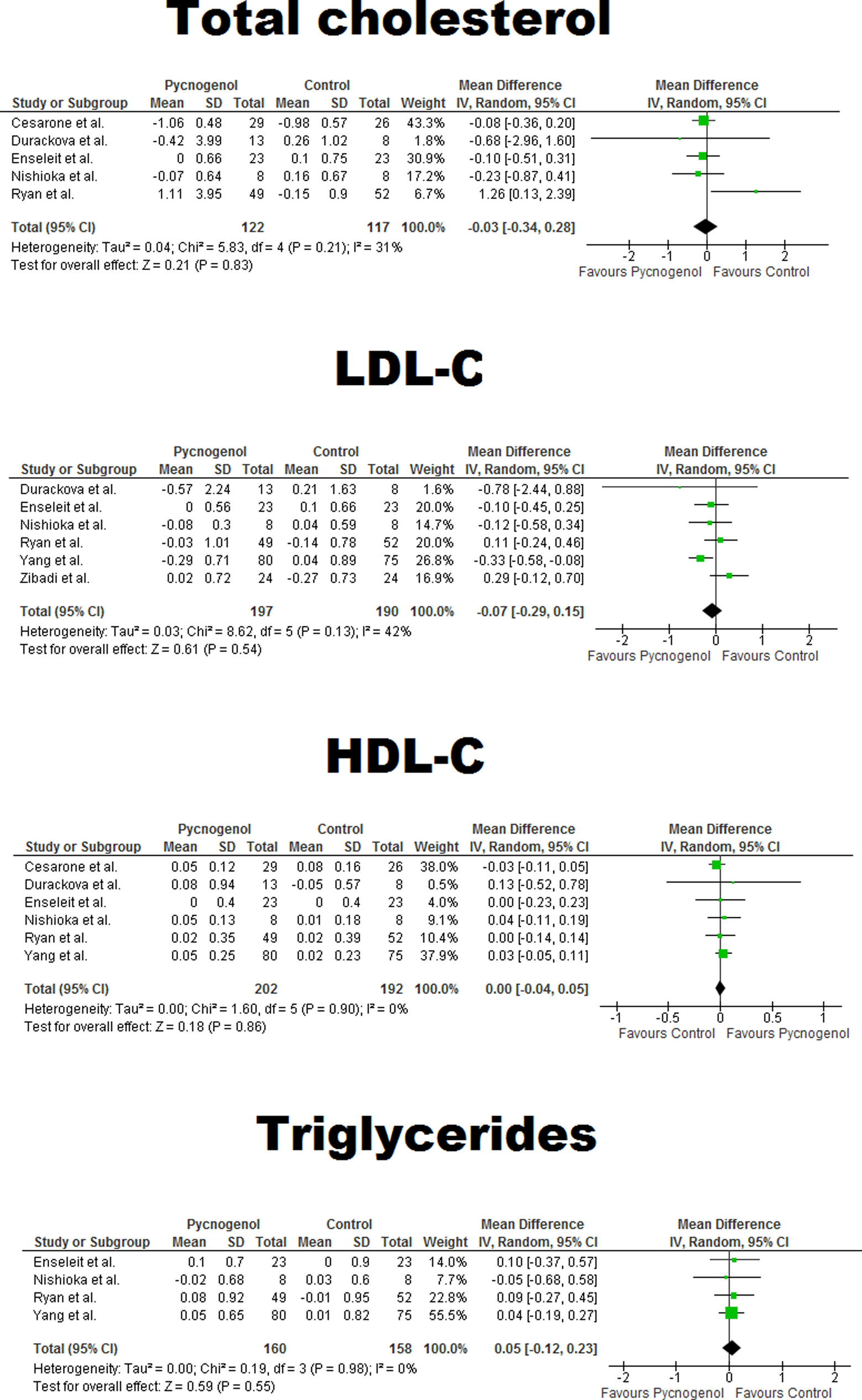
Forest plot detailing weighted mean difference and 95% confidence intervals for the impact of Pycnogenol on plasma lipids. LDL-C indicates low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.
