Abstract
Spinal cord injury leads to disruption in autonomic control resulting in cardiovascular, bowel, and lower urinary tract dysfunctions, all of which significantly reduce health-related quality of life. Although spinal cord stimulation shows promise for promoting autonomic recovery, the underlying mechanisms are unclear. Based on current preclinical and clinical evidence, this narrative review provides the most plausible mechanisms underlying the effects of spinal cord stimulation for autonomic recovery, including activation of the somatoautonomic reflex and induction of neuroplastic changes in the spinal cord. Areas where evidence is limited are highlighted in an effort to guide the scientific community to further explore these mechanisms and advance the clinical translation of spinal cord stimulation for autonomic recovery.
Keywords
Introduction
Spinal cord injury (SCI) disrupts crucial communication between the spinal cord circuits and supraspinal control centers, resulting in a broad spectrum of sensorimotor and autonomic impairments. Among individuals with SCI, restoration of autonomic function is a high priority for recovery (Anderson 2004; Simpson and others 2012). This is not surprising given the deterioration in health-related quality of life, dignity, autonomy, and increased mortality due to autonomic dysfunctions (Wheeler and others 2018). Until recently, the majority of research focused on only “visible” sensorimotor dysfunction (e.g., motor paralysis). Despite the unmet needs of individuals with SCI, these “invisible” disabilities—such as cardiovascular, bowel, and lower urinary tract (LUT) dysfunctions—remain relatively underprioritized in research and clinical practice. While there are currently limited effective treatments for mitigating these dysfunctions (Krassioukov and others 2021), spinal cord stimulation (SCS) is emerging as a leading approach for restoring autonomic function following SCI.
SCS is a neuromodulation approach where electrical current is applied over the spinal cord to promote functional recovery in the injured nervous system. Originally developed for pain management by leveraging the gate control theory of pain (Melzack and Wall 1965), potential applications of SCS have increased exponentially in recent years. Aside from pain management, there is a growing body of evidence suggesting that SCS aids in the recovery of the following functions: cardiovascular (Harkema and others 2018; Phillips and others 2018; Sachdeva and others 2021; West and others 2018), bowel (DiMarco and others 2021; Kreydin and others 2022; Walter and others 2018), and LUT (Gad and others 2018; Herrity and others 2018; Herrity and others 2022). This review aims to explore and summarize plausible mechanisms underlying the effect of SCS in restoring these autonomic functions following SCI. We developed a literature search strategy with selected keywords related to our topic of interest (e.g., SCI, epidural SCS [eSCS], transcutaneous SCS [tSCS], autonomic dysfunction) in MEDLINE, Embase, Scopus, and CHINAHL. In addition to the search, we added relevant important studies in the field. On the basis of the selected literature, we discuss two overarching mechanisms of SCS underlying autonomic recovery: activation of somatoautonomic reflexes and facilitation of neuroplasticity of spinal autonomic pathways. We include preclinical and clinical evidence on cardiovascular, bowel, and LUT functions.
Activation of Somatoautonomic Reflexes
Somatoautonomic reflex activation is one of the most plausible mechanisms for how SCS affects autonomic functions. Somatoautonomic reflex is defined as a reflex induced by sensory afferent inputs, such as cutaneous stimulation, manifesting as an alteration of autonomic nervous system function (Sato and others 1997). The physiologic phenomenon of visceral responses to cutaneous stimulation is well established. The blood vessels, gastrointestinal tract, and LUT all respond to tactile and thermal stimulation of the skin. The somatoautonomic reflex arc also receives central regulation from supraspinal structures such as the rostral ventrolateral medulla and Barrington nucleus (Lebedev and others 1984). Similar to the effect of cutaneous stimulation, electrical stimulation via SCS can activate the somatoautonomic reflex arc. Herein, we describe potential mechanisms involved in activating the somatoautonomic reflex arc via SCS, including activation of primary afferent fibers, spinal interneurons, and autonomic efferent pathways that result in targeted responses in visceral organs (Figure 1).
