Abstract
Severe aortic stenosis due to calcification of the aortic valve is the most common indication for aortic valve replacement in the United States and Europe. The standard therapy for symptomatic patients with severe aortic stenosis is replacement of the valve. Some of the risk factors and pathophysiologic mechanisms in atherosclerosis play an important role in the development of calcific aortic stenosis. In the last few years, there have been an increased number of publications regarding the use of medications in order to delay the progression of aortic stenosis. These medications include statins, angiotensin-converting enzyme inhibitors, and biphosphanates. This article describes and summarizes some of the medical approaches that have emerged to alter the progression of aortic stenosis. Currently, only statins have been evaluated in randomized, placebo-control trials. Furthermore, statins have not proven to alter the progression of aortic stenosis. Ongoing randomized controlled trials with the use of angiotensin-converting enzyme inhibitors, statins, and biphosphonates will determine the use of these medications to delay the progression of aortic stenosis.
Calcific aortic valve stenosis (AS) is the most common reason for valvular replacement in the United States and Europe. 1 It is commonly associated with a trileaflet aortic valve in patients with risk factors for atherosclerotic disease 2 (Figures 1 and 2). Currently, 2% to 3% of the population greater than 75 years of age will develop AS. 2,3 Once symptoms develop, the prognosis is poor with a mortality rate of 25% per year if the valve is not replaced. 3 The standard therapy for symptomatic severe calcific AS is aortic valve replacement, which has a 2% to 5% surgical risk. In order to delay the progression of the disease, many different modifiable and nonmodifiable pathophysiologic mechanisms and risk factors have been identified. 4 The risk factors for atherosclerosis such as hyperlipidemia, hypertension, male gender, diabetes mellitus, and renal dysfunction play an important role in the development of both atherosclerosis and AS. 1,5 Early valve lesions of calcific AS resemble lesions of atherosclerosis, which contain inflammatory cells, deposition of atherogenic lipids, and calcification 6 (Figure 3).

Calcified aortic valve leaflets.

Calcified aortic valve leaflet (haematoxylin & eosin, original magnification ×200). Calcifications (C) of small vessels (V) with inflammation and extravasated erythrocytes (arrow).
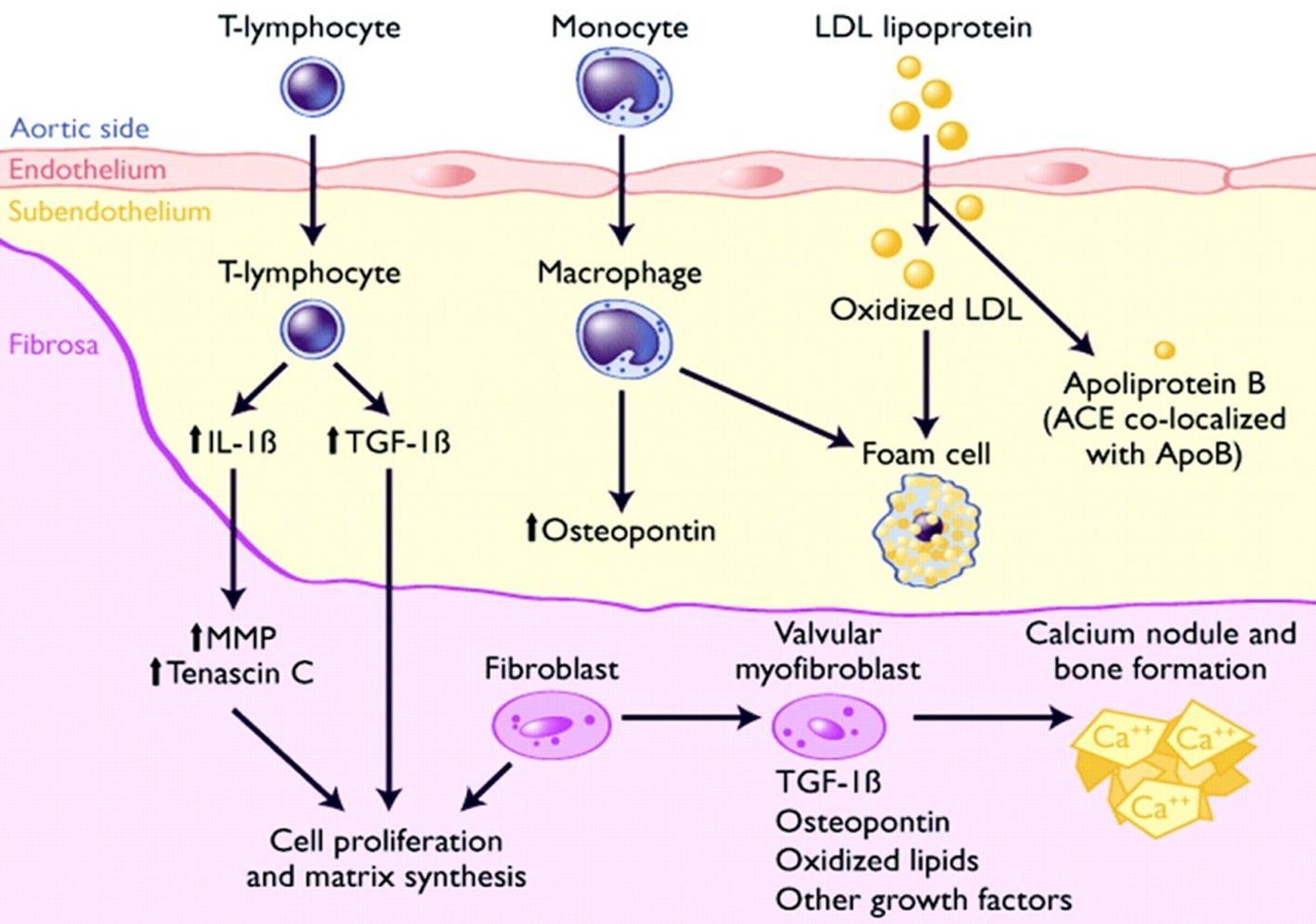
Potential pathways depicting calcific aortic valve disease. T lymphocytes and macrophages infiltrate endothelium and release cytokines that act on valvular fibroblasts to promote cellular proliferation and extracellular matrix remodeling. A subset of valvular fibroblasts within fibrosa layer differentiate into myofibroblasts that possess characteristics of smooth muscle cells. Low-density lipoprotein (LDL) that is taken into the subendothelial layer is oxidatively modified and taken up by macrophages to become foam