Calculated effect sizes for the impact of Pycnogenol on all of the lipid indices were robust in sensitivity analyses. The results of sensitivity analyses are summarized in Table 2.
Leave-One-Out Sensitivity and Heterogeneity Analyses for the Impact of Quercetin Supplementation on Plasma Lipids.
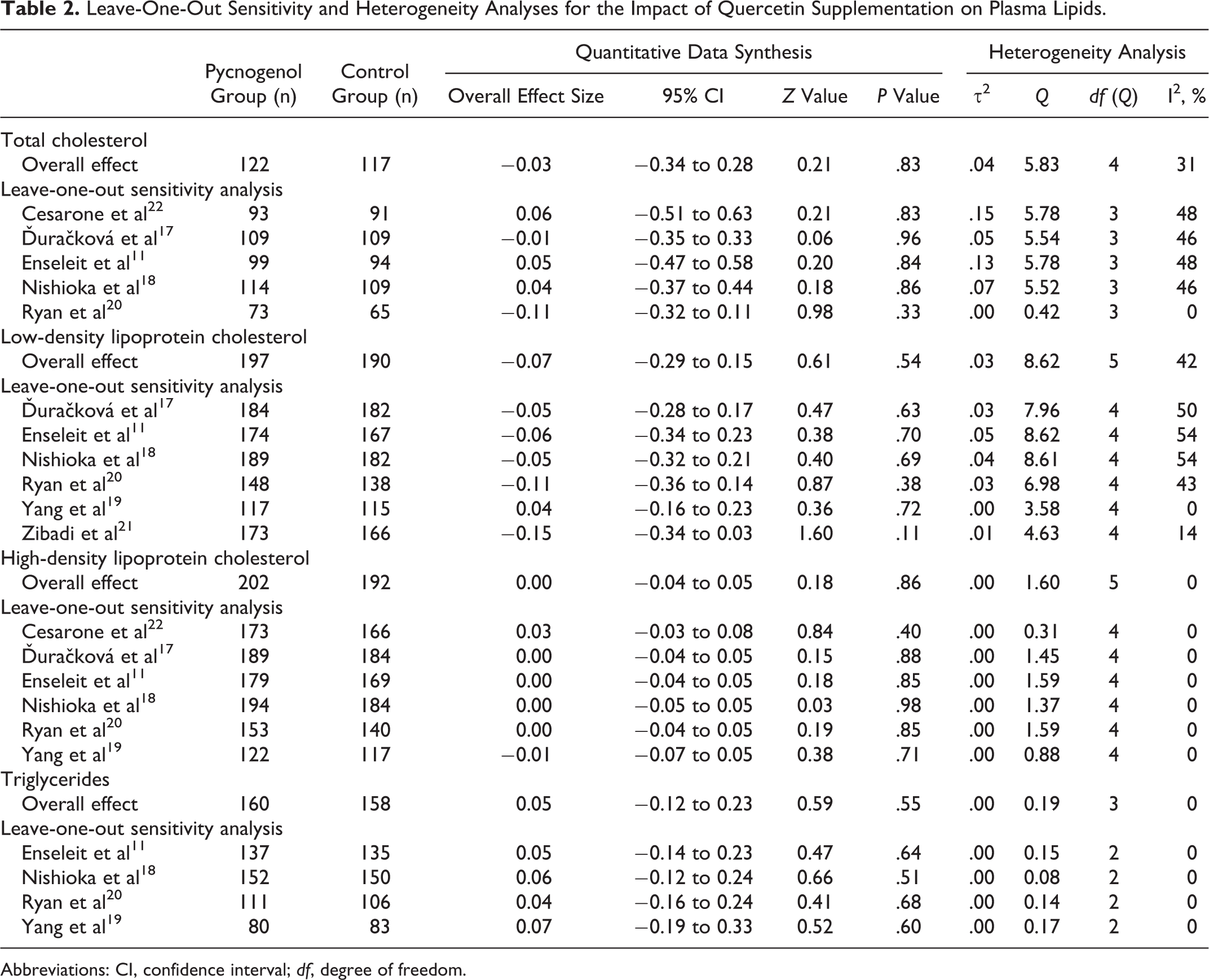
Abbreviations: CI, confidence interval; df, degree of freedom.