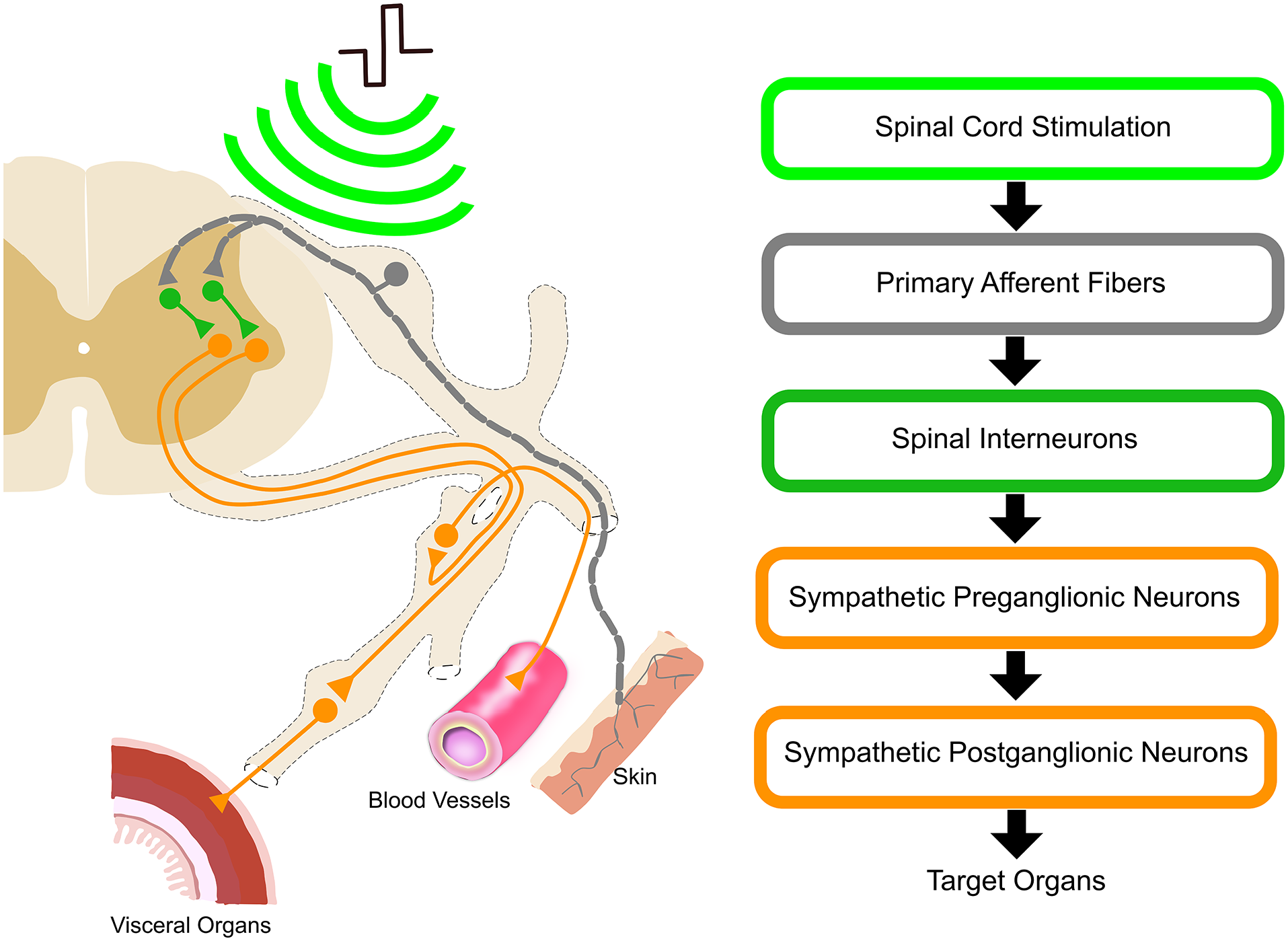
Somatoautonomic reflex evoked by SCS. The somatoautonomic reflex is initiated by SCS of the dorsal roots (light green) activating primary afferent fibers (gray). The inputs from the primary afferent fibers elicit the activation of spinal interneurons (dark green), leading to the activation of autonomic efferent pathways (orange), which consist of sympathetic pre- and postganglionic neurons. In turn, the efferent neurons induce activation of the visceral organs, skin, and blood vessels. SCS, spinal cord stimulation.
