Abstract
The pathological role of adipose derived fatty acids following myocardial infarction has long been hypothesized. However, most methods for reducing adipocyte lipolysis have significant non-adipose effects. Atglistatin, a direct inhibitor of the initial lipase in the lipolysis cascade, has been recently shown to inhibit adipose tissue lipolysis after oral administration. To explore the ability of Atglistatin to impact the pathophysiology of cardiac ischemia we performed prophylactic treatment of mice with Atglistatin for 2 days before 1-hour cardiac ischemia. After 7 days of reperfusion, hearts of Atglistatin treated mice showed significantly improved systolic pump function while infarct and scar size were unaffected. Strain analysis of echocardiographic data revealed an enhanced performance of the remote myocardium as cause for overall improved systolic function. The present study provides evidence that inhibition of adipocyte adipose triglyceride lipase (ATGL) using Atglistatin is able to improve cardiac function after MI by targeting the remote myocardium.
Introduction
A myocardial infarction (MI) is followed by significant cardiac remodeling, characterized by necrosis, apoptosis, inflammation, fibrosis, and neovascularization, and the extent of this remodeling is the major determinant of patient outcomes. 1 The loss in cardiac output from an MI leads to a systemic activation of the sympathetic nervous system, 2 and the renin-angiotensin-aldosterone system. These responses aim to increase cardiac contractility, blood pressure, and salt and fluid retention to compensate for cardiac failure and ensure proper perfusion in peripheral organs. However, these compensatory mechanisms also increase maladaptive cardiac remodeling, such as fibrosis and hypertrophy, 3 as well as myocardial oxygen consumption. 2 Drug based therapy for ischemic heart disease has therefore targeted β-adrenergic receptors by using β-blockers, or the renin-angiotensin-aldosterone system by using Angiotensin converting enzyme inhibitors or Angiotensin II receptor 1 blockers. In addition to structural remodeling processes, cardiac metabolism also undergoes severe changes during and after MI. Cardiac metabolism is highly oxygen reliant, with approximately 95% of the ATP in the healthy heart coming from oxidative phosphorylation (OXPHOS). 4 During ischemia, glycolysis is drastically increased in an attempt to meet the energetic needs of the heart, which leads to excessive proton production and subsequent ionic disturbances. After ischemia, OXPHOS is reinstalled, however with a shift in substrate preference to fatty acid oxidation (FAO) over glucose oxidation (GOX). 4 This shift further increases ionic disturbances and has an oxygen wasting effect. 5 The use of metabolic modulators to improve myocardial metabolism after MI has emerged as a useful treatment strategy. Metabolic modulators aim to decrease FAO and increase GOX by targeting key metabolic regulators such as pyruvate dehydrogenase, enzymes of β-oxidation, carnitine palmitoyltransferase 1, or peroxisome proliferator-activated receptor transcription factors. 4
The systemic activation of the sympathetic nervous system during and after MI 6 impacts several other organs, including peripheral adipose tissue. The classical breakdown cascade of triacylglycerol (TAGs) is initiated by β-adrenergic stimulation and activation of protein kinase A. The first and rate limiting enzyme of TAG hydrolysis is adipose triglyceride lipase (ATGL). 7 Genetic knockout 8 or pharmacological inhibition of ATGL using the small molecule inhibitor Atglistatin 9 leads to a profound inhibition of adipose tissue lipolysis. The crosstalk between adipose tissue and the heart has been demonstrated in the setting of pressure overload-induced heart failure. By blocking lipolysis with either an adipocyte specific knockout of ATGL 10 or Atglistatin, 11 cardiac function was preserved after transverse aortic constriction and cardiac remodeling processes were attenuated. Since adipose tissue lipolysis is also increased in the setting of MI, 6 inhibiting lipolysis might serve as a new treatment strategy to improve cardiac metabolism and function post-MI. In the present study, we investigated the impacts of inhibiting lipolysis with the small molecule inhibitor Atglistatin in an ischemia/reperfusion mouse model on cardiac function. We demonstrate for the first time that inhibiting adipose tissue lipolysis is able to improve cardiac function after ischemia/reperfusion by enhancing contractile function of the remote myocardium.