Meta-Regression
Possible association between the net effect of Pycnogenol on lipid indices and putative moderators (Pycnogenol dose and duration of supplementation) was assessed using random-effects meta-regression analysis. There was no association between Pycnogenol dose and its effect on total cholesterol (slope: −0.002; 95% CI: −0.011 to 0.007; P = .69), HDL-C (slope: 0.0009; 95% CI: −0.001 to 0.003; P = .36), and triglycerides (slope: −0.0006; 95% CI: −0.009 to 0.008; P = .89). However, a significant dose–effect association was found for LDL-C (slope: −0.007; 95% CI: −0.012 to −0.001; P = .01; Figure 3).
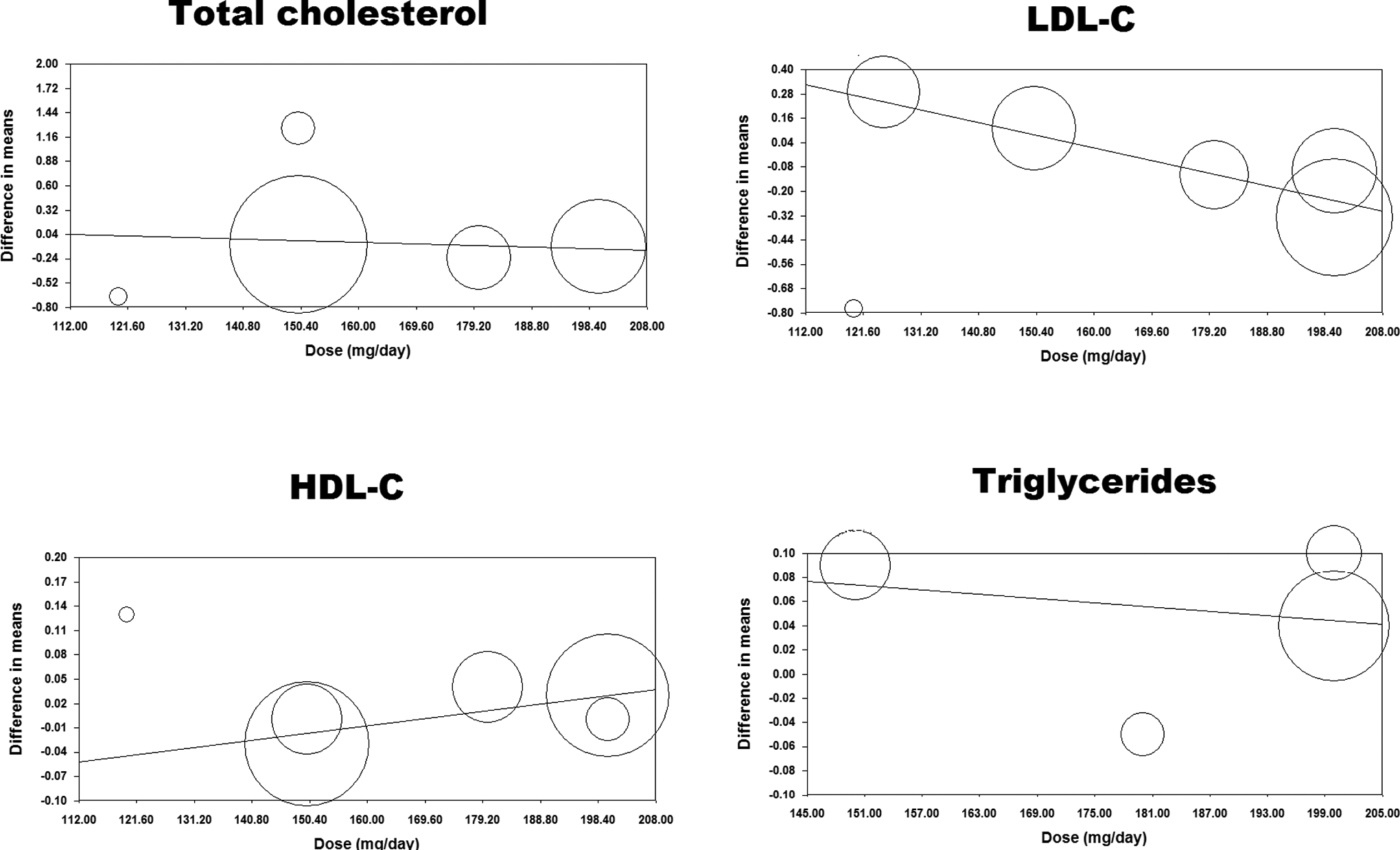
Meta-regression plots of the mean change in plasma lipid concentrations according to the administered Pycnogenol dose. The size of each circle is inversely proportional to the variance of change. LDL-C indicates low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.
The effects of Pycnogenol on none of the lipid indices were found to be significantly associated with the duration of supplementation: total cholesterol: slope: 0.002; 95% CI: −0.022 to 0.025; P = .88; LDL-C: slope: −0.016; 95% CI: −0.034 to 0.003; P = .10; HDL-C: slope: −0.001; 95% CI: −0.007 to 0.005; P = .64; and triglycerides: slope: −0.0003; 95% CI: −0.022 to 0.021; P = .98 (Figure 4).

Meta-regression plots of the mean change in plasma lipid concentrations according to the duration of treatment with Pycnogenol. The size of each circle is inversely proportional to the variance of change. LDL-C indicates low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.
Publication Bias
Visual inspection of the funnel plot asymmetry suggested potential publication bias for the effects of Pycnogenol on plasma total cholesterol and LDL-C (Figure 5). However, Begg rank correlation test and Egger linear regression tests did not indicate significant publication bias for any of the lipid indices (Figure 5).

Funnel plots detailing publication bias in the studies selected for analysis. Results of Begg rank correlation and Egger weighted regression tests are presented next to the respective figure. LDL-C indicates low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.
Discussion
