Abstract
Background:
Inhibiting the growth of small abdominal aortic aneurysms (AAAs) is a clinically valuable goal and fills an important therapeutic void. Based on studies in animals and humans, inhibition of the activity of elastolytic matrix metalloproteinases (MMPs) has the potential to slow AAA expansion and limit morbidity and the need for surgery. Previous attempts to make use of the synthetic MMP inhibitors in the treatment of chronic conditions have been limited by intolerable side effects. The limited-spectrum synthetic MMP inhibitor, XL784, was well tolerated and devoid of side-effects associated with other nonspecific MMP inhibitors in phase I studies. We hypothesized that clinically relevant doses of XL784 would be effective at inhibiting aneurysm development in a mouse model.
Methods:
The 14-day elastase-perfusion model of AAA in mice was used. An initial screening study of XL784 (50 [n = 17], 125 [n = 17], and 250 mg/kg [n = 18]) administered via gavage daily until harvest. Controls received diluent alone (n = 18) or doxycycline in drinking water (n = 19). Aortic diameter was measured pre-perfusion (ADpre) and at harvest (ADhar). A second study used XL784 (250 [n = 9]; 375 [n = 9], and 500 mg/kg [n = 14]) and diluent alone (n = 9) administered via gavage. The percentage dilatation [%ΔAD = [(ADhar − ADpre)/ADpre] ×100] was calculated and elastin and inflammatory content was scored.
Results:
All mice tolerated the treatments similarly. Control mice all developed aneurysms with a mean %ΔAD of 158.5% ± 4.3%. Treatment with all doses of XL784 and doxycycline were effective in inhibiting aortic dilatation. There was a clear dose–response relationship between XL784 and reductions in aortic dilatation at harvest (50 mg/kg 140.4% ± 3.2%; 125 mg/kg 129.3% ±5.1%; 250 mg/kg 119.2% ± 3.5%; all Ps < .01 compared to control). This continued with the higher doses (375 mg/kg 88.6% ± 4.4%; 500 mg/kg 76.0% ± 3.5%). The highest 2 doses of XL784 tested were more effective than doxycycline (112.2% ± 2.0%, P < .05) in inhibiting maximal dilatation of the aorta after elastase perfusion.
Introduction
Abdominal aortic aneurysms (AAAs) of less than 5.5 cm in maximal diameter have a very low risk of morbidity so that the risk of leak or rupture is so low as to make surgical intervention of no net benefit to this population. Unfortunately, over time, most aneurysms continue to increase in size with progressive weakening of the aortic wall. Surgical treatment of these larger aneurysms significantly reduces subsequent morbidity but is itself accompanied by risk and significant expense. Medical treatment to prevent the progressive expansion of aortic aneurysms could offer a novel therapeutic approach to reduce the risk of aneurysm rupture while limiting the need for preventive surgery.
Multiple studies have suggested that certain members of the family of matrix metalloproteinases (MMPs) which are capable of degrading elastic fibers may contribute to the progression of aneurysm disease. Therefore, clinical inhibition of MMP could be an attractive therapeutic approach to prevent the progressive expansion of small AAAs and thus reduce morbidity and the need for surgical intervention. Studies in multiple animal models have shown that a variety of MMP inhibitors are effective at preventing growth of model AAA, including the anti-infective doxycycline. Doxycycline and modified tetracyclines without antibiotic activity have been shown to inhibit MMP activity and aneurysm development in animal models. Limited clinical studies have also suggested that doxycycline at typical anti-infective doses may be effective clinically, although there may be important limits to the long-term tolerability of the drug.