Methods and Material
Animal Experiments
All animal experiments were performed according to local ACUC guidelines (ACUC—PHS Assurance # A3245-01, protocol # 3616).
For testing Atglistatin via gavage, mice were fasted overnight and received in the morning 100 µmol/kg BW Atglistatin dissolved in peanut oil or oil alone as vehicle. Blood was taken from the tail vein before administration as well as 4 and 8 h after gavage. 9
Atglistatin treatment in the myocardial infarction model was as follows: 12-week old C57BL/6 J male mice received 2 mg Atglistatin (#S7364, Selleckchem)/ 5 g chow 2 days prior to ischemia, while control mice received normal chow. Atglistatin cookies were prepared, based on a protocol by Schweiger et al, 12 by grinding the food, mixing the Atglistatin powder with the ground food and forming into cookies by adding a small amount of drinking water. Control mice received similar prepared cookies without Atglistatin. Mice were administered an additional bolus by oral gavage 3 h prior to ischemia with either 100 µmol Atglistatin/kg bodyweight in peanut oil, or peanut oil alone as a vehicle control. For ischemia, mice were anesthetized by i.p. injection with 120 mg/ 12 mg/kg bodyweight Ketamine/Xylazine, and orally intubated and ventilated using a small animal ventilator (MiniVent, Harvard Apparatus). Anesthesia was maintained throughout the remainder of the procedure with 1.5% isoflurane. The chest was opened in the 3rd intercostal space, and the left anterior descending (LAD) artery was identified and ligated for 60 minutes. Ischemia was verified for all experimental mice by ECG ST-segment elevation. For postoperative analgesia, mice received 0.05-0.1 mg/kg bodyweight Buprenorphine.
Echocardiography was performed on animals anesthetized with 1.5% isoflurane after 7 days of reperfusion using a Vevo 2100 small animal ultrasound device (Visual Sonics) with a MS400 probe. Access to Vevo 2100 was provided by Dr. John A. Hossack, Biomedical Engineering, UVA. Data were analyzed using Simpsons protocol 13 and Vevo Strain Software.
For organ harvest, mice were sacrificed by cervical dislocation.
Glycerol Assay
Measurement of plasma glycerol levels was performed using Free Glycerol Reagent (Sigma, #F6428) according to the manufacturers’ protocol.
Histology
WGA-FITC staining and analysis of scar size was performed on 8 µm slices of snap frozen raw tissue as previously described. 14 To assess the area at risk and infarct size, mice were euthanized by cervical dislocation after 24 h of reperfusion. The LAD was ligated again using the original suture, and hearts were perfused with a 5% phthalocyanine blue solution in physiological saline (Sigma, #252980). Hearts were then sectioned in 1 mm slices and stained with freshly prepared triphenyltetrazolium chloride (TTC) (Cayman, #17342). Briefly, heart sections were immersed in TTC staining solution (0.75% TTC in 100 mM Tris-HCl, pH 7.4) for 15 min at 37° C, followed by fixation with 10% formaldehyde solution for 2 h. Both apical and basal sides of each section were imaged, and ImageJ analysis software was used to manually demarcate and quantify the size of the area at risk (blue), healthy (red), and infarcted (unstained) tissue and calculated as a percentage of the total cross-sectional area.
Statistical Analysis
For statistical comparison of 2 groups with unequal standard deviations unpaired, 1-tailed Welch’s unequal variances t-test was used. For statistical comparison of 2 groups over different time points, 2-way ANOVA with Sidak’s multiple comparison test was used.
Results
Atglistatin Inhibits Lipolysis After Oral Gavage
We first tested the ability of Atglistatin, the ATGL inhibitor to block lipolysis. Mice were fasted overnight and received either Atglistatin or vehicle via oral gavage the next morning as previously described. 9 Plasma glycerol levels were reduced 4 and 8 h after administration of Atglistatin but not vehicle, indicating that Atglistatin inhibits lipolysis already 4 h after administration (Figure 1A). Atglistatin can also be administered via food, and oral administration of Atglistatin at these concentrations leads to inhibition of ATGL activity in adipose tissue with little impact on cardiac ATGL activity. 12 To ensure sufficient levels of Atglistatin were achieved, we established the following protocol for the ischemia experiments: 2 days prior to the induction of ischemia, mice received Atglistatin via chow while control mice received normal chow. 3 h prior to ischemia mice received an additional single dose of Atglistatin or vehicle via oral gavage (Figure 1B). Food intake measured over the 2 days of administration was comparable between the 2 groups (Figure 1C).
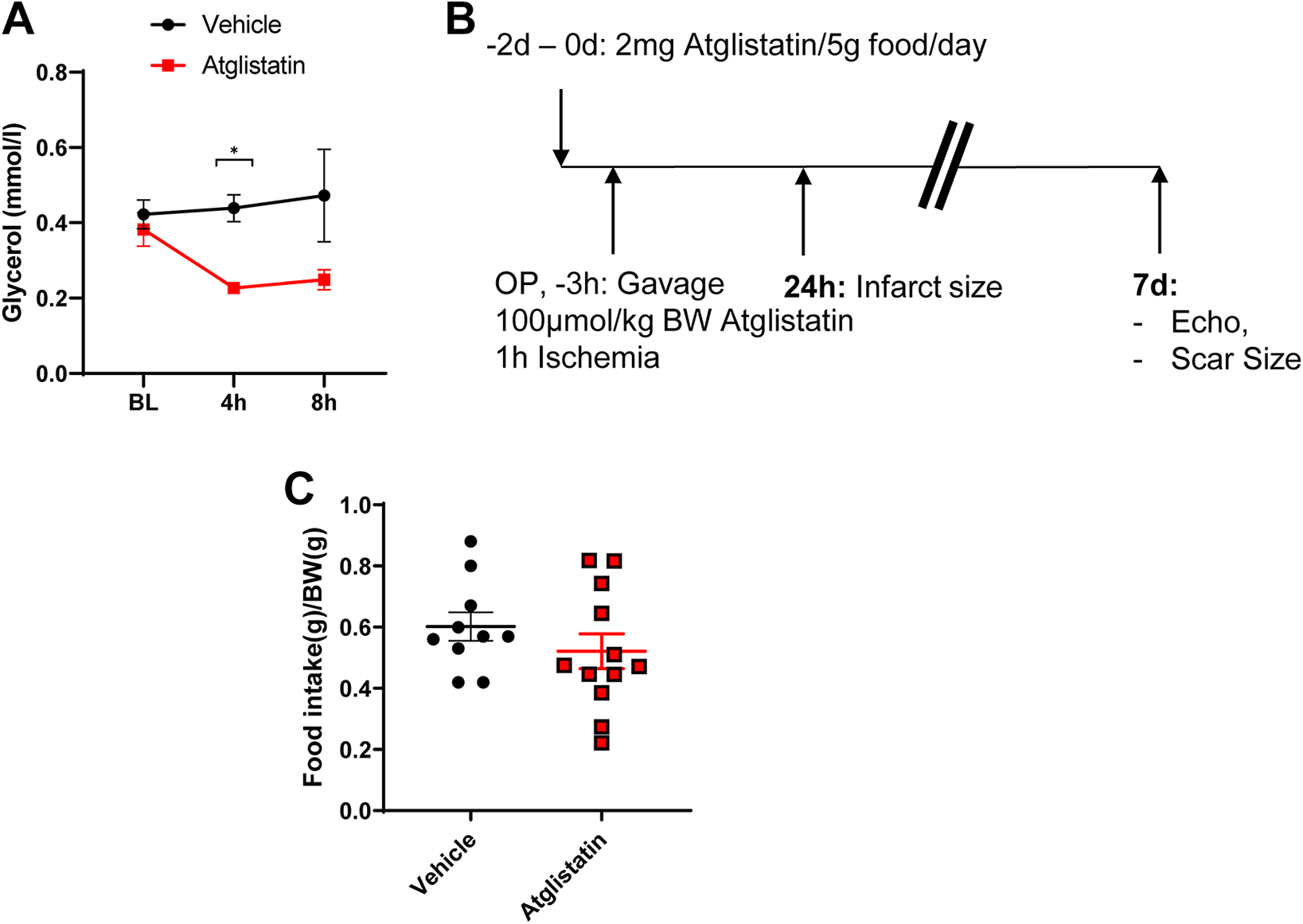
Atglistatin reduces glycerol levels. (A) Plasma glycerol levels after overnight fasting and oral gavage of Atglistatin or vehicle the next morning. n = 3-4, 2-Way ANOVA with Sidak’s multiple comparison test, *P < 0.05. (B) Treatment schematic. (C) Food intake over 2 days of Atglistatin treatment. Unpaired 1-tailed t-test with Welch’s correction, no statistical significant differences.