The first step in the somatoautonomic reflex is the activation of myelinated afferent fibers. eSCS and tSCS likely deliver electrical current to the dorsal roots (Ladenbauer and others 2010). Yet, there is still debate on whether SCS can directly activate interneurons in the dorsal horn without the activation of primary afferent fibers (Lee and others 2020; Rogers and others 2022). eSCS and tSCS have demonstrated similar electromyography (EMG) patterns in the same individuals with SCI (Hofstoetter and others 2018). However, these differ in the proportion of electrical current reaching the dorsal roots (Ladenbauer and others 2010) and in the spatial selectivity of the activated spinal segments (Capogrosso and others 2013; Wagner and others 2018).
The recruitment order of dorsal root nerve fibers by SCS follows two rules (Grill and Mortimer 1995). First, large-diameter afferent fibers are preferentially activated by SCS. Stimulation induces transmembrane potential changes in larger-diameter nerve fibers, with larger spacing between nodes of Ranvier, as compared with smaller-diameter nerve fibers. Second, as opposed to distant nerve fibers, nerve fibers in multiple dorsal roots, near electrodes, are preferentially activated by SCS. Furthermore, SCS activates multiroot responses that induce monosynaptic excitation with additional polysynaptic responses incorporated with spinal interneurons (Capogrosso and others 2013). This indicates that SCS activates multiple dorsal roots due to the expansion of an electrical field. A recent simulation study showed that eSCS electrical fields activated sympathetic preganglionic neurons (SPNs) via multiple dorsal roots, without direct activation of dorsal column, intraspinal, or dorsal horn neurons (Squair and others 2021). The dorsal rhizotomy led to elimination of the effect of eSCS on blood pressure (BP), indicating that eSCS relies on dorsal root activation to induce the somatoautonomic reflex (Squair and others 2021). Thus, eSCS most likely activates the primary afferent fibers in the multiple spinal segments near the stimulation electrode, leading to the functional changes via spinal cord neuromodulation.
The autonomic nervous system is segmentally organized. Different spinal segments constituting sympathetic and parasympathetic preganglionic neurons innervate different organs (Rabchevsky 2006). The activation of the dorsal afferents typically results in the activation of local spinal segments for specific organs in the intact spinal cord. Following the activation of primary afferent fibers, spinal interneurons play a key role in bridging these somatic afferent pathways and autonomic efferent pathways from SPNs (Schramm 2006). Interneurons relay sensory inputs to other interneurons, preganglionic neurons in laminae V and VII, and supraspinal centers. Signal transmission crosses the midline via commissural interneurons or via local interneurons in nearby and distant spinal segments through short and long propriospinal interneurons (Conta and Stelzner 2004). These propriospinal neurons synapse with preganglionic neurons in laminae VII and X.
Dorsal afferent inputs produce polysynaptic potentials in SPNs. Excitatory interneurons convey afferent information to preganglionic neurons through polysynaptic excitatory postsynaptic potentials (Deuchars and others 2005). Inhibitory interneurons in the dorsal horn are also activated by afferent inputs, thereby inhibiting noxious input signals. Specifically, GABA-ergic interneurons in the dorsal gray commissure presynaptically regulate SPNs by inhibiting excitatory interneurons (Deuchars and others 2005). Similarly, glycinergic interneurons evoke inhibitory postsynaptic potentials in SPNs (Dun and Mo 1989). Therefore, it has been suggested that dorsal afferent inputs activate interneurons in order to control autonomic efferent pathways (Gad and others 2018; Kreydin and others 2022; Squair and others 2021; West and others 2018). The activation of the somatoautonomic reflex potentially inhibits or excites sympathetic pre- and postganglionic neurons for each autonomic function, as discussed in the following sections: Spinal Cord Neuromodulation for Cardiovascular Function, Spinal Cord Neuromodulation for Gastrointestinal Function, and Spinal Cord Neuromodulation for Lower Urinary Tract Function.