cells. Angiotensin-converting enzyme (ACE) is colocalized with apolipoprotein B (ApoB) and facilitates conversion of angiotensin II (Ang II), which acts on angiotensin 1 receptors (AT-1R), expressed on valvular myofibroblasts. A subset of valvular myofibroblasts differentiate into osteoblast phenotype that is capable of promoting calcium nodule and bone formation. IL indicates interleukin; TGF, transforming growth factor; MMP, matrix metalloproteinases. Reproduced with permission from Freeman et al. 23
Multiple trials using medications that may slow the progression of the disease have been completed in the last few years. 7,8 The purpose of this article is to describe some of the different approaches that have emerged as potential medical therapies to alter the course of the progression of AS.
Effects of Statins on Calcific AS
A clear relationship between cholesterol levels and the progression of AS has not been established. 9 However, the use of statins to delay the progression of AS has generated much interest due to the pathologic similarities of early atherosclerotic lesions and the earliest lesions of degenerative aortic valve disease. The early lesion of calcific AS is an active inflammatory process with a similar pathophysiological mechanism to atherosclerosis (deposits of calcium and atherogenic lipoproteins) and prominent mineralization with presence of smooth muscle cells. 10
Retrospective case series have demonstrated contradictory findings when compared with randomized prospective trials regarding the role of statins in preventing the progression of AS. 11 Novaro et al, 5 retrospectively studied 174 patients with mild to moderate AS, of which 57 patients took statins. They found a diminution in the aortic valve area (AVA) of −0.06 ± 0.16 cm2/year in the statin group compared with −0.11 ± 0.18 cm2/year in the nonstatin group, P = .03. In another study of 180 patients with coronary risk factors for statins who had mild AS, the patients who took statins had a reduction in the increase of the peak aortic systolic gradient, P = .0098. 12
In a prospective, nonrandomized study by Bellamy et al, 13 38 of 156 patients took either simvastatin or lovastatin, with a follow-up of at least 6 months. They demonstrated that even though changes in the cholesterol levels did not correlate with the progression of AS, patients who received statins had a slower reduction in the AVA, of 0.04 ± 0.15 cm2/year compared with −0.09 ± 0.17 cm2/year in the placebo group, P < .01.
The Rosuvastatin Affecting Aortic Valve Endothelium (RAAVE) trial 14 was an open-label prospective study of 121 patients with moderate-to-severe asymptomatic AS that compared the effects of rosuvastatin (61 patients) versus placebo, with a follow-up of 18 months. A reduction in the AVA of −0.05 ± 0.1 cm2 was noted in the rosuvastatin group compared with −0.10 ± 0.1 cm2 in the placebo group (P = .041) and an increase in the peak aortic valve velocity of +0.24 ± 0.3m/s per year in the placebo group compared with +0.04 ± 0.38 m/s per year in the rosuvastatin group (P = .007).
