Abstract
Objective:
Synthetic class A amphipathic helical peptide analogs of apolipoprotein-AI (apoAI; with varied phenylalanine residues) are emerging therapeutic approaches under investigation for atherosclerosis. Utilizing retroinverso sequencing, we designed reverse-D4F (Rev-D4F) peptide with 18
Materials/Methods:
ApoE-null mice were fed a chow diet and administered water (control), Rev-D4F, or L4F mimetic peptides (0.4 mg/mL, equivalent to 1.6 mg/d) orally in drinking water for 6 weeks. Aortic root atherosclerotic lesion area, lesion macrophage content, and the ability of plasma high-density lipoprotein (HDL) to influence monocyte chemotaxis were measured.
Results:
Rev-D4F significantly decreased aortic sinus atherosclerotic lesion area and lesion macrophage content without affecting plasma total and HDL-cholesterol levels in apoE-null mice. The HDL from Rev-D4F-treated mice showed enhanced anti-inflammatory monocyte chemotactic activity, while low-density lipoprotein (LDL) exhibited reduced proinflammatory activity. In in vitro studies, Rev-D4F inhibited LDL oxidation, endothelial cell vascular cell adhesion molecule 1 (VCAM-1), and monocyte chemotactic factor 1 (MCP-1) expression, and monocyte adhesion to aortic endothelial cells.
Conclusions:
The Rev-D4F inhibits atherosclerosis by inhibiting endothelial inflammatory/oxidative events and improving HDL function. The data suggest that Rev-D4F may be an effective apoAI mimetic peptide for further development in preventing atherosclerosis.
Introduction
Increased high-density lipoprotein (HDL) cholesterol is protective against the development of atherosclerotic coronary heart disease. Antiatherogenic properties of HDL are attributed to the ability of apolipoprotein-AI (apoAI; the major protein of HDL) containing HDL particles to stimulate cellular cholesterol efflux through reverse cholesterol transport pathway as well as to inhibit low-density lipoprotein (LDL) oxidation and inflammatory events. 1 , 2 Studies on apoAI gene therapy or apoAI transgenic animals provided convincing evidence for the antiatherosclerotic actions of HDL and apoAI in animal models and generated enormous interest in HDL-/apoAI-based therapy for atherosclerotic cardiovascular disease. 3,4
In this regard, the original investigations on apoAI mimetic synthetic peptides containing class A amphipathic helical sequence which mimic functional properties of apoAI have been under considerable investigation as therapeutic agents for atherosclerosis.
5
Previously several 18-amino acid peptides containing
Since subtle differences in apoAI mimetic peptide structure and its amino acid orientation result in varied lipid-associating and anti-inflammatory biological activity in artery wall cells, we used retroinversal sequencing of the parent L4F peptide to design a modified L4F apoAI mimetic peptide with inverse chirality containing
Materials and Methods
Preparation of L4F and Rev-D4F apoAI Mimetic Peptides
L4F peptide contained the following amino acid sequence: Ac-DW
Animals and Treatment
Female apoE-null mice (4 weeks old) on C57BL/6J background were purchased from Jackson Laboratories and maintained on a chow diet (Ralston Purina, St Louis, Missouri). All animal studies were approved by the VA Long Beach Subcommittee on Animal Studies and Research & Development. Four groups of 4-week-old female apoE-null mice (12 mice per group, except 15 mice in the control) were fed a chow diet and administered water (control), Rev-D4F, or L4F mimetic peptides (0.4 mg/mL, equivalent to 1.6 mg/d) orally in drinking water for 6 weeks. The dosage of Rev-D4F (0.4 mg/mL in the drinking water) was selected based on the previously reported effective dose of D4F to inhibit atherosclerosis in mice. 9 Based on previous publications on the effect of apoAI mimetics on atherosclerosis in apoE-null mice and power analysis, we initially estimated that the 12 mice/group used in our studies would be adequate to attain power for statistical significance.
Measurement of Atherosclerotic Lesion Area and Lesion Macrophage Content
Atherosclerotic lesion areas of the aortic root were measured by established procedures. 10 Briefly, 6-µm serial sections (aortic root region embedded in OCT) were cut through a 250-µm segment of the aortic root, where all 3 valve leaflets are present. Quantitative morphometry of aortic root cross-sections stained with oil-red O was performed using Image Pro Plus software (Leica, Buffalo Grove, Illinois). Immunohistochemical procedures were used to measure the macrophages content and monocyte chemotactic factor 1 (MCP-1) localization and content in aortic root lesions using rat monoclonal to Monocyte + Macrophages (MOMA-2) antibody (Fitzgerald Industries International, Inc, Concord, Massachusetts) and anti-MCP-1 antibody (R&D Systems, Inc, Minneapolis, Minnesota), respectively.
Mouse Plasma Lipoproteins Isolation and Measurements of Lipid Hydroperoxide and Paraoxonase 1 Activity
Mouse plasma lipoproteins (HDL and LDL) were separated from the pooled plasma samples of 3 mice in each group by fast protein liquid chromatography (FPLC/AKTA, Amersham Bioscience, Piscataway, New Jersey). Pooled plasma, 300 µl, was loaded onto a Superose 6 column (Pharmacia, Bridgewater, New Jersey) and eluted with 0.1 mol/L Tris-HCl (pH 7.5) at a constant flow rate of 0.35 mL/min. The HDL-rich particles were located in the HDL size range fractions (fractions 27-29), the LDL-rich particles were located in the fractions of 20 to 23. The pooled fraction rich in lipoprotein, which contains HDL or LDL, was concentrated to a volume of 100 µL with a Centricon 30 (Pharmacia) for use within 3 days. Plasma total and HDL cholesterol concentrations were determined using enzymatic assay kits (Wako Chemical Co, Richmond, Virginia). Paraoxonase 1 activity was measured according to the previously described procedures 11 ; and plasma lipid hydroperoxide content was assayed using Cayman assay kit.
Cell Culture and Treatment
Human primary aortic endothelial cells (HAECs) and human aortic smooth muscle cells (HASMCs; Cambrex Bioscience, Walkersville Inc, Walkersville, Maryland) were cultured in Endothelial cell Growth Medium-2 or Smooth Muscle Growth Medium-2 (EMG-2; SMGM-2) growth medium, respectively, according to the supplier’s instructions. Both HAECs and HASMCs were used at 4 to 6 passages. Human monocytic cells (Acute Monocytic Leukemia cell line [THP-1], ATCC) were grown in 1640 medium containing 5% fetal bovine serum (FBS).
