Abstract
Objectives:
We aimed to characterize different cellular effects of angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin 1 (AT1) receptor blockers (ARBs) as mono- or combination therapy in cardiac pressure overload.
Methods and Results:
C57B1/6 mice received either the ACEI ramipril (2.5 mg/kg body weight), the ARB telmisartan (20 mg/kg body weight), or the combination. In all groups, pressure overload was induced by transverse aortic constriction (TAC). Cardiac hypertrophy (heart weight/tibia length) induced by TAC was reduced in all 3 treatment groups, with the most pronounced effect in the telmisartan group. The cardiomyocyte short-axis diameter and cardiac fibrosis were increased by TAC and similarly reduced by ACEI, ARB, and the combination therapy. The TAC-induced increase in the number of proliferating Ki67pos cardiomyocytes and noncardiomyocytes was reduced more potently by ACEI than by ARB. Four days of drug treatment induced a significant increase in Scalpos/VEGFR1pos endothelial progenitor cells (EPCs) in all animals in the treated SHAM groups. After 1 day of aortic constriction, only ramipril increased EPC numbers; after 5 weeks, telmisartan monotherapy did not change the EPC levels compared to vehicle or the combination therapy but raised it compared to ramipril. Neither TAC nor one of the therapies changed the number of cardiac capillaries per cardiomyocytes.
Conclusions:
ACE inhibition and AT1 receptor blockade have beneficial effects in remodeling processes during cardiac pressure overload. There are small differences between the 2 therapeutical approaches, but the combination therapy has no additional benefit.
Keywords
Introduction
The activity of the renin–angiotensin system (RAS) is involved in the pathogenesis of blood pressure, atherosclerosis, and myocardial hypertrophy. Two of the major drug classes used to reduce RAS activity are angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II receptor blockers (ARBs). The ACEI blocks the conversion of angiotensin I to angiotensin II and inhibits the breakdown of bradykinin. A disadvantage of ACEI is the “ACE escape” phenomenon, which is related to the tissue production of angiotensin II by enzymes other than ACE. 1 The human α-chymase is able to convert angiotensin I into angiotensin II and is assumed to be responsible for more than 80% of tissue angiotensin II formation in the human heart. 2 The ACE is also identical to kininase II, the enzyme that metabolizes bradykinin to inactive fragments. 3 Therefore, ACE inhibition leads to attenuation of bradykinin metabolism and to an increase in the local concentration of this potent vasodilator. The ARBs block RAS activity at the AT1 receptor, thereby also targeting angiotensin II produced by chymases. Since both classes differ, one could speculate that a combination of ACEI and ARB would have complementary effects.
The well-documented antihypertensive and antihypertrophic properties of ACEI and ARBs are associated with beneficial cardiovascular effects. 4 In the heart, increased afterload leads to cardiac hypertrophy and fibrosis and to upregulation of EPCs from the bone marrow which may contribute to myocardial angiogenesis. 5,6 We showed earlier that ACE inhibition using ramipril in a dose of 5 mg/kg body weight was ameliorating these effects in cardiac overload in mice. 7 In patients after acute coronary syndrome, treatment with an ARB led to similar numbers of EPC in the peripheral blood as treatment with an ACEI. 8
We therefore hypothesized that ACE inhibition and AT1 receptor blockade may have similar effects in increased cardiac afterload and wanted to test whether the combination of both drugs would be beneficial. We used doses of ramipril 2.5 mg/kg body weight and telmisartan 20 mg/kg body weight to mimic the 1:8 ratio used in large studies such as ONTARGET and TRANSCEND. 9,10
Methods
Animals, Drug Treatment, and Transverse Aortic Constriction
The study was approved by the animal ethics committee of the Universität des Saarlandes and conforms to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (National Institutes of Health Pub. No. 85-23, revised 1996). Ten-week-old male C57/ Bl6 (Charles River Laboratories, Sulzfeld, Germany) were housed under standard conditions. Animals received at 10 weeks of age either ramipril at a dose of 2.5 mg/kg body weight in the form of pulverized tablets per kg body weight (Aventis, Frankfurt, Germany) introduced into the diet or telmisartan at a dose of 20 mg/kg body weight in the drinking water (Boehringer-Ingelheim, Ingelheim, Germany) or both drugs or a matched placebo diet (sniff, Germany) and normal drinking water. Three days after starting drug treatment or placebo diet, the animals were anesthetized with ketamine (100 mg/kg body weight, intraperitoneally [IP]) and xylazine (10 mg/kg, IP) for transverse aortic constriction (TAC). After orotracheal intubation using a 20-gauge catheter, the tube was connected to a volume-cycled rodent ventilator (Harvard Apparatus, Holliston, Massachusetts) on supplemental oxygen with a tidal volume of 0.2 mL and respiratory rate of 110 per min. The chest cavity was entered in the second intercostal space at the left upper sternal border through a small incision and aortic constriction was performed by tying a 7-0 nylon suture ligature against a 27-gauge needle to yield a narrowing 360 µm in diameter and a TAC of 65% to 70%. Control mice underwent a sham operation. After 5 weeks, animals were anesthetized with ketamine (100 mg/kg body weight, IP) and xylazine (10 mg/kg, IP) and a 1.4F pressure-transducing catheter (Mikro Tip Catheter, Millar Instruments, Houston, Texas) was used for left ventricular (LV) pressure measurements. After the LV pressure measurements, hearts were rapidly excised. Hearts were partly snap-frozen and stored at −80°C, partly fixed and embedded in paraffin. Peripheral blood was sampled for further preparation.