The present study is the first to meta-analyze clinical trial findings on the impact of Pycnogenol on plasma lipid levels. Pooling the data retrieved from all qualified controlled trials indicated that none of the lipid indices (total cholesterol, LDL-C, HDL-C, and triglycerides) is significantly altered following supplementation with Pycnogenol. Moreover, meta-regression analyses revealed that this lack of significant effect is, in most cases, independent of Pycnogenol dosage and duration of supplementation. Owing to the increased population size, this finding is more powerful and reliable compared to individual studies.
Aside from inherent efficacy, a possibility for the lack of significant effect of Pycnogenol could be the baseline characteristics of populations studied. Changes in lipid concentrations were not among the primary outcome measures of any of the included studies because none of the selected studies was originally designed to investigate the lipid-modulating properties of Pycnogenol. Therefore, baseline lipid levels were mainly within the normal range in the included studies (LDL-C being elevated [>130 mg/dL] only in 1 trial, 20 while all studies had baseline HDL-C >40 mg/dL). As a consequence of this, none of the studies controlled dietary intake of lipids during the course of the trial. Hence, interstudy variations in terms of diet might have potentially confounded the results on lipids. Thus far, clinical evidence on the lipid-modulating effects of Pycnogenol has been presented by 3 studies. Devaraj et al 7 reported a significant decline in plasma LDL-C and elevation in plasma HDL-C following 6 weeks of supplementation with Pycnogenol (150 mg/d) in healthy participants. However, the results of this study are biased by lack of including a control group. In the second study, significant reductions in total cholesterol and LDL-C were reported in patients with chronic venous insufficiency treated with Pycnogenol (360 mg/d) for a period of 4 weeks. 16 Again this study was flawed by looking at the within-group changes and lack of appropriate controlling for Pycnogenol effects. The third study, conducted by Ďuračková et al, 17 was a randomized double-blind placebo-controlled trial also included in the present meta-analysis. In the mentioned trial, significant reductions in total cholesterol and LDL-C were reported in patients having erectile dysfunction. Nevertheless, this study was a small-scale pilot trial recruiting merely 21 patients.