Monocyte Transmigration Assay
Cocultures of human aorta wall cells and monocyte transmigration assays were performed according to the previously described procedures. 12 In brief, HASMCs were seeded onto collagen-coated transwell plates at 1.5 × 105 cells/cm2 and cultured for 2 days. The HAECs were then seeded on top of the HASMC monolayer at 2.5 × 105 cells/cm2 and allowed to grow to form a confluent monolayer. After 5 days of growth, fluorescently labeled THP-1 monocytes (BCECF-AM-labeled monocytes) were added at 2.5 × 105 cells/0.5 mL per cm2 and incubated for 60 minutes at 37°C. Nonmigrated monocytes were removed by washing 5 times with phosphate-buffered saline (PBS). The cocultures were fixed with 10% buffered formalin for 24 hours, and the number of monocytes in the subendothelial space (beneath the endothelial cells) was determined in a minimum of 9 fields under 400 magnification using a fluorescence microscope (Nikon) in a blinded manner.
Low-Density Lipoprotein and Phospholipid Oxidation Assays
Human plasma LDL (100 µg protein/mL, human plasma LDL was commercially obtained from Calbiochem, La Jolla, California) was incubated with HAECs in a serum-free medium in the absence or presence of apoAI mimetic peptides (50 µg/mL) for 24 hours at 37°C. Endothelial cell-induced oxidation of LDL was assessed by measuring the thiobarbituric acid reactive substances (TBARs). Phospholipid oxidation was determined by incubating a micelle substrate (3 mmol/L of 1-palmitoyl-2-linoleoylphosphatidylcholine dispersed in 2.7 mmol/L of borate and 6 mmol/L of deoxycholate) with lipo-oxygenase (5 units/µl) and then measuring the conjugated diene (lipid peroxidation) formation during 20 minutes of incubation at 234 nm. 13
Monocyte Adhesion Assay
Monocyte adhesion assay was performed as previously described, 14 with minor modifications. The HAECs were grown to confluence in 24-well plates and pretreated with the peptides for 18 hours and then stimulated with oxidized-LDL for 6 hours. The THP-1 cells were labeled with a fluorescent dye, 2',7'-bis(2-carboxyethyl)-5(6)-carboxyfluorescein acetoxymethyl ester (BCECF-AM) by incubation with 10 µmol/L BCECF-AM at 37°C for 1 hour in RPMI-1640 medium and were subsequently washed with Endothelial cell Basal Media-2. The labeled THP-1 cells (2 × 105 cells/200 µL) were added to each well of HAECs. The THP-1 cells were allowed to adhere to HAECs by incubation at 37°C for 60 minutes, and unbound THP-1 cells were removed by washing (3 times, 5 minutes). The THP-1 cells bound to HAECs were counted under fluorescent microscope. The numbers of adherent monocytes were photographed and determined by counting 4 fields per well using fluorescent microscopy (Nikon) in a blinded manner.
Preparation of RNA and real-time polymerase chain reaction (PCR) analysis for vascular cell adhesion molecule 1 (VCAM-1) and MCP-1 mRNA expression
Total RNA samples were isolated by the TRIzol method (Life Technologies, Grand Island, New York). First-strand complementary DNA (cDNA) templates were generated with oligo(dT) using the Reverse Transcription kit (Omniscipt RT kit) as described in the manufacturer’s instruction. The amount of messenger RNA (mRNA) for each sample was quantitatively determined by running a SYBRgreen real-time PCR using the Bio-Rad iCycler System. Previously described primers of VCAM-1 and MCP-1 15,16 were obtained from Sigma Genosys. These primers include VCAM-1, 5′-GGGAACGAACACTCTTACCT-3′ (sense) and 5′-ATCCTCCAGAAATTCCTGAC-3′ (antisense); MCP-1, 5′-CCCAGTCACCTGCTGTTAT-3′ (forward) and 5′-TGCTGCTGGTGATTCTTCT-3′ (reverse); and β-actin, 5′-ACATCCGCAAAGACCTGT-3′ (sense) and 5′-CCTTCACCGTTCCAGTTT-3′ (antisense). Quantitative analysis was performed using the 2(-Delta Delta C(T)) method as described in the Bio-Rad iCycler System manual. The mRNA expression levels were normalized to the expression of β-actin before they were compared to the control. The levels of VCAM-1 and MCP-1 mRNA are expressed relative to the control.
Statistical Analysis
In vivo atherosclerosis data in mice were analyzed by nonparametric test (for skewed distribution) using the Wilcoxon Rank Sum test to examine the difference between groups. For other data, the Student t test was used to assess the statistical significance between the control and treatment groups.
Results
Rev-D4F Maintains Exact Mirror Image and Superimposable Characteristics to that of the Parent L4F Peptide
To synthesize Rev-D4F, retroinversal sequencing of the parent L4F peptide was utilized in order to design a modified L4F apoAI mimetic peptide with
Rev-D4F and L4F ApoAI Mimetic Peptides did not Alter Plasma Lipid Profile or Body Weight
Oral administration of apoAI mimetic peptides Rev-D4F or L4F (added in the drinking water at 0.4 mg/mL equivalent to 1.6 mg peptide/d per mouse) to apoE-null mice for 6 weeks did not alter body or liver weight (Table 1). Plasma total cholesterol levels in each group exceeded 400 mg/dL, and there was no difference in cholesterol levels between control and apoAI mimetic peptide (L4F and Rev-D4F)-treated mice (Table 1). Similarly, apoAI mimetic peptides did not affect HDL-cholesterol or non-HDL-cholesterol levels (Table 1). Fast-performance liquid chromatography analysis of cholesterol distribution in the plasma lipoproteins revealed similar patterns in control and apoAI mimetic peptide-treated groups (data not shown). No significant differences were noted in the amount of water consumed between control and apoAI mimetic peptide-treated mice (data not shown).
Effect of ApoA-I Mimetic Peptides on Body Weight, Liver Weight, and Plasma Cholesterol Levels
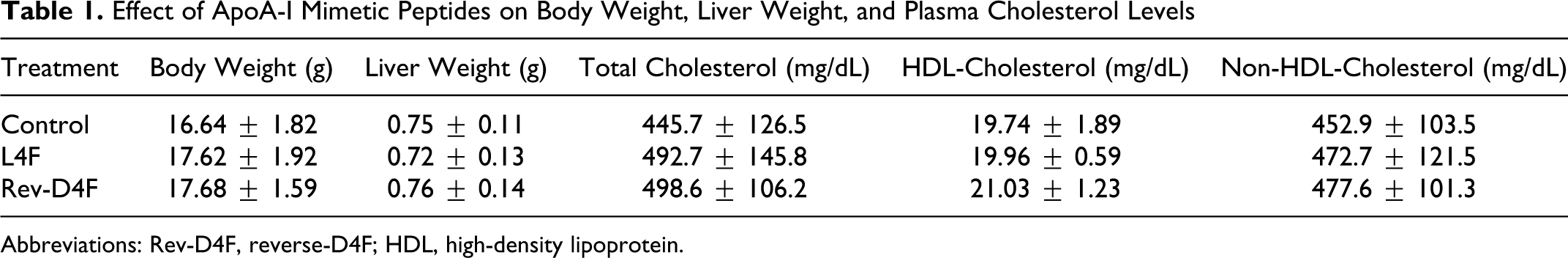
Abbreviations: Rev-D4F, reverse-D4F; HDL, high-density lipoprotein.