These promising results led to several randomized prospective trials. The Scottish Aortic Stenosis and Lipid Lowering Trial, Impact on Regression (SALTIRE) was a double-blind prospective study of 155 patients with moderate-to-severe AS. 15 The patients were randomized to receive atorvastatin 80 mg (77 patients) or placebo and were followed for 25 months. The results showed no significant difference between the 2 groups in the peak aortic jet velocity or in the progression in valve calcification as measured by helical computed tomography. This study demonstrated that atorvastatin does not regress nor stop the progression of AS.
The Simvastatin and Ezetimibe in Aortic Stenosis (SEAS) trial, 16 a double-blind trial, compared simvastatin and ezetimibe with placebo in patients with mild-to-moderate AS. It consisted of 1873 patients, of which 944 belonged to the study group, and they were followed for 52 months. There were no differences in the peak aortic jet velocity or AVA noted between the 2 groups. However, there was a reduction in the incidence of ischemic cardiovascular events with the use of statins.
Finally, the most recently published trial is the Aortic Stenosis Progression Observation: Measuring Effects of Rosuvastatin (ASTRONOMER). 17 This double-blind prospective trial randomized 269 asymptomatic patients with mild-to-moderate AS with no indications for lipid-lowering agents to receive 40 mg/d of rosuvastatin (134 patients) or placebo (135 patients) for 3.5 years. There were no significant differences noted in the transaortic gradients or AVA in the rosuvastatin group compared with placebo (Table 1).
Nonrandomized and Randomized Prospective Trials Utilizing Statins in Patients With Aortic Stenosis
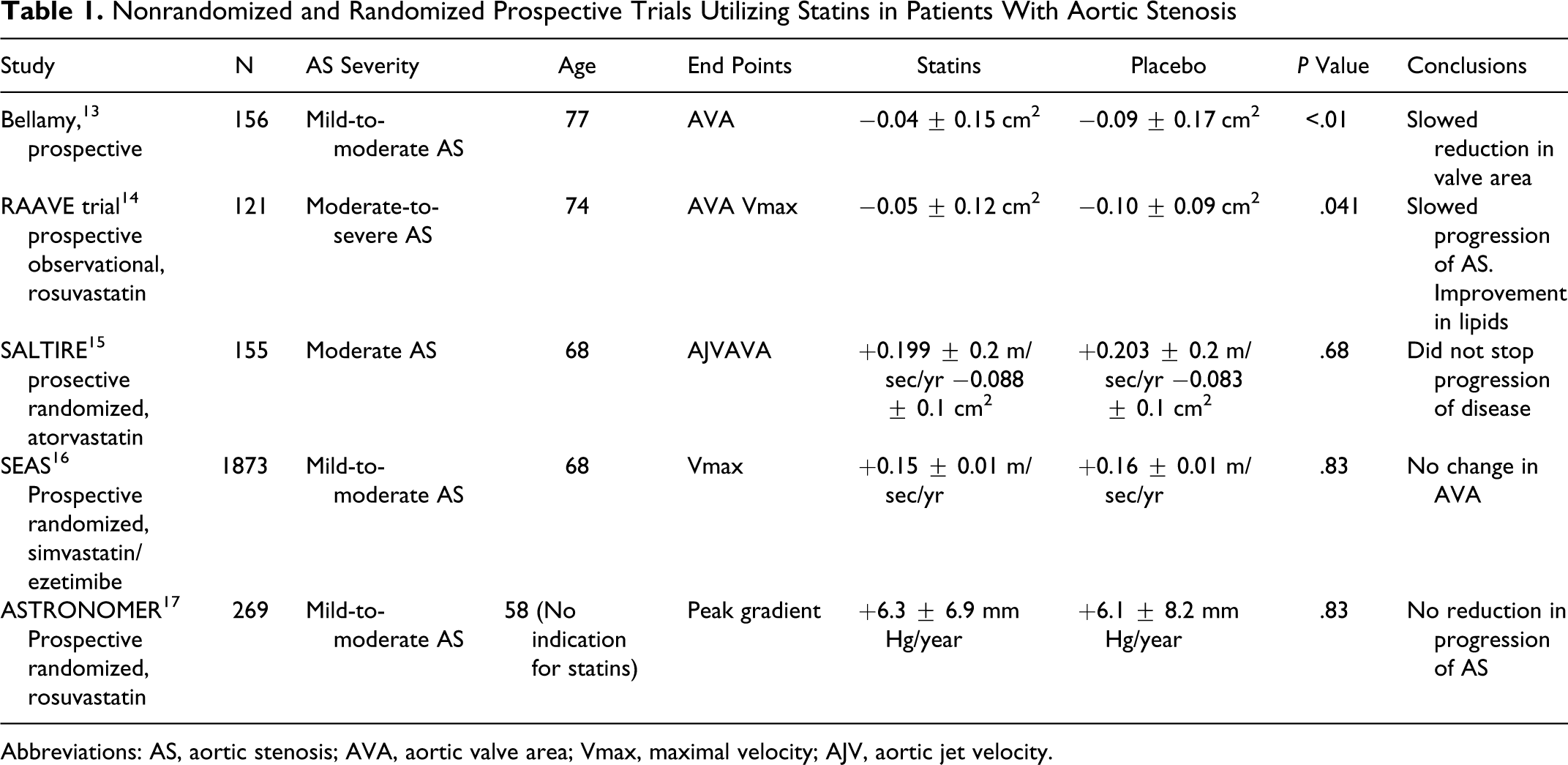
Abbreviations: AS, aortic stenosis; AVA, aortic valve area; Vmax, maximal velocity; AJV, aortic jet velocity.
Ongoing Trials With Statins
