Abstract
Background:
Heart rate (HR) reduction with ivabradine has been proved to reduce hospitalization and death from heart failure (HF). We sought to investigate whether pyridostigmine would effectively reduce HR in patients with chronic HF as compared with ivabradine.
Methods:
Twenty-one patients with HF who were in sinus rhythm with a resting HR over 70 bpm, despite optimal medical treatment, were included in a randomized, double-blind study comparing pyridostigmine versus ivabradine. The initial dose of ivabradine was 5 mg twice daily to reach a target HR between 50 and 60 bpm and could be titrated to a maximum of 7.5 mg twice daily. Pyridostigmine was used in a fixed dose of 30 mg 3 times daily.
Results:
The baseline HR for ivabradine and pyridostigmine groups was 89.1 (13.5) and 80.1 (7.2) bpm, respectively (P = .083). After 6 months of treatment, HR was significantly reduced to 64.8 (8.3) bpm in the ivabradine group (P = .0014) and 63.6 (5.9) bpm in the pyridostigmine group (P = .0001). The N-terminal pro-B-type natriuretic peptide was reduced in the ivabradine group (median: 1308.4 [interquartile range: 731-1896] vs 755.8 [134.5-1014] pg/mL; P = .027) and in the pyridostigmine group (132.8 [89.9-829] vs 100.7 [38-360] pg/mL; P = .002). Inflammatory markers interleukin-1, interleukin-6, and tumor necrosis factor were reduced in both groups. Exercise capacity was improved in both groups, with increments in volume of oxygen utilization (
Conclusion:
Both drugs significantly reduced HR, with improvements in exercise capacity and in neurohormonal and inflammatory profiles.
The treatment of heart failure (HF) has improved substantially with the introduction of angiotensin-converting enzyme (ACE) inhibitors, mineralocorticoid receptor antagonists, and β-blockers. 1 -3 Among many effects related to β-blockers, the reduction in heart rate (HR) has been recognized for a long time. Although many patients with HF take β-blockers, some of them remain with a HR above 70 bpm, despite maximum doses of these medications. Since resting HR in HF is related to increased cardiovascular risk, 4 it is clinically relevant to search for alternatives to reduce HR.
In the Systolic Heart failure treatment with the If inhibitor ivabradine Trial (SHIFT), 4 ivabradine was compared with placebo in patients with HF and HR above 70 bpm despite optimal medical treatment. Ivabradine use reduced HR and was associated with improved outcomes, defined as cardiovascular death or hospital admission for HF. Thus, the SHIFT trial confirmed the important role of HR in the pathophysiology of HF.
Pyridostigmine is a reversible cholinesterase inhibitor and thus is an indirect cholinergic agonist that has been proved to enhance vagal tonus both in normal patients 5,6 and patients with HF. 7,8 As a consequence, pyridostigmine reduces HR and increases HR variability. Pyridostigmine has been in the market for a long period of time and is a less expensive option.
In the SHIFT trial, the benefits of ivabradine regarding the primary outcome were driven mainly by a reduction in HF hospitalizations and no significant effects were observed on either all-cause mortality or cardiovascular mortality. The parasympathetic modulation observed with pyridostigmine might have additional effects beyond those observed with a selective HR reduction, with possible benefits on mortality. Given this background, we hypothesized that the use of pyridostigmine in patients with HF and persistent elevated resting HR despite optimal medical treatment would lead to similar HR reduction as in those observed with ivabradine, resulting in improved functional capacity and neurohormonal profile which would support a new pathway in the treatment of HF.
Methods
Enrollment and Study Design
We designed a randomized, double-blind, double-dummy study comparing ivabradine with pyridostigmine in patients with chronic HF. Inclusion criteria were the presence of overt HF, sinus rhythm, ejection fraction less than 50% as assessed by echocardiography (Simpson method), and resting HR over 70 bpm despite optimal medical treatment, including maximum tolerated doses of β-blockers. Exclusion criteria were patients with pacemakers, serum creatinine level higher than 3 mg/dL, acute myocarditis, active myocardial ischemia, asthma, glaucoma, urinary obstruction, thyroid dysfunction, and patients expected to be submitted to myocardial revascularization or device implantation in the next 6 months. Patients were already being followed for at least 1 year at the HF clinic of our hospital and were on stable doses of HF medication, with no change in medication in the last 3 months. Resting HR was assessed at the bedside, after an at least 5-minute rest, on 2 consecutive visits before randomization. Our study complies with the Declaration of Helsinki, and the ethics committee of our hospital approved the study protocol. Eligible patients provided a consent form after receiving verbal and written information about the study. The study was registered at Plataforma Brasil (http://plataformabrasil.saude.gov.br), number 01824312.5.0000.5243.