Rev-D4F, but not L4F, Inhibits Aortic Sinus Atherosclerosis
Representative histological images of atherosclerotic lesion areas of the aortic root indicated the presence of intimal lipid staining lesions in control and L4F-treated apoE-null mice (Figure 1, upper panel). In apoE-null mice treated with Rev-D4F, intimal lipid stained lesions were considerably minimal when compared to control or L4F-treated mice (Figure 1, upper panel). Quantitative morphometry of aortic root lesions showed that during the 6 weeks of the study, apoE-null mice on a chow diet exhibited early aortic sinus lesions, and the mean lesion area was 46 911 μm2 (Figure 1, lower bar diagram). Oral administration of L4F to apoE-null mice did not significantly alter aortic sinus lesion area compared to controls (Figure 1, lower bar diagram). Rev-D4F significantly decreased aortic sinus lesions by 46% as compared to controls (P = .015; Figure 1, lower bar diagram). Since aortic lesion data in Figure 1 are not normally distributed, we adopted nonparametric test (for skewed distribution) using Wilcoxon Rank Sum test to determine the statistical differences between groups.
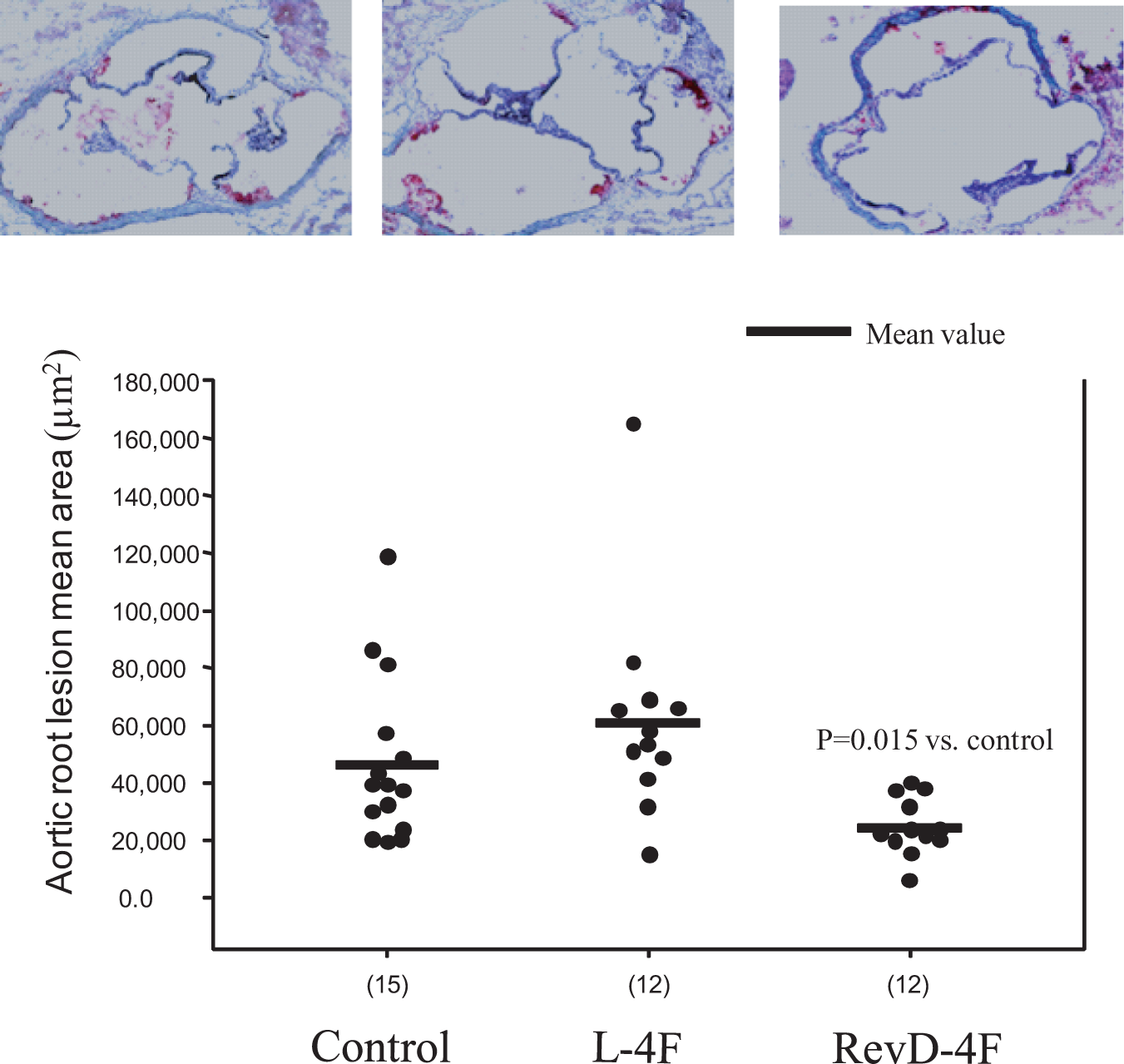
Effect of apoA-I mimetic peptides on atherosclerosis in apoE-deficient mice. The aortic sinus sections were stained with oil-red O and counterstained with hematoxylin and light green to quantify atherosclerotic lesions in aortic roots as described in Methods section. Upper panel shows the representative histological images of atherosclerotic lesions in control, L4F-, and Rev-D4F-treated mice. Lower bar diagram shows the quantitative analysis of atherosclerotic lesions in control, L4F-, and Rev-D4F-treated mice. Values in the lower bar diagram are mean ± SEM of 12 mice in each group (15 mice in control). Statistical significance using Wilcoxon Rank Sum test were control vs Rev-D4F, P = .015. apoAI indicates apolipoprotein-AI; Rev-D4F, reverse-D4F; SEM, standard error of the mean.