Currently there is no sufficient evidence to support the use of statins to delay the progression of calcific AS. The Effect of Statin Therapy on the Progression of Calcific Valvular Aortic Stenosis (STOP AS) trial 18 will evaluate the effects of atorvastatin (40 mg/d) in patients with mild-to-moderate AS. The study is an open-label trial that uses a historical group as a control group. It will measure the rate of change in the AVA at a 2-year follow-up period. Another ongoing trial is the Efficacy of Fluvastatin on Inflammatory Markers in the Hemodynamic Progression of Degenerative Aortic Stenosis (AORTICA). 19 This is a double-blind randomized study that proposes that fluvastatin (80 mg/d) can decrease inflammatory markers in patients with AS and halt the progression of the disease. A third study is the Statin Therapy in Asymptomatic Aortic Stenosis (STAAT) trial, 20 which is a double-blind study that will evaluate the progression of AS in asymptomatic patients with mild-to-moderate AS. It will randomize patients to fluvastatin (40 mg/d) or placebo and will have a median follow-up of 18 months.
Effects of Angiotensin-Converting Enzyme Inhibitors
Angiotensin-converting enzyme (ACE) protein and angiotensin II are present in the aortic valve leaflets in patients with AS. Angiotensin-converting enzyme inhibitors have been shown to suppress ventricular fibrosis and inhibit angiotensin II type 1 receptor in the cardiomyocytes and therefore decrease systolic and diastolic dysfunction in patients with left ventricular hypertrophy and AS. 21
Rosenhek et al 22 showed that the use of ACE inhibitors did not change the hemodynamic progression of AS in 211 patients with moderate AS after at least 6 months of treatment. However, this study demonstrated a significant reduction in the progression of AS in patients taking a combination of ACE inhibitors with statins (0.10 ± 0.4 m/s per year peak aortic jet velocity) compared with patients taking ACE inhibitors alone (0.39 ± 0.4 m/s per year, peak aortic jet velocity). Angiotensin-converting enzyme inhibitors did not provide additional benefit to slow the progression of the valvular disease. 24
Another retrospective study by Obrien et al 25 of 123 patients evaluated the aortic valve calcium score by electron beam computed tomographic scans. Of the 123 patients, 43 were taking ACE inhibitors. There was an absolute median change rate reduction in aortic valve calcium score of 25.1%/year versus 12.2%/year, P = .02, in the patients taking ACE inhibitors compared with patients that did not, respectively.
