Abstract
Introduction:
Clinical data are scarce for furosemide administered as a low-dose (<160 mg/24 hours) continuous intravenous infusion in acute heart failure (HF). Our purpose was to evaluate the efficacy and safety of low-dose continuous infusion of furosemide on diuretic response, renal function, and patient outcomes.
Methods:
A retrospective study of patients with acute HF who received furosemide administered as a continuous infusion after initial therapy with intermittent boluses (usually 40-80 mg every 12 hours). End points included mean hourly urine output, incidence of acute renal injury, and outcome disparities of patients who developed acute renal injury. Comparison of patients with preserved and reduced left ventricular ejection fraction (LVEF) was also performed.
Results:
The study included 150 patients (age 57 ± 13 years, male gender 61%, admission weight 87 ± 32 kg, LVEF 37 ± 15%, 28% preserved LVEF). Mean initial and maximum furosemide doses were 5.1 ± 1.1 mg/h and 6.2 ± 2.2 mg/h, respectively. Mean duration of therapy was 51.4 ± 67.5 hours. Continuous infusion of furosemide was associated with a significant increase in mean hourly urine output compared to baseline (150 ± 77 mL/h vs 116 ± 69 mL/h, P < .001). Acute renal injury developed in 19% of patients, with 70% of those occurring within the first 48 hours of therapy. Mean serum creatinine (baseline 1.55 ± 1.50 mg/dL vs at discharge 1.64 ± 1.61 mg/dL, P = .20) and estimated glomerular filtration rate (baseline 67 ± 39 mL/min vs at discharge 67 ± 43 mL/min, P = .89) did not significantly change over the course of the hospitalization. Development of acute renal injury was associated with poorer outcomes, higher furosemide dose, and longer duration of furosemide therapy. Diuretic response and safety were not different between patients with preserved or reduced LVEF.
Conclusions:
In patients with acute HF, furosemide administered as a low-dose continuous infusion was effective in achieving diuresis and was not associated with a detectable effect on renal function. This diuretic approach appeared to be similarly effective and safe in patients with preserved LVEF.
Introduction
Loop diuretics remain the most common modality for the management of fluid balance in patients with heart failure (HF). 1 High-dose loop diuretics (≥160 mg furosemide equivalents) are needed in approximately one fourth of patients hospitalized for acute HF. 1 Data from the recent DOSE-AHF study comparing lower doses (median 358 mg over 72 hours) to higher doses (median 773 mg over 72 hours) suggested a modest advantage in patients’ global assessment of symptoms and net fluid loss with the higher dose. 2 However, the controversy over the comparative efficacy and safety of low versus high doses remains due to mixed data on other outcomes. 3 In the DOSE-AHF, efficacy achieved with the higher doses was at the expense of greater incidence of worsening renal function, at least in the short term. In addition, the literature supports an association between high bolus dose diuretics and poorer outcomes. In the Acute Decompensated Heart Failure Registry, higher diuretic dose was associated with worse renal function, increased hospital length of stay (LOS), and increased mortality compared with patients who received lower doses. 1
An alternate approach that is commonly employed to enhance diuresis without escalating doses is administration of loop diuretics via continuous intravenous infusion. This approach may be superior to intermittent bolus administration as it negates the potential for acute postdiuretic compensatory sodium reabsorption. 4 –7 In addition, since high peak drug concentrations are avoided, there may be a reduction in diuretic-induced neurohormonal activation and a lower propensity for adverse effects on electrolytes and blood pressure. 8,9
Studies investigating the comparative efficacy and safety of loop diuretics administered as intermittent boluses versus continuous infusion have been far from conclusive. A pooled analysis of prospective, randomized, controlled studies prior to 2004 concluded an advantage to continuous infusion in terms of increased urine output and decreased incidence of tinnitus. 3 However, the studies included in the analysis were quite small (N = 254 total), heterogeneous in design, and the advantage was lost when 2 studies utilizing hypertonic saline were excluded. Recent studies have failed to reach a consensus. Allen et al 10 found no advantage with continuous infusion, whereas Thomson et al 11 did find a significant increase in urine output with equivalent doses of furosemide administered via continuous infusion compared to intermittent boluses. The DOSE-AHF study found no differences in patient global assessment of symptoms, net fluid loss, change in weight, renal safety, or hospital LOS based on the mode of administration. 2 However, whether there was truly no difference between the bolus and continuous infusion regimens is equivocal as there was a trend toward lower total diuretic dose in the continuous infusion group.