Rev-D4F, but not L4F, Inhibits Aortic Root Macrophage Abundance and MCP-1 Content
Serial cross-sections of the aortic sinus region were examined for macrophage abundance using macrophage-specific antibody MOMA-2 staining. As shown in Figure 2 upper panel, representative immunohistological images indicated that macrophage-positive cells were abundantly seen in the subendothelial area of atherosclerotic lesions in the control apoE-null mice. While L4F had no effect on aortic lesion macrophage content when compared to controls, Rev-D4F treatment considerably reduced macrophage positive cells (Figure 2, upper panel). Quantitative analysis showed that Rev-D4F significantly inhibited macrophage content by 47% as compared to controls (Figure 2, lower bar diagram).
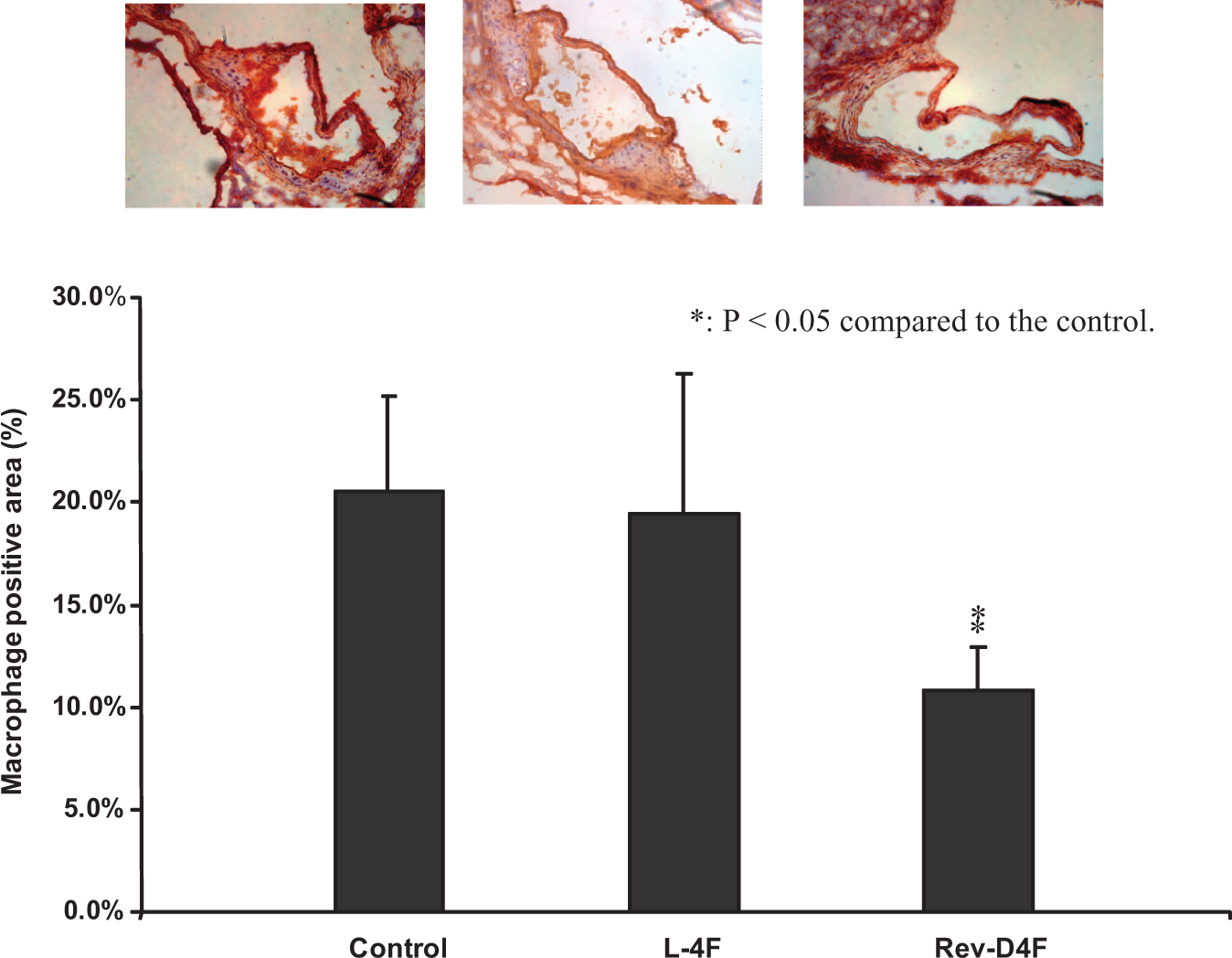
Macrophage accumulation in aortic root sinus lesions of apoE-null mice. Frozen sections of aortic root sinus plaques were stained with antimacrophage-specific antibody rat monoclonal to Monocyte + Macrophages (MOMA-2). Computer-assisted image analysis was used to determine the relative amounts of macrophages in atherosclerotic lesion. Upper panel shows the representative histological images of the presence of macrophage-positive areas in control, L4F-, and Rev-D4F-treated mice. Lower bar diagram shows the quantitative analysis of macrophage accumulation in control, L4F-, and Rev-D4F-treated mice. Values in the lower bar diagram are mean ± SEM of 12 mice in each group (15 mice in control). apoE indicates apolipoprotein E; Rev-D4F, reverse-D4F; SEM, standard error of the mean.
Representative immunohistological images for aortic root MCP-1 content and localization showed that MCP-1-positive areas (stained red) were abundant in control and L4F-treated apoE-null mice (Figure 3, upper panel). Noticeably reduced MCP-1-positive areas were seen in aortic root sections of Rev-D4F-treated apoE-null mice (Figure 3, upper panel). Quantitative analysis of aortic root MCP-1 content indicated that Rev-D4F significantly reduced aortic root MCP-1 content by 32% as compared to controls (Figure 3, lower bar diagram). L4F had no effect on aortic root MCP-1 content (Figure 3, lower bar diagram).