Drug Regimens
Patients were randomized via online software (www.randomization.com) to one of the study drugs. The initial dose of ivabradine was 5 mg twice daily to reach a target HR between 50 and 60 bpm and could be either titrated to a maximum of 7.5 mg twice daily or reduced to 2.5 mg twice daily. Pyridostigmine was used in a fixed dose of 30 mg 3 times daily. The medications were dispensed monthly, after the medical visit, by a coinvestigator. The intervals between doses of the 2 drugs were different (ivabradine was administered twice daily and pyridostigmine 3 times daily). Therefore, to maintain the double blindness of the study, a second placebo medication, in the opposite dosage of the real drug, was necessary in each of the groups (double-dummy).
Follow-Up
Medical visits took place every month until study closure. The primary investigator, who was blinded to the randomization plan, provided all the medical treatment during the study. At each follow-up visit, the primary investigator could maintain the study drug dose or adjust the dose according to HR. The recommendation to adjust drug dose was passed on to the coinvestigator responsible for drug dispensation, who was the only investigator aware of the randomization plan. Data were assessed before and after 6 months of treatment with one of the drugs.
Patients’ Evaluation
Quality of life was assessed using the Minnesota Living With Heart Failure Questionnaire. The N-terminal pro-B-type natriuretic peptide (NT-proBNP) was analyzed in whole blood, within 6 hours after collection. The dosage of NT-proBNP was made by the Elecsys system (Roche, Basel, Switzerland). The detection range of the system is 5 to 35 000 pg/mL. Inflammatory markers (interleukin 1 [IL-1], IL-6, and tumor necrosis factor [TNF]) were measured in blood samples, which were centrifuged and frozen in a freezer at −80°C. Dosages were performed using enzyme-linked immunosorbent assay in EMD Millipore assays (Merck KGaA, Darmstadt, Germany).
Two experienced echocardiographers, without previous knowledge of the results of the other tests, using the Siemens Acuson Cypress 20 Cardiovascular System (Munich, Germany), performed transthoracic echocardiograms. Left ventricular ejection fraction (LVEF) was calculated according to the Simpson method. The examinations were performed according to the recommendations of the European Association of Echocardiography and the American Society of Echocardiography.
Patients underwent a maximal cardiopulmonary exercise test on the treadmill using the MedGraphics (MGC) VO2000 metabolic analyzer (Imbrasport, Porto Alegre, Rio Grande do Sul State, Brazil), together with the Ergo PC Elite 13 system and Centurion 300 treadmill (Micromed, Brasília, DF, Brazil). The gas analyzer was precalibrated before each test by the Autocal system in a ventilated setting. The peak volume of oxygen utilization (
Cardiac sympathetic activity was assessed using 123-iodine-metaiodobenzylguanidine (123I-MIBG) myocardial scintigraphy. Early and late myocardial anterior planar images were, respectively, acquired at 30 minutes and 4 hours after the radiotracer infusion. Derived from the scintigraphic planar images, semiquantitative myocardial 123I-MIBG uptake and washout reflected functional/structural cardiac innervation and sympathetic tone, respectively.
Bioelectrical impedance vector analysis was used to assess congestion. This method utilizes the EFG Renal software (Akern, Pontassieve, Florence, Italy) for estimating the parameters of resistance, reactance, and phase angle. Then, the hydration index (HI) is calculated to estimate total body water. The normal HI range is 72.7% to 74.3%. Values above this range indicate congestion, and values below this cutoff indicate dehydration.
Statistical Analysis
Data are presented as mean (standard deviation; SD), except for variables with non-Gaussian distribution, for which median and interquartile ranges are provided. Categorical variables were analyzed using the χ2 test or McNemar test. For comparison of numerical data, Student t test for independent samples or the Mann-Whitney U test (nonparametric) was used. The homogeneity of the variance was tested by the Levene test. In the comparisons between the same individual, the paired Student t test or the Wilcoxon test was used for variables with or without normal distribution, respectively. Nonparametric methods were used, since some variables did not present normal distribution, due to the great dispersion and rejection of the normality hypothesis according to the Kolmogorov-Smirnov test. In the analysis of the inflammatory cytokines, in the cases of non-Gaussian distribution, the logarithmic transformation was made for the analysis. Heart rate within groups was compared using repeated-measures analysis of variance. To verify the percentage differences between the baseline and the end of 6 months of intervention, the delta variation (Δ%) reported as median and interquartile range was used and the Mann-Whitney U test was performed. The criterion for determining significance was 5%. Statistical analysis was performed by SAS System, version 6.11 (SAS Institute, Inc, Cary, North Carolina).
