Abstract
Background:
Mesenchymal stromal cells (MSCs) improve tissue repair but their mechanism of action is not fully understood. We aimed to test the hypothesis that MSCs may act via macrophages, and that specifically, human cardiac adipose tissue-derived mesenchymal stromal cells (AT-MSCs) can polarize human macrophages into a reparative, anti-inflammatory (M2) phenotype.
Methods and Results:
We isolated and grew AT-MSCs from human cardiac adipose tissue obtained during cardiac surgery. Macrophages were grown from CD14+ monocytes from healthy donor blood and then cocultured with AT-MSCs, with and without transwell membrane, for 1 to 14 days. In response to AT-MSCs, macrophages acquired a star-shaped morphology, typical of alternatively activated phenotype (M2), and increased the expression of M2 markers CD206+, CD163+, and CD16+ by 1.5- and 9-fold. Significantly, AT-MSCs modified macrophage cytokine secretion and increased the secretion of anti-inflammatory and angiogenic cytokines: interleukin (IL)-10 (9-fold) and vascular endothelial growth factors (3-fold). Moreover, AT-MSCs decreased macrophage secretion of inflammatory cytokines such as IL-1α (2-fold), tumor necrosis factor α (1.5-fold), IL-17 (3-fold), and interferon gamma (2-fold). Remarkably, the interaction between AT-MSCs and macrophages was bidirectional and macrophages enhanced AT-MSC secretion of typical M2 inducers IL-4 and IL-13. Notably, AT-MSCs decreased macrophage phagocytic capacity. Finally, IL-6 mediates the M2 polarization effect of AT-MSCs on macrophages, by increasing M2-associated cytokines, IL-10 and IL-13.
Conclusions:
Human cardiac AT-MSCs can polarize human macrophages into anti-inflammatory phenotype. Our findings suggest a new mechanism of action of AT-MSCs that could be relevant to the pathogenesis and treatment of myocardial infarction, atherosclerosis, and various cardiovascular diseases.
Mesenchymal stromal cells (MSCs) are the nonhematopoietic multipotent progenitor cells found in various adult tissues. 1 They are characterized by their reparative and immunomodulatory properties, but their mechanism of action is complex and not fully understood. Adipose tissue is now recognized as an accessible, abundant, and reliable source for the isolation of stem cells. 2 Particularly, MSCs derived from adipose tissue (AT-MSCs) have shown promising potential for proliferation and differentiation into multiple cell lineages, secretion of protective factors, and creation of matrix for tissue regeneration. 3–5
Although MSCs have been shown to have immunomodulation properties, particularly on T and B lymphocytes, relatively little attention has been paid to possible interaction with tissue macrophages. 6 We previously hypothesized that MSCs may improve infarct repair via macrophages. 7 Indeed, a few recent reports have suggested that MSCs may act via macrophages and can switch macrophages from inflammatory to anti-inflammatory, reparative phenotype. 8–12 However, the influence of human cardiac MSCs, particularly those derived from cardiac fat, on human macrophage activation has not been investigated.
Macrophages possess unique versatility and plasticity and respond to environmental signals with different forms of polarization. 13 This can be categorized generally as classical (M1), and nonclassical, or alternatively activated (M2) polarization. 13 , 14 M1 macrophages are pro-inflammatory 13 , 14 and have been implicated in the early phases of atherosclerosis, 15 as well as in acute inflammation after myocardial infarction (MI). 16 , 17 M2 macrophages, on the other hand, are immunosuppressive and reparative 14 and are implicated in the advanced stages of atherosclerosis, 18 MI healing, 16 , 17 and in the development of pulmonary hypertension (PHT). 19
In view of the major role of both MSCs and macrophages in the pathogenesis and treatment of cardiovascular disease, the aim of the present study was to determine the potential interaction between human cardiac AT-MSCs and human macrophages. The rational for studying cardiac AT-MSCs was first, their superior paracrine and proliferative capacity compared with that of bone marrow (BM)-derived MSCs. 3 , 20 Second, due to their proximity to the heart, the potential interaction between AT-MSCs and macrophages could be relevant to the pathogenesis of atherosclerosis, ischemic and inflammatory heart disease, and the development of novel immunomodulatory and regenerative therapies for these disorders.