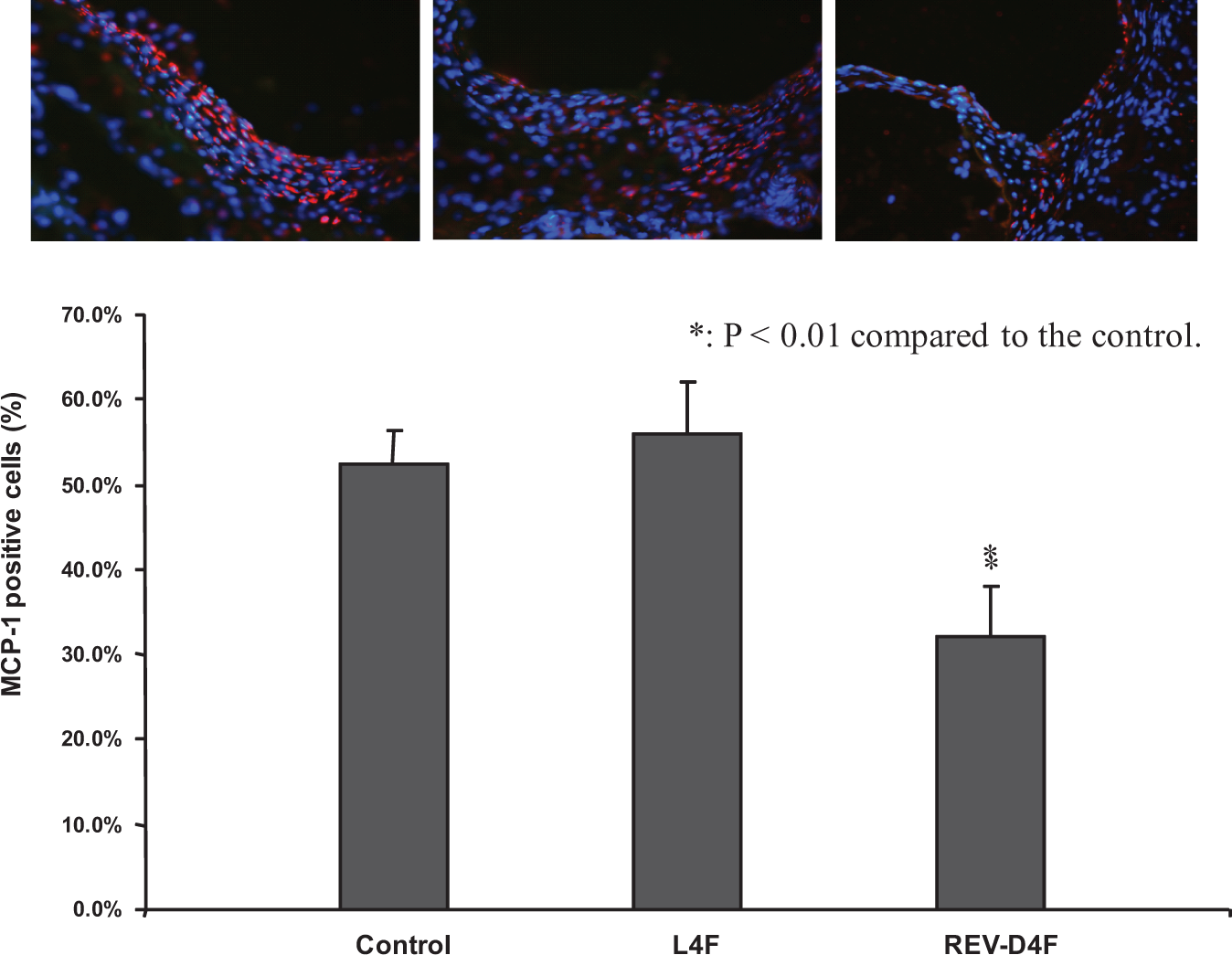
Monocyte chemotactic factor 1 (MCP-1) expression in aortic root sinus lesions of apoE-null mice. Frozen sections of aortic root sinus plaques were stained with anti-MCP-1-specific antibody and visualized by red fluorescent dye. Computer-assisted image analysis was used to determine the relative amounts of MCP-1 expression in atherosclerotic lesion. Upper panel shows the representative histological images of the presence of MCP-1-positive areas in control, L4F-, and Rev-D4F-treated mice. Lower bar diagram shows the quantitative analysis of the aortic root MCP-1 expression in control, L4F-, and Rev-D4F-treated mice. Values in the lower bar diagram are mean ± SEM of 12 mice in each group (15 mice in control). Rev-D4F indicates reverse-D4F; SEM, standard error of the mean.
Functional Properties of HDL and LDL Isolated From Plasma of Mice Treated With apoAI Mimetic Peptides on Monocyte Chemotaxis in CoCultures of Human Aorta Wall Cells
In order to examine whether apoAI mimetic peptides influenced functional properties of lipoproteins, we isolated HDL and LDL from plasma of control and apoAI mimetic peptide-treated mice by FPLC. In the first study, we tested the protective capacity of HDL to inhibit LDL-induced monocyte chemotaxis in cocultures of human aortic endothelial smooth muscle cells. As shown in Figure 4A, HDL isolated from control and L4F-treated mice did not show protection against LDL-induced monocyte chemotaxis. In contrast, HDL isolated from mice treated with Rev-D4F significantly inhibited LDL-induced monocyte chemotaxis by 81% (Figure 4A). These data clearly indicated the protective capacity of HDL fractions isolated from Rev-D4F-treated mice. Additionally, Rev-D4F treatment significantly increased HDL paraxonase activity by about 40% when compared to control or L4F treatment (353 ± 25, 256 ± 39, and 295 ± 25 units per microgram of HDL-cholesterol, respectively).