Although the DOSE-AHF study was well designed and is widely considered a landmark trial, additional data with alternate regimens are still needed to help clinicians decide on an optimal diuretic regimen for a given patient with acute HF. All the above studies, with the exception of DOSE-AHF, reflect daily doses of furosemide in excess of 160 mg/24 h. In DOSE-AHF, an analysis of patients who received lower doses and as a continuous infusion was not reported. Therefore, clinical data for furosemide administered as a low-dose continuous intravenous infusion in acute HF remain limited. Considering the potential clinical advantages of utilizing lower doses of furosemide, the purpose of our study was to further characterize the effects of low-dose continuous infusion of furosemide on diuretic response, renal function, and patient outcomes in a wide range of patients with acute HF.
Study Design and Methods
Study Design
A retrospective analysis of data from consecutive hospitalized patients with HF admitted to the cardiac intensive care unit (CICU) at the Los Angeles County and University of Southern California (LAC + USC) Medical Center was conducted. The study was approved by the LAC + USC Medical Center Institutional Review Board.
Patient Population
Adult patients admitted to the CICU at LAC + USC Medical Center between April 2009 and July 2010 were screened for inclusion. Patients were included if they were treated with a continuous intravenous infusion of furosemide for at least 3 hours after being initially treated with intermittent boluses of furosemide. Patients were excluded if there was missing data regarding urine output while on intermittent bolus furosemide (no baseline data), or if the continuous infusion of furosemide was not administered for at least 3 consecutive hours without interruption. Patients who received additional diuretics in combination (ie, metolazone) with the continuous infusion of furosemide were excluded. Patients presenting with a baseline serum creatinine ≥5 mg/dL or those who received renal replacement therapy at anytime during the hospitalization were also excluded.
Data Collection
Data were collected from the Affinity and Eclipsys medical charting systems which are used in the CICU at the LAC + USC Medical Center. Data related to the continuous infusion of furosemide regimen included initial, maximum, and minimum doses, and duration of therapy. The doses and durations of therapy were decided by the primary team caring for each patient. Patient demographic data included patient characteristics (past medical history of ischemic heart disease, hypertension, arrhythmia, dyslipidemia, diabetes, current smoker, and/or current alcohol intake), HF characteristics (ischemic or nonischemic etiology, New York Heart Association Functional Classification [NYHA FC], left ventricular ejection fraction [LVEF]), other concomitant cardiovascular medications received during the continuous infusion of furosemide therapy (nitroglycerin, nesiritide, nitroprusside, dobutamine or milrinone, dopamine, norepinephrine, epinephrine, phenylephrine, vasopressin, other vasopressors, beta-blockers, calcium channel blockers, angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, hydralazine, oral nitrates, digoxin, spironolactone, and amiodarone), and patient disposition (length of hospitalization and in-hospital mortality). Data collected related to the study end points including hourly vital statistics, urine output, net fluid balance, and serum chemistries.
Statistical Analysis
The primary efficacy end point of the study was the difference in mean hourly urine output during continuous infusion of furosemide compared to baseline (intermittent bolus furosemide). Mean difference in net fluid balance was also evaluated. The primary safety end point was the mean difference in serum creatinine at discharge compared to baseline. This end point was chosen to reflect whether continuous infusion of furosemide therapy was associated with any lasting effects on renal function. Secondary renal safety end points included mean difference in serum creatinine at 24 hours compared to baseline, mean difference in peak serum creatinine during continuous infusion of furosemide therapy compared to baseline, mean difference in blood urea nitrogen at 24 hours, and at discharge compared to baseline, and mean difference in glomerular filtration rate (GFR), estimated by the Modified Diet in Renal Disease formula, at peak serum creatinine compared to the baseline. The incidence of acute renal injury, defined as an absolute increase in serum creatinine of 0.3 mg/dL or greater, or a 25% increase above baseline, was also assessed. Other safety end points included incidence of electrolyte abnormalities (hyponatremia [<135 mEq/L], hypokalemia [<3.5 mEq/L], hypomagnesemia [<1.7 mEq/L]) and incidence of hypotension (systolic blood pressure less than 80 mm Hg or mean arterial pressure less than 50 mm Hg) during continuous infusion of furosemide therapy. The primary efficacy and primary safety end points were analyzed using a paired samples t test. Significance was set at P < .05. Secondary end points were reported only as descriptive statistics.
To explore the effect of acute renal injury on the efficacy and safety of low-dose continuous infusion of furosemide, each of the above end points was compared between cohorts of patients who did and did not meet criteria for acute renal injury. In addition, it is well documented that the pressure–volume hemodynamics of patients with HF with preserved LVEF are dissimilar to patients with reduced LVEF. 12,13 To explore whether outcomes varied based on LVEF, each end point was also evaluated based on left ventricular function. The comparison of end points between the cohorts described above was conducted with an independent samples t test or chi-square test (or Fisher exact test) for continuous and nonparametric data, respectively. Significance was set at P < .05.
Results
Patient Population
A total of 150 patients were included in the study. Mean age was 57 ± 13 years, mean weight was 87 ± 32 kg, and 61% were male. The majority (73%) of patients had HF of nonischemic etiology. The mean LVEF was 37± 15%, and 28% of patients had a preserved LVEF (LVEF ≥50%). The most common comorbid conditions included hypertension (72%) and diabetes (45%). The mean duration of hospitalization was 34.8 ± 86.7 hours (median of 12 hours) before the low-dose continuous infusion of furosemide was initiated. Patients were treated with low-dose continuous infusion of furosemide (mean initial, maximum, and minimum doses of 5.1 ± 1.1 mg/h, 6.2 ± 2.2 mg/h, and 4.8 ± 1.3 mg/h, respectively) for a mean duration of 51.4 ± 67.5 hours (median of 30 hours). Intravenous vasoactive therapy was commonly used (75%), with the majority receiving intravenous nitroglycerin (55%), nesiritide (19%), or dopamine (11%). The mean hospital LOS was 7.3 ± 7.5 days, with an in-hospital mortality rate of 2%. A summary of the demographic data and in-hospital medication use are provided in Tables 1 and 2.
Baseline Characteristics of Study Population

Abbreviations: HTN, hypertension; IHD, ischemic heart disease; LOS, length of stay; LVEF, left ventricular ejection fraction.
In-Hospital Use of Medication

Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker.
Continuous Infusion Efficacy and Safety
Compared to baseline (intermittent bolus administration), continuous infusion of furosemide was associated with a significant increase in mean hourly urine output (150 ± 77 mL/h continuous infusion vs 116 ± 69 mL/h baseline, P < .001). The mean difference in hourly urine output and net negative fluid balance between continuous infusion of furosemide and baseline was 34 ± 88 mL/h and 28 ± 117 mL/h, respectively (Table 3).
Effect on Urine Output and Fluid Balance

Abbreviation: UO, urine output.
a P < .001 versus mean hourly UO at baseline.
In terms of safety, continuous infusion of furosemide was not associated with a significant change in renal function (Table 4). The mean serum creatinine at 24 hours (1.55 ± 1.26 mg/dL) and peak serum creatinine during therapy (1.63 ± 1.42 mg/dL) was not different than baseline (1.55 ± 1.50 mg/dL). Mean serum creatinine at discharge (1.64 ± 1.61 mg/dL) was also not different compared to baseline. Mean estimated GFR determined using the peak serum creatinine during therapy (60 ± 30 mL/min) was not different from baseline (67 ± 39 mL/min). Blood urea nitrogen concentrations during therapy were not significantly changed compared to baseline. Overall, 19% of patients met criteria for acute renal injury, as defined by an increase in serum creatinine of 0.3 mg/dL or 25% from baseline, with 70% of these occurring within the first 48 hours of therapy. Hyponatremia (38%), hypokalemia (25%), and hypomagensemia (15%) were common in our study population. Of note, 19% of the patients were hyponatremic (<135 mEq/L) at baseline. Baseline mean systolic and diastolic blood pressures were 119 ± 24 and 72 ± 15 mm Hg, respectively. Both systolic and diastolic blood pressures tended to be slightly lower during therapy with continuous infusion of furosemide, with a 22% incidence of patients experiencing at least 1 systolic blood pressure less than 80 mm Hg.