Materials and Methods
The protocol of human cell collection and informed consent were reviewed and approved by the Sheba Medical Center’s institutional review board.
Collection and Isolation of Human Cells
We obtained samples of human cardiac adipose tissue from 3 patients undergoing open heart surgery. We excised small samples (1 or 2) of tissue from the fat pad covering the aortic root, the coronary artery groove, and the pericardium, with a surgical scalpel to avoid thermal injury to the tissue. We then minced the tissue samples into small pieces and incubated them with an enzymatic digestion mixture, as described previously. 21
Macrophages were isolated from leukocyte blood units, from healthy young donors (aged 18-30 years), using Ficoll membrane tubes (NovaMed, Chicago, Illinois) and subsequently treated with red blood cell lysis solution (Biological Industries, Beit Haemek, Israel). CD14+ monocytes were positively selected from mononuclear cells (MNCs) using MACS (Miltenyi Biotec, Bergisch Gladbach, Germany). CD14-negative cells served as an MNCs control group. We cultured the isolated human monocytes for 5 days in RPMI (Gibco-Invitrogen, Carlsbad, California) supplemented with 10% fetal bovine serum ([FBS]; Biological Industries, Beit Haemek, Israel), and 1% penicillin streptomycin (pen-strep; Biological Industries, Beit Haemek, Israel). On the fifth day, the medium was refreshed and adherent macrophages were cocultured for 1 to 14 days with passage 2 to 4 AT-MSCs or CD14neg MNCs (1:4 ratio of AT-MSC/MNCs:macrophage), directly or separated by a transwell membrane (1 μm, Millipore, Billerica, Massachusetts).
Cell Characterization
To characterize macrophage phenotype, we harvested cultured macrophages using a scraper and then washed and stained with saturating concentrations of monoclonal antibodies against M2 markers CD206, CD163, and CD16 (BioLegend, San-Diego, California; mouse anti-human CD206-FITC/CD163-APC or CD16-APC).
To characterize MSC phenotype, we used cells after the third passage. Cells were analyzed using the following fluorescent anti-human antibodies: CD105-APC (eBioscience, Frankfurt, Germany), CD73-PE (BD Pharmingen, San Diego, California), CD90-PE (BioLegend, San Diego, California), CD34-PE, CD45-PE, and C-kit-APC (Dako, Copenhagen, Denmark). Labeled cells (1×106) were analyzed using FACS Calibur Cytofluorimeter (Cytek Development, Fremont, California) running with Flowjo software (Tree Star, Ashland, Oregon).
To determine the multipotency of isolated MSCs, we carried out in vitro assays for differentiation into osteoblasts, adipocytes, and myogenic lineage, as described previously. 21
Immunohistochemistry
To further characterize the cells, cultured macrophages were fixated by incubation for 20 minutes with 4% formaldehyde solution and 1 minute with hematoxylin and then washed with phosphate-buffered saline (PBS). Following fixation, cells were incubated overnight with a human polyclonal immunoglobulin G antibody for blocking and then incubated 1 hour with monoclonal antibodies against dendritic cell markers CD1a and CD83 (BioLegend, San-Diego, California; mouse anti-human APC-CD1a and FITC-CD83) to rule out dendritic cell differentiation.
Enzyme-Linked Immunosorbent Assay
To determine the levels of cytokine secretion from cells, we collected culture medium at different time points and kept frozen at −80°C. We then measured cytokine levels in triplicate with commercially available kits (R&D, McKinley Place NE, Minneapolis, Minnesota) of sandwich enzyme-linked immunosorbent assays (ELISAs) and Q-Plex arrays (Quansys Biosciences Multiplex ELISA, West Logan, Utah) and incubated them with capture antibodies overnight. After fluid addition, a biotinylated detection antibody was added, followed by streptavidin-horseradish peroxidase and substrate solution addition for optical density measurements by a spectrophotometer or light measurements and analysis using Q-View imager and software.
Effect of AT-MSC Cytokines on Human Macrophages
To identify the potential inducer of macrophage polarization, we incubated macrophages with various cytokines secreted by AT-MSCs for 1 to 3 days, in concentrations of 0.25 and 4 times those detected in AT-MSC cultures. We examined interleukin (IL)-6, IL-8, vascular endothelial growth factors (VEGFs), IL-4, IL-10, and tissue inhibitor of matrix metalloproteinases (TIMP-1 and TIMP-2, Pepro Tech, Rocky Hill, New Jersey). We also used control groups of cultured macrophages without cytokine exposure and macrophages incubated with M2-polarizing concentration of IL-13.