Results
Baseline Characteristics and Medications Adjustments in the Follow-Up
The study flowchart is depicted in Figure 1. Four hundred nine patients with HF were screened and 19 patients were included in the final analysis. Baseline characteristics are shown in Table 1. Despite the randomization, more men were allocated to the ivabradine group. At baseline, all patients were taking carvedilol (mean daily dose 47.3 [7.8]), 12 (57%) were taking ACE inhibitors, 9 (43%) were taking angiotensin receptor blockers, 19 (90.5%) were taking spironolactone, 17 (81%) were taking furosemide, 8 (38%) were taking a combination of isosorbide dinitrate and hydralazine, and 5 (24%) were taking digoxin. There was no change in the baseline medication regimen during the study, except for suspension of diuretics in 3 patients after 3 months of treatment with the study drugs (2 in the ivabradine group and 1 in the pyridostigmine group). In addition, daily doses of furosemide were reduced from 80 to 40 mg in 4 patients (2 in the ivabradine group and 2 in the pyridostigmine group). In 2 patients, the ivabradine dose was increased to 7.5 mg twice daily, and in 1 patient, it was reduced to 2.5 mg twice daily.

Screening and randomization.
Baseline Characteristics.

Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin-receptor blockers; BIVA, bioelectrical impedance vector analysis; LV, left ventricle; NT-proBNP, N-terminal pro-B-type natriuretic peptide; NYHA, New York Heart Association.
a Data presented as median and interquartile range.
Effects on HR
The baseline HR for the ivabradine and pyridostigmine groups was 89.1 (13.5) and 80.1 (7.2) bpm, respectively (P = .083). As depicted in Figure 2, in the first month of follow-up, there was a decrease in HR in both groups, although the magnitude of the decrease was greater in the ivabradine group (mean reduction of 24 vs 8 bpm; P = .01). After 6 months of treatment, HR was significantly reduced to 64.8 (8.3) bpm in the ivabradine group (P = .0014 as compared with baseline) and to 63.6 (5.9) bpm in the pyridostigmine group (P = .0001).
