Abstract
Background:
Furosemide is associated with poor prognosis in patients with heart failure and reduced ejection fraction (HFrEF).
Aim:
To evaluate the association between daily furosemide dose prescribed during the dry state and long-term survival in stable, optimally medicated outpatients with HFrEF.
Population and Methods:
Two hundred sixty-six consecutive outpatients with left ventricular ejection fraction <40%, clinically stable in the dry state and on optimal heart failure therapy, were followed up for 3 years in a heart failure unit. The end point was all-cause death. There were no changes in New York Heart Association class and therapeutics, including diuretics, and no decompensation or hospitalization during 6 months. Furosemide doses were categorized as low or none (0-40 mg/d), intermediate (41-80 mg/d), and high (>80 mg). Cox regression was adjusted for significant confounders.
Results:
The 3-year mortality rate was 33.8%. Mean dose of furosemide was 57.3 ± 21.4 mg/d. A total of 47.6% of patients received the low dose, 42.1% the intermediate dose, and 2.3% the high dose. Receiver operating characteristics for death associated with furosemide dose showed an area under the curve of 0.74 (95% confidence interval [CI]: 0.68-0.79; P < .001), and the best cutoff was >40 mg/d. An increasing daily dose of furosemide was associated with worse prognosis. Those receiving the intermediate dose (hazard ratio [HR] = 4.1; 95% CI: 2.57-6.64; P < .001) or high dose (HR = 19.8; 95% CI: 7.9-49.6; P < .001) had a higher risk of mortality compared to those receiving a low dose. Patients receiving >40 mg/d, in a propensity score–matched cohort, had a greater risk of mortality than those receiving a low dose (HR = 4.02; 95% CI: 1.8-8.8; P = .001) and those not receiving furosemide (HR = 3.9; 95% CI: 0.07-14.2; P = .039).
Conclusion:
Furosemide administration during the dry state in stable, optimally medicated outpatients with HFrEF is unfavorably associated with long-term survival. The threshold dose was 40 mg/d.
Introduction
The past decades have witnessed major advances in the diagnosis and treatment of heart failure (HF). Optimal treatment of HF with a reduced ejection fraction (HFrEF) requires receiving the recommended medication at the highest tolerated dose. 1,2
Furosemide, a loop diuretic with potent natriuretic properties, has a Class I Level of Evidence C and is administered for relief of symptoms in patients with congestion. 1,2 This agent has been proved to be effective in managing congestion 3 and is a common chronic therapy in patients with HF. 4 Most patients with acute HF decompensation remain stable on a low maintenance dose of furosemide after clinical stabilization with a high dose. 5 Several studies have shown that increasing the dose of loop diuretic therapy has a greater risk of mortality. 6 -11 Others have suggested that the dose of diuretics is only a sign of clinical instability, which is in turn a prognostic marker. 10 The question remains what should be furosemide optimal dose, in the long term, for the symptomatic control of ambulatory patients with HFrEF. To clarify this issue, patients should be in a recompensated “dry state.” The ideal “dry” state for these patients should not be an overzealous euvolemic state with the consequent dehydration 5 but an “optivolemic” state reflecting an optimal balance between the signs and symptoms of congestion and a low cardiac output.
The main objective of this study was to evaluate the association between the daily furosemide dose, prescribed during the dry state, and the long-term survival in stable outpatients with HFrEF on optimized therapy.
Methods and Materials
Study Cohort
A total of 266 consecutive patients were enrolled in the present study according to the inclusion criteria. Patients were recruited from a single specialized outpatient heart failure clinic in a university hospital between May 2010 and October 2013.