Synthetic MMP inhibitors with a broad spectrum of activity have been studied to treat a number of diseases which may result from the inappropriate activity of these enzymes. Unfortunately side effects have limited the long-term tolerability of many of these compounds. The synthetic MMP inhibitor, XL784, has a more limited spectrum of activity against the MMP enzymes which act predominantly as collagenases but is relatively more effective at inhibiting elastolytic MMPs. 1 This limited spectrum appears to reduce the side-effects of this molecule. 2 , 3
We hypothesized that an MMP inhibitor with this spectrum of activity would be particularly effective at inhibiting AAAs, while its limited side-effect profile makes it an attractive long-term therapeutic agent to prevent clinical aneurysm growth. Therefore, we evaluated the efficacy of this compound in a well-established mouse model of AAA and compared it to treatment with doxycycline.
Methods
Experimental Animals
All mice used in the experiment were commercially obtained C57/Bl6 (Jackson Labs, Bar Harbor, Maine) inbred strain mice. Throughout the experimental course, mice were allowed free access to food and water. Animals were housed in a controlled animal facility, and all mouse care and treatment occurred under protocols approved by the Washington University School of Medicine Animal Studies Committee. The experiments described below were begun in April 2010 and were completed by June 2011.
Elastase Perfusion Model
Adult male mice were subjected to transient perfusion of the abdominal aorta as described previously. 4 Briefly, after sedation and preparation, the aorta was approached through a midline laparotomy. The infrarenal aorta was dissected free of surrounding tissues and the diameter was measured under magnification with a micrometer. A segment of infrarenal aorta was isolated and a 5-minute perfusion of this segment was performed through an arteriotomy at 100 mm Hg with a solution containing type I porcine pancreatic elastase (PPE 0.16 U/mL; E-1250, Sigma-Aldrich, Inc, St Louis, Missouri). All of the experiments were performed with a single PPE preparation derived from the same commercial source and lot.
Following aortic perfusion the arteriotomy was repaired, the laparotomy was closed, and the animal was allowed to completely recover before returning to their standard housing. The animals were maintained in standard housing with ad libitum access to standard food and water for 14 days.
Final Aortic Diameter Measurement and Specimen Collection
Two weeks following elastase perfusion, the mice were again anesthetized, the laparotomy incision was reopened, and the final aortic diameter was measured in vivo prior to sacrifice. The animals were humanely sacrificed, and the entire perfused segment of aorta was harvested for further analysis as described below.
XL784 Treatment Protocol 1
A total of 89 animals underwent aortic perfusion. Beginning the day of perfusion, animals were treated with the study drug, a negative control of the drug diluent, or a known aneurysm growth-inhibiting control agent (doxycycline). All animals were kept in the same assigned group until sacrifice. Of the initial 89 animals that underwent perfusion, a total of 76 animals survived to sacrifice and were included in the analysis.
Animals treated with the experimental agent, XL784, received gavage daily with the agent diluted in 0.1 ml of Cremophor, a nonionic castor oil-based solubilizer and emulsifying agent (BASF Corporation, Lindburgerhof, Germany). Three doses of the drug were used, 50 (n = 17), 125 (n = 17), and 250 mg/kg per d (n = 18) administered as a single daily dose. Control animals were similarly treated with daily gavage of the Cremophor diluent only (n = 18). The fifth group of mice did not receive a gavage treatment but were treated with doxycycline (n = 19) in their drinking water at a concentration intended to deliver 100 mg/kg per d based on the known water consumption of the animals, as previously described. 5 , 6
Two model surgeons performed the procedures and each performed equal numbers of animals in each group. Evaluation of aortic diameters was also performed by the model surgeons who were not blinded to the treatment group.
XL784 Treatment Protocol 2
In the second treatment protocol, a total of 50 animals underwent aortic perfusion and 47 animals survived for analysis at 14 days. The 5 treatment groups were XL784 at 250, 375, or 500 mg/kg, Cremaphor diluent alone, or doxycycline 100 mg/kg. Animals were assigned in groups of 3 to a treatment group rotating randomly through each treatment group until there were 9 animals in each group except for the 500 mg/kg per d group which totaled to 14 animals. All animals were treated with the selected therapy with daily gavage. A single animal surgeon performed all of the procedures and gavage. A different animal surgeon performed all of the measurements of aortic diameter, and that individual was unaware of the treatment group of each animal.