In addition, we incubated macrophages and AT-MSCs with 50 μg/mL of neutralizing antibodies against both IL-6 and its receptor CD126 (Cayman Chemicals, Ann Arbor, Michigan). After 1 and 3 days, we collected the culture medium and measured cytokine levels in triplicate by Q-Plex arrays as described above.
Phagocytosis Assay
To determine the influence of MSCs on macrophage phagocytic capacity, we measured the percentage of the macrophages that engulfed fluorescent beads and the geometric mean of their fluorescent intensity. As stock suspension, we used aqueous suspension with red fluorescent latex beads (2.5% solids, mean diameter 1 μm, amine-modified polystyrene; Sigma-Aldrich, Rehovot, Israel). Working solution was prepared by adding 50 μL of stock solution to 10 mL PBS. Six well plates with cultured macrophages were washed with PBS and 2 mL of fresh RPMI was added (10% FBS, 1% PS), with 22 μL of working suspension per 106 adherent cells (5 beads per cell). Plates were incubated at 37°C for 3 hours in the dark and then washed and scraped. Single-cell suspension was prepared for analysis using a flow cytometer.
Statistical Analysis
Data are presented as mean ± standard error of the mean. Differences among groups were compared with 2-way analysis of variance and Bonferroni posttest. GraphPad Prism version 5 was used for analysis (GraphPad, San Diego, California).
Results
Human AT-MSCs were characterized by the expression of classical MSC markers such as CD105 (98%), CD73 (97%), and absence of hematopoietic cell markers CD45 (near 0%) and CD34 (3%). The ability of human AT-MSCs to differentiate into bone, fat, and myogenic cells was confirmed, as described previously. 21
Cardiac AT-MSCs Increased the Percentage of M2 Macrophages
Human cardiac AT-MSCs significantly affected human macrophage morphology and induced a star-shaped morphology, with 5 to 7 cytoplasmatic extensions, after coculture through a transwell membrane, for 3 to 7 days (Figure 1). This new morphology resembled M2 macrophages created by treatment with M2 inducers IL-4 or IL-13. 22 Immunostaining of the cells for dendritic cell markers, CD1a and CD83, was negative, ruling out a dendritic phenotype. In contrast, coculturing of human macrophages with MNCs induced a different, fibroblastic-like elongated shape (Figure 1), which has been reported in M1 human macrophages. 23