Effect on Renal Function, Electrolytes, and Blood Pressure

Abbreviations: BUN, blood urea nitrogen; DBP, diastolic blood pressure; GFR, estimated glomerular filtration rate; HR, heart rate; MAP, mean arterial pressure; SBP, systolic blood pressure; Scr, serum creatinine.
Comparison for Acute Renal Injury
Acute renal injury developed in 19% patients. At baseline, there were no differences in age, gender, past cardiac medical history, or in HF characteristics between those who did or did not develop acute renal injury (data not shown). Prognosis was worse in patients who developed acute renal injury indicated by longer duration of hospitalization (10.8 ± 10.2 days vs 6.4 ± 6.5 days, P = .035) and higher in-hospital mortality (7% vs 1%, P = .09). Patients who developed acute renal injury also received a higher maximum dose of furosemide than those who did not develop acute renal injury (7.4 ± 2.8 mg/h vs 5.9 ± 1.9 mg/h, P = .01) despite the initial doses (5.0 ± 1.0 mg/h vs 5.1 ± 1.2 mg/h, P = .743) being similar. Acute renal injury was also associated with a longer average duration of diuretic treatment (103 ± 125 h compared to 40 ± 37 h, P = 0.01). Use of intravenous inotropes (18% vs 5%, P = .018) and norepinephrine (11% vs 1%, P = .021) were more common in patients who developed acute renal injury. Frequency of use of all other medications was not different.
Patients not developing acute renal injury had a greater urine output at baseline (124 ± 67 mL/h vs 82 ± 67 mL/h, P = .003) and on treatment (160 ± 80 mL/h vs 106 ± 47 mL/h, P = .001) compared with those who developed acute renal injury (Table 5). However, the difference in urine output between baseline and on treatment was not different for both groups (P = .498). The difference in net fluid balance with treatment was also not different.
Outcomes Stratified by Development or Absence of Acute Renal Injury
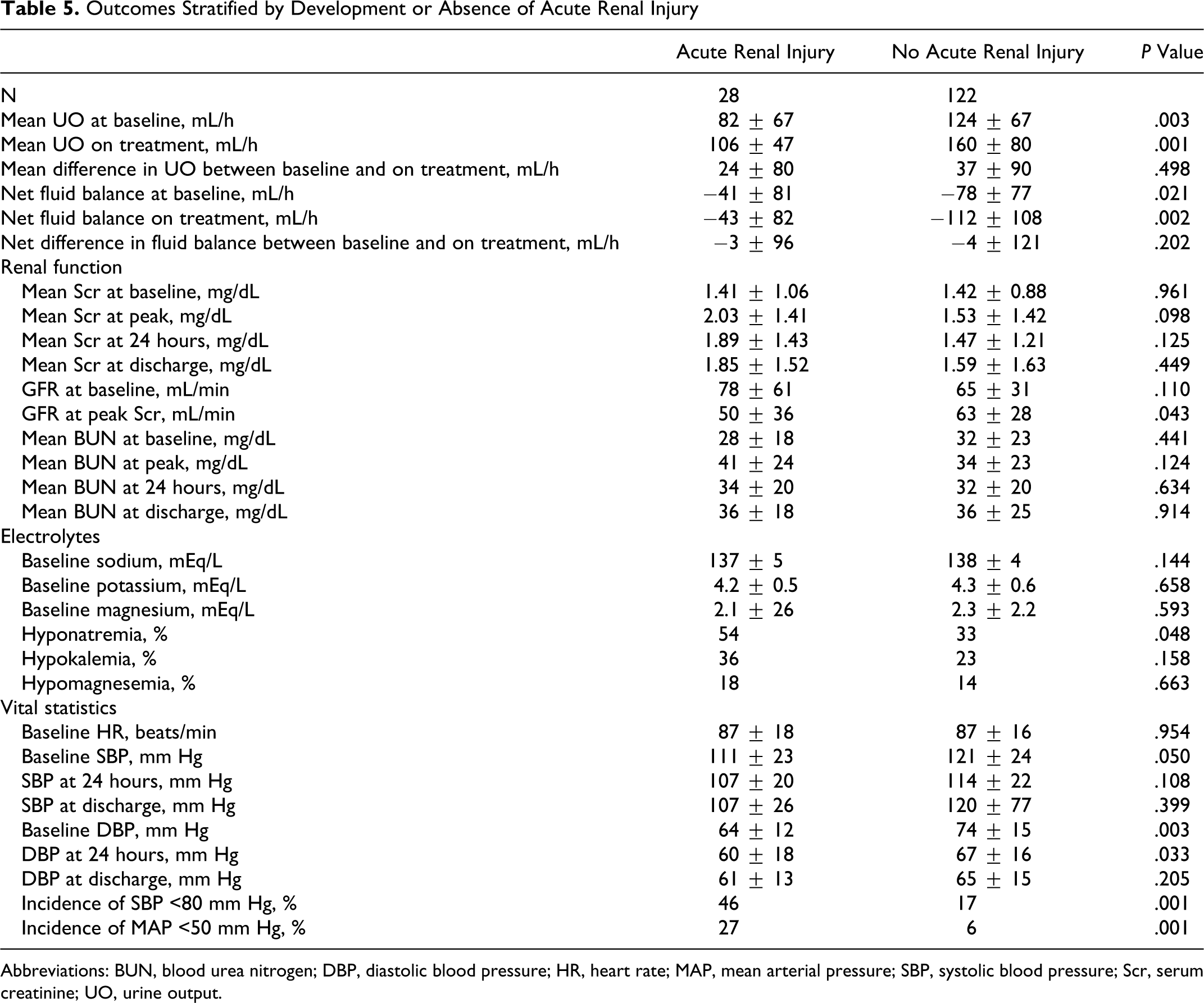
Abbreviations: BUN, blood urea nitrogen; DBP, diastolic blood pressure; HR, heart rate; MAP, mean arterial pressure; SBP, systolic blood pressure; Scr, serum creatinine; UO, urine output.