Peripheral Blood Pressure
Peripheral blood pressure was obtained on the tail artery of mice 4 weeks after sham surgery or aortic constriction on 5 consecutive days using at least 20 repeating measurements per day (BP 2000 Series II Blood Pressure Analysis System, Visitech Systems, North Carolina).
Fluorescence-Activated Cell Sorter Analysis
Blood and bone marrow were analyzed as described. 5 –7,11 The viable lymphocyte population was analyzed for stem cell antigen 1 (Sca-1)-FITC (E13–161.7, Pharmingen, Germany) and vascular endothelial growth factor receptor 2 (VEGFR-2; Flk-1; Avas12α1, Pharmingen) conjugated with the corresponding phycoerythrin-labeled secondary antibody (Sigma, Germany). Isotype-identical antibodies served as controls (Becton Dickinson, Germany).
Immunofluorescence Analysis
For pretreatment slices with 3-µm-thick sections were placed in Coplin jars with 0.05% citraconic anhydride solution, pH 7.4 for 1 hour at +98°C and then incubated overnight at 4°C with the first antibody, followed by the appropriate secondary antibody at 37°C for 1 hour. Immunofluorescence studies were performed by applying monoclonal antibodies against α-sarcomeric actin (clone5c5; Sigma-Aldrich, Schnelldorf, Germany) to detect cardiomyocytes. Sections were co-stained for capillary detection (Podocalyxin; R&D Systems, Wiesbaden, Germany). Co-immunostaining for the antigen Ki67 (Novocastra Laboratories LTD, Newcastle upon Tyne, UK) was used to detect cycling cells. A green fluorescent protein (GFP)-specific antibody (Abcam, Cambridge, UK) was used to enhance GFP fluorescence of tie2-positive bone marrow-derived cells in bone marrow-transplanted animals. The fluorescein isothiocyanate-, tetramethyl rhodamine isothiocyanate-, biotin-, and peroxidase- (Dianova, Hamburg, Germany) conjugated anti-mouse immunoglobulin (Ig)M, anti-mouse IgG, anti-rabbit IgG, and anti-goat IgG were used as secondary antibodies and for amplification if necessary. Sections were counterstained with DAPI (Calbiochem, Darmstadt, Germany) and washed and mounted with fluorescent mounting medium (Vectashield; Vector Laboratories, Burlingame, California) for fluorescence microscopic analysis. All sections were evaluated using a Nikon E600 epifluorescence microscope (Nikon, Düsseldorf, Germany) with appropriate filters.
Tissue Morphometry
For morphometric analyses, LV tissue sections (3 μm) were examined. Cardiomyocyte short-axis diameter, degree of cardiac fibrosis, percentages of cycling cardiomyocytes, numbers of cycling cardiomyocytes, and ratio of capillaries to cardiomyocytes were evaluated. The procedures used for morphometric analyses were provided in detail elsewhere. 6
Statistical Analysis
Results are presented as mean ± standard error of the mean. One-way analysis of variance with a Fisher least significant difference post hoc test or Kruskal-Wallis test with a Mann-Whitney post hoc test were used where applicable. Values of P < .05 were considered significant.