Unlike some herbal products, Pycnogenol is readily absorbed and has considerable oral bioavailability. Major flavonoid constituents of Pycnogenol including catechin, caffeic acid, ferulic acid and taxifolin, and its major metabolite δ-(3,4-dihydroxy-phenyl)-γ-valerolactone (M1) have been detected in plasma of healthy volunteers within 14 hours of ingestion (single- and multiple-dose pharmacokinetics of maritime pine bark extract (Pycnogenol) after oral administration to healthy volunteers). Therefore, lack of lipid-modulating effects cannot be attributed to the poor bioavailability of Pycnogenol.
Lack of detecting a significant effect of Pycnogenol on lipid profile does not rule out the cardioprotective effects of this flavonoid-rich natural product or its potential antiatherosclerotic properties. As referred earlier, Pycnogenol favorably affects several factors important for cardiovascular health including endothelial function, 11 blood pressure, 14 systemic antioxidant capacity, 7 while minimizing inflammation as an underlying pathologic factor for ACVD. 9,10 Besides, it must be pointed out that antiatherosclerotic effects are not merely exerted via alteration of plasma concentrations of LDL and other lipoproteins, but mitigation of LDL oxidation is an important mechanism for the prevention of atherosclerosis. Procyanidins possess antioxidant activity and have been shown by in vitro studies to inhibit chemically induced oxidation of LDL. 33 –35 Therefore, future studies are warranted to explore whether Pycnogenol can cause any reduction in circulating levels of oxidized LDL, oxidized fatty acids, and other pathogenic by-products of lipoprotein oxidation.
Aside from lack of control for dietary intake of lipids in the included studies, a number of other limitations need to be acknowledged for the present meta-analysis. First, although the present meta-analysis looked at the changes in lipid levels in a considerably larger population compared to single studies, it might not still be sufficiently powered due to the small number of studies included. Although the latter possibility cannot be excluded, it is worth noting that the observed effect sizes in the present analysis for the impact of Pycnogenol on plasma lipids were too small to be clinically relevant, even in case of possible statistical significance in larger populations. As another limitation, none of the studies assessed oxidized LDL as an important contributor in atherogenesis, which has been previously shown to be affected by Pycnogenol. 36 Most of the included studies did not provide exact details regarding concomitant lipid-lowering therapy; yet from the inclusion/exclusion criteria provided it is conceivable that lipid-lowering agents were not used in most of the included studies. The exception is the study by Enseleit et al in which the majority of patients were on statin treatment. However, sensitivity analyses (Table 2) revealed that exclusion of this study from the analyses does not change the overall effect size. Finally, interstudy variations regarding the applied inclusion criteria might have confounded the results.
Conclusion
In summary, results from this meta-analysis do not favor any clinical efficacy of supplementation with Pycnogenol in improving lipid profile. Along with the findings obtained for some other phytochemicals and herbal supplements, care should be taken in translating the lipid-modulating and cardioprotective properties of flavonoids observed in experimental studies into clinical practice. To obtain more conclusive evidence, future studies are warranted to explore the lipid-modifying effects of Pycnogenol in dyslipidemic populations, and plasma lipid levels need to be a priori designed as the primary outcome measure.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