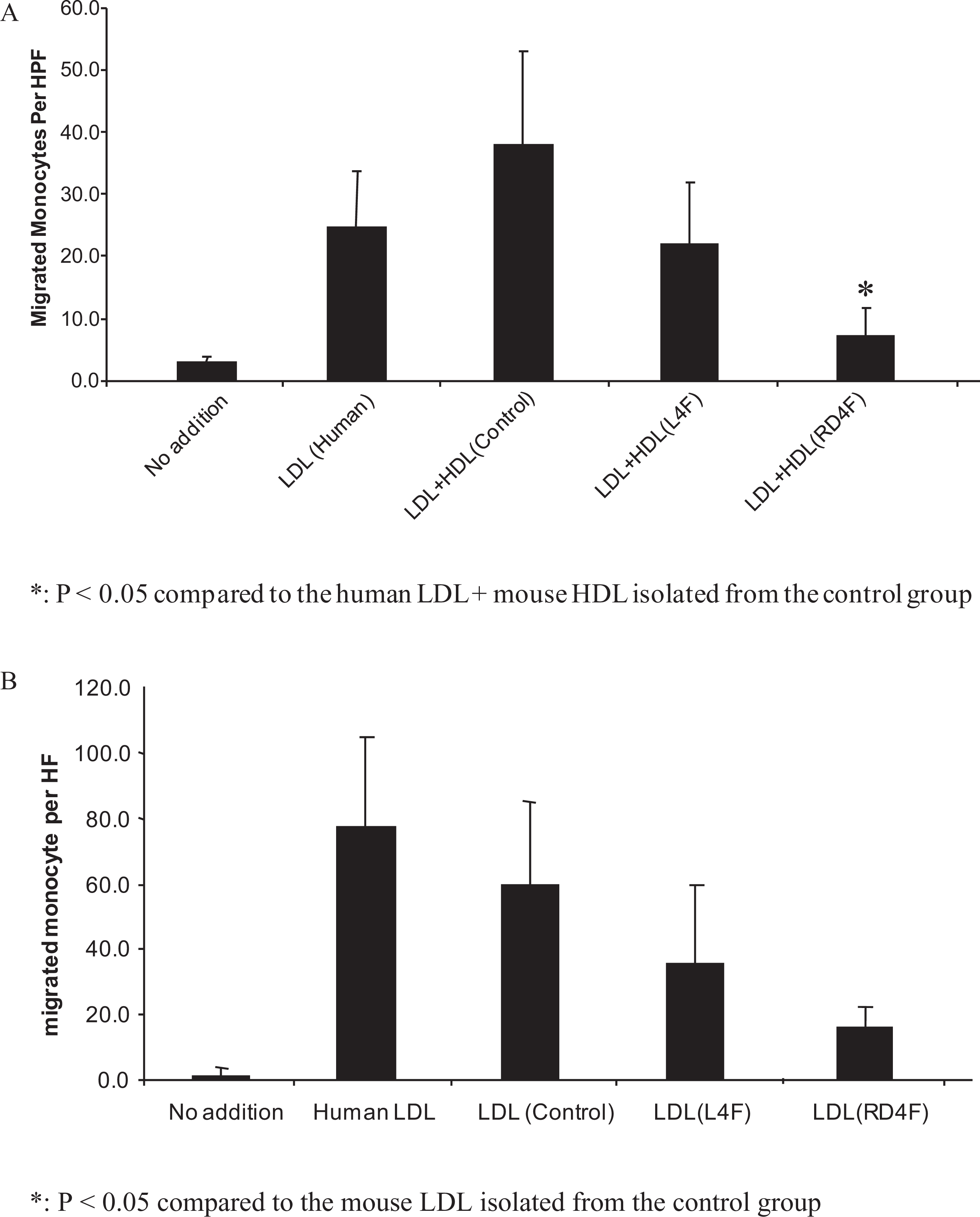
Effect of HDL and LDL fractions isolated from apoE-null mouse plasma on monocyte chemotaxis in human aorta wall cell coculture model. A, HDL protective capacity for LDL-induced monocyte chemotaxis. Human LDL at 100 µg/mL was added to cocultures alone (LDL) or together with mouse HDL at 50-µg cholesterol/mL taken from mice that received either no apoAI mimetic peptides (control) or peptide L4F (LDL + HDL, L4F) or Rev-D4F (LDL + HDL, RD4F) and monocyte chemotactic activity was determined. The data shown are the mean ± SD of 3 experiments. *P < .05 compared with LDL + no peptide-treated mouse HDL. B, Monocyte chemotaxis activity in cocultures was assessed using mouse LDL (100 µg cholesterol/mL) isolated from mice that did not receive apoAI mimetic peptide (control) or from mice that received apoAI mimetic peptides L4F or Rev-D4F. No addition represents control coculture without incubation with LDL. Human plasma LDL was added only to the second group in Figure 4B which serves as a positive control. The data shown are the mean ± SD of 3 experiments.*P < .05 compared with no peptide-treated mouse LDL. HDL indicates high-density lipoprotein; LDL, low-density lipoprotein; Rev-D4F, reverse-D4F; SEM, standard error of the mean.
Since LDL is known to induce monocyte chemotaxis, in the second study we examined whether LDL fractions isolated from mice treated with apoAI mimetic peptides would modulate its monocyte chemotactic property. Low-density lipoprotein isolated from both control and L4F-treated mice markedly induced monocyte chemotaxis as assessed in cocultures of aorta wall cells (Figure 4B). Conversely, LDL fractions isolated from mice treated with Rev-D4F markedly inhibited monocyte chemotaxis compared to LDL fractions from control mice (Figure 4B). These data indicate that LDL fractions from Rev-D4F-treated mice exhibited reduced pro-chemotactic property compared to LDL from control mice.
In Vitro Effects of ApoAI Mimetc Peptides on Phosphlipid and LDL Oxidation, VCAM-1 and MCP-1 Expression, and Monocyte Adhesion
Since atherosclerosis is regulated by oxidative and inflammatory processes, we have undertaken a series of in vitro studies using cell-free system or human aortic endothelial cells to understand mechanisms of action of Rev-D4F compared to its parent L4F. Using cell-free system, we have shown that Rev-D4F and the parent L4F apoAI peptides similarly inhibited lipo-oxygenase-catalyzed phosphatidylcholine micelle oxidation (Figure 5A). Similarly, Rev-D4F and L4F apoAI peptides significantly inhibited human aortic endothelial cell-mediated LDL oxidation (Figure 5B).