Results
Telmisartan Prevents Cardiac Hypertrophy to a Greater Extent Than Ramipril
Transverse aortic constriction (360 µm, 35 days, n = 19; sham group [SHAM] n = 20) increased the ratio of heart weight to tibia length (mg/mm) to 13.4 ± 1 versus 7.9 ± 0.4 in sham-operated mice (P < .001). All therapies decreased this parameter in the mice receiving TAC compared to the vehicle group (R-TAC 11 ± 0.5, T-TAC 8.8 ± 0.3, RT-TAC 9.2 ± 1.3, all P < .05 vs TAC), while telmisartan monotherapy had a more pronounced effect than the ramipril treatment (P < .05; Figure 1A).

Effect of ramipril, telmisartan, and their combination on left ventricular (LV) and peripheral blood pressure and on cardiac hypertrophy. Effect of ramipril (2.5 mg/kg body weight/d PO), telmisartan (20 mg/kg body weight/d PO), the combination of ramipril and telmisartan and transverse aortic constriction (TAC, 360 µm, 35 days) on LV systolic pressure (A), on tail artery systolic pressure (B), and on the ratio of heart weight to tibia length (C). Significant (P < .05): * versus SHAM; ** versus R-SHAM, *** versus T-SHAM, **** versus RT-SHAM, # versus TAC, $ significant versus R-TAC. SHAM indicates mice receiving sham surgery; TAC, mice receiving transverse aortic constriction; R, mice receiving treatment with ramipril; T, mice receiving treatment with telmisartan; RT, mice receiving combination therapy of ramipril and teslmisartan; PO, postoperatively.
Left ventricular systolic pressure increased to 119 ± 7 mm Hg in TAC versus 79 ± 4 mm Hg in sham-operated mice (P < .0001). This TAC-induced increase in left ventricular systolic pressure was significantly reduced by the combination therapy of telmisartan 20 mg and ramipril 2.5 mg/kg body weight (RT-SHAM 45 ± 2 mm Hg, n = 4, RT-TAC 86 ± 6 mm Hg, n = 6; P < .05 vs TAC) but not by one of the monotherapies (R-SHAM 67 ± 12 mm Hg, n = 4, R-TAC 115 ± 6 mm Hg, n = 5, T-SHAM 50 ± 3 mm Hg, n = 4, R-TAC 103 ± 7 mm Hg, n = 7; Figure 1B). Systolic blood pressure of the tail artery decreased significantly in mice after aortic ligation to 96 ± 2 mm Hg compared to 113 ± 3 mm Hg in sham-operated mice (P < .001). Ramipril alone did not significantly influence this parameter in both groups (R-SHAM 116 ± 6 mm Hg, R-TAC 106 ± 4 mm Hg), while telmisartan and the combination therapy reduced the blood pressure significantly in the respective SHAM and TAC groups (T-SHAM 88 ± 6 mm Hg, T-TAC 79 ± 4 mm Hg, RT-SHAM 77 ± 5 mm Hg, RT-TAC 80 ± 3 mm Hg, all P > .05 versus respective SHAM and TAC-group; T-TAC and R-TAC P < .05 vs RT-TAC; Figure 1C).
Ramipril and Telmisartan and Their Combination Do Not Differ Concerning Prevention of Cardiomyocyte Hypertrophy and Cardiac Fibrosis
Transverse aortic constriction for 35 days increased cardiomyocyte short-axis diameter (TAC 15.5 ± 0.5 µm) compared to sham-operated animals (SHAM 10.6 ± 0.3 µm, P < .000001 vs TAC). Both monotherapies and the combination therapy attenuated this effect compared to the respective vehicle SHAM (R-SHAM 9.8 ± 0.2, T-SHAM 9.5 ± 0.5, RT-SHAM 9 ± 0.3, all P < .05 vs SHAM) and vehicle TAC group (R-TAC 10.8 ± 0.1, T-TAC 10.7 ± 0.3, RT-TAC 10.3 ± 0.3, all P < .05 vs TAC; Figure 2A).