There were no differences in mean serum creatinine, estimated GFR, or blood urea nitrogen concentrations between groups at baseline (Table 5). Mean peak serum creatinine was numerically higher (2.0 ± 1.4 mg/dL vs 1.5 ± 1.4 mg/dL, P = .098) and mean decrease in GFR was significantly greater (−28 ± 28 mL/min vs −2 ± 11 mL/min, P < .001) in patients who developed acute renal injury, however, serum creatinine and GFR at discharge was not different between groups. Mean blood urea nitrogen concentrations were not significantly different at any measured time point. There were no differences in electrolytes at baseline, however, patients who developed acute renal injury were more likely to also develop hyponatremia (P = .048). Baseline systolic and diastolic blood pressures were lower in the acute renal injury group. The incidence of hypotension was more frequent in patients who developed acute renal injury.
Comparison Between Preserved Versus Reduced LVEF
Efficacy and safety outcomes were compared for patients with preserved LVEF (LVEF ≥50%) and reduced LVEF (LVEF <50%). Of the 150 patients in the study, 28% had preserved LVEF (Table 6). Mean LVEF was 58 ± 5% and 29 ± 9% in the preserved and reduced LVEF groups, respectively. Patients with preserved LVEF were shorter (163 ± 10 cm vs 168 ± 12 cm, P = .044), and more commonly female (57% vs 32%, P = .004) and diabetic (64% vs 38%, P = .004). Age, other comorbid conditions, and HF characteristics were not different (data not shown). In-hospital mortality was not different; however, the duration of hospitalization was numerically longer in those with preserved LVEF (8.9 ± 9.4 days vs 6.6 ± 6.5, P = .147). There were no significant differences between groups in mean initial (5.3 ± 1.3 mg/h vs 5.0 ± 1.0 mg/h, P = .16) or maximum (6.2 ± 2.0 mg/h vs 6.2 ± 2.3 mg/h, P = .919) dose of furosemide administered. Use of other cardiovascular medications was also not different between groups, with the exception of beta-blockers being used less (36% vs 55%, P = .033) and calcium channel blockers being more common (29% vs 9%, P = .003) in patients with preserved LVEF.