At the present time, there are no published randomized prospective studies using ACE inhibitors to delay the progression of calcific aortic stenosis. There is, however, published data showing a favorable hemodynamic response with the use of ACE inhibitors in patients with preserved left ventricular function and mild-to-moderate AS and also in patients with hypertension and moderate-to-severe AS. The afterload reduction effect of the ACE inhibitors is compensated by an incremental increase in the transaortic pressure gradient with preserved or even higher diastolic pressure resulting in no evidence of limited coronary flow. 26,27 A randomized double-blind prospective trial using captopril and trandolapril compared with placebo, to determine whether ACE inhibitors improve acute hemodynamic parameters in patients with severe symptomatic AS is ongoing. 28 There is also a trial assessing the effects of an angiotensin II receptor blocker on AS. This is an ongoing double-blind placebo-controlled prospective study entitled, The Potential of Candesartan to Retard the Progression of Aortic Stenosis (ROCK-AS), which will analyze the degree of inflammation, calcification, lipid accumulation, and fibrosis in patients taking candesartan compared with placebo. 29
Use of Bisphosphonates in Aortic Stenosis
The inflammatory process in AS includes osteoblast expression, cell proliferation, and calcification of the valve, with deposits of hydroxyapatite, osteopontin, and other proteins submerged in a bone-like collagen matrix. 4 There is evidence of osteoblast bone formation in the calcific aortic valve. 7 The presence of osteoblast-like cellular phenotype and upregulated receptors (present in normal bone formation-signaling pathway) may indicate that the valvular cells potentially differentiate into bone-forming cells. 30
Studies have linked the loss of bone-mineral density with progressive vascular and valvular calcification through different mechanisms of ossification. 31 It is well recognized that osteoporosis and aortic stenosis share certain associated conditions including dyslipidemia, estrogen deficiency, chronic inflammation, and abnormalities of vitamin D metabolism. 32 Studies on vitamin D receptor polymorphism have shown that individuals with alterations of calcium homeostasis (parathormone, proteins, and second messengers) might trigger calcification of extraosseus structures, such as the aortic valve. 33
Bisphopshonates inhibit bone turnover by blocking farnesyl pyrophosphate synthetase in the 3 hydroxy 3 methylglutaryl CoA reductase pathway. This impedes the formation of farnesyl pyrophosphate and geranylgeranyl pyrophosphate, both of which are precursors for the formation of prenylated proteins. These molecules are essential in the binding of proteins for the formation of the osteoclast cell membrane. It is important to mention that bisphosphonates share some similarity with statins when inhibiting the 3 hydroxy 3 methylglutaril CoA reductase pathway in the production of cholesterol 35 (Figure 4).

Schematic representation of the mevalonate pathway and the effects of nitrogen-containing bisphosphonates. Copyright 2004 by ALPHAMED PRESS, INC. Reproduced with permission of ALPHAMED PRESS, INC. 34
In addition to inhibiting the mevalonate pathway, bisphosphonates can prevent the deposition of calcium in bioprosthetic porcine aortic valve cusps and aortic wall. Rapoport et al 36 reported that the concomitant use of bisphosphonates and triglicydil amine showed no significant calcification in long-term subdermal implanted bioprosthetic porcine aortic valves in rats. They demonstrated that pretreatment with bisphosphonates can potentiate the anticalcification efficacy of triglicydil amine cross-linked bioprosthetic valves likely due to reduction in alkaline phosphatase activity. The bisphosphonates also inhibit in vitro bone nodule formation and induce osteoblast apoptosis, as well as interfere with bone alkaline phosphatase in the osteogenic process. 37
Skolnick et al 38 retrospectively evaluated 55 patients with AS, at an average of 2.4 years, who were on medical therapy for osteoporosis. Of the 55 patients, 18 were receiving osteoporosis medications; 12 were on bisphosphonates, and the remainder on calcitonin or selective estrogen receptor modulators. The reduction in AVA was significantly less in the 18 patients receiving osteoporosis drugs compared with the 37 patients who were not being treated. At follow-up, the AVA had decreased by −0.10 cm2 in the osteoporosis-treated group, and −0.22 cm2 in the untreated group (P = .025). In multivariable analyses, only the osteoporosis drug therapy emerged as being independently related to the delay of AS progression (P = .043).
Other Studies
Future randomized prospective studies could bring answers about the use of agents such as statins, bisphosphonates, ACE inhibitors, and angiotensin receptor blockers as a relatively safe way to decrease the progression of the disease. There are ongoing trials evaluating the use of calcification-inhibiting agents such as phytate (inositol 6-phosphate) in order to delay the progression of cardiovascular calcification. 39 A German trial is going on to assess the role of vitamin K supplementation as a matrix protein activator to slow the progression of valvular calcification. This trial is not yet recruiting patients. 40
Conclusions
As the pathophysiology of calcific AS becomes elucidated, new treatment alternatives may be developed. At the present time, there is no conclusive evidence that medical therapy slows the progression of aortic valve stenosis. Surgery is still the standard therapy in the management of symptomatic calcific AS. Answers may arise in the next few years that will guide the medical prevention of progression of calcific AS.
Footnotes
The author(s) declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