Effect of ramipril, telmisartan, and their combination on cardiomyocyte size and cardiac fibrosis. Effect of ramipril, telmisartan, and the combination therapy on cardiomyocyte short-axis diameter (A) and on cardiac fibrosis evaluated by collagen content (expressed as percentage of myocardial content) (B). Significant (P < .05): * versus SHAM, *** versus T-SHAM, **** versus RT-SHAM, # versus TAC. (C) to (F) show sirius red staining of SHAM (C), TAC (D), R-TAC (E), T-TAC (F), and RT-TAC (G) mouse heart sections. SHAM indicates mice receiving sham-surgery; TAC, mice receiving transverse aortic constriction; R, mice receiving treatment with ramipril; T, mice receiving treatment with telmisartan; RT, mice receiving combination therapy of ramipril and teslmisartan.
While TAC markedly increased cardiac fibrosis quantified morphometrically as fractional area of collagen content in percentage of myocardial content using picrosirius red staining (SHAM 0.7 ± 0.2%, TAC 2.6 ± 0.5%; P < .05), all 3 therapies abolished this effect significantly (R-TAC 0.5 ± 0.1%, T-TAC 0.5 ± 0.2%, RT-TAC 0.8 ± 0.4%, all P < .05 vs TAC; Figure 2B-F).
Telmisartan Shows a More Pronounced Effect on the Reduction of Cycling Cardiomyocytes and Noncardiomyocytes Than Ramipril
Ki67, a nuclear protein which is expressed during all stages of the cell cycle excluding G0, was used to measure the number of cardiomyocytes and noncardiomyocytes undergoing cell division (n ≥ 4 in each group). The Ki67 expressing noncardiomyocytes increased significantly from 3 ± 0.6 per mm2 in the sham group to 17.4 ± 2.6 per mm2 after aortic constriction (P < .001, Figure 3A and C). In the TAC groups treated with the ACEI, the ARB or the combination, this effect was significantly attenuated ([Ki67pos noncardiomyocytes per mm2]; R-TAC 4 ± 1.6, T-TAC 0.5 ± 0.2, RT-TAC 3.7 ± 1.5, all P < .05 vs TAC), the ARB monotherapy inducing a stronger increase than the 2 other therapies (P < .05 vs both).

Effect of ramipril, telmisartan, and their combination on cycling cardiac cells. Ramipril, telmisartan, and their combination differentially decrease cycling noncardiomyocytes (A) and cardiomyocytes (B). Significant (P < .05): * versus SHAM, **** versus RT-SHAM, # versus TAC, $ versus R-TAC, § versus RT-TAC. (C) and (D) show examples of cardiac sections from mice after transverse aortic constriction with co-immunostaining for the myocyte marker α-sarcomeric actin (green) and Ki67 (red). Proliferating noncardiomyocyte nuclei (C) and cardiomyocyte nuclei (D) are marked by arrows. Nuclei are stained blue by DAPI. Bars = 10 µm. SHAM indicates mice receiving sham surgery; TAC, mice receiving transverse aortic constriction; R, mice receiving treatment with ramipril; T, mice receiving treatment with telmisartan; RT, mice receiving combination therapy of ramipril and teslmisartan; DAPI, 4′,6-diamidino-2-phenylindole.
While there were no cycling cardiomyocytes in sham-operated mice, 0.1% ± 0.02% of cardiomyocytes per mm2 expressed Ki67 in mice after TAC (P < .001). All drug-receiving sham groups also showed no Ki67-positive cardiomyocytes. Compared to the vehicle-treated TAC mice, all drug-receiving animals showed a lower percentage of cycling cardiomyocytes (R-TAC 0.02% ± 0.007%, T-TAC none, RT-TAC 0.005% ± 0.002%, all P < .05 vs TAC), but the telmisartan treatment induced a more pronounced effect—since abolishing all cycling cardiomycoytes—than ramipril (P < .05) or the combination therapy (P < .05; Figure 3B and D).
The EPC Upregulation by Ramipril but not by Telmisartan Occurs Early
Aortic constriction elevated EPC levels after 1 day compared to base levels in sham to 134% ± 10% in peripheral blood (P < .05; Figure 4A). The ACE inhibition and the combination therapy—but not AT1-receptor-antagonism alone—for 3 days prior to surgery increased EPC numbers in sham (R-SHAM 146% ± 12%, T-SHAM 126% ± 11%, RT-SHAM 181% ± 21%; R-SHAM and RT-SHAM P < .05 vs SHAM) and TAC mice (R-TAC 329% ± 61%, T-TAC 124% ± 12%, RT-TAC 182% ± 11%, R-TAC and RT-TAC P < .001 vs TAC; R-TAC P < .001 vs T-TAC, RT-TAC P < .05 vs T-TAC). After 5 weeks, TAC led to upregulation of EPC numbers to 137% ± 14% (P < .05) of baseline in the peripheral blood (Figure 4B). At this time point, neither EPC from sham animals treated with ramipril or telmisartan nor the combination therapy differed from baseline (R-SHAM 108% ± 8%, T-SHAM 121% ± 14%, RT-SHAM 91% ± 19%). However, after TAC, telmisartan treatment did not change the EPC level in the animals compared to vehicle or the combination therapy—but raised it compared to ramipril monotherapy (R-TAC 102% ± 5%, T-TAC 130% ± 10%, RT-TAC 101% ± 23%, T-TAC P < .05 vs R-TAC).
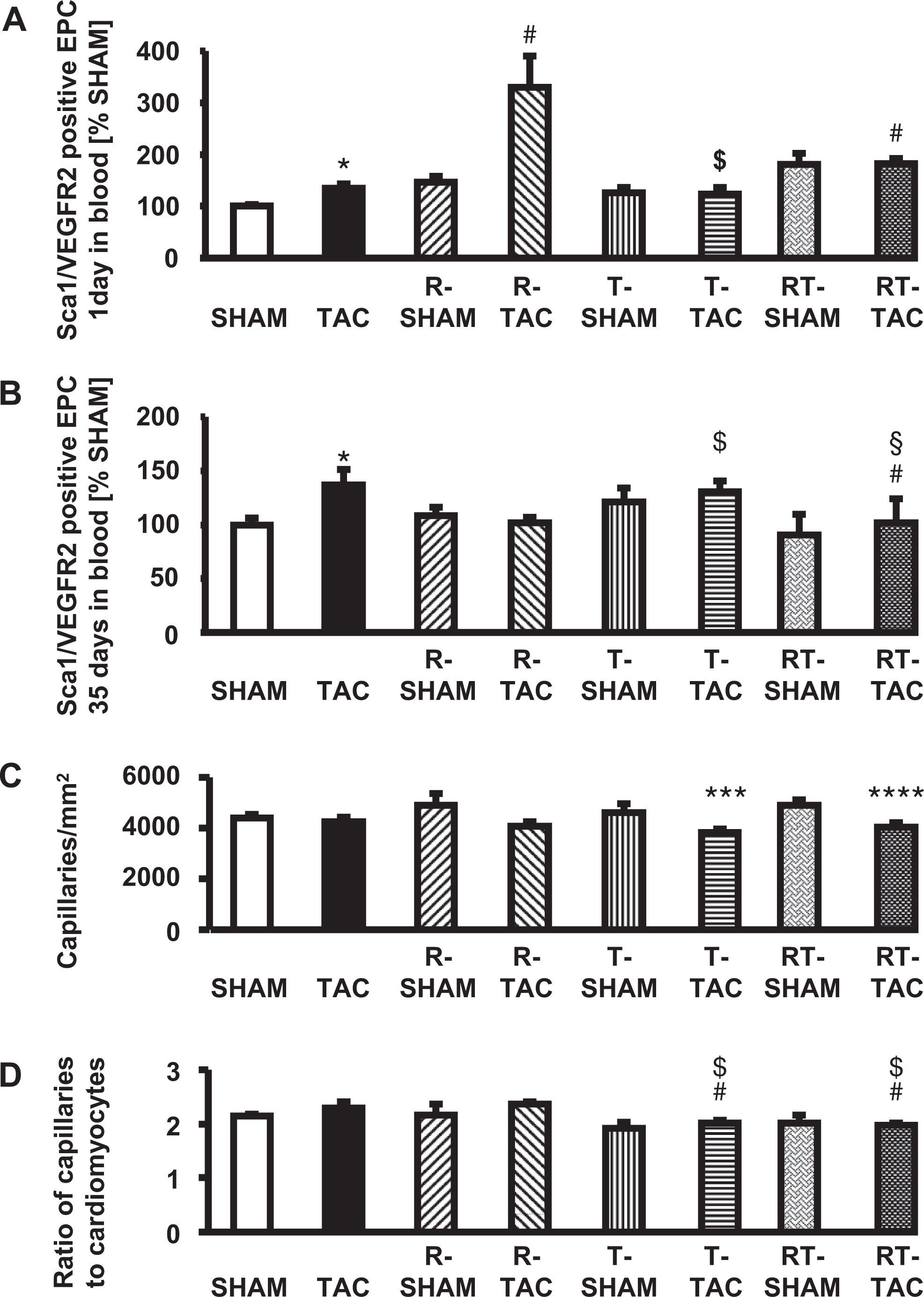
Effect of ramipril, telmisartan, and their combination on EPC numbers and cardiac capillarization. Influence of ramipril, telmisartan, and the combination of both drugs on EPC numbers in peripheral blood after 1 day (A) and after 35 days (B). The EPCs were measured by double labeling for Sca-1 and VEGF receptor 2 by fluorescence activated cell sorting analysis. Numbers are expressed as percentage of the EPC number measured in the relevant sham group. The effects of ramipril, telmisartan, and their combination on capillary density are expressed as number of podocalyxin-stained capillaries per mm2 (C) and as ratio of capillaries per cardiomyocytes (D). Significant (P < .05): * versus SHAM, *** versus T-SHAM, **** versus RT-SHAM, # versus TAC, $ versus R-TAC, § versus RT-TAC. SHAM indicates mice receiving sham surgery; TAC, mice receiving transverse aortic constriction; R, mice receiving treatment with ramipril; T, mice receiving treatment with telmisartan; RT, mice receiving combination therapy of ramipril and teslmisartan; EPC, endothelial progenitor cell; VEGF, vascular endothelial growth factor receptor 2.
Ramipril, Telmisartan, and Combination Therapy Do Not Increase Cardiac Capillarization
Cardiac capillaries were detected by immunostaining for podocalyxin. The number of capillaries per squared millimeter did neither change after 5 weeks of pressure overload nor by telmisartan, ramipril, or combination therapy (S 4404 ± 120, TAC 4250 ± 187, R-SHAM 4881 ± 493, T-SHAM 4622 ± 336, RT-SHAM 4877 ± 222). After TAC, the capillary number was not changed in mice treated with the ACEI but was even decreased in mice receiving the AT1-receptor antagonist or the combination treatment compared with the corresponding SHAM groups (R-TAC 4076 ± 174, T-TAC 3824 ± 155, RT-TAC 4032 ± 185; T-TAC P < .05 vs T-SHAM; RT-TAC P < .05 vs RT-SHAM; Figure 4C). Regarding the ratio of capillaries to cardiomyocytes, there was only a significant decrease between telmisartan- and combination therapy-treated TAC groups compared to the TAC group receiving vehicle (S 2.1 ± 0.1, TAC 2.3 ± 0.1, R-SHAM 2.2 ± 0.2, T-SHAM 1.9 ± 0.1, RT-SHAM 2.0 ± 0.1, R-TAC 2.2 ± 0.1, T-TAC 2 ± 0.1, RT-TAC 2 ± 0.1; R-TAC & T-TAC P < .05 vs TAC; Figure 4D).
Discussion
Mechanical pressure overload leads to cardiomyocyte hypertrophy as an adaptive response to the increase in wall stress induced by the increased external load. We aimed to know whether ACE inhibition and AT1 receptor blockade have similar effects in increased cardiac afterload and wanted to test whether the combination of both drugs would be beneficial.
We showed before that ACE inhibition reduces cardiac hypertrophy in transverse aortic constriction. 7 In this study, telmisartan and the combination therapy showed a similar effect. Telmisartan in renovascular hypertensive rats also led to a reduced cardiomyocyte cross-sectional area which was dose dependent. 12 This was believed to be caused by the reduction in the blood pressure which was dose dependent. In our study, telmisartan and the combination therapy reduced blood pressure significantly after TAC, which was not the case in the group receiving ramipril monotherapy as seen before using an even higher dose of ramipril. 7 Therefore, the reduction in cardiomyocyte hypertrophy by treatment with ramipril seems to be independent of blood pressure. On the other side, telmisartan monotherapy and the combination therapy reduced cardiac hypertrophy, measured as heart weight per tibia length, stronger than the ramipril treatment which could be related to the differences in the blood pressure. So there may be additional effects of ACEI causing reduction in cardiac and cardiomyocyte hypertrophy apart form peripheral blood pressure.
Telmisartan as well as ramipril and the combination treatment abolished the extent of fibrosis induced by pressure overload to a similar extent. Therefore, this effect could be independent of blood pressure as described recently in hypertensive rats given excess of salt. 13 On the other side, after TAC, LV systolic pressure was only reduced by the combination therapy. Since this reduction was followed neither by further reduction in cardiomyocyte size nor by a decrease in cardiac fibrosis, neither the greater inhibition of angiotensin II by the combination therapy nor the decrease in LV systolic pressure could be beneficial or both mechanisms abrogate each other.
The ACEI treatment was shown to decrease the induction of collagens I and III, laminin, and fibronectin as well as the ratio of the gelatinolytic matrix metalloproteinase 2 to the tissue inhibitor 14,15 of matrixmetalloproteinases 4. In transforming growth factor-β1 transgenic mice receiving telmisartan, we also observed a reduction in interstitial fibrosis due to a normalized ratio of matrixmetalloproteinases and their inhibitors. 16
We observed a greater extent of early EPC upregulation by ramipril than by telmisartan. We have not examined possible underlying mechanisms in this study but one could speculate that this could be due to a mobilization at different time points since after 5 weeks, no effect of ramipril or any other drugs on animals with pressure overload was observed, but telmisartan at this time point led to significant higher numbers of EPC than ramipril in TAC mice. Differences in a study performed in patients after acute coronary syndrome showing similar increases in EPC by ARB and ACEI 8 might be explained by the different setting of acute ischemia versus the pressure overload model used in our study. Heart size and cardiac function are angiogenesis dependent and disruption of coordinated tissue growth and angiogenesis in the heart contributes to the progression from adaptive cardiac hypertrophy to heart failure in myocardial hypertrophy. 17 In animals with pressure overload induced by TAC receiving 5 mg ramipril per kg body weight, we observed an amelioration of the capillary–cardiomyocyte ratio. 7 But in this study, neither ramipril at the lower dose of 2.5 mg nor telmisartan or the combination therapy were significantly influencing the cardiac capillary density in pressure overload compared to vehicle treatment. The difference in capillary density in contrast to our previous study using the higher dose of ramipril might be dose related. On the other side, the 2 drugs and their combination decreased cardiomyocte size significantly in aortic-ligated mice leading to a reduced diffusion distance which is important for an adequate supply of oxygen and nutrient substrates. 18 Therefore, the capillary–cardiomyocyte mismatch was ameliorated in the mice receiving treatment compared to the vehicle-fed TAC group even if the number of capillaries did not decrease. The difference in capillary density in contrast to our previous study using the higher dose of ramipril properties might be dose related. In the literature, both proangiogenic 19 –21 and antiangiogenic 20,22 –24 properties of ACEIs and AT1-receptor blockers have been described. The possible mechanisms for this phenomenon remain unresolved since ACE inhibition and AT1-receptor blockade also hamper neovascularization in other tissues. 20
There was no additional effect of the combination therapy on top of the most efficient of the 2 drug classes. This was true for all the parameters tested in this study apart from the LV pressure. A magnetic resonance tomography study in a subgroup of high-risk patients with cardiovascular disease in the ONTARGET study revealed similar effects of telmisartan and ramipril on LV mass and volume, and the combination therapy was not more effective in high-risk patients with cardiovascular disease. 25
In conclusion, ACE inhibition and AT1-receptor blockade in remodeling processes during cardiac pressure overload showed similar beneficial effects concerning cardiac hypertrophy, fibrosis, cell cycling, and capillarization. Differences between the 2 therapeutic approaches were small, but the combination therapy had no additional benefit.
Footnotes
Acknowledgments
We thank E. Becker, S. Jäger, and I. Hartmann for their excellent technical assistance.
Authors' Note
The authors state that the animal protocol was approved by an Institutional Animal Care and Use Committee.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors hereby disclose all other current and foreseeable financial and personal relationships that might inappropriately influence their actions and create a conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Boehringer/Ingelheim (PM, MB), the Hans & Gertie Fischer-Stiftung, Essen (PM) and from the Deutsche Forschungsgemeinschaft (KFO 196, MB, UL).