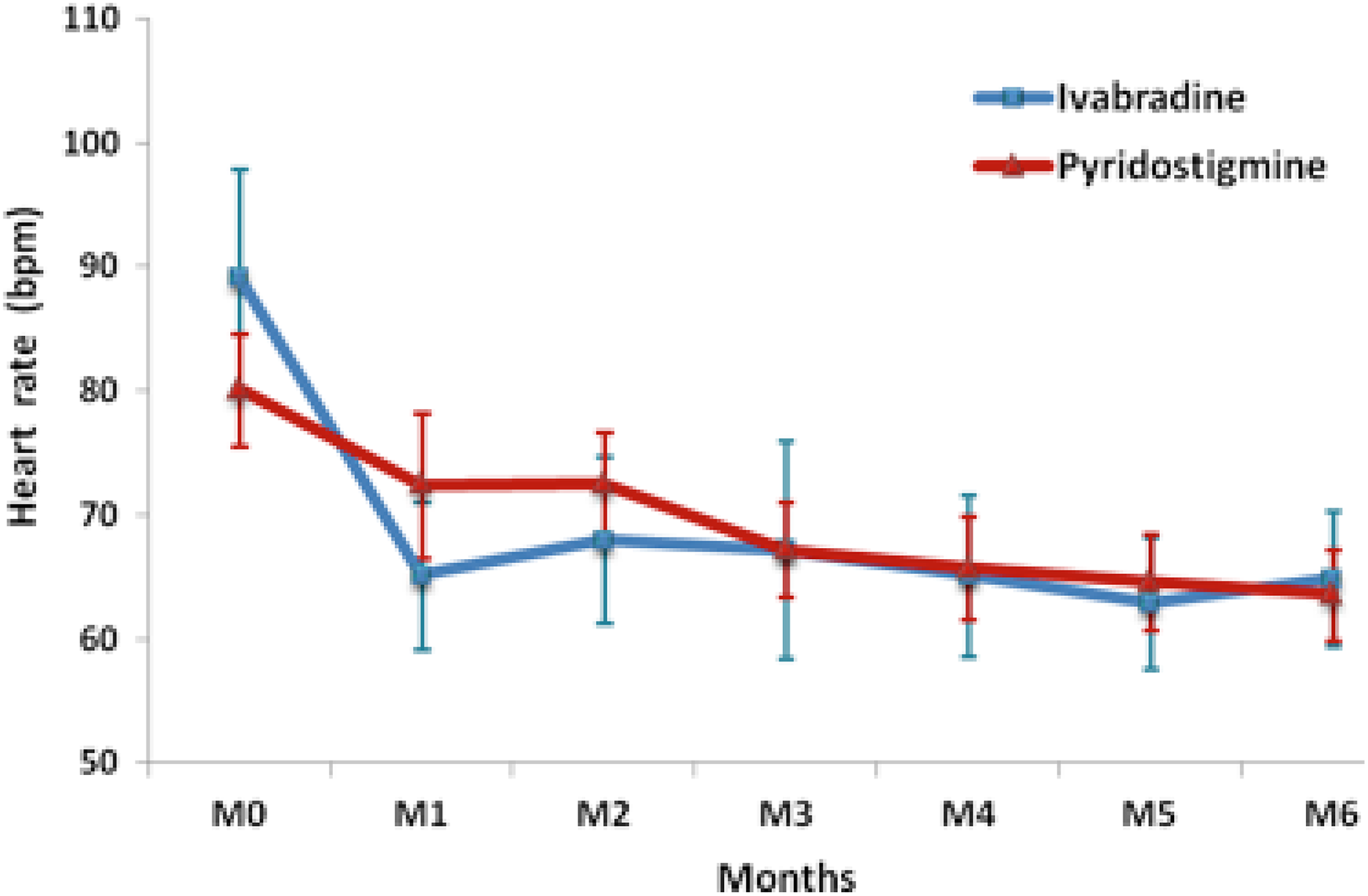
Mean heart rate during the study, by allocation group.
Group Comparisons
The comparison between group treatments is shown in Table 2. Both groups showed a reduction in natriuretic peptides (Figure 3) and inflammatory markers and an increase in peak
Effects on Clinical, Echocardiographic, Neurohormonal, and Inflammatory Variables.
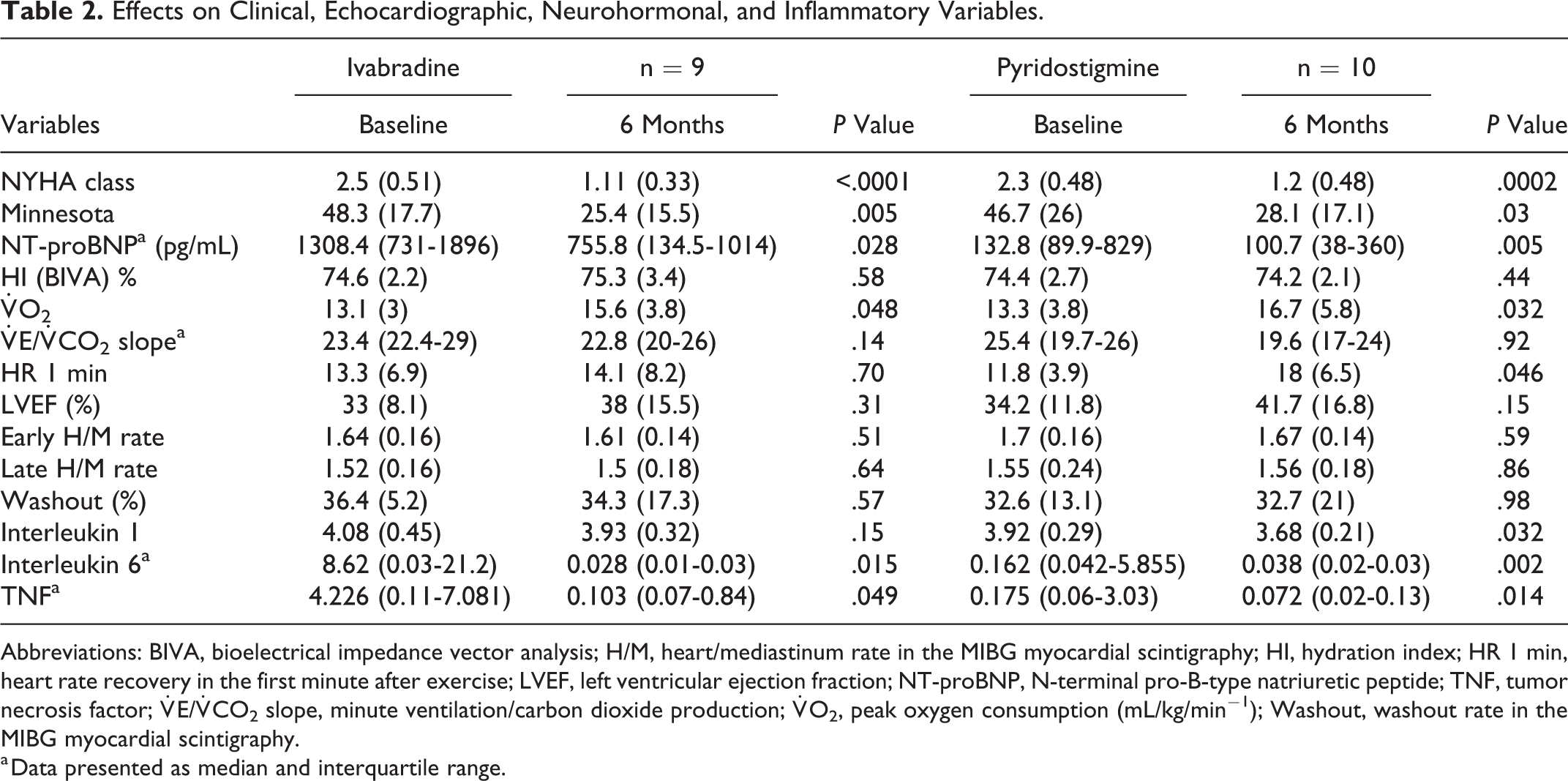
Abbreviations: BIVA, bioelectrical impedance vector analysis; H/M, heart/mediastinum rate in the MIBG myocardial scintigraphy; HI, hydration index; HR 1 min, heart rate recovery in the first minute after exercise; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal pro-B-type natriuretic peptide; TNF, tumor necrosis factor;
a Data presented as median and interquartile range.

The NT-proBNP levels before and after drug use (P = .028 for ivabradine group and P = .005 for pyridostigmine group as compared with baseline). NT-proBNP indicates N-terminal pro-B-type natriuretic peptide.

Heart rate recovery in the first minute after exercise according to study drug.
Delta Variation (Δ%) Between Groups.a

Abbreviations: BIVA, bioelectrical impedance vector analysis; H/M, heart/mediastinum rate in the MIBG myocardial scintigraphy; HR 1 min, heart rate recovery in the first minute after exercise; IQ, interquartile range; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal pro-B-type natriuretic peptide; TNF, tumor necrosis factor;
a Data presented as median and interquartile range.