Inclusion criteria were as follows: systolic HF according to the European Society of Cardiology (ESC) guidelines, 12 including evidence of left ventricular systolic dysfunction with an ejection fraction <40% by transthoracic echocardiography; patients completed a nurse-led HF education program; patients achieved optimal HF therapy (according to 2 cardiologists of the unit); and patients were clinically stable. Clinical stability was defined as no hospital admission or HF decompensation (no additional consultation in the HF clinic) and no change in New York Heart Association (NYHA) class and the furosemide dosage during a 6-month period. These conditions ensured the dry state, clinical stability, and optimal treatment (medicine and devices). This study conformed to the principles outlined in the 1964 Declaration of Helsinki. The protocol was approved by the institutional review board, and written consent was obtained from all patients.
Definition of Optimal Medical Treatment
Heart failure therapy (medicine and devices) followed the ESC guidelines for chronic HF. 1 Titration to either the target dose (TD) or the maximum tolerated dose was performed by medical staff and by nurses specialized in HF under medical supervision at the HF unit and according to the guidelines. Target doses for angiotensin-converting enzyme inhibitors (ACEi), angiotensin receptor blockers (ARBs), and β-blockers (BBs) were based on the ESC guideline recommendations, and the percentage of the TD was calculated. The cohort was categorized based on the following cutoff values: (1) not using furosemide, (2) <50% of the TD, and (3) ≥50% of the TD. Because the main focus of medical treatment in HF is a combination of drugs to induce neurohormonal blockage, the following subgroups were used: (1) ACEi/ARB + BB (yes/no therapy), (2) ACEi/ARB + BB ≥50% of the TD (both drugs ≥50% of the TD), and (3) ACEi/ARB + BB + spironolactone.
The furosemide dose was arbitrarily stratified according to 2 cutoff values: (1) 40 mg/d (0, 1-40 mg/d, >40 mg/d) and (2) 80 mg/d (0, 1-40 mg/d, 41-80 mg/d, >80 mg/d). These doses correspond to 1 (40 mg/d) and 2 or more (80 mg/d) furosemide tablets.
Baseline Characteristics
At the baseline visit, several parameters, including clinical, laboratory, electrocardiographic, and echocardiographic determinants, were collected. Demographic and clinical characteristics from medical charts, including age, sex, body mass index, medical history (comorbidities and etiology), treatment and doses, NYHA functional class, laboratory data (hemoglobin, renal function, including estimated glomerular filtration rate [GFR; as defined by the Modification of Diet in Renal Disease method], electrolytes, Nt-ProBNP levels), heart rhythm, and echocardiographic variables (left ventricular ejection fraction [LVEF] and tricuspid annular plane systolic excursion), were collected. All laboratory variables were analyzed, as a routine protocol, by the Clinical Chemistry Laboratory at the Lisbon North Hospital Centre.
Follow-Up and Clinical Outcome Data
Patients were assessed at scheduled clinical visits in the HF clinic during a 3-year follow-up. The regular schedule included a minimum of quarterly visits to the HF unit. Those who did not attend were contacted by telephone to ensure adherence. All-cause death was the main outcome. The circumstances of the incidents were determined from the hospital records and/or interviews with the relatives. However, we were not able to discriminate the exact cause of death in some patients because of out-of-hospital death.
Objectives
The main objective of this study was to evaluate the association between the daily furosemide dose, prescribed during the dry state, and the long-term survival in stable outpatients with HFrEF on optimized therapy. The secondary objectives were as follows: (1) to evaluate the relationship between furosemide therapy and comorbidities, namely renal function, (2) to evaluate the relationship between furosemide dose and optimal medical therapy, and (3) to evaluate the primary objective using propensity score matching for a low dose (≤40 mg/d) and a high dose (>40 mg/d) of furosemide.
Statistical Analysis
Statistical analysis was performed using the statistical package IBM SPSS 22.0 for Windows. Continuous variables are expressed as mean ± standard deviation or median and interquartile range depending on whether the distribution was normal. Categorical variables are expressed as percentages. Comparative analysis between variables was performed using the required tests for each variable (dichotomous or continuous) and according to whether the distribution was normal (χ2 test, Student t test, Mann-Whitney U test). Significance was considered at P < .05.
The association between medical therapy and mortality was analyzed using Kaplan-Meier survival curves comparing logarithmic ranges (log-rank test) and the Cox regression model. Survival time was calculated from the baseline visit until the date of death or last follow-up. Survival was plotted, and univariable and multivariable (covariate-adjusted) Cox proportional hazards regression models were used to estimate the effects for each covariate. We performed simultaneous adjustment for potential confounders in multivariable models. Proportional hazards assumptions were verified. Significant covariates in the univariate analysis and clinically significant confounders included for adjustment in multivariable models were as follows: age, LVEF, atrial fibrillation, arterial hypertension, ischemic etiology, chronic obstructive pulmonary disease, and estimated glomerular function (<60 mg/mL/m2). As stated in the previous section, the daily furosemide dose was categorized according to 2 cutoff values (40 and 80 mg/d). Because of the small number of patients receiving >80 mg/d, for some calculations, we used only the cutoff of 40 mg. Patients were classified as receiving no furosemide, a low dose (1-40 mg/d), or a high dose (>40 mg/d). We used a propensity score–matched (PSM) cohort for each pair combination (none vs low, none vs high, and low vs high). The PSM was used to account for the probability (propensity) of receiving a certain furosemide dose at baseline. Patients were then matched. Because only a few patients did not receive furosemide, 2 cohorts of patients were constituted, the first being for the comparison of patients not receiving furosemide with those receiving the low or the high dose. We were able to match 1:1 86% of patients not receiving furosemide with those receiving either dose. The second cohort matched 1:1 patients receiving low-dose furosemide with those receiving the high dose—55% of those receiving the low dose and 56% of those receiving the high dose were matched. An absolute standardized difference of <10% was considered for balance. Cox proportional hazards regression analysis was conducted based upon the matched subgroups. Comparison between the low and high doses was adjusted for confounders, while comparison with no furosemide was not adjusted because of the small sample.
Results
Table 1 shows the patients’ demographics, clinical characteristics, and treatment. In brief, there were 90 (33.8%) deaths. One-third of the patients were in NYHA class III, 40.6% were ischemic, and 47.4% had renal dysfunction (GFR <60 mg/mL/m2). With regard to medical therapy, 94% received ACEi/ARBs, 83.8% received BBs, and 80.8% received both. One-third (34.6%) of the patients had a cardiac resynchronization therapy device implanted. Furosemide daily dose was 57.3 ± 21.4 mg. Only 22 (8.3%) patients were not treated with furosemide, almost half (47.6%) received a low dose (≤40 mg/d), 42.1% received 41 to 80 mg/d, and only 2.3% received >80 mg/d.
Demographics, Comorbidities, Outcome, and Therapy in All Population and According to the Furosemide Dose Level.

Abbreviations: ACEi, angiotensin-converting enzyme inhibitors; AHT, Arterial hypertention; ARB, angiotensin receptor blocker; BB, β-blocker; BMI, body mass index; COPD, chronic obstructive pulmonary disease; CRT, cardiac ressynchronization therapy; DM, Diabetes Mellitus; GFR, glomerular filtration rate; Hb, hemoglobin; HR, hazard ratio; Ht, hematocrit; LVEF, left ventricular ejection fraction; NS, not significant; NYHA, New York Heart Association; RDW, red blood cell distribution width; Spir, spironolactone; TD, target dose.
a P value for the comparison between furosemide subgroups—F test by analysis of variance (ANOVA) between the 3 doses subgroups in continuous variables and from χ2 test by cross tabulation in categorical variables.
b P for comparison between the 2 subgroups 1 to 40 mg/d versus >40 mg by ANOVA with Bonferroni correction and Fisher exact test for 2 × 2 cross tabulation.
Relationship Between Furosemide Dose, Demographics, Comorbidities, Laboratory Data, and Therapy
There was a strong association between furosemide dose and the patients’ clinical characteristics (Table 1). A greater furosemide dose (Table 1; none vs 1-40 vs >40 mg/d) led to (1) worse outcome (P < .001) and a greater proportion of patients with NYHA class III (P < .001) and atrial fibrillation (P < .001); (2) increasing values of creatinine (P < .001) and Nt-ProBNP (P < .001) and worse GFR (P < .001); (3) a progressive decrease in LVEF (P = .018); and (4) a lower proportion of patients receiving ACEi/ARBs, BBs, and combinations thereof.
Furosemide Dose and Outcome
The receiver operating characteristic (ROC) curve for all-cause death was associated with an increasing daily dose of furosemide (Supplemental Figure 1; area under the curve = 0.74; 95% confidence interval [CI]: 0.68-0.79; P < .001), and a dose of >40 mg/d was identified as the optimal cutoff value (sensitivity = 73.3, specificity = 70.5; Youden index: 0.4379) to identify patients with higher probability of death, meaning that a patient receiving high-dose furosemide (>40 mg/d) has a greater chance of dying (true positive), with fewer false positives.
A furosemide increase of 1 mg/d was significantly associated with survival (Table 2). The adjusted survival curve (Figure 1) was associated (log-rank, P < .001; hazard ratio [HR] = 1.96; 95% CI: 1.22-3.15; P < .001) with the furosemide dose (from 0 to >80 mg/d). The subgroup not receiving furosemide and the low-dose subgroup had almost superimposed adjusted survival curves (Figure 1). Therefore, we merged these subgroups (Table 2). Those receiving 41 to 80 mg/d (HR = 2.12; P = .005) or >80 mg/d (HR = 5.81; P = .005) had a greater risk of mortality than the combined subgroup receiving furosemide 0 to 40 mg/d. All of these results were adjusted for significant confounders (Table 2).
Adjusted Mortality According to the Furosemide Dose Levels, Taking Furosemide 0 to 40 mg/d Dose as Reference.a,b

Abbreviations: ACEi, angiotensin-converting enzyme inhibitors; ARB, angiotensin receptor blocker; BB, β-blocker; CI, confidence interval; COPD, chronic obstructive pulmonary disease; GFR, glomerular filtration rate; HR, hazard ratio; NYHA, New York Heart Association.
aStatistical model adjusted model for significant confounders—age, COPD, ischemic, hypertensive, NYHA, atrial fibrillation, GFR, and ACEi/ARB + BB therapy.
bOther independent predictors of survival, in the adjusted model, were age (HR: 1.02, 95% CI: 0.9-1.04; P = 0.083), COPD (HR: 0.33, 95% CI: 0.16-0.69; P = .002), ischemic (HR: 1.7, 95% CI: 1.0-3.0; P = .042), and ACEi/ARB + BB (HR: 0.55, 95% CI: 0.32-0.93; P = .026).
cDose levels: 0 to 40 mg/d, 41 to 80 mg/d, and >80 mg/d.

Adjusted survival curve according to furosemide dose level (from 0 to >80 mg/d).
Furosemide Dose and Outcome in a PSM Cohort
The clinical and laboratory data and the therapeutic profiles of the PSM cohorts are shown in Supplemental Table 1. In the PSM (Table 3) cohort, patients receiving furosemide >40 mg/d had a greater risk of mortality than those not receiving furosemide (HR = 3.9; P = .039) or those who were receiving a low dose (HR = 4.023; P = .001). There was no significant difference in the hazard risk between patients receiving a low dose and those not receiving furosemide.
Patient Survival According to Furosemide Daily Dose Groups in the Propensity Score–Matched Cohort.a

Abbreviations: ACEi, angiotensin-converting enzyme inhibitors; ARB, angiotensin receptor blocker; BB, β-blocker; CI, confidence interval; COPD, chronic obstructive pulmonary disease; HR, hazard ratio.
aOther independent predictors of survival, in the adjusted model, were COPD (HRcox: 0.11; 95% CI: 0.02-0.55; P = .007) and NYHA (HRcox: 7.7; 95% CI: 2.8-20.8; P < .001)
bNonadjusted model because of the small sample size.
cAdjusted model for age, COPD, ischemic, hypertensive, NYHA, atrial fibrillation, GFR, and ACEi/ARB + BB therapy, log Nt-ProBNP.
Effect of the Clinical and Therapeutic Profiles on the Negative Effect of Furosemide Dose on Survival
For each dose (<50% of the TD and ≥50% of the TD) of ACEi/ARBs, BBs, or their combination, the 3-year mortality rate (Supplemental Figure 2) significantly increased with the furosemide dose (from 0 to >40 mg/d). The same trend was observed for patients in NYHA class II (P = .009) but not for those in NYHA class III.
Patients in NYHA class II and those receiving a high dose (≥50% of the TD) of ACEi/ARBs, BBs, or both had a greater risk of mortality with an increasing dose of furosemide (Figure 2). However, the furosemide dose had a neutral effect on the prognosis of patients in NYHA class III or those receiving a low dose of ACEi/ARBs, BBs, or their combination. These models were adjusted for significant confounders.
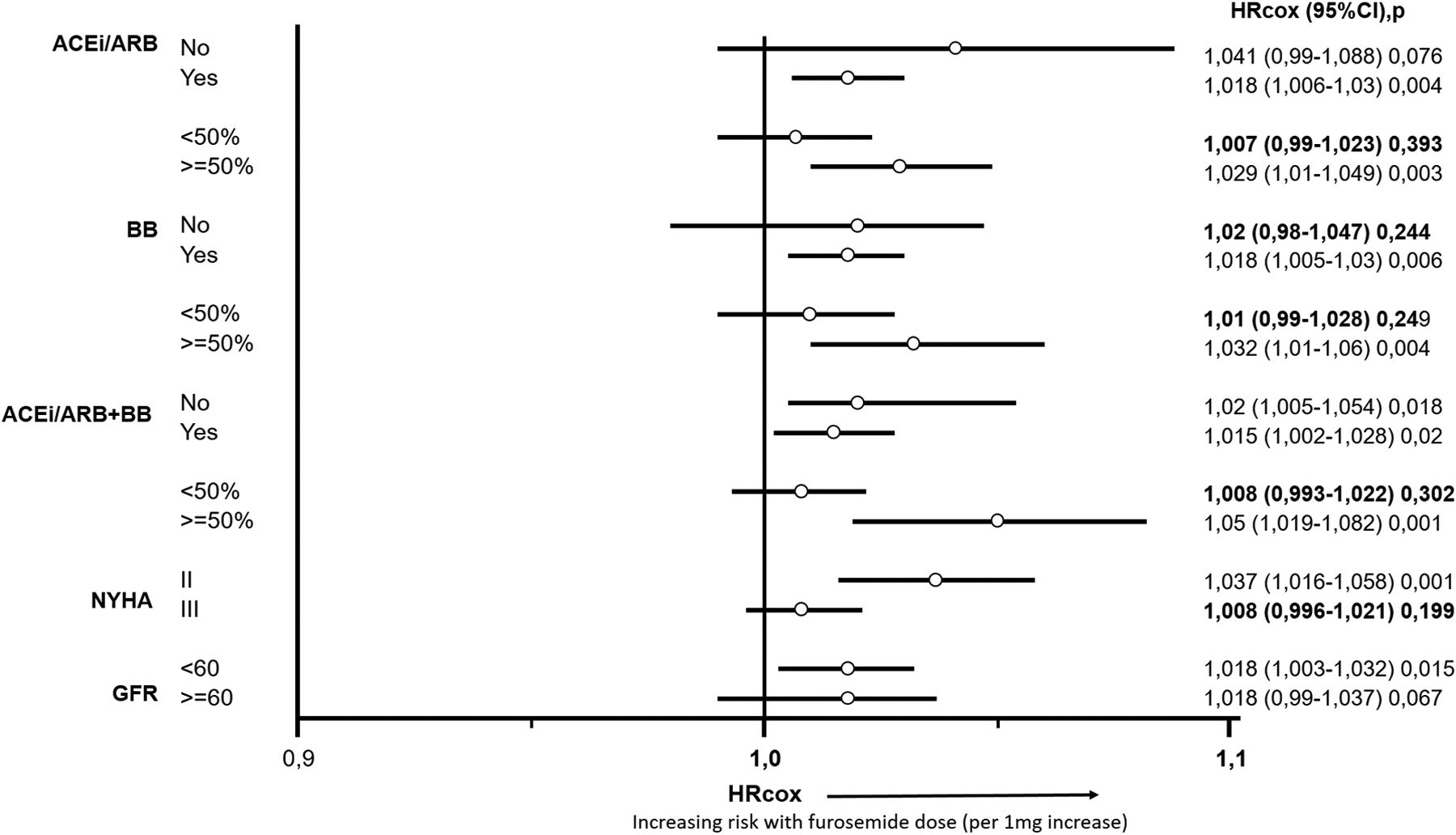
Adjusted hazard risk of death with the daily furosemide dose (per 1 mg/d) according to optimal medical therapy dose level, New York Heart Association (NYHA) class, and renal function. Highlighted in bold are those subgroups with a neutral effect of furosemide dose on survival.
Discussion
In the present study, the daily furosemide dose prescribed during a 6-month period of the dry state in optimally stable outpatients with HFrEF was associated with 3-year survival. This observation was valid in the adjusted model for significant confounders and a PSM population. Moreover, NYHA class and the optimal medical therapy dose appeared to interact with this association.
The relationship between loop diuretics and outcome has previously been described in HF. 6 -8 However, there is disagreement on the optimal cutoff values and the physiopathological explanation for this association. Some authors considered 50 mg as a cutoff, 8 whereas others used 80 mg. 7 We chose the arbitrary values of 40/80 mg, similar to other reports. 6 This choice was based on the dose of 1 furosemide tablet (40 mg) and the HF clinic protocol for furosemide scaling doses in ambulatory patients with congestion. This value also corresponded to the best cutoff value of the ROC curve for death.
Several hypotheses have been proposed to explain the association between loop diuretics and outcome. Diuretics stimulate the renin–angiotensin–aldosterone system and the sympathetic nervous system, 13,14 which are associated with a worse prognosis. 12 These effects should be counteracted by the use of ACEi and BBs. 15,16 Therefore, the prescription pattern and titration level of these drugs need to be evaluated, as well as the effect of loop diuretics on survival. With regard to renal dysfunction, high doses of loop diuretics are associated with increased incidence of renal dysfunction in hospitalized 17 and ambulatory patients. 6 Hypotension and fatal arrhythmia by electrolyte imbalance 18 represent another potential mechanism. In contrast, some authors argue that the dose of loop diuretics is only a consequence of the clinical status, which in turn is the true prognostic factor. In the absence of randomized clinical trials, the selection bias of observational studies may explain in part the associations of furosemide, worse clinical status, and outcome. Despite the uncertain effect on mortality, diuretics are necessary in all stages of chronic HF to maintain symptomatic control and the patient’s clinical stability. However, overzealous congestion drainage may lead to dehydration, with renal dysfunction and activation of the renin–angiotensin–aldosterone system. 5 The change from an overvolemic to a volume-depletion state, and the corresponding weight loss, could be influenced by the body mass index paradox. Obese and overweight patients with HFrEF have better survival than their counterparts of normal weight. 19 In some patients with HFrEF, in order to maintain a good balance between the need to achieve a dry state, reduce congestion and dyspnea, and avoid dehydration, it is necessary to retain slight peripheral edema in patients while keeping them being free from symptoms during usual effort. In the present study, we evaluated the impact on survival of several furosemide doses in patients, including those not receiving furosemide. In all populations and in the PSM cohort, the subgroup not receiving furosemide and the low-dose subgroup had almost identical adjusted survival curves. On the other hand, patients receiving high-dose furosemide had a greater risk of death than those receiving the low dose or no furosemide. Most patients with chronic HF stabilized with furosemide at high doses remained stable on lower doses. 5
In our cohort, a simple explanation for the association of furosemide with outcome is that patients who were ill with a worse outcome had to receive higher doses of furosemide. This possibility is supported in part by the baseline clinical and laboratory characteristics of the population. Patients receiving higher doses of furosemide were older and had poor LVEF, NYHA class, and renal function. They also received and tolerated lower doses of medical therapy. However, in the adjusted survival model that included significant clinical and laboratory variables and a PSM cohort, higher furosemide doses remained associated with a worse prognosis. Moreover, the association of furosemide with survival was not universal in all subgroups of patients. Low-risk groups were especially sensitive to the adverse effect of furosemide on survival. Patients in NYHA class II or those receiving higher doses of medical therapy had a greater risk of mortality with an increasing daily furosemide dose. However, no effect of furosemide on survival was apparent in patients in NYHA class III or those receiving suboptimal medical therapy. New York Heart Association class III includes patients at lower risk for an event. This raises the question of why these patients were receiving furosemide during the dry state if they were clinically stable. Chronic furosemide administration affects renal function, 6 but the adverse effect of furosemide on survival affects patients with or without renal dysfunction.
The daily furosemide dose taken during a long period of the dry state by stable, optimally medicated outpatients with HFrEF appears to have a deleterious effect on long-term survival, especially in a low-risk subgroup of patients. This finding reemphasizes the need to decrease or use furosemide with caution in stable outpatients who are on optimal medical therapy. Furosemide should only be prescribed to control symptoms, while more emphasis should be placed on prescribing and uptitrating neurohormonal blockers, which have a favorable effect on survival. 1
Clinical Implications
There is no evidence-based recommendation for chronic administration of loop diuretics in stable outpatients with HFrEF. Therefore, administration of loop diuretics depends on caregivers’ knowledge, experience, and possibly preference. The present study also suggests that in stable outpatients with HFrEF on optimal medical therapy, furosemide prescription and dose are associated with a worse prognosis in all populations and especially in subgroups of patients at lower risk. Furosemide overuse should be avoided, especially in patients in NYHA class II and those receiving higher doses of ACEi/ARBs plus BBs. Therefore, until the physiopathological mechanism of chronic furosemide administration in stable outpatients is fully understood, furosemide administration should be conducted with caution and at doses as low as possible and only to avoid symptoms of congestion. The threshold dose is ≤40 mg/d (1 tablet or less per day).
Study Limitations
We acknowledge several limitations of our study. The sample size was small, and the furosemide dose was only determined at baseline and not over time. This was an observational study, and thus, the role of treatment on prognosis is not directly measurable, as occurs in randomized clinical trials. Besides the low sample size (ie, in the subgroup not on furosemide), we attempted to create PSM subgroups according to furosemide doses of 0, 1 to 40 mg/d, and >40 mg/d. Therefore, by matching patients and balanced clinical and laboratory characteristics, we were able to estimate the effect of furosemide on survival but in a cohort with a small number of patients.
Conclusion
In our cohort of stable outpatients with HFrEF on optimal therapy, the use of furosemide during the dry state was associated with an increased risk of mortality, especially in specific low-risk subgroups of patients (NYHA class II and those on higher doses of medical therapy for HF).
Footnotes
Authors’ Note
Institution where the work reported was done: Heart Failure Unit, Cardiology Department, Lisbon North Hospital Centre, Pulido Valente Hospital, Lisbon, Portugal.
Author Contributions
L. Sargento contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. A. Vicente Simoes contributed to design, acquisition, and analysis and drafted the manuscript. S. Longo contributed to conception and interpretation. N. Lousada contributed to conception, analysis, and interpretation. R. Palma Reis contributed to conception and design and contributed to analysis and interpretation. All authors critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