Measures of Plasma Levels of XL784
To determine the plasma levels of the drug in the mouse, C57Bl/6 mice were treated with daily gavage of XL784 in Cremophor diluent. In one experiment, there were 5 different doses of the drug used (50, 125, 250, 375, and 500 mg/kg per d), and 6 mice per dose were treated for 3 days. In a second experiment, all mice received 500 mg/kg per d of XL784 with 20 mice being treated for 3 days and 18 mice being treated for 14 days. Half of each group of mice were sacrificed at 1 hour following the final dose, and half were sacrificed 24 hours later. Blood was collected into lithium heparin containing phlebotomy tubes and centrifuged for 10 minutes at 5000g at 4°C. The plasma is collected and then centrifuged for 5 minutes at 5000g at 4°C to remove platelets. The plasma was then stored in 100 µL aliquots at <−70°C until processed.
Plasma levels of XL-784 were assayed using a liquid chromatography with tandem mass spectrometric detection (LC-MS/MS) method on Sciex QTRAP 5500. This assay was contracted with Covance Laboratories, Inc (Madison, Wisconsin). Results are reported in ng/mL.
Light Microscopy
The aortas from 3 mice of each experimental group were perfusion fixed with 10% neutral-buffered formalin, removed, and placed in additional formalin for a minimum of 24 hours prior to processing for paraffin embedding. Following paraffin embedding, aortic specimens were cut into sections of 5 μm and mounted on glass slides. Each specimen was stained with hematoxylin and eosin to evaluate the inflammatory cell infiltration and Accustain Elastic Stain kit (HT25A-1KT; Sigma-Aldrich, Inc) to assess the degree of elastin degradation. Photomicrographs of serial sections were obtained using an Olympus BX60 light microscope equipped with CV12 video capture camera.
At least 3 serial sections from the central portion of the harvested aorta were collected and randomly labeled. These photomicrographs were scored by 6 experienced individuals unaware of the experimental group as previously described. 7 Both the elastin content and the degree of inflammatory infiltration were evaluated. The scale for elastin content ranged from 0 (no stainable elastin) to 4 (normal stainable amounts of elastin without breaks in the lamellae), and the scale for inflammation ranged from 0 (no inflammatory cells seen) to 4 (extensive inflammation in all layers of the aortic wall), and the mean (± standard error [SE]) score was calculated.
Statistical Analysis
The change in aortic diameter between baseline and harvest are expressed as the mean percentage increase from baseline ± standard error of the mean (SEM). Statistics used for multiple sample experiments include 1-way analysis of variance (ANOVA) or Welch ANOVA for unequal variances when appropriate and Tukey-Kramer multiple comparison posttest. Presence of an aneurysm at harvest was defined as an increase of ≥100% over pre-perfusion aortic diameter. Chi-square (or Fisher exact test when average cell size was less than 5) was used to test for differences in incidence of AAA between groups.
Results
Screening the Effect of XL784 on Model Aneurysm Development (Experiment 1)
In an initial screening study, AAA model initiation was performed on a total of 89 mice. After aortic perfusion the mice were assigned to 1 of the 5 treatment groups for 2 weeks of treatment. Three of the groups of mice were treated with XL784 50, 125, or 250 mg/kg daily by oral gavage. Two additional groups of mice served as the controls. Untreated control mice received daily gavage of the carrier solution for the experimental drug. A second control consisted of mice who received a concentration of doxycycline in their drinking water which was calculated to result in a daily dose of 100 mg/kg based on typical water consumption habits.
There was no appreciable toxicity noted in any of the animals. We found no significant difference in the numbers of animals surviving for 2 weeks following surgery, with 2 animal deaths after perfusion but prior to harvest in each of the experimental drug treatment groups, 3 animal deaths in the diluent-only treated groups, and 4 animal deaths in the doxycycline-treated group. The remainder of the animals underwent repeat laparotomy after 14 days and the aortic diameter was measured prior to sacrifice and compared to the pre-perfusion diameter of the aortas.
In untreated control animals, the mean %ΔAD was 158.5% ± 4.3%, and all of the animals had a %ΔAD which was greater than 100%—our definition of aneurysm development in this model. As expected, treatment with doxycycline resulted in a mean %ΔAD significantly less than the untreated control animals at 112.2% ± 2.0% (P < .0001). Doxycycline treatment also resulted in 13% of animals not reaching the threshold designated for aneurysms in the model. All dose regimens of XL784 resulted in significant inhibition of mean aneurysm diameter (Figure 1A). A box-and-whisker plot is shown in Figure 1B. The median %ΔAD diminished with increasing drug dosage of XL784. Only 1 animal treated with XL784 (at the 125 mg/kg dose) had a %ΔAD of greater than the median of the control animals.

Effect of increasing doses of XL784 treatment via daily gavage on aortic dilatation 14 days following isolated aortic elastase perfusion. Control mice were gavaged with the drug carrier solution (Cremophor) for the XL784 on a schedule matching the treated mice or received doxycycline in their drinking water at a dose calculated to result in a intake of 100 mg/kg per d (n = 15 in each group except the 250 mg/kg group with n = 16). A, Results are reported as mean ± standard error (SE). Data were compared with analysis of variance (ANOVA) using Tukey correction for multiple comparisons among the treatment groups. Significant differences are indicated by a line connecting the 2 groups with a significance value over the line. All significant (P < .05) comparisons are shown. B, Results shown in box and whisker plot format to demonstrate the interquartile and overall range of the aortic dilatation based on treatment.
Blinded Evaluation of XL784 on Model Aortic Aneurysms (Experiment 2)
Based on the screening evaluation, a second set of XL784 treatment experiments in elastase perfusion model mice was performed. In this set of experiments, a single mouse surgeon performed all of the procedures as well as the treatment gavage, but a second individual—blinded to treatment group—measured the aortic diameters. There were 4 treatment groups: XL784 250, 375, and 500 mg/kg and untreated controls receiving drug diluent.
There was no appreciable toxicity in any of the treated animals, there were a total of 50 animals that underwent aortic elastase perfusion, and 3 animals did not survive the 2 weeks until harvest, 1 in the untreated control group and 1 each in the XL784 375 and 500 mg/kg groups. The mean %ΔAD was calculated based on the aortic diameters measured for all of the remaining mice prior to harvest at 14 days. All doses of XL784 and doxycycline treatment significantly inhibited aneurysm development compared to the untreated control group (P < .01; Figure 2). In the 250, 375, and 500 mg/kg XL784 treatment groups, the dilatation of the aorta achieved the aneurysm threshold (maximal %ΔAD>100) in 33%, 13%, and 0% of the animals at harvest (P < .001 compared to the untreated of all 4 groups).

Effect of an increased treatment dose of XL784 on the development of aneurysms in mice 14 days following elastase perfusion (XL784 250 mg/kg n = 9, 375 mg/kg n = 8, 500 mg/kg n = 13, vehicle n = 8, and doxycycline [gavage n = 9, drinking water n = 15 from Figure 1]). Bars depict the mean ± standard error (SE) percentage increase in aortic diameter compared to baseline, and the line graph depicts the percentage of animals with an aneurysm (%ΔAD > 100%) at harvest. All treatment regimens resulted in significant reduction of %ΔAD compared to vehicle-treated controls, and animals treated with 375 and 500 mg/kg per day of XL784 had a significantly smaller mean increase in aortic diameter than the doxycycline-treated animals (†P < .05 compared to control, ‡P < .05 compared to doxycycline [Drinking] treated.) In addition, significantly fewer animals in the XL784 treatment groups achieved aneurysmal dimensions compared to both control and doxycycline (drinking)-treated animals (P < .05 by chi-square).
The inhibition of aneurysm development in mice treated with 100 mg/kg per d of doxycycline by gavage was significantly greater than the inhibition when the doxycycline is administered in the drinking water. There are also fewer AAA developed in animals receiving doxycycline by gavage (11.1%) rather than in the drinking water (86.7%). When compared to doxycycline administered in the drinking water, the animals treated with XL784 at 375 and 500 mg/kg developed significantly less dilatation of the aorta and fewer aneurysms (P < .001). However there were no significant differences in aneurysm development between any of the XL784 groups and doxycycline administered by gavage in this experiment.
Aortic Histology
Three aortas from each group (except the XL784 250 and 375 mg/kg groups in experiment 2) were fixed in formalin following aortic harvest and processed into paraffin blocks. During harvest only the maximally dilated segment of the aorta is taken, and serial sections of the block are made to assure that the most dilated segment of aorta is imaged. These maximally dilated segments are stained with hematoxylin and eosin stains as well as an elastin highlighting stain (Verhoff-Von Giesen [VVG]).
As shown by the representative images in Figure 3, in the absence of MMP inhibitor therapy, aortas from control animals had severe medial elastic fiber destruction associated with an appreciable mononuclear cellular infiltration. Treatment with doxycycline following elastase perfusion results in the preservation of the medial elastin, but there continues to be a modest cellular infiltrate. By treating with the XL784 agent (Figure 4), the apparent degree of elastin damage and inflammatory cell inflammation inversely correlates with the dose of drug administered.
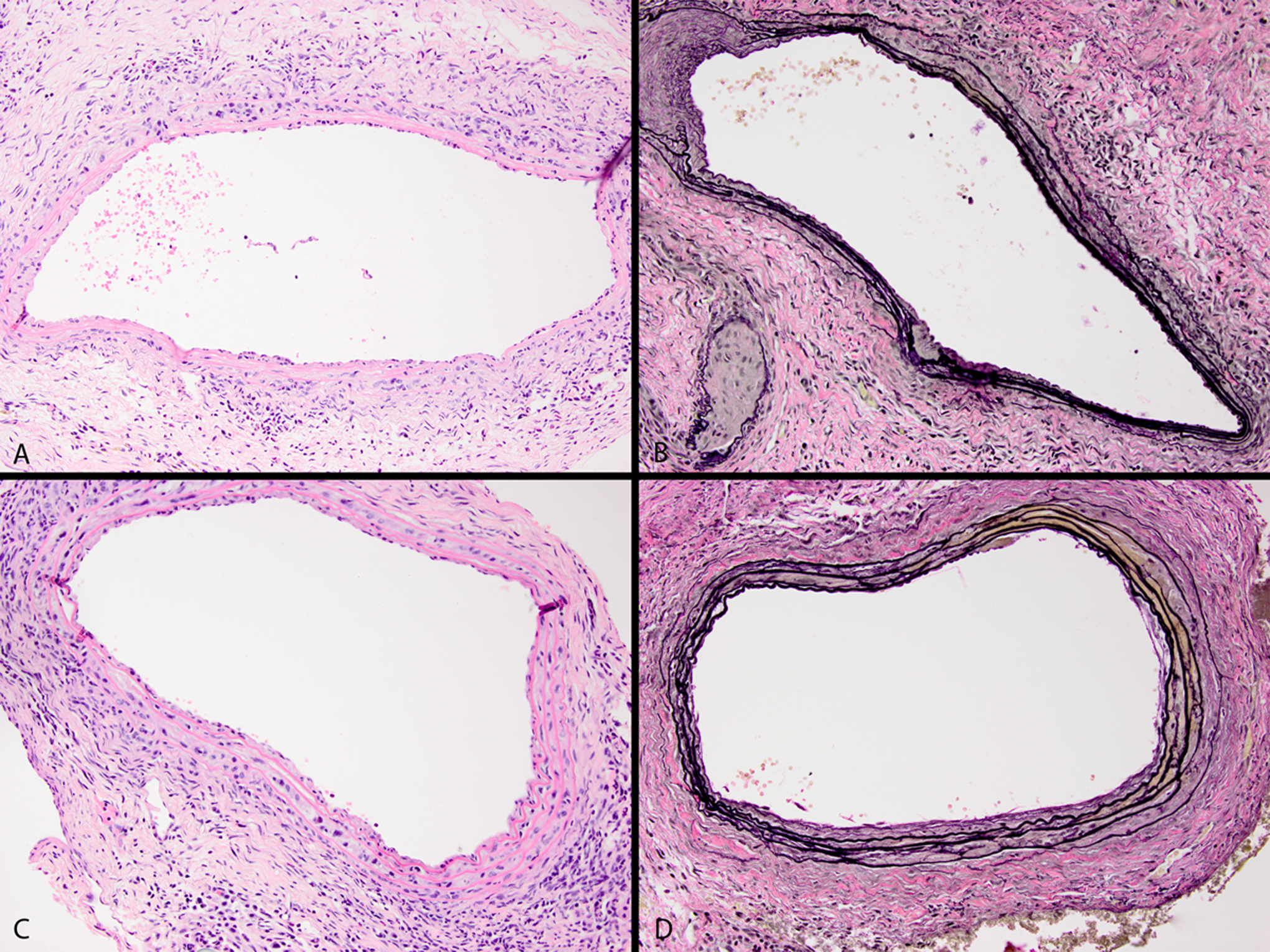
Aortic histology of control (panels A and B) and doxycycline-treated aortas (panels C and D) at ×60 objective magnification 14 days following isolated aortic perfusion with elastase. Panels A and C are sections stained with hematoylin and eosin. B and D, Sections of aortas stained with the elastin-specific VVG stain. The aorta from the control mouse shows extensive inflammation and appreciable thinning and fragmentation of the elastic fibers in the media. The doxycycline-treated sections of the elastic fibers are reasonably well preserved, although there is still a moderate amount of adventitial inflammation.

Aortic histology of mice of 14 days following brief elastase perfusion to initiate aneurysm development (×60 objective magnification). Animals were treated with XL784 at 50 mg/kg (panels A and B), 125 mg/kg (panels C and D), and 250 mg/kg (panels E and F) imaged at ×60 objective magnification 14 days following isolated aortic perfusion with elastase. Panels A, C, and E are sections stained with hematoylin and eosin. (B, D, and F) Sections of aortas stained with the elastin-specific VVG stain.
Semiquantitative analysis of the elastin and inflammatory cell content in aortic sections by blinded histologic evaluation on a scale from 0 to 4 confirms the qualitative impression (Figure 5). At the lowest dose of XL784, there was significantly reduced elastin content and significantly greater aortic inflammation than all other groups. Treatment of the mice with both the intermediate and highest dose of the XL784 agent resulted in aortic elastin content comparable to that of doxycycline treatment. The highest dose of the agent suppressed the inflammatory infiltration similar to that of doxycycline, as well. Interestingly, at the doses tested, the aortas never completely regained the histology of a normal aorta and had significantly less elastin and greater inflammation.

Semiquantitative analysis of representative histology sections from aortas of mice not perfused with elastase as well as elastase-perfused mice from both experiment 1 and experiment 2 treated with daily XL784 (50, 125, 250, and 500 mg/kg), doxycycline (100 mg/kg in drinking water or via daily gavage), or vehicle control. The quantity of elastin and inflammation was rated on a 0 (absent) to 4 (maximal) scale by 6 independent reviewers blinded to the treatment group. All reviewers evaluated the same 3 serial sections from 3 animals in each group. Aortas from both doxycycline-treated groups and the XL784-treated group at 500 mg/kg per d showed significantly more intact elastic fiber by histology than the control vehicle treatment group and were not statistically different than unperfused, normal aorta. However, all groups had significantly more inflammation than normal aorta, and none were significantly different from the inflammation seen in the vehicle-treated group.