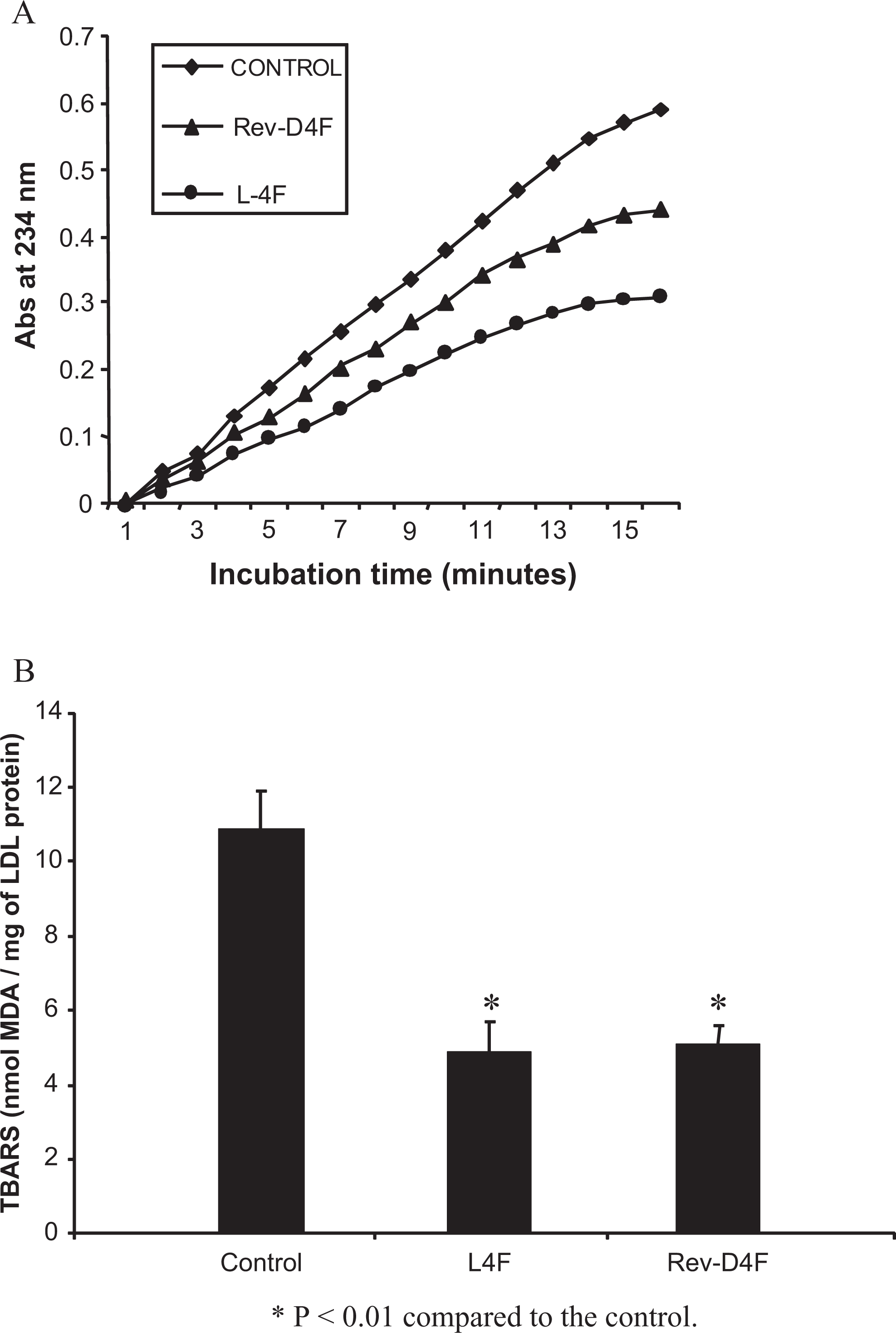
Effect of Rev-D4F in inhibiting lipo-oxygenase-mediated lipid peroxidation (A) and LDL oxidation (B). A, Phospholipid micelles were exposed to soybean lipo-oxygenase (5 U/µL) in the presence of L4F or Rev-D4F and lipid peroxidation was determined as described in the Methods section. Results are representative of 3 identical experiments. B, Human aortic endothelial cells were incubated with LDL in the absence or presence of apoAI mimetic peptides, and LDL oxidation was assessed by TBARS as described in Methods section. Data are mean of 3 separate experiments. *P < .05 compared to control. apoAI indicates apolipoprotein-AI; LDL, low-density lipoprotein; Rev-D4F, reverse-D4F; SEM, standard error of the mean.
In additional studies, we investigated the in vitro effects of apoAI mimetic peptides on the expression of oxidation-sensitive inflammatory genes VCAM-1 and MCP-1 and the adhesion of monocytes to endothelial cells. Preincubation of human aortic endothelial cells with Rev-D4F similarly reduced ox-LDL-induced VCAM-1 and MCP-1 expression as compared to L4F (Figure 6A and B). Parallel to VCAM-1 and MCP-1 expression, apoAI mimetic peptides (Rev-D4F and L4F) also significantly inhibited ox-LDL-induced monocyte adhesion to aortic endothelial cells (Figure 6C).
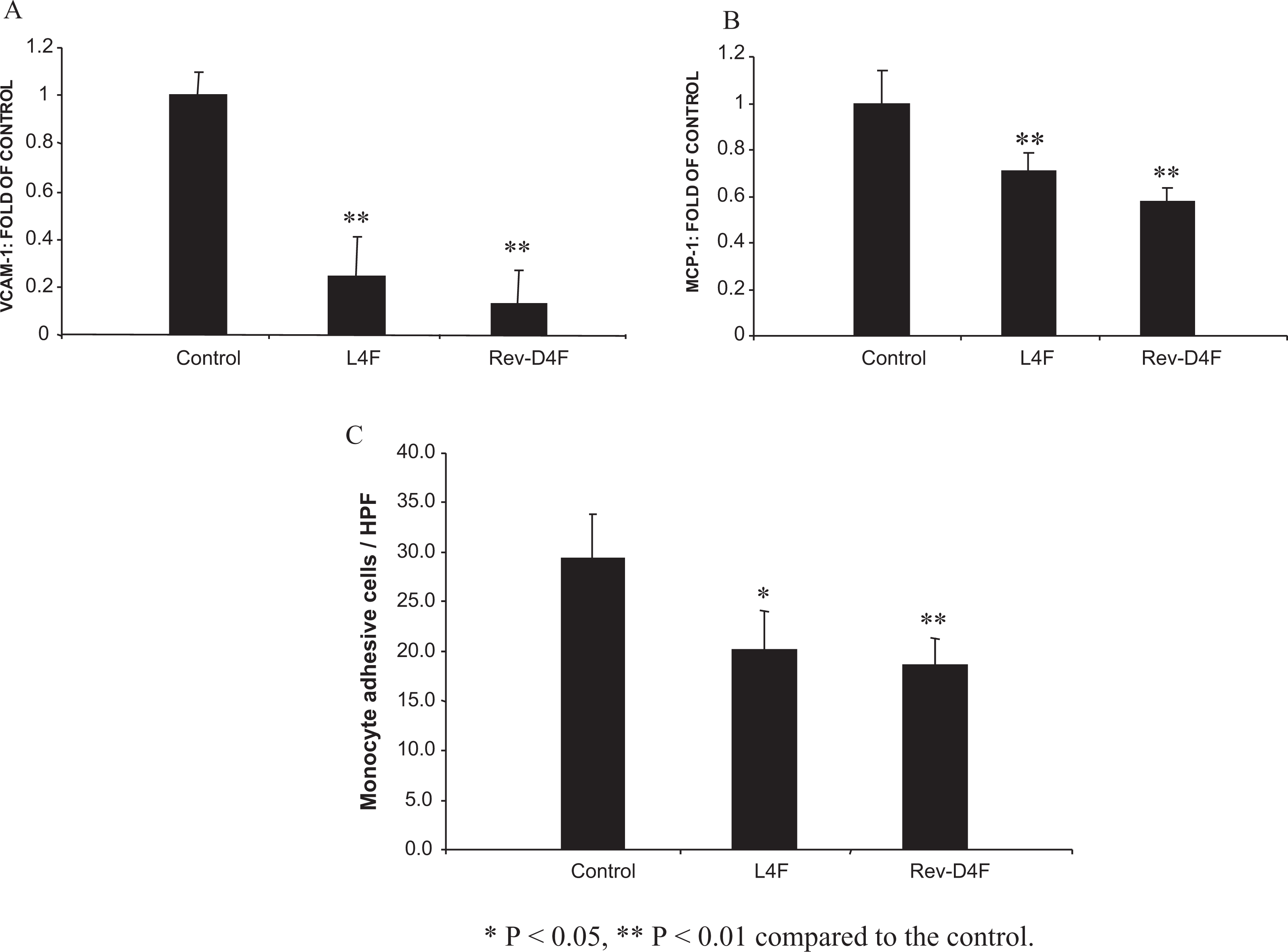
Effect of Rev-D4F to inhibit LDL-mediated VCAM-1 and MCP-1 expression and monocyte adhesion in HAECs. mRNA expression of VCAM-1 (A) and MCP-1 (B) were measured by quantitative real-time RT-PCR as described in Methods section. The mRNA expression levels were normalized to the expression of β-actin before they were compared to the control. Both VCAM-1 and MCP-1 mRNA levels are expressed relative to the control. C, Monocyte adhesion to human aortic endothelial cells induced by oxidized LDL. Human aortic endothelial cells were pretreated with L4F or Rev-D4F for 18 hours and stimulated with ox-LDL for 6 hours. A fluorescent dye (BCECF-AM)-labeled Acute Monocytic Leukemia cell line (THP-1) cells were added to each well of confluent HAECs. The THP-1 cells bound to HAECs were counted under fluorescent microscope. Data are mean of 3 separate experiments. *P < .05 compared to control. Rev-D4F indicates reverse-D4F; LDL, low-density lipoprotein; VCAM-1, vascular cell adhesion molecule 1; MCP-1, monocyte chemotactic factor 1; mRNA, messenger RNA; HAECs, human primary aortic endothelial cells.