Promoting Neuroplasticity of Spinal Autonomic Pathways
The other plausible overarching mechanism is that SCS facilitates neuroplastic changes within spinal autonomic circuits. Herein we review preclinical studies investigating neuroplastic changes of autonomic spinal circuits with long-term SCS, including myelination and regeneration.
First, SCS potentially promotes myelin preservation and remyelination around the lesion for autonomic recovery (Gris and others 2004). Following SCI, there is a limited capacity of spontaneous oligodendrocyte proliferation and differentiation from endogenous neural stem cells and progenitor cells for remyelination in the spinal cord (Horky and others 2006). In rats, eSCS over the lesion site, shortly after injury, has been shown to protect spinal myelin following SCI (Li and others 2020). This study also showed that myelin protection was potentially derived from increased endogenous oligodendrocyte progenitor cell differentiation into oligodendrocytes instead of astrocytes, by inhibiting the bone morphogenetic protein signaling pathway. In addition, transspinal SCS—a different form of SCS, with electrodes over the spinous processes of the lesion—induced myelin preservation that may result from less apoptosis, as indicated by decreased intracellular calcium marker expression (Tian and others 2016). Conversely, remyelination and inhibition of astrocyte differentiation may not be necessary for functional recovery (Duncan and others 2018). Further investigation is warranted to test the significance of SCS on remyelination associated with autonomic function.
Clinical evidence has shown sustained motor recovery after long-term SCS in individuals with SCI (Wagner and others 2018). One potential mechanism supporting the long-term recovery after SCS treatment is the promotion of axon sprouting and short-distance regeneration in cases of chronic SCI. By using a staggered hemisection rat SCI model, lumbosacral eSCS with locomotor training and monoamine receptor agonists significantly improved walking function (van den Brand and others 2012). Abolished corticospinal projections to the lumbosacral locomotor center were compensated by corticospinal tract branching, which crossed the midline around the lesion and recrossed to propriospinal neurons (van den Brand and others 2012). Following the same eSCS intervention, glutamatergic cortical neurons projected on ventral gigantocellular reticular nuclei and reticulospinal pathways carried cortical commands across the lesion (Asboth and others 2018).
Finally, SCS may improve afferent fiber function by reversing maladaptive sprouting. Following SCI, the aberrant sprouting with CGRP-dominant fibers (calcitonin gene–related peptide) increases due to receptivity to afferent inputs (Krenz and Weaver 1998), which can cause autonomic dysfunctions (Krassioukov and others 2002). Only one study showed that CGRP was downregulated after tSCS at the S1 vertebral level for LUT function in a rat SCI model (Elkelini and others 2012). Additional research is required to reveal the effect of SCS on afferent fiber plasticity for autonomic recovery.
Spinal Cord Neuromodulation for Cardiovascular Function
The heart receives sympathetic innervation from the upper thoracic spinal cord (T1-T5) and parasympathetic innervation via the vagus nerve from the medulla. The neural control of blood vessels is continuously modulated via the sympathetic efferent pathway from the thoracolumbar spinal cord. Specifically, sympathetic outflow from T1-T5 segments regulates upper body blood vessels, while T6-L2 segments control lower body blood vessels. Blood vessels throughout the body receive no parasympathetic innervation (Krassioukov and Weaver 1996). The splanchnic bed, controlled via sympathetic innervation (T6-L2), plays a significant role in controlling systemic BP, especially in rapid response to positional changes. Continuous sympathetic control of peripheral vasculature is mediated by supraspinal control from C1 catecholaminergic neurons in the rostral ventrolateral medulla (Brown and Guyenet 1985). Central baroreceptors respond to reductions in vessel stretch to control the vagal and sympathetic outflow.