Plasma Levels
After 3 doses of the drug, the mean peak plasma levels of XL784 ranged from 245 ± 17 ng/mL in animals treated with 50 mg/kg per d to 10 458 ± 1795 ng/mL in animals treated with 500 mg/kg per d. The corresponding trough levels were 2.6 ± 0.1 ng/mL (50 mg/kg per d) to 34.3 ± 4.2 ng/mL (500 mg/kg per d). In animals treated for 14 days with 500 mg/kg per d dose of XL784 (Figure 6), we found peak and trough levels which were not statistically different than after 3 days of treatment (peak: 7066 ± 952 ng/mL, trough: 28.2 ± 2.5 ng/mL).

Circulating levels of XL784 in mice sacrificed after 3 and 14 days of treatment with daily gavage of XL784 500 mg/kg. Peak levels (1 hour after gavage) are shown in A and trough levels (24 hours after gavage) are shown in B. There were no significant differences in either the peak or trough levels at 3 days versus 14 days of treatment.
Discussion
Pharmacologic therapy for AAAs holds great promise to revolutionize the management of this stealthy but deadly disease. The disease affects the elderly individuals and can be easily detected long before the wall strength has been appreciably reduced. These small aneurysms are a negligible clinical concern since the risk of rupture is low. Unfortunately, the natural history of an untreated small aortic aneurysm is one of progressive growth and decreased wall strength, leading to a substantial risk of rupture. A pharmacologic means of stabilizing the aorta at these small sizes would potentially obviate the need for surgical/anatomic exclusion of these aneurysms; currently, the only proven means to prevent rupture.
The pathophysiology of AAA is believed to be related to loss of medial elastin fibers. 8 , 9 Multiple studies have shown that elastolytic enzymes of the MMP family are likely critical mediators of the process which results in the dilatation of the aorta. Metalloproteinase activity has been consistently found in nearly every model to be associated with the aortic dilation. 6,10–12 These enzymes are appreciably elevated in human aortic aneurysm tissue. 13–16 One of the first enzymes of this class to be convincingly associated with aortic aneurysm disease was MMP-2. 14 Circulating MMP-9 levels have been shown to correlate with the size of the aneurysm and there is evidence that increasing levels of plasma MMP-9 may indicate a more rapidly progressive aneurysm. 17,18
There have been extensive studies of MMP inhibition in animal models of AAA. Several different broad-spectrum synthetic MMP inhibitors, including BB-94, 19 RS-113456, 20 RS132908, 21 and Marimastat, 22 have shown consistent success in preventing AAA progression. However, because of clinical side effects of these early novel synthetic agents, they have not been translated to clinical use. Considerable focus has also been paid to the use of the antibiotics in the tetracycline class. This class of drugs is recognized to inhibit the activity of many metalloproteinases through the chelation of Zn2+ necessary for the enzymatic activity of the proteins, with doxycycline having the greatest MMP inhibitory effect. 23 Early studies by Golub et al have shown that the MMP inhibitory properties of the tetracycline antibiotics is independent of their antibiotic activity. 24 Animal studies have consistently shown effective inhibition of AAA with doxycycline and non-antibiotic chemically modified tetracycline derivatives. 5,12,25–27
Ultimately the goal of medical therapy for a patient with an aortic aneurysm is prevention of aortic rupture without the need for surgical intervention. Clinical translation of any MMP inhibitor will, at a minimum, need to prevent the further dilatation of an established aneurysm. It is likely that chronic therapy to suppress the matrix destroying protease activity will be necessary. Any potential agent will need to have few side-effects (in an elderly patient population) to maximize compliance and therefore efficacy. Therefore, a narrow specificity of action is likely to be advantageous.
Doxycycline has been one of the first MMP inhibitors to be considered for translation to AAA therapy. In essentially all animal studies, oral doxycycline is administered in the drinking water with dosing based on the estimates of drinking habits. Serum steady state levels of doxycycline (100 mg twice daily [bid]) in humans appears to be intermediate between serum levels of 50 mg/kg per d and 100 mg/kg per d in mice. 5 Our finding of significantly enhanced AAA inhibitory activity of doxycycline administered via gavage compared to in drinking water suggests that treatment in drinking water may overestimate the dose actually delivered to mice.
Some early translational studies have shown evidence of AAA growth inhibition with doxycycline, 28–31 but all of the published studies are underpowered to determine the clinical utility of this treatment and additional clinical studies are planned. 32 While doxycycline is considered a potential therapeutic option, there are limitations to doxycycline therapy that may limit its usefulness in the population. The drug is well recognized to cause skin photosensitivity, 33 photo-oncholysis, 34 gastrointestinal discomfort, altered drug metabolism, 35 decreased seizure threshold, 36 altered bacterial flora, 37 and tooth discoloration 38 , 39 as side-effects. In a 6-month study of doxycycline (100 mg orally [po] bid) treatment of patients with AAA, it was found that reasonably good compliance of 92% could be achieved despite significant treatment-related effects in 14%. 32 However, the number of patients with side-effects thought to be related to treatment was 61% throughout the trial. Only 2 patients developed infectious complications during this trial, but the antibiotic properties of doxycycline in this patient population is unnecessary. Overall, the tolerability of this drug for chronic use at this dose has not been determined, and higher doses may be needed to obtain optimum effectiveness based on animal studies.
The studies reported here demonstrate that XL784 as a novel, narrow-spectrum MMP inhibitor is able to successfully inhibit model AAA in a dose-dependent fashion. Treatment with XL784 appears to remarkably inhibit the loss of elastic fibers and the aortic dilatation but has a relatively mild reduction of aortic inflammation in this model. The finding that a narrow profile MMP inhibitor with a high degree of specificity for MMP-2 can also effectively inhibit aneurysms in an animal model is a unique finding. This is important since prior broad-spectrum MMP inhibition has been limited by the occurrence of the musculoskeletal syndrome. 40
While full pharmacodynamic studies were not performed, the peak plasma levels of the XL784 achieved in these mice were also achieved without clinical side effects at lower than the maximal doses evaluated in phase 1 studies in humans. Further the drug half-life of 3 hours in the mice is appreciably shorter than the approximately 7 to 9 hours seen in human studies. Therefore, the potential to achieve clinical aneurysm inhibition with this novel therapeutic appears feasible.
This drug has also undergone prior phase II clinical evaluation in diabetic patients with renal disease at a dose of 200 mg/d in over 60 participants for 12 weeks. There were no differences in the occurrence of adverse events between the XL784-treated participants and placebo-treated participants. There was no discontinuation of the treatment due to musculoskeletal syndrome. Overall, the drug appeared well tolerated by this patient group with moderately severe underlying medical disease after 12 weeks of therapy.
Whether XL784 would ultimately be effective for clinical use to prevent the growth of small AAA depends on several factors, including its longer term side effect profile and, of course, its effectiveness in preventing progressive dilatation of an established aneurysm. The relatively specific mechanism of action of XL784 on the elastolytic proteases may impart a lower risk of undesirable side-effects and thus improve its tolerability for chronic use, as has been suggested by shorter term studies.
Conclusion
Specific and well-tolerated therapeutic options are needed to prevent the growth and rupture of small AAAs. We have shown that a novel metalloproteinase inhibitor with relative specificity for MMP-2 is at least as effective as a full-spectrum MMP inhibitor in preventing the growth of a mouse model of aortic aneurysm disease. Further studies of this agent should be undertaken to determine its potential for clinical use in AAA therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by a grant from Renavance Pharmaceuticals.