Pre-Ischemic Inhibition of Lipolysis Improves Cardiac Function After Myocardial Ischemia
To test the impact of adipose tissue lipolysis on cardiac function post-MI, we administered Atglistatin or vehicle control to 12-week old C57BL/6 J male mice. After 1 h of ischemia and 7 days of reperfusion, vehicle-treated mice displayed reduced systolic cardiac function as depicted by an ejection fraction (EF) of 26.5 ± 2.1%, a fractional shortening (FS) of 5.5 ± 0.9% and a fractional area change (FAC) of 20.6 ± 2.1% compared to untreated, unoperated control animals and demonstrating the success of the procedure. In contrast, Atglistatin-treated mice showed a marked improvement in cardiac function as depicted by an EF of 41.1 ± 1.7%, a FS of 8.2 ± 0.9% and a FAC of 41.1 ± 1.7% in comparison to vehicle treated mice (Figure 2A-C). Left ventricular dilatation and compensatory hypertrophy was comparable between the 2 groups, as there were no significant differences in end diastolic volume (Figure 2D) or heart weight to body weight ratios (Figure 2G).

Prophylactic Atglistatin treatment improves cardiac function after myocardial ischemia. (A-C) Parameters of cardiac function: Ejection fraction (EF) (A), fractional shortening (FS) (B) and fractional area change (FAC) (C) after 1 h ischemia/7 d reperfusion. (D-F) Cardiac volume parameters: end diastolic (EDV) (D), end systolic (ESV) (E), and stroke volume (SV) (F) after 1 h ischemia/7 d reperfusion. (G) Heart weight to body weight ratio, calculated from parasternal long axis images. n = 5. Data are presented as mean ± SEM. “Pre-treatment” group were untreated and unoperated 12 week old C57BL/6 J male mice, n = 10. Statistical comparison between “Atglistatin” and “Vehicle” groups using unpaired 1-tailed Welch’s unequal variances t-test, *P < 0.05, **P < 0.01.
Atglistatin Treatment Does Not Reduce Infarct Size
To investigate the underlying cause for the improved cardiac function after Atglistatin treatment in ischemic hearts, we analyzed scar size and infarct size. After 7 days of reperfusion, scar size was comparable between the treatment and vehicle groups (Figure 3A and B). Consistent with these results, the area at risk (AAR) and infarct size (IS) after 24 h of reperfusion also showed no significant differences between the 2 groups (Figure 3C and D).
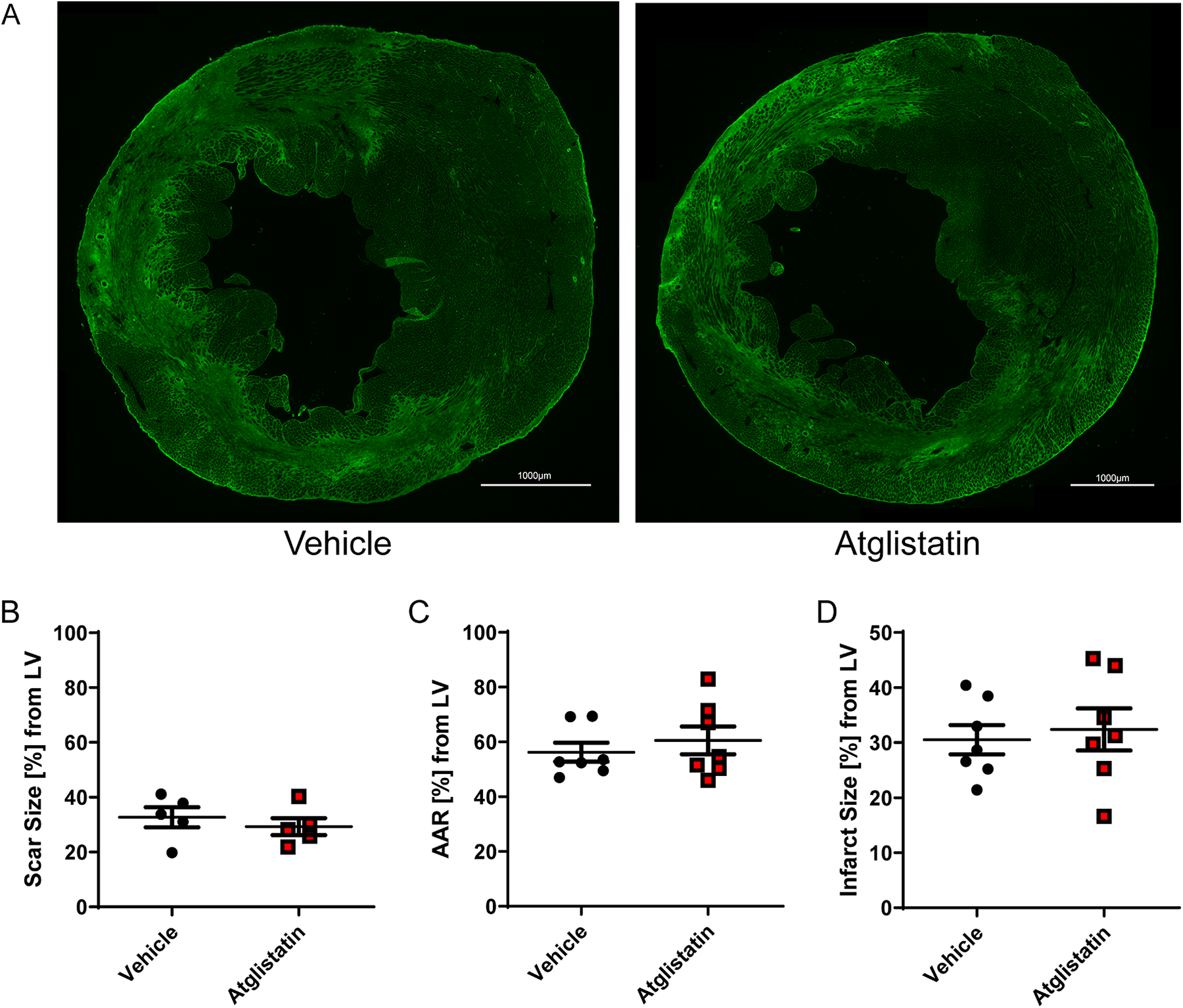
Atglistatin treatment does not affect infarct size. (A) WGA-FITC staining of hearts after 1 h ischemia and 7 d reperfusion after treatment of mice with either vehicle (left panel) or Atglistatin (right panel). (B) Quantification of scar size after 1 h ischemia/7 d reperfusion in hearts of mice either treated with vehicle or Atglistatin. (C-D) Quantification of C. area at risk (AAR), and (D) infarct size (IS) in hearts of mice treated either with vehicle or Atglistatin after 1 h ischemia and 24 h reperfusion using phthalo blue and TTC staining.
Atglistatin Treatment Improves Performance of Basal Wall Segments
Since there were no significant differences in infarct size or scar size between the 2 groups, we next investigated the performance of the remote myocardium. We measured peak radial strain and strain rate in different regions of the heart as a proxy for assessing changes in intrinsic myocardial contractility (Figure 4A and B). 15 In agreement with our results on infarct and scar size, peak radial strain (Figure 4C) and strain rate (Figure 4D) were similar in the apical and middle wall segments of the hearts for both groups. In contrast, peak radial strain for both, anterior and posterior basal wall segments was significantly increased in the Atglistatin treated group. Peak radial strain rate was also significantly higher in the posterior basal segment and strongly tended to significance in the anterior basal wall segment. These results indicate that the improvement in overall cardiac function from Atglistatin treatment is due to increased contractility in the remote myocardium.

Atglistatin improves performance in basal wall segments. A. Schematic of wall segments. B. Example graphic display of cardiac long axis radial strain of vehicle and Atglistatin treated mice. Anterior base: green, Posterior base: blue, Anterior mid: yellow, Posterior mid: white, Anterior apex: purple, Posterior apex: light blue. C. Radial strain peak (%) of all 6 wall segments for vehicle and Atglistatin treated mice. D. Radial strain rate (1/s) of all 6 wall segments for vehicle and Atglistatin treated mice.
Discussion
Cardiovascular disease is the number one cause of mortality worldwide, with ischemic heart disease accounting for over one-third of those deaths. 16 New, targeted therapeutic options are needed to improve patient outcomes after MI. Metabolic modulators are a class of drugs targeting cardiac metabolism to improve metabolic processes and thereby cardiac function. 17 In the present study, we used Atglistatin, a small molecule inhibitor of ATGL, as a metabolic modulator to improve cardiac function after MI by inhibiting peripheral adipose tissue lipolysis. During and after MI, the sympathetic nervous system is strongly activated; in addition to increasing cardiac contractility and promoting vasoconstriction, this activation also impacts other organs such as adipose tissue. 6 In adipose tissue, β-adrenergic activation stimulates the Gαs-coupled pathway to activate PKA and subsequently initiate TAG hydrolysis. The classical breakdown cascade starts with hydrolysis of TAGs to DAGs via the enzyme ATGL. DAGs and MAGs are then further hydrolyzed by hormone sensitive lipase (HSL) and monoglyceride lipase (MGL). Importantly, the hydrolysis of TAGs is the critical and rate limiting step in the breakdown cascade, and while other enzymes such as HSL are capable of hydrolyzing TAGs, genetic deletion of ATGL results in drastic reductions in lipolytic rates. 18 Whole body knockout of ATGL leads to severe defects in lipolysis and lipid accumulation in several tissues, including pathological TAG accumulation in the heart. 8 As a result of sympathetic nervous system activated adipocyte lipolysis, levels of circulating fatty acids are increased during myocardial ischemia. 19,20 Further, the availability of circulating fatty acids is a major determinant of the rate of fatty acid uptake by the heart. 21 There are several key ways in which increased FFA uptake by the heart has been shown to be detrimental to the heart and contribute to contractile dysfunction. First, increased oxidation of FA has an oxygen wasting effect, as β-oxidation consumes more oxygen per ATP molecule generated than glucose. 22 Next, due to the Randle cycle, 23 substrate preference for oxidation is shifted further toward acetyl-CoA from FA, thereby uncoupling glycolysis from glucose oxidation resulting in proton accumulation. 24 Finally, TAGs accumulate within cardiomyocytes which correlates with an increase in lipotoxic products such as DAGs and ceramides. 25,26 This hypothesis is supported by reports which show detrimental effects of lipid accumulation in cardiac tissue 8,27,28 or improved cardiac function and cardioprotection in the setting of increased cardiac lipolysis. 29 -31 Therefore, blocking the release of FFA by adipose tissue and thus preventing increased FA uptake by the heart may be a useful treatment strategy. Indeed, there are several reports confirming an inter-organ crosstalk between adipose tissue and the heart. Foryst-Ludwig et al showed that adipose tissue lipolysis is important for exercise induced cardiac hypertrophy, and that inhibiting lipolysis within adipose tissue reduces cardiac FA uptake. 32 Additionally, they identified C16:1 palmitoleic acid as a mediator of myocardial metabolism. 32 Other reports have shown in the TAC model that inhibiting lipolysis is beneficial for contractile function and is able to correct changes in the cardiac lipidome which occur in the pathological setting of pressure overload induced heart failure. 10,11 To block lipolysis, these studies used either adipocyte specific ATGL deficient transgenic mice or the small molecule inhibitor of ATGL, Atglistatin. As mentioned before, inhibiting ATGL is highly sufficient to block peripheral lipolysis, however inhibition of cardiac ATGL is detrimental and leads to cardiomyopathy due to excessive lipid accumulation. 33 Atglistatin has a half-life of 8 h, and due to its lipophilic properties it accumulates mainly in adipose tissue and liver thereby preventing these detrimental cardiac effects. 12
In the present study, we used Atglistatin in a preventive treatment strategy with oral administration prior to ischemia. With this protocol, we demonstrated that inhibiting lipolysis is able to improve outcomes after myocardial infarction. Pretreatment with Atglistatin significantly improved cardiac function 7 days after ischemia. Interestingly, this effect was not due to a decreased infarct size. Next, we further analyzed the function of the remote myocardium using strain analysis of segmental wall movement. We found that the basal wall segment displayed a higher peak radial strain and strain rate, indicating better contractile function of the remote myocardium after Atglistatin treatment. Not much is known about the metabolic state of the non-ischemic remote myocardium during and after ischemia. As the remote myocardium has to compensate for the loss of ischemic tissue, it undergoes several adaptation processes such as increased passive stiffness via titin domain phosphorylation. 34 Metabolically oxidative phosphorylation is increased immediately after acute MI, 35 yet decreased in later stages after MI. 36 As for substrate preferences, one could speculate that the remote myocardium resembles the pressure overloaded failing heart, as the workload of the myocardium is pathologically increased in both settings. The hallmark of the failing heart is an overall decrease of oxidative capacity and thereby reduced fatty acid oxidation and glucose oxidation. It has been shown that a decrease of FAO and a subsequent increase in GOX is beneficial in the setting of pressure overload. 29 However, the precise metabolic processes are still unclear and need to be elucidated. The present study gives a first hint that inhibiting lipolysis is beneficial in the setting of myocardial infarction, and that this modulation mainly targets the function of the remote myocardium. At present, it remains unclear whether prophylactic inhibition of lipolysis is necessary to produce the beneficial effects within the remote myocardium, or whether inhibition just prior to reperfusion may produce similar results. Importantly, we would expect the administration of β-blockers to provide these same beneficial effects through the inhibition of lipolysis. While most patients are placed on β-blocker therapy within 24 h of primary percutaneous coronary intervention or fibrinolytic therapy, 37 currently β-blockers are not recommended for early pre-procedural intervention as there is no clear evidence for their benefit. 38 Further, there are a number of groups with contraindications for β-blocker therapy, such as patients presenting with acute heart failure, hemodynamic instability, or AV block among others. 37 Isolating the inhibition of lipolysis from the inotropic effects of β-blockers may therefore provide a more effective therapeutic approach during the early management of STEMI and for patients with contraindications. Taken together, the inhibition of adipose tissue lipolysis may serve as a new metabolic modulator to treat ischemic heart disease.
Footnotes
Authors’ Note
No human studies were carried out by the authors for this article.
Acknowledgments
We thank Dr. John Hossack for access to Vevo 2100 small animal echocardiography device. The Vevo 2100 was funded by grant 1S10RR027333. We thank Dr. Axel Gödecke for access to Vevo Strain Software.
Author Contributions
K.B. and T.E.H. planned the study. K.B., M.E.G. and V.O. performed the experiments and analyzed the data. K.B., M.E.G and T.E.H. wrote the manuscript. T.E.H. and J.W.F. edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.E.H. is an inventor on a patent application relevant to this study filed by the University of Virginia through the UVA Licensing & Ventures Group (15/760,990).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health research grants R01 DK101946 to T.E.H. and T32 GM008715 to M.E.G.