Following SCI at T6 or above, loss of supraspinal inputs from the rostral ventrolateral medulla causes impaired sympathetic vasomotor control in the splanchnic bed and lower extremity blood vessels. Although the baroreceptor function and parasympathetic control of the heart remain anatomically intact, the impaired cardiovascular regulation leads to a range of serious complications after SCI due to disrupted supraspinal autonomic control. These challenges include low resting BP, significant BP drops when moving to upright positions defined as orthostatic hypotension (OH), and episodes of uncontrolled hypertension termed autonomic dysreflexia (AD; Guttmann and Whitteridge 1947). These hypo- and hypertensive events result from insufficient and excessive vasoconstriction, respectively. Recent preclinical and clinical studies suggest that real-time SCS alleviates OH via activation of the sympathetic efferent pathway (Supplemental Table S1) and mitigates AD via inhibitory mechanisms for visceral and/or noxious afferent inputs (Supplemental Table S2).
Activation of Sympathetic Spinal Cord Circuits and Mitigation of Orthostatic Hypotension
A preclinical study demonstrated that eSCS could prevent OH by activating somatoautonomic reflexes (Squair and others 2021). Transsynaptic retrograde tracing revealed the existence of glutamatergic spinal interneurons synapsing with primary afferent neurons and splanchnic sympathetic ganglia (Squair and others 2021). In the sympathetic efferent pathway, SPNs play a key role in the activation of sympathetic postganglionic neurons modulating responses of target organs. The density of SPNs labeled by retrograde traces from the splanchnic sympathetic ganglia, responsible for BP control, reached maximum in T11-T13 spinal segments following a Gaussian distribution (Squair and others 2021). The mapping of the pressor response to eSCS showed the highest activation via eSCS at T11-T13 spinal segments, which was highly correlated to the density of SPNs in rat contusion SCI models. eSCS at the T12 spinal segment also induced cFOS expression in tyrosine hydroxylase–positive neurons in the superior mesenteric ganglia in a rat T3 SCI model. When these neurons were silenced via optogenetic inhibition with the inhibitory opsin eNpHr3.0, the pressor response to eSCS was eliminated. This supports the connections between afferent neurons and SPNs synapsing on the superior mesenteric ganglia (Squair and others 2021). Yet, there is no direct evidence that SCS induced activation of excitatory interneurons synapsing on SPNs.
Clinical studies support the effect of SCS for increasing resting BP and improving orthostatic tolerance following SCI. Midthoracic tSCS induced acute BP pressor responses at rest and during an orthostatic challenge in individuals with cervical or upper thoracic SCI (Phillips and others 2018). Similarly, lumbosacral eSCS prevented OH during an orthostatic challenge (Aslan and others 2018; Darrow and others 2019; Harkema and others 2018; West and others 2018). A recent case report presented that stimulating the T10-T11 spinal cord altered the BP pressor responses (Squair and others 2021). eSCS at T10-T11 spinal segments was more effective for elevating systolic BP than eSCS at T12-L1 spinal segments in an individual with complete cervical SCI. The authors found increased plasma norepinephrine and peroneal nerve muscle sympathetic nerve activity during eSCS.
In summary, while the evidence is limited to one preclinical study and five clinical studies, there is some evidence supporting that SCS activates the somatoautonomic reflex, resulting in increased sympathetic outflow to mitigate OH (Figure 2A). It has been suggested that SCS excites the sympathetic efferent pathway consisting of SPNs and sympathetic postganglionic neurons (Figure 2A, sympathetic efferent pathways in orange; Aslan and others 2018; Harkema and others 2018; Phillips and others 2018) potentially through glutamatergic interneurons (Figure 2A, excitatory interneurons in green; Squair and others 2021). It is still important to validate this potential mechanism by testing sympathoexcitatory neurons, including excitatory interneurons, and SPNs via advanced methods, such as imaging techniques, chemogenic and optogenetic approaches, and single-nucleus RNA sequencing analysis.