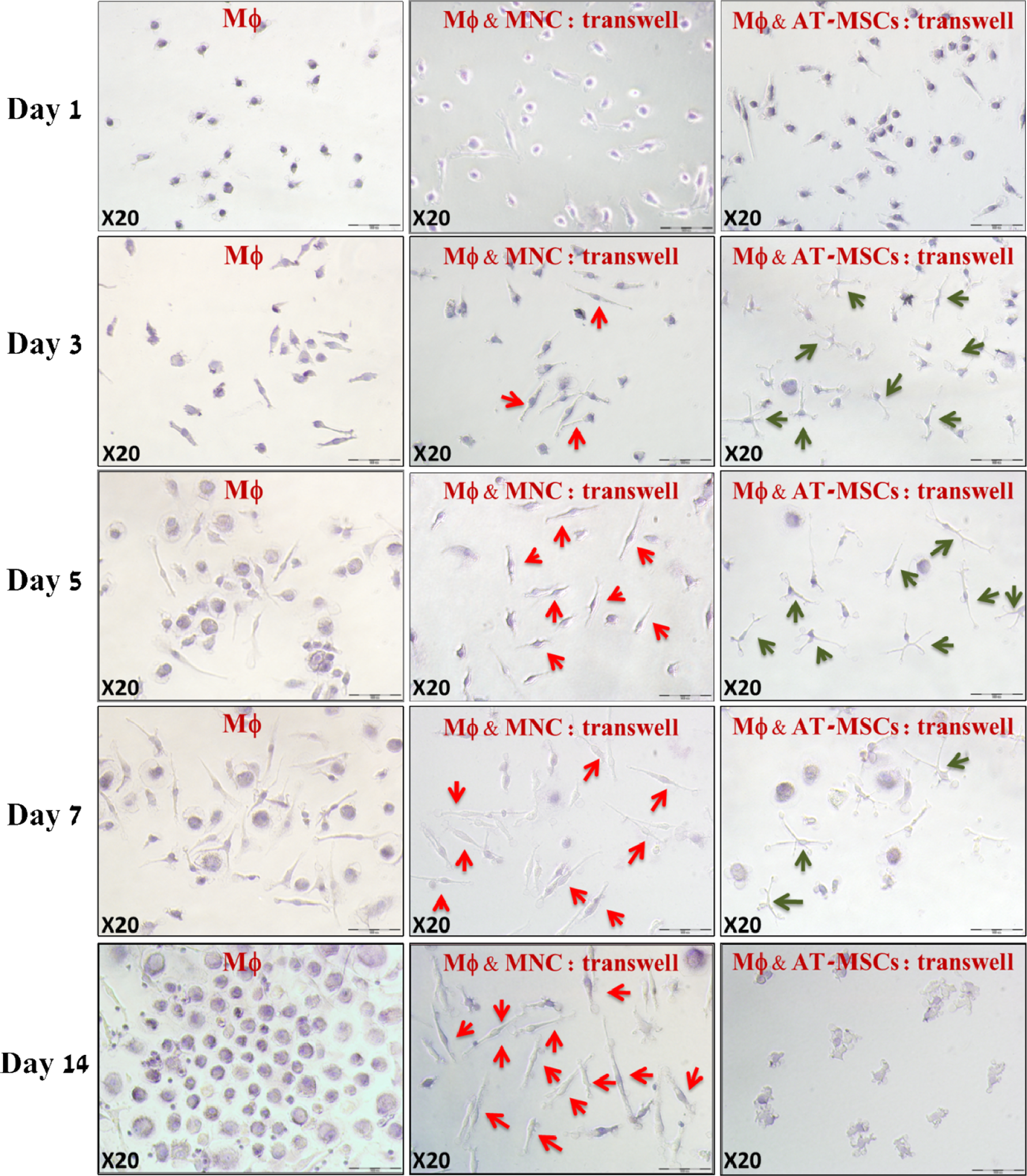
Temporal changes in macrophage morphology during coculture with cardiac AT-MSCs or MNCs. Cultured human macrophages (left column) change their morphology from small cells with thin extensions on day 1 to a mixture of elongated fibroblast-like and large, round cells on days 3 to 7, and finally to large round cells on day 14. Macrophages cocultured with MNCs with transwell membrane (middle column) developed a fibroblast-like spindle shape, which became more dominant over time (red arrows). Coculturing human macrophages with MSCs, through transwell membrane (right column), induced a star-shaped morphology, with 5 to 7 cytoplasmatic extensions (green arrows), particularly on days 3 and 5 of incubation. The effect of AT-MSCs on macrophage morphology diminished from day 7 and practically disappeared on day 14 (HE staining; magnification ×200). MΦ indicates macrophage; AT-MSC, adipose tissue-derived mesenchymal stromal cell; MNCs, mononuclear cells.
As markers of M2 macrophages, we used surface receptors CD206, CD163, and CD16. Indeed, the percentage of macrophages expressing M2 markers was increased after incubation with AT-MSCs, particularly on days 3 and 5 (Figure 2A and B). For example, CD206 and CD163 were increased up to 1.5-fold and CD16 up to 9-fold. On the other hand, coculturing macrophages with MNCs had a lesser or no effect on M2 marker expression. Thus, AT-MSCs but not MNCs can reprogram macrophages toward M2 phenotype.

Cardiac AT-MSCs increase percentage of macrophages expressing M2 markers CD206, CD163, and CD16. A, Incubating macrophages with AT-MSCs increased the percentage of macrophages expressing M2 markers CD206 and CD163 (1.5-fold), especially on days 3 and 5, while coculturing with MNCs did not.B, Incubating macrophages with AT-MSCs increased the percentage of macrophages expressing the human M2 marker CD16 (up to 8-fold), especially on days 3 and 5, while coculturing with MNCs did not. AT-MSC, adipose tissue-derived mesenchymal stromal cell; MNCs, mononuclear cells.
The AT-MSCs Modulate Macrophage Cytokine Secretion and Phagocytic Capacity
To assess the effect of AT-MSCs on macrophage cytokine secretion and vice versa, we incubated the cells separately, together, or with a cell-conditioned medium. To adjust to the increasing number of cells in the coculture group, we also calculated the total amount of cytokine secretion from AT-MSCs only plus macrophages only (Table 1).
Cytokine Secretion of Human Macrophages Cocultured With Human Cardiac Adipose Tissue-Derived AT-MSC by ELISA Results (pg/mL)

Abbreviations: ELISA, enzyme-linked immunosorbent assay; MΦ, macrophages; IL, interleukin; AT-MSC, adipose tissue-derived mesenchymal stromal cells.
a P < .01 (compared to sum of MΦ + AT-MSC).
b P < .001 (compared to sum of MΦ + AT-MSC).
c P < .05 (compared to sum of MΦ + AT-MSC).
dReached nondetectable concentrations.
The AT-MSCs significantly modified the profile of cytokine secretion (Table 1, Figure 3). For example, after 1 day of coculturing, there was a significant increase in the secretion of M2-related cytokines: IL-13 (up to 2.5-fold), IL-4 (up to 2-fold), VEGF (up to 3-fold), and IL-10 (up to 9-fold). Significantly, the level of IL-10, an anti-inflammatory cytokine typical of M2 macrophages, was increased with direct (9-fold, P < .001) and indirect (5-fold, P < .001) coculturing with AT-MSCs, on days 1 and 3 (Figure 4). Furthermore, AT-MSCs reduced macrophage secretion of M1-related cytokines such as IL-1α (2-fold, P < .01), TNF-α (1.5-fold, P < .001), IL-12 (1.5-fold, P < .001), IL-17 (3-fold, P < .001), IL-23 (16-fold, P < .01), and interferon gamma (2-fold, P < .001). Surprisingly, the interaction between AT-MSCs and macrophages inhibited the secretion of trophic and angiogenic cytokines, such as angiopoietin-II, fibroblast growth factor, and hepatocyte growth factor (Table 1). Interestingly, direct cell contact was significant for increased secretion of cytokines such as IL-4, IL-13, and VEGF but not for secretion of other cytokines (Table 1, Figure 3). Finally, incubation of AT-MSCs with macrophages created a mutual effect. For example, the macrophage-condition medium stimulated the secretion of typical M2 inducers, such as IL-4 and IL-13, from AT-MSCs (Table 1, Figure 3). Thus, the interaction between AT-MSCs and macrophages was bidirectional.

Changes (%) in cytokine secretion of macrophages and AT-MSCs cocultured for 24 hours. While coculturing macrophages with AT-MSCs significantly increased macrophage secretion of M2-associated cytokines IL-10 and VEGF the secretion of M1-associated cytokines IL-1, TNF-α, IL-12, IL-17, IL-23, and IFN-γ decreased. Cell contact between macrophage and AT-MSCs enhanced this effect. In addition, macrophages increased MSC secretion of the M2 inducers IL-4 and IL-13, in a contact-dependent manner. AT-MSC indicates adipose tissue-derived mesenchymal stromal cell; VEGF, vascular endothelial growth factor; IL, interleukin; IFN, interferon; TNF, tumor necrosis factor.

Effect of human cardiac AT-MSCs on macrophage IL-10 secretion. The IL-10 secretion, which is strongly associated with M2 macrophage function, was significantly increased with direct and indirect coculturing with AT-MSCs on day 1 (9-fold, P < .001) and day 3 (5-fold, P < .001). However, from day 7 and onward, levels decreased in cocultured groups. The MSCs exposed to macrophage-conditioned medium and MSCs alone secreted low to nondetected amounts of IL-10, supporting the notion that IL-10 is produced mainly by macrophages and not by MSCs and increases in response to coculturing with MSCs, rather than the opposite. AT-MSC indicates adipose tissue-derived mesenchymal stromal cell; IL, interleukin.
To determine the influence of AT-MSCs and MNCs on macrophage phagocytic capacity, we incubated macrophages with red fluorescent latex beads. Change in the phagocytic capacity was assessed by a change in the number of red fluorescent latex beads phagocytized per cell, which was measured by the fluorescent intensity of macrophages that phagocytized beads. Indirect exposure to AT-MSCs decreased macrophage phagocytic capacity after 3 days of incubation (by 2-fold), while exposure to MNCs increased it (by 1.5-fold, Figure 5).