Outcomes Stratified by Left Ventricular Ejection Fraction
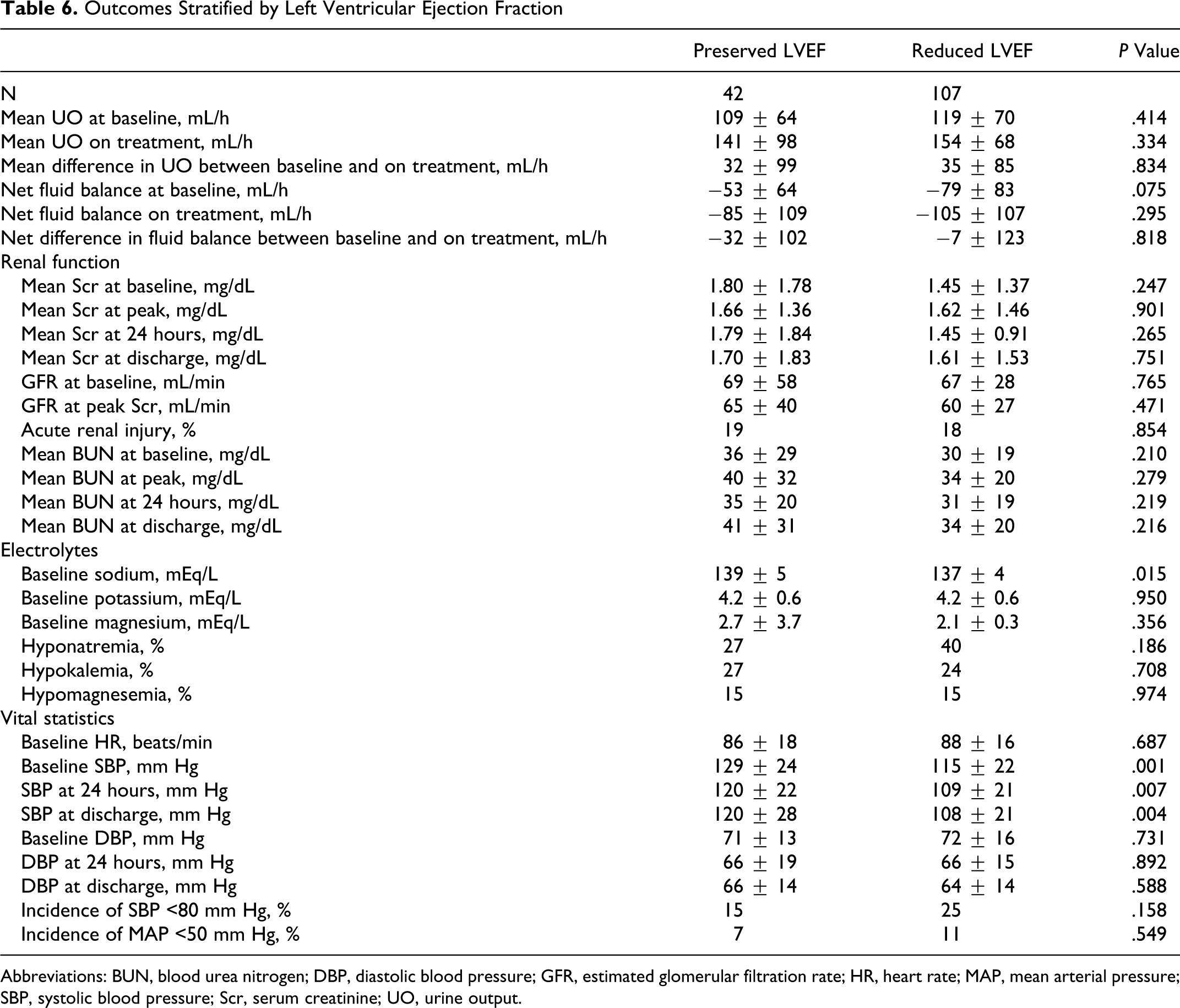
Abbreviations: BUN, blood urea nitrogen; DBP, diastolic blood pressure; GFR, estimated glomerular filtration rate; HR, heart rate; MAP, mean arterial pressure; SBP, systolic blood pressure; Scr, serum creatinine; UO, urine output.
There were no significant differences in baseline or on treatment mean hourly urine outputs based on LVEF (Table 6). Net fluid balance was also not different between groups. Baseline serum creatinine and blood urea nitrogen concentrations tended to be worse in patients with preserved LVEF, although the estimated GFR was not different. There were no significant differences in mean serum creatinine, GFR, or blood urea nitrogen concentrations during treatment with low-dose continuous infusion of furosemide or upon discharge between patient with or without preserved LVEF. Incidence of acute renal injury was also not different (19% preserved LVEF vs 18% reduced LVEF, P = .85). Systolic blood pressure, but not diastolic blood pressure, was significantly higher in patients with preserved LVEF (129 ± 24 vs 115 ± 22 mm Hg, P = .001) at baseline. Incidence of hypotension was not significantly different between groups, although the frequency of patients experiencing a systolic blood pressure <80 mm Hg was numerically lower in those with a preserved LVEF.
Discussion
Determination of the optimal method for using diuretics in the management of fluid volume in patients with acute HF remains an ongoing issue. Most, but not all, available literature suggests that using the lowest effective dose of a loop diuretic to achieve the appropriate clinical response is desirable. Administration via continuous infusion may facilitate this goal as this strategy reflects the application of pharmacokinetic properties of loop diuretics and the pathophysiology underlying diuretic resistance. However, available evidence on the efficacy and safety of lower doses of furosemide administered as a continuous infusion in HF are limited. Our analysis of a large cohort of patients hospitalized for acute HF who were initially treated with intermittent bolus furosemide suggests that low-dose continuous infusion of furosemide is effective in achieving diuresis with a favorable safety profile. This was reflected in an increase in mean hourly urine output, and no detectable difference in serum creatinine or blood urea nitrogen concentrations, or estimated GFR over both the course of the infusion and the duration of the hospitalization. Mean net negative fluid balance was also numerically higher on the low-dose continuous infusion of furosemide. Importantly, the mean initial and peak doses of furosemide were only 5.1 and 6.2 mg/h, respectively, suggesting little dosage titration was necessary.
Although our study was not prospective and randomized, our results are in general agreement with existing literature comparing continuous infusion to intermittent bolus furosemide, while providing evidence for a dose range which has not been well studied. In terms of efficacy, our data suggest a small advantage with continuous infusion (mean difference 34 mL/h). Prior to 2010, there were only small studies (≤20 participants) which had compared furosemide administered as a continuous infusion versus intermittent bolus. 5,14 –16 The majority of these studies found small but statistically significant greater diuresis and natriuresis with continuous infusion. A pooled analysis found a weighted mean difference of 271 cc/24 h which is slightly less, but comparable, to the mean difference we observed. 3 There was significant heterogeneity in these studies, with the continuous infusion hourly dose of furosemide ranging from 2.5 to 86 mg/h, and the duration of infusion varying from 4 to 48 hours. In a more recent prospective, randomized, parallel-group study involving 56 patients with HF, the difference in mean urine output was 523 mL/24 h with continuous infusion (mean dose furosemide 8 mg/h) versus intermittent bolus which was also close to our finding. 11 However, a single-center prospective, open-label study of 41 patients with HF who were randomized to a mean dose of furosemide of approximately 7.5 mg/h administered as either a continuous infusion or twice-daily bolus injections, found no difference in cumulative urine output after 3 days. 10 The most contemporary and largest prospective, randomized study to compare continuous infusion to bolus furosemide every 12 hours, also found no difference in net fluid loss at 72 hours. 2 However, there was a trend toward a greater median total dose of furosemide equivalents with bolus administration (592 mg bolus group vs 480 mg continuous infusion group, P = .06), which could potentially have confounded the net fluid loss end point.
In terms of safety, we did not detect a significant effect of low-dose continuous furosemide on renal function as assessed by changes in serum creatinine, blood urea nitrogen concentrations, and estimated GFR. The recent DOSE-AHF and Thomson studies also found no difference in the incidence of acute worsening of renal function during the course of therapy. 2,11 No previous study reported a difference in effect on blood pressure, which is also in agreement with our findings.
Influence of Acute Renal Injury on Outcomes
Development of worsening renal function remains an important prognostic indicator in acute HF. In the current study, acute renal injury, defined by an increase in serum creatinine of 25% over baseline or an absolute increase of ≥0.3 mg/dL, occurred in 19% of these patients who were diuresed with low-dose continuous infusion of furosemide. Consistent with previous literature, acute renal injury was associated with a longer hospital LOS and a trend toward increased in-hospital mortality. 17,18 These patients required higher diuretic doses and longer durations of diuretic therapy. Use of intravenous inotropes and vasopressors was also more common which could have been a consequence of the increased incidence of hypotension seen in the acute renal injury cohort. Interestingly, the development of renal injury tended to occur early, within the first 48 hours of treatment. Our rate of renal injury is consistent with other acute HF studies which report an incidence varying between 17% and 45%. 2,11,17 –20 Although we did not detect significant changes in mean serum creatinine and blood urea nitrogen in this study, the incidence and timing of the observed acute renal injury suggest that the use of low-dose continuous infusion of furosemide does not preclude the risk of renal injury and that close clinical monitoring remains an essential part of managing diuretic therapy.
Influence of LVEF on Outcomes
The physiologic relationship between left ventricular end-systolic pressure and volume is much steeper in patients with HF and preserved LVEF, or diastolic dysfunction, and left ventricular end-diastolic pressure tends to be elevated. 13 Therefore, small changes in volume can lead to large changes in left ventricular pressures and as a consequence, HF symptoms may present without substantial volume overload in these patients. Limited data exist evaluating loop diuretics in HF with preserved LVEF. 21 They have been shown to improve the quality of life and reduce hospitalizations in the setting of chronic HF with preserved LVEF. 21 In an observational study of hospitalized patients with HF with preserved LVEF, absence of diuretic therapy upon admission was predictive of poor in-hospital survival. 22 From 1 registry, intravenous diuretics are used in the vast majority of hospitalized patients with HF regardless of LVEF and with similar frequency. 23 However, no study has reported whether diuretic response, dosage requirements, or acute safety of loop diuretics is different in acute HF with reduced versus preserved LVEF. In our analysis, there were no significant differences in any of the efficacy and safety end points for low-dose continuous infusion of furosemide between patients with reduced or preserved LVEF. This strategy appears to provide similar diuresis in HF with preserved LVEF without increasing the risk of overdiuresis and/or hypotension that can lead to acute worsening of renal function.
Limitations
The study has limitations. First, the observational nature of the study means that the furosemide dose and duration of therapy was at the discretion of the managing physician. Therefore, it cannot be confirmed whether the overall safety and lack of observed effect on renal function were due to other factors such as close clinical monitoring by the managing health care team. The lack of randomization to parallel treatment strategies also precludes firm conclusions regarding the superiority of the continuous infusion over intermittent bolus administration. In addition, the exact intermittent bolus doses reflecting the baseline phase were not ascertainable as doses given in the emergency department were not readily retrievable, so it is possible that the greater diuresis was a result of higher drug exposure. However, as described above, the results are consistent with much of the existing prospective study literature while providing “real-world” experience with a dose range with limited data. Second, the only loop diuretic included in the analysis was furosemide, thus the conclusions cannot be extrapolated to the more potent loop diuretics torsemide and bumetanide. Although furosemide is the most widely used loop diuretic, it remains controversial whether the more potent (on a mg per mg basis) loop diuretics should be used first line. 24 The rationale for the use of the other more potent loop diuretics is related to their potential superiority in achieving diuresis without a signal for increased adverse events. 25 –27 Finally, the exclusion of patients who received renal replacement therapy may have resulted in our incidence of acute renal injury being an underestimate of the true risk. However, it was felt that these patients should be excluded as it would confound the majority of the other laboratory-based end points.
Conclusions
In a cohort of diverse patients with acute HF, furosemide administered as a low-dose continuous infusion was effective in achieving clinically significant diuresis and was not associated with a detectable adverse effect on renal function. It was also found to be similarly effective and safe in patients with preserved LVEF. Low-dose continuous infusion of furosemide may be a rational choice for initial diuretic therapy in many patients with acute HF.
Footnotes
Acknowledgments
The authors would like to thank Tesfa Ghebreyesus for his help in extracting study data from the CICU computer system.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tien Ng has received compensation as a consultant and research funding from Otsuka America Pharmaceutical, Inc. Uri Elkayam has received research funding from Amgen, Nile, Zensun, Otsuka, and NIH.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