Discussion
Our studies demonstrated that the Rev-D4F treatment for 6 weeks significantly inhibited the early aortic sinus atherosclerosis and lesion macrophage content in apoE-null mice without affecting plasma or HDL-cholesterol. Because mammalian enzymes degrade peptides synthesized from
Based on our mechanistic studies assessing the HDL function in ex vivo vascular cell coculture studies (Figure 4A), we suggest that apoA1 mimetic peptide Rev-D4F provides protection against atherosclerosis through improving HDL function independent of HDL levels. Previous studies indicated that plasma D4F was mainly found in HDL fraction (85%) after oral administration of apoAI mimetic peptide D4F to apoE-null mice. 17 Since D4F is a parent peptide of Rev-D4F, we predict that in our studies also Rev-D4F would be mainly incorporated into HDL fraction. However, in this initial study we have not performed these analyses in our studies with Rev-D4F, and such analyses would be performed in future studies.
In addition to the role of apoAI in reverse cholesterol transport, the ability of apoAI to remove low levels of highly potent oxidized lipids (so-called seeding molecules) required for oxidation of LDL has been extensively investigated to understand the mechanisms by which apoAI inhibits atherosclerotic processes.
12
,18,19
ApoAI mimetic peptides have been reported to remove these “seeding molecules” involved in LDL oxidation and improved the ability of HDL to protect LDL against oxidation.
12
,18,20
In order to define the mechanisms by which Rev-D4F inhibits atherosclerosis, we have investigated ex vivo assessment of lipoprotein monocyte chemotactic functions and in vitro endothelial–monocyte interaction. While treatment of mice with L4F had no effect on the functional properties of HDL or LDL, Rev-D4F treatment markedly increased the protective capacity of HDL particles and decreased the ability of LDL to induce monocyte chemotactic activity in human vascular wall coculture system. Parallel to the atherosclerosis findings, as L4F with
In order to define underlying mechanisms by which these apoAI mimetic peptides affect atherosclerosis, we performed in vitro studies using primary cultures of human aortic endothelial cells. The data indicated that both Rev-D4F and L4F significantly inhibited the mRNA expression of VCAM-1 and MCP-1 associated with endothelial–monocyte adhesion and monocyte chemotaxis. Additionally, these apoAI mimetic peptides effectively inhibited LDL oxidation induced by endothelial cells or transitional metal ions. Since L4F in these in vitro studies did not undergo enzymatic degradation (as it occurs when administered orally to animals), L4F effectively inhibited VCAM-1 and MCP-1 expression, monocyte adhesion, and LDL oxidation. Additionally, these data suggest that Rev-D4F retained beneficial lipid-associating properties and oxidized lipids-seeding molecule removal capacity of the parent peptide and directly exhibited anti-inflammatory properties specifically involved in endothelial monocyte adhesion and chemotaxis, key primary events involved in early atherosclerosis. In addition to antioxidation and anti-inflammatory properties, it is likely that Rev-D4F may also utilize other atheroprotective properties including cholesterol transport from macrophages which would be investigated in future investigation. Additional future studies are needed to determine the effect of Rev-D4F on cholesterol transport from macrophages.
Although the findings of the current investigation suggest that Rev-D4F may be a promising apoAI mimetic peptide for the prevention of atherosclerosis, additional detailed studies on the safety profile of Rev-D4F in humans are required. Bloedon and colleagues have recently reported that the oral administration of apoAI mimetic peptide D4F (30-500 mg) to patients with coronary heart disease or equivalent risk did not cause significant trends in any safety parameters during the follow-up visits 2 to 30 days. 21 Overall, there were no unexpected adverse events, and none were thought to be due to the administration of D4F. 21 Future similar investigations on the safety profile of Rev-D4F in humans would be necessary for further clinical development of Rev-D4F for preventing atherosclerosis.
In summary, we have shown that Rev-D4F inhibits early atherosclerosis by directly inhibiting endothelial inflammatory/oxidative events and improving HDL anti-inflammatory function. Our studies suggest that Rev-D4F is an effective apoAI mimetic peptide which can be developed for oral administration for preventing atherosclerosis. Additionally studies on the effect of Rev-D4F on established or advanced atherosclerosis in apoE-null mice and/or LDLR-null mice would be important in further development of Rev-D4F as a therapeutic agent for atherosclerotic coronary artery disease.
Footnotes
Part of this work was presented in abstract form at the American Heart Association Meeting, Dallas 2005 and Chicago 2006. Arisaph Pharmaceuticals designed and synthesized Rev-D4F and L4F, and licensed to Kos Pharmaceuticals, Inc. The authors VSK and MLK are senior coauthors of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Bachovchin and Lai are also staff members in Arisaph Pharmaceuticals, Boston. All other authors have no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported through a grant from Kos Pharmaceuticals, Inc, which was acquired by Abbott Laboratories.