Effect of human cardiac AT-MSCs on human macrophage phagocytic capacity. A, Red fluorescent latex beads were used to assess the AT-MSCs effect on macrophage phagocytic capacity. After 3 days of incubation, indirect exposure to MSCs decreased macrophage phagocytic capacity by 2-fold, while exposure to MNCs increased it by 1.5-fold.B, A representative image of macrophages engulfing red fluorescent beads (composed image with and without red fluorescence filter). AT-MSC, adipose tissue-derived mesenchymal stromal cell; MNCs, mononuclear cells.
IL-6 as AT-MSC Mediator of M2-polarization
To identify the AT-MSC mediator of macrophage polarization, we incubated human macrophages with various cytokines for 1 to 3 days, in concentrations similar to those secreted by AT-MSCs in our cell cultures. While many of the examined cytokines (IL-8, VEGF, IL-4, IL-10, TIMP-1, and TIMP-2) had no significant effect on macrophage cytokine secretion (data not shown), incubating macrophages with IL-6 significantly increased the secretion of IL-13 (up to 15-fold, P < .0001, Figure 6A) and IL-10 (day 3, up to 2-fold, P < .001, Figure 6B) and both are classical inducers and biomarkers of M2 macrophages. Significantly, blocking IL-6 and its receptor CD126 in macrophage and AT-MSC coculture, decreased IL-13 secretion to near zero (P < .0001, Figure 6C) and decreased the proportion of M2 macrophages by one-third (CD206+, P < .01, Figure 6D). These findings suggest that IL-6, secreted in high amounts from AT-MSCs, control the switch to M2 phenotype, probably through IL-10 and IL-13 secretion.

Effect of IL-6 on macrophage cytokine secretion and M2 polarization. Stimulating macrophages with IL-6 increased secretion of IL-13 by up to 15-fold (A, P < .0001), and IL-10 by up to 2-fold (B, P < .001). Blocking IL-6 in coculture of macrophages and AT-MSCs strikingly decreased IL-13 secretion by 18-fold (C, P < .0001), and the proportion of M2 macrophages (CD206+) by one-third (D, P < .01). Together, these findings suggest IL-6 participation in M2 macrophage polarization induced by AT-MSCs. AT-MSC indicates adipose tissue-derived mesenchymal stromal cell; IL, interleukin.
Discussion
The new findings of the present study suggest that human cardiac AT-MSCs can reprogram macrophages to M2-like, anti-inflammatory phenotype. Concomitantly, AT-MSCs decrease the secretion of inflammatory cytokines and increase the secretion of inflammatory cytokines. Significantly, we show for the first time, a bidirectional interaction: macrophages stimulate AT-MSCs to secrete M2 inducers, cytokines such as IL-4 and IL-13. Additionally, we found that AT-MSCs decreased macrophage phagocytic capacity after 3 days, while MNCs increased it. Overall, the importance of these findings is that they provide further insight into the potential mechanism behind the therapeutic effects of MSC therapy in cardiovascular disease. Furthermore, the interaction between AT-MSCs and macrophages could be relevant to the pathogenesis of atherosclerosis, MI, valvular disease, cancer, and PHT.
The present study adds to recent reports, 8 –12 showing that MSCs, from BM, gingiva, and umbilical cord, can switch monocytes/macrophages to an anti-inflammatory activation state that may be associated with improved cardiac remodeling and function. 11,24 Importantly, the present study differs from previous reports by providing detailed, temporal, cytokine secretome analysis and by demonstrating bidirectional interaction between cardiac AT-MSCs and macrophages.
Programming macrophages from M1 toward M2 phenotype by MSCs could be beneficial to control and resolve inflammation after tissue injury. For instance, one of the major challenges in the management of acute MI is to improve the healing process. It has been proposed that MSCs can improve infarct repair, 25 but their mechanism of action is complex and not fully understood. We and others have suggested that MSCs might interact with macrophages during infarct healing. 1,11,26
Particularly, our findings might be relevant to the pathogenesis and treatment of coronary artery disease. The role of monocyte and macrophage subsets in atherogenesis has been a topic of great interest throughout the last decade. 15 A recent report has shown that the number of M1 macrophages in cardiac adipose tissue correlates with the severity of coronary artery atherosclerosis in humans. 27 In mice, hypercholesterolemia is associated with an increase in inflammatory monocytes referred to as Lychi that have functional characteristics of M1 macrophages. 15
Of interest is the effect of MSCs on macrophage phagocytic capacity. We found that MSCs decreased macrophage phagocytic capacity after 3 days, while MNCs increased it. M1 cells exhibit phagocytic, proteolytic, and pro-inflammatory functions that help digest damaged tissue 17 and kill intracellular pathogens. Therefore, a decrease in phagocytic capacity might reflect M2 polarization.
In the present study, cardiac AT-MSCs increased IL-10 secretion from macrophages by up to 10-fold. The IL-10 suppresses the acute inflammatory response and inflammatory cytokine secretion by M1 and plays a significant role in extracellular matrix remodeling. 28 These findings are important because IL-10 has both anti-atherosclerotic and cardioprotective properties. 29,30 Another significant mediator is VEGF, a growth factor for vascular endothelial cells that promotes angiogenesis, and has anti-apoptotic properties. While our cardiac AT-MSCs secreted large amounts of VEGF, macrophages did not. However, after coculture, macrophages significantly increased VEGF secretion in a contact-dependent manner.
In an attempt to identify the cytokines which mediate M2 polarization, we further examined the possible paracrine mediators secreted by MSCs. A putative mediator is IL-6 but its role in macrophage polarization has been controversial. While IL-6 has been suggested to promote alternative M2 polarization, 10 , 31 a recent study has shown opposite effect. 32 Temporal blockade of IL-6 signaling after spinal cord injury in mouse reduced inflammatory activity and promoted functional recovery by promoting the formation of alternatively activated M2 macrophages. 32 In the present study, we found that incubating macrophages with IL-6, which is secreted in high amounts from MSCs, increased the secretion of IL-13. The latter is an anti-inflammatory cytokine and inducer of M2 macrophages. This effect was completely abolished by IL-6 receptor antagonist. Our current findings suggest that IL-13, secreted from IL-6 stimulated macrophages, induced M2 polarization, after which M2-polarized macrophages secreted IL-10, a typical M2 and anti-inflammatory cytokine. Thus, IL-6 can induce M2 polarization. However, the therapeutic potential of IL-6 as a macrophage polarization agent in vivo and its effect on tissue repair warrant further research.
Finally, the present study shows a bidirectional interaction between MSCs and macrophages: macrophages stimulate AT-MSCs to secrete cytokines such as IL-4 and IL-13, M2 inducers, that accelerate macrophage polarization. These interactions are likely to play an important role in tissue inflammation and may provide insight into the role of MSCs in inflammation and tissue injury. 33 For example, through the release of soluble factors, macrophages stimulate MSC migration to tumors and sites of injury and alter their cytokine secretion profile. 34
Limitations
We are aware of several limitations in our study. First, our findings and conclusions are based on in vitro experiments. Although we used human cells that would need the use of immune-compromised animals, in vivo experiments could strengthen our conclusions. Indeed, we and others have shown that mouse BM MSCs can switch macrophages toward M2 phenotype in mouse models of MI and sepsis. 11 , 12 , 24 Second, MSCs and macrophages were produced from different donors: macrophages from young healthy donors and MSCs from cardiac patients who are sick. It is possible that using macrophages and MSCs from the same patient could control for interpersonal allogeneic-associated variables.
Conclusions
Our findings suggest that human cardiac AT-MSCs polarize macrophages toward an M2 anti-inflammatory phenotype. Our study highlights the potential of human macrophages in the emerging field of regenerative medicine by demonstrating the ability to reprogram macrophages by AT-MSCs. Thus, the concepts described here may also be extended to several other human disorders, enabling translational research into disease mechanisms and therapies.
Footnotes
Authors' Note
Jonathan Leor has developed a technology to isolate stem cells from tissues.
Acknowledgments
We thank Yuval Shimrat for his guidance in multiplex-ELISA experiments and Vivienne York for her skillful English language editing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grants from the Legacy Heritage Fund of New York; Tel-Aviv University PhD Student Scholarship for Stem Cell Research, funded by the Legacy Heritage Fund (TBM); the Seventh Framework Program, European Commission-Cardiovascular Disease (JL).