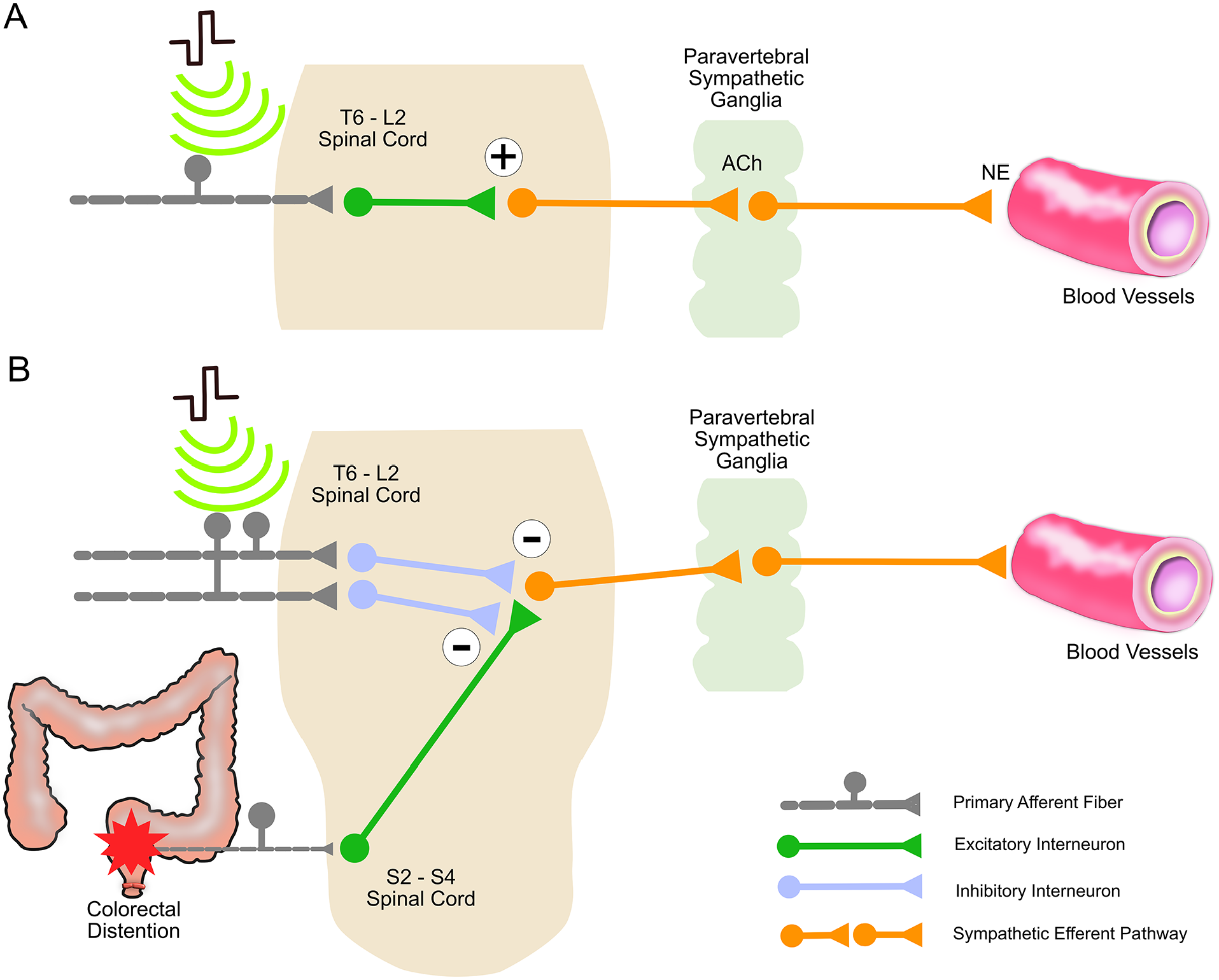
Putative mechanisms of spinal cord stimulation to improve cardiovascular function following spinal cord injury. (A) Mechanism of mitigating orthostatic hypotension in the injured spinal cord at or above T6. The activation of primary afferent fibers (gray) is followed by the activation of excitatory interneurons (green), resulting in the excitation of sympathetic efferent pathways (orange) and leading to the release of norepinephrine. Norepinephrine induces vasoconstