Abstract
Aim:
To investigate the mechanism of rosuvastatin in preserving cardiac function after acute myocardial infarction (AMI) in a rat model.
Methods:
Sprague-Dawley rats were randomized to receive either rosuvastatin (5 mg/kg every day) or placebo (0.5% CMC-Na), respectively, by daily gavage from 7 days before AMI. Acute myocardial infarction (AMI) model was induced by left anterior descending coronary artery ligation through a lateral thoracotomy in rats. The expressions of stromal-cell-derived factor 1 (SDF-1), chemokine motif CXC receptor 4 (CXCR-4), vascular endothelial growth factor (VEGF), and intercellular adhesion molecule 1 (ICAM-1) in peri-infarction region and nonischemic region at different time points were determined by the Western blot analysis. Immunohistochemistry analysis was performed on the 28th day after AMI to investigate the accumulation of CD90+, CD133+, and c-kit+ progenitor cells in the peri-infarction region. Masson staining and echocardiograph were performed to evaluate the left ventricular remodeling and postinfarction cardiac function 4 weeks after AMI.
Results:
Western blot analysis showed that rosuvastatin could change the cytokine expressions in the peri-infarction region by upregulating the SDF-1 expression and downregulating the expressions of CXCR-4, ICAM-1, and VEGF in 4 to 14 days after AMI. Immunohistochemistry analysis showed that rosuvastatin treatment was associated with increased accumulation of CD90+, CD133+, and c-kit+ progenitor cells in the peri-infarction region. Masson staining and echocardiograph confirmed that rosuvastatin could attenuate left ventricular remodeling and improve postinfarction systolic function.
Conclusion:
The data suggest that rosuvastatin can protect the heart from ischemic injury and preserve the cardiac function in rats in vivo. The changing expressions of SDF-1, CXCR-4, ICAM-1, and VEGF, and the accumulation of progenitor cells were involved in this process.
Introduction
Postinfarction heart failure is one of the major causes of death and disability in the developed world, with a dismal 5-year mortality rate of 50% to 70% in symptomatic patients. 1 Recent study showed that the ischemic myocardium could express several cytokines that play an important role in tissue repair and heart remodeling. 2 Gene expression analysis after myocardial infarction (MI) revealed increased levels of several genes including stromal-cell-derived factor 1 (SDF-1), vascular endothelial growth factor (VEGF), intercellular adhesion molecule 1 (ICAM-1), and so on. 3,4
Statins are 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA) reductase inhibitors, extensively used to lower plasma cholesterol levels. Additionally, statins can protect the heart against ischemic injury and preserve the postinfarction function through mechanisms independent of their lipid-lowering potencies. 5 –9 However, the mechanisms of the statins, especially rosuvastatin, in protecting tissues against ischemic attack remain unclear.
The present study is designed to address the relationship between oral rosuvastatin treatment and the changing levels of cytokines, namely SDF-1/chemokine motif CXC receptor 4 (CXCR-4) axis, ICAM, and VEGF, expressing in different heart sites in rats after acute myocardial infarction (AMI). We also investigated the accumulations of progenitor cells and the improvement in heart function by rosuvastatin treatment.
Methods and Materials
Rat Myocardial Infarction Model
Male Sprague-Dawley rats (250-300 g) of about 6 weeks old were obtained from the laboratorial animal center of the Chinese Academy of Sciences and housed in the animal facilities of Zhongshan Hospital, Fudan University, Shanghai. All the experimental procedures were approved by the Care of Experimental Animals Committee of Fudan University, Shanghai, China. Rats were anesthetized with ketamine hydrochloride (100 mg/kg intraperitoneally), intubated, and ventilated with a small animal ventilator. The heart was exposed through a lateral thoracotomy with a sterile technique. Acute myocardial infarction (AMI) model was induced by permanent occlusion of the left anterior descending coronary artery at 2 mm from the tip of the left auricle. Before chest closure, myocardial infarction was confirmed by observation of a demarcation of injury with blanching of the myocardium and immobility of the left anterior wall. The rats in the sham group underwent thoracotomy and cardiac exposure without coronary artery ligation.
Study Design
Rats were randomly assigned to the rosuvastatin group (22 in the AMI group and 20 in the sham group) and the placebo group (21 in the AMI group and 20 in the sham group). The rosuvastatin group included the d0 (the day of operation) group (4 in the AMI group and 4 in the sham group), d4 (the 4th day after operation) group (4 in the AMI group and 4 in the sham group), d7 (the 7th day after operation) group (4 in the AMI group and 4 in the sham group), d14 (the 14th day after operation) group (4 in the AMI group and 4 in the sham group), and d28 (the 28th day after operation) group (6 in the AMI group; 4 in the sham group). The placebo group included the d0 group (4 in the AMI group and 4 in the sham group), the d4 group (4 in the AMI group and 4 in the sham group), the d7 group (4 in the AMI group and 4 in the sham group), the d14 group (4 in the AMI group and 4 in the sham group), and the d28 group (5 in the AMI group and 4 in the sham group). After randomizing, the animals received either rosuvastatin (5 mg/kg every day) or placebo (0.5% CMC-Na) by daily gavage 7 days before the AMI surgery. The administration of either rosuvastatin or placebo continued at the same dose after AMI establishment until the day before sacrifice.
Western Blot
The protein levels of SDF-1, CXCR-4, ICAM-1, and VEGF were determined by Western blot analysis. Myocardial samples of anterior wall and posterior wall of the left ventricle were harvested on days 0, 4, 7, and 14 after surgery. Protein was isolated from the following standard protocol. Protein concentrations were determined by the bicinchoninic acid (BCA) method. Each sample (50 μg of protein/lane) was separated by sodium dodecyl sulfate–polyacrylamide gel (12.5%) electrophoresis followed by electrophoretic transfer of protein from the gel to a nitrocellulose membrane. Membranes were treated with the blocking buffer for 1 hour at room temperature, followed by incubation with primary antibodies at 4°C overnight. Primary antibodies used here included anti-SDF-1 (1:500 dilution; Santa Cruz Biotechnology, USA), anti-CXCR-4 (1:1000 dilution; Abcam, UK), anti-ICAM-1 (1:200 dilution; BioVision, USA), and anti-VEGF (1:500 dilution; Santa Cruz Biotechnology). Normalization of results was determined by running parallel Western blots for detecting glyceraldehyde 3-phosphate dehydrogenase protein (GAPDH). The optical density was quantified using an image processing analysis program.
Masson Staining and Immunohistochemistry
Rats were sacrificed on d28 after myocardial infarction (n = 5 in the placebo group and n = 6 in the rosuvastatin group) and myocardial samples of the left ventricle were fixed in 4% formaldehyde for histological procedures. Masson staining was performed to analyze the myocardial infarction size (percentage of left ventricle perimeter) and collagen volume fraction. To evaluate the progenitor cells, which may contribute to cardiomyogenesis and neoangiogenesis in the ischemic region, the myocardial sections were immunolabeled with antibodies against CD90 (1:100 dilution; Abcam), c-kit (1:100 dilution; Santa Cruz Biotechnology), and CD133 (1:500 dilution; Abcam). For quantification of positively stained cells and vessels, 3 randomly chosen high-power fields (HPFs) in every chosen section were examined blindly by the investigators. The mean optical density values of CD90-positive and c-kit-positive regions were analyzed using Image-pro plus 6.2 software. The CD133-positive vessels were counted and expressed as the number of vessels per HPF.
Echocardiograph
Echocardiographic evaluations were performed on the 10th day (d10) and the 28th day (d28) after surgery (n = 5 in the AMI placebo group; n = 4 in the sham placebo group; n = 6 in the AMI rosuvastatin group; and n = 4 in the sham rosuvastatin group), with a Vevo 770 high-resolution imaging system (VisualSonics, Canada) and Vevo analysis software (VisualSonics). End-systolic volume and end-diastolic volume of left ventricle were measured to evaluate the heart remodeling after surgery. Postoperation systolic function was determined by the left ventricle ejection fraction (LVEF) and left ventricle short-axis fractional shortening (LVFS) assessment.
Statistical Analysis
All data are expressed as mean ± standard deviation. Comparisons between 2 groups were made using the unpaired Student t test. Comparisons between 3 or more groups were evaluated by one-way analysis of variance. A probability value of <.05 was considered significant. Data were collected and analyzed using the SPSS software (version 16.0).
Results
Rosuvastatin Attenuates Left Ventricular Remodeling and Improves Postinfarction Systolic Function
Two-dimensional (Figure 1) and M-mode (Figure 2) ultrasound cardiographic measurements demonstrated that the end-systolic volume and end-diastolic volume of the left ventricle were decreased in the rosuvastatin group compared with the placebo group (291.52 ± 46.92 μL vs 379.04 ± 48.14 μL, P < .05; 483.22 ± 59.96 μL vs 597.78 ± 76.26 μL, P < .05; Table 1) on d10. The reducing tendency of the left ventricle volume was maintained until d28 but was of no statistical significance (376.44 ± 113.27 μL vs 498.30 ± 97.27 μL, P = .05; 603.59 ± 104.98 μL vs 624.41 ± 111.14 μL, P > .05; Table 1). Masson staining confirmed that the size of myocardial infarction was decreased (32.58% ± 8.15% vs 44.28% ± 5.11%, P < .05; Figure 3) and the collagen fibrosis was attenuated in the rosuvastatin-treated group (19.68% ± 4.61% vs 39.61% ± 6.36%, P < .01; Figure 4) 4 weeks after AMI. Compared with the placebo-treated animals, the rosuvastatin-treated ones retained the postinfarction left ventricle function by improving LVEF (38.58% ± 9.85% vs 27.13% ± 3.05%, P < .05; Table 2) and LVFS (20.09% ± 5.95% vs 13.43% ± 1.64%, P < .05; Table 2) on d28. There was no statistical change in either the left ventricle volume or systolic heart function between rosuvastatin and placebo treatment in the sham groups after surgery (Tables 1 and 2).
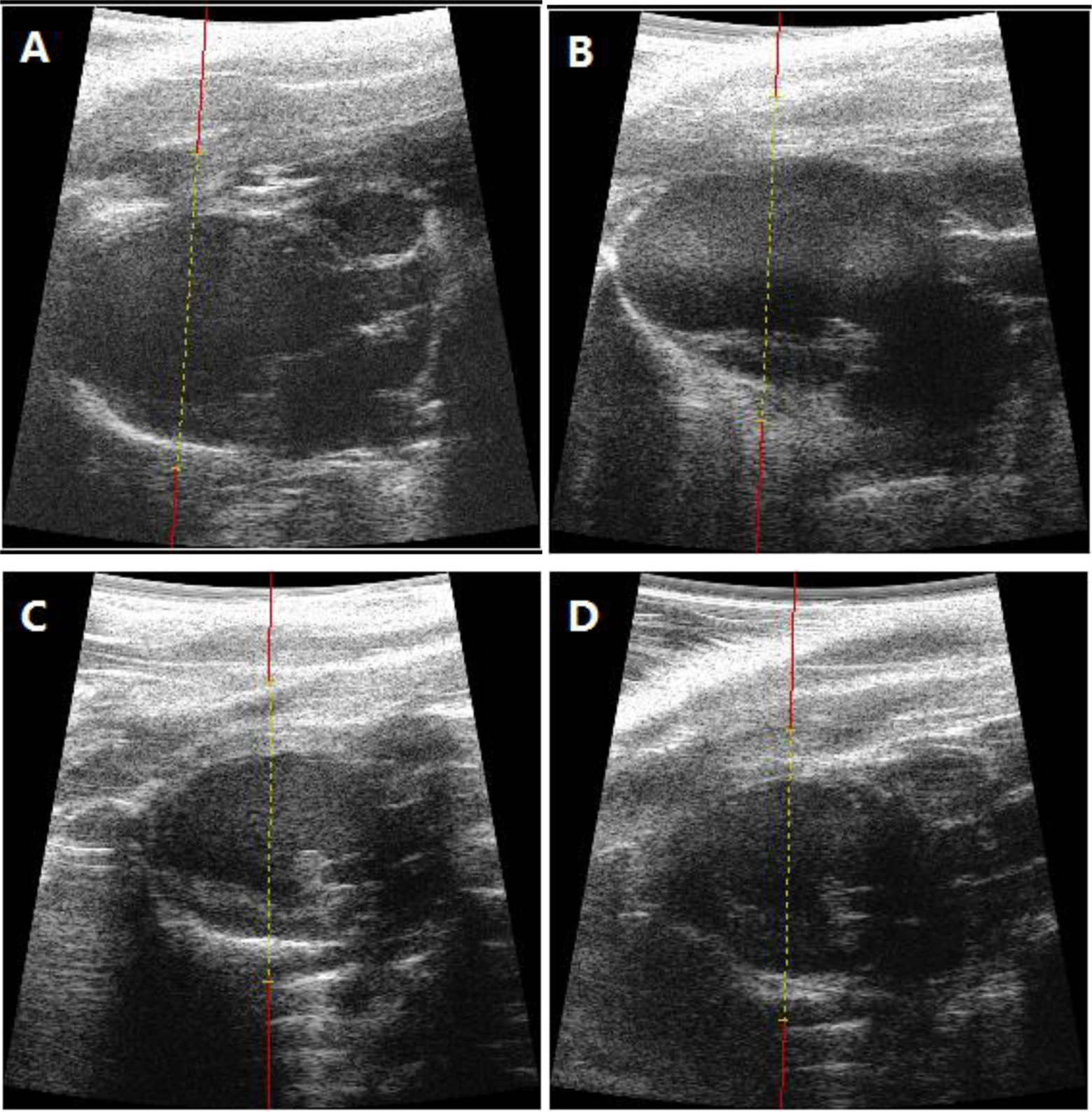
Two-dimensional echocardiographic measurements after acute myocardial infarction (AMI). (A) The placebo group on day 10; (B) the placebo group on day 28; (C) the rosuvastatin-treated group on day 10; (D) the rosuvastatin-treated group on day 28.
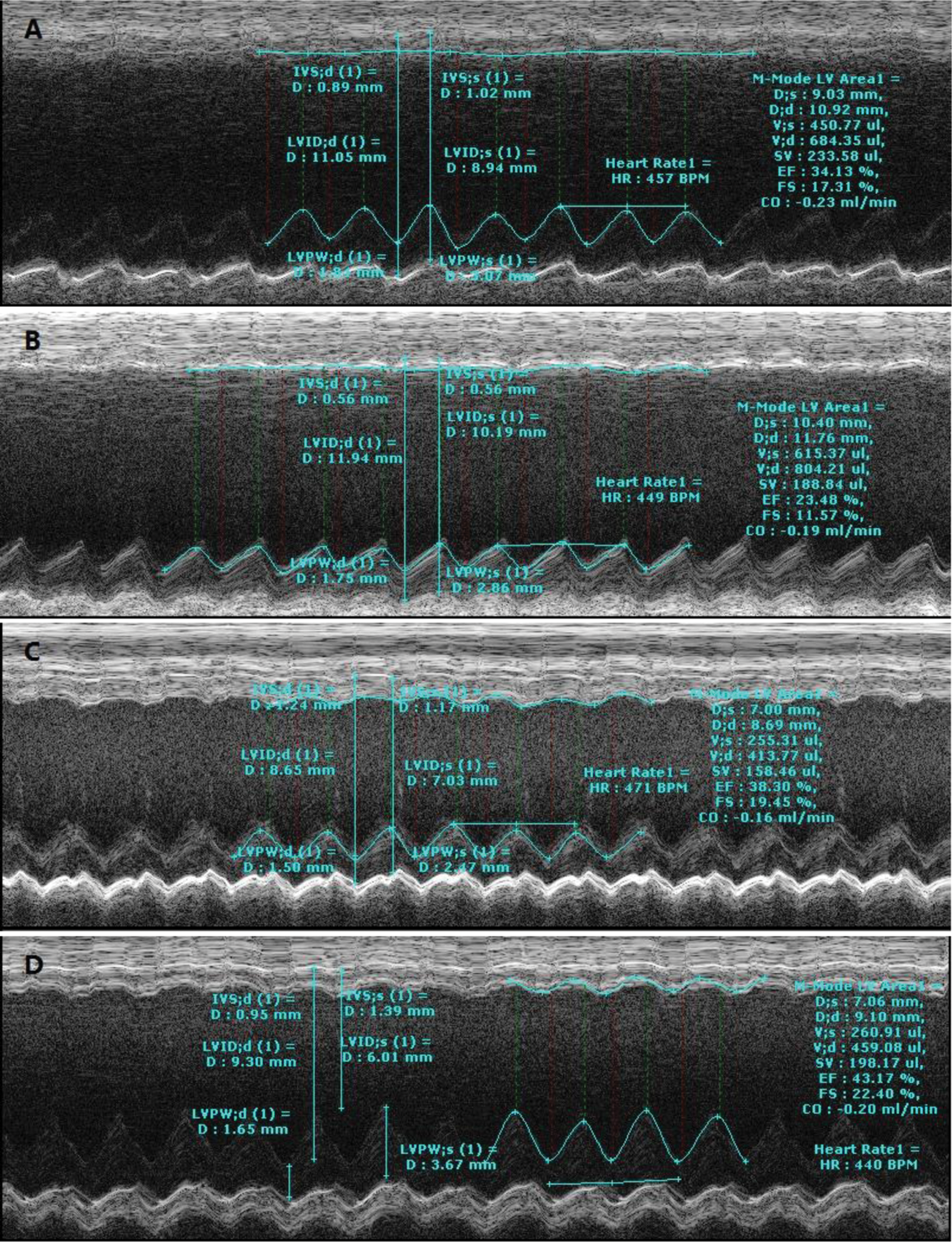
M-mode echocardiographic measurements after acute myocardial infarction (AMI). (A) The placebo group on day 10; (B) the placebo group on day 28; (C) the rosuvastatin-treated group on day 10; (D) the rosuvastatin-treated group on day 28.

Comparison of myocardial infarction size by Masson staining. (A) The placebo group on day 28; (B) the rosuvastatin-treated group on day 28; (C) myocardial infarction size compared between two groups (*P < .05).

Comparison of collagen volume fraction (CVF) in infarcted territory by Masson staining. (A) The placebo group on day 28; (B) the rosuvastatin-treated group on day28; (C) CVF compared between two groups (*P < .01).
End-Systolic and End-Diastolic LV Volume After AMI.

Abbreviations: AMI, acute myocardial infarction; EDLV, end-diastolic left ventricle volume; ESLV, end-systolic left ventricle volume; LV, left ventricle.
a P < .05.
b P < .05.
The LV Systolic Function After AMI.

Abbreviations: AMI, acute myocardial infarction; LV, left ventricle; LVEF, left ventricle ejection fraction; LVFS, left ventricle short-axis fractional shortening.
a P < .05.
b P < .05.
Rosuvastatin Changes the Local Expressions of Cytokines in Peri-infarction Myocardium
We performed Western blot on the days 0, 4, 7, and 14 to verify the changing cytokine expressions by rosuvastatin treatment after surgery. Western blot analysis showed that rosuvastatin treatment could upregulate the SDF-1 (P < .05; Figure 5) expression and downregulate the expressions of CXCR-4 (P < .05; Figure 6), ICAM-1 (P < .05; Figure 7), and VEGF (P < .05; Figure 8) in the anterior wall of the left ventricle compared with the placebo group after AMI. However, there was no significant change in cytokine expressions in the posterior wall of the left ventricle between the rosuvastatin group and the placebo group after AMI (P > .05; Figures 9 –12). In the sham groups, the changes in cytokine expressions between the rosuvastatin and placebo groups were of no statistical significance (P > .05; Figures 5 –12).

Myocardial expression of stromal-cell-derived factor 1 (SDF-1) in anterior wall of left ventricle on day 0, 4, 7, and 14 after operation in rats.

Myocardial expression of chemokine motif CXC receptor 4 (CXCR-4) in the anterior wall of the left ventricle on days 0, 4, 7, and 14 after operation in rats.

Myocardial expression of intercellular adhesion molecule 1 (ICAM-1) in the anterior wall of the left ventricle on days 0, 4, 7, and 14 after operation in rats.

Myocardial expression of vascular endothelial growth factor (VEGF) in anterior wall of left ventricle on days 0, 4, 7, and 14 after operation in rats.

Myocardial expression of stromal-cell-derived factor 1 (SDF-1) in the posterior wall of The left ventricle on days 0, 4, 7, and 14 after operation in rats.

Myocardial expression of chemokine motif CXC receptor 4 (CXCR-4) in the posterior wall of the left ventricle on days 0, 4, 7, and 14 after operation in rats.

Myocardial expression of intercellular adhesion molecule 1 (ICAM-1) in the posterior wall of the left ventricle on days 0, 4, 7, and 14 after operation in rats.

Myocardial expression of vascular endothelial growth factor (VEGF) in the posterior wall of the left ventricle on days 0, 4, 7, and 14 after operation in rats.
Rosuvastatin Treatment Recruits CD90+ and c-kit+ Cells and Promotes Angiogenesis in Ischemic Territory
In order to analyze the recruitment of progenitor cells and neoangiogenesis in peri-infarction territory by the rosuvastatin treatment, the expressions of CD90, 10,11 c-kit, 1,12 and CD133 1,10,11,13,14 were detected by immunohistochemical test performed on the myocardial tissue harvested on d28. Compared with the placebo group, there were significantly more CD90+ cells (P < .05; Figure 13) and c-kit+ cells (P < .05; Figure 14) in the infarcted region in the rosuvastatin-treated group. The number of CD133+ vessels per HPFs was also found more in the rosuvastatin-treated animals with statistical significance (6.47 ± 1.92 vs 2.18 ± 0.98, P < .01; Figure 15).

The expression of CD90 in the peri-infarction region on the 28th day after acute myocardial infarction (AMI) in rats. (A) The placebo group; (B) the rosuvastatin-treated group; (C) the comparison of mean optical density (MOD) between two groups (*P < .05).

The expression of c-kit in the peri-infarction region on the 28th day after acute myocardial infarction (AMI) in rats. (A) The placebo group; (B) the rosuvastatin-treated group; (C) The comparison of mean optical density (MOD) between two groups (*P < .05).

Neoangiogenesis in the peri-infarction region on the 28th day after acute myocardial infarction (AMI) in rats. (A) The expression of CD133 in the placebo group; (B) the expression of CD133 in the rosuvastatin-treated group; (C) the comparison of CD133+ vessels number (per HPF) between two groups (*P < .01).
Discussion
Ischemic attack will change the local expression of several chemotactic cytokines. SDF-1, also known as chemokine motif CXC ligand 12 (CXCL-12), is the ligand for its receptor, CXCR-4. It is known that SDF-1 plays a critical role in trafficking hematopoietic cells, lymphopoietic cells, and progenitor cells and in maintaining hematopoietic stem cell niches in the bone marrow. 15,16 Recent evidence showed that preconditioning by SDF-1 could improve the survival of bone marrow mesenchymal stem cells and endothelial progenitor cells. 17,18 The SDF-1 also can induce the progenitor cells to myocardial and endothelial development. 19 It has been reported that postinfarction myocardial SDF-1 expression increased and peaked at the early stage and decreased thereafter. The effect of cell transplantation differs at different times after myocardial infarction owing to the time course of the expression of cytokines in the ischemic myocardium. 20 –22 It is believed that SDF-1 is a potential target in promoting the tissue repair after AMI. Our findings of the dynamic changes in the local expressions of SDF-1/CXCR-4 pathway, VEGF, and ICAM-1 after AMI in the present study are consistent with the reported literature.
However, the biological mechanism of SDF-1/CXCR-4 pathway is complicated. The overexpression of SDF-1 in the ischemic region is associated with scar formation and ventricle remodeling after myocardial infarction via the CXCR-4 receptor pathway. The specific antagonist of CXCR-4, AMD3100, reduces the infarct size, attenuates the hypertrophic response of the left ventricle, and concomitantly improves ventricular contractile function in 1-week postinfarction rats. 23 Furthermore, SDF-1 traffics not only normal stem cells but also tumor stem cells that express CXCR-4 and may facilitate tumor vasculature development and metastasis. 24 –26 These side effects of the SDF-1/CXCR-4 mechanism prohibit the clinical use of SDF-1 in the therapeutic practice.
In this study, we found that the oral administration of rosuvastatin (5 mg/kg every day) can change the local expression of cytokines in the ischemic region after AMI. Rosuvastatin upregulates the SDF-1 expression and downregulates the CXCR-4 expression in the peri-infarction tissue. This action of rosuvastatin can last as long as 2 weeks after AMI. The expressions of VEGF and ICAM in the peri-infarction region were downregulated after AMI in the treated animals, which were probably attributed to containing inflammation and decreasing infarction size by rosuvastatin administration. However, rosuvastatin makes no difference in cytokine expressions in the posterior wall of the AMI group and in the sham group, which may be attributed to less tissue inflammation and cell necrosis in the nonischemic tissue. To our knowledge, the time course and regional difference in the cytokine response to rosuvastatin intervention are reported for the first time in literature.
It has been proved that statin intervention is associated with increasing the number and the function of the circulating progenitor cells, improving neovascularization in mice with AMI. 2 The use of HGM-CoA reductase inhibitors has been claimed to mobilize endothelial progenitor cells and to facilitate their incorporation into the neovasculature by supporting differentiation and inhibiting apoptosis. 27 –29 Atorvastatin treatment increases the mobilization of CD34+/CD133+ progenitor cells into the circulation. 30 Pravastatin improves function in hibernating myocardium by mobilizing CD133+ and c-kit+ bone marrow progenitor cells and promoting myocytes to re-enter the growth phase of the cardiac cell cycle. 31 Simvastatin can stimulate rat bone marrow stem cells (BMSCs) to express endothelial-specific genes and proteins, inducing the capillary tube-like formation of the BMSCs. 32 In our study, we also confirmed that the CD90+ (the marker of bone marrow mesenchymal cell 10 ,11 ), c-kit+ cells (the marker of cardiac stem cell 1,12 ), and CD133+ (the marker of endothelial progenitor cell 1,10,11,13,14 ) vessels in the peri-infarction region were significantly more in the rosuvastatin-treated group compared with those in the placebo group, suggesting the recruitment of the progenitor cells by the rosuvastatin treatment in vivo. Recruitment of these multipotent progenitor cells and enhancement of angiogenesis by rosuvastatin treatment may contribute to the tissue repair and function preservation after the ischemic attack.
Echocardiographic measurement also confirmed the morphology improvement and function preservation by statin treatment in the present study, which was consistent with the previously reported studies. 33,34 The echocardiographic results showed that the morphology improvement by rosuvastatin treatment was significant on the 10th day after AMI and the left ventricle volume maintained a reducing trend on the 28th day after AMI. Meanwhile, the function improvement due to rosuvastatin treatment was significant on the 28th day after AMI. The time lag may be attributed to the different response latencies to rosuvastatin treatment between morphology change and function improvement. Further investigation will be done to optimize the rosuvastatin administration to maximize its favorable effect. In conclusion, the data suggest that rosuvastatin can protect the heart from ischemic injury and preserve the cardiac function in rats in vivo. The favorable effect of rosuvastatin treatment is associated with the change in SDF-1, CXCR-4, ICAM-1, and VEGF expressions and the accumulation of progenitor cells. The results of the study provide evidence in support of the therapeutic potency of rosuvastatin in cytokine modulation in cellular cardioplasty for ischemic heart disease.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [81000043]; and the Research Fund for the Doctoral Program of Higher Education of China [20100071120072].