In general, both drugs were well tolerated. There was one case of asymptomatic bradycardia in one patient randomized to the ivabradine group, and it was resolved with a dose reduction to 2.5 mg twice daily. Three patients in the pyridostigmine group had diarrhea. In 2 cases, there was complete remission in a few days, and in one case, the patient withdrew consent to study participation. No deaths occurred during follow-up, and one patient in the ivabradine group was hospitalized due to HF decompensation.
Discussion
Our results demonstrate that pyridostigmine is able to reduce HR in patients with chronic HF, compared with the standard drug for this purpose, ivabradine. Reduction of HR by parasympathetic modulation was as effective as selective reduction of HR, although the magnitude of HR decrease was greater with ivabradine. This reduction was accompanied by improvement in exercise capacity and reduction in neurohormonal and inflammatory markers, confirming the hypothesis of the study. However, no effects were observed in cardiac sympathetic activity as assessed by 123I-MIBG cardiac scintigraphy.
The reduction of HR with ivabradine was very pronounced in the first month. This most likely results from the mechanism of action of the drug, acting directly on ion channels of the sinus node. The reduction in HR with pyridostigmine was significant, but it occurred more smoothly over time, reaching the HR of the ivabradine group only in the third month of treatment. This is expected because the action of pyridostigmine depends on the modulation of the autonomic system, and not on direct action on the sinus node, which requires time, as observed with sympathetic modulation. It is important to note that, in the same way as Serra et al, 8 we demonstrated that pyridostigmine improves recovery HR 1 min, an effect not observed with ivabradine. This variable is linked to parasympathetic modulation and is considered an important prognostic factor and can be used as a therapeutic end point. 9 This finding demonstrates that ivabradine reduces resting HR, but does not act on postexertion HR, which depends on vagal modulation.
The effects of HR reduction with ivabradine in patients with HF were already known. Its use promotes improvement in cardiac function and hemodynamics, 10,11 reduction in natriuretic peptides, 12 and improvement in exercise capacity, 12 with the clinical result being a reduction in mortality and hospitalizations for HF. 4 The new finding in the present study was to demonstrate that some of these benefits also occur with the use of pyridostigmine. The reduction of HR alone explains the benefits observed with pyridostigmine, but other mechanisms may be involved as a result of parasympathetic modulation. Pyridostigmine inhibits the enzyme acetylcholinesterase, with a consequent increase in the release of endogenous acetylcholine and activation of the muscarinic receptors in the cardiac muscle, resulting in increased parasympathetic activity. 13 There is abundant experimental material in the literature suggesting benefits in the parasympathetic modulation in cardiovascular diseases, including HF. In rats, the use of pyridostigmine has been shown to improve autonomic function following acute myocardial infarction, 14 and pyridostigmine also reduces postinfarction inflammation. 15,16
Vagal action has also been reported in numerous experimental studies in HF. 17 -21 Cholinergic activity inhibits the remodeling in the heart of rats submitted to stress with angiotensin II, 17 and conversely, the reduction of cholinergic activity promotes cardiac remodeling and HF. 18 Interestingly, in the latter study, administration of pyridostigmine for 2 weeks reversed the HF phenotype. Finally, the inhibition of the enzyme acetylcholinesterase with donepezil, a medication used to treat Alzheimer disease, prolonged survival in rats with chronic HF. 19 -21
Serra et al evaluated the effects of pyridostigmine for 48 hours before performing dynamic exercise in patients with chronic HF. 8 They demonstrated that the use of pyridostigmine improved reserve HR, recovery HR at the first minute after exertion, and increased oxygen pulse, an indicator of ventricular systolic volume. Taken together these data suggest that parasympathetic modulation may play a role in the therapy of patients with HF, deserving further studies in this area. Due to parasympathetic modulation, it is possible that pyridostigmine has greater effects on clinical outcomes than observed with ivabradine.
Pyridostigmine has been used for a long time in the treatment of myasthenia gravis, with a safe profile. 22 -24 In one study, after a 9.6-year follow-up period, only one patient had to stop pyridostigmine therapy, because of stomach complaints. 22 Common adverse effects with pyridostigmine, mostly mild, are due to cholinergic stimulation of muscarinic (smooth muscle or autonomic glands) or nicotinic receptors (skeletal muscle). The most frequent muscarinic adverse events are gastrointestinal disorders, mainly due to gut hypermotility, including stomach cramps, nausea, vomiting, and diarrhea. Three of our patients had diarrhea with pyridostigmine use. It disappeared spontaneously in 2 patients within a few days and another patient decided to stop the drug and withdrew informed consent. In our study, the dose of pyridostigmine was lower than that used in the treatment of myasthenia gravis. As a consequence, the side effects were infrequent and mild.
The main limitation of the present study is the small number of patients included. This may have caused β-type errors, due to lack of study power, and may have interfered with the homogeneity of the baseline characteristics between the groups. As a matter of fact, NT-proBNP, IL-6, and TNF were higher in ivabradine than in pyridostigmine group suggesting that, as a play of chance, more severe patients were randomized to ivabradine group. However, we believe this may have favored ivabradine, since sicker patients have more room for improvement. We screened more than 400 patients, but only 64 fulfilled the inclusion criteria. This difficulty arises from the fact that most patients with HF using β-blockers already have HR below 70 bpm. Due to these limitations, our results should be considered preliminary data and need to be confirmed in bigger trials.
Conclusion
This is a unique study in the medical literature. For the first time, pyridostigmine was used for 6 months in patients with HF. The study confirmed the known benefits of ivabradine and raised favorable hypotheses regarding pyridostigmine. The preliminary data presented here add information to the short-term beneficial effects demonstrated in previous studies with pyridostigmine and support future studies with a greater number of patients using pyridostigmine in patients with chronic HF.
Footnotes
Author Contributions
A.S.V. and H.V. contributed to conception or design; contributed to acquisition, analysis, or interpretation; and drafted the manuscript. J.A.C., B.C.P., P.B.P., L.U.R., M.N., A.R.X., S.K., and C.T.M. contributed to acquisition, analysis, or interpretation. A.C.L.d.N contributed to conception or design. All authors critically revised manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Foundation for Research Support of the State of Rio de Janeiro (Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro—FAPERJ) [E-26/110.529/2012].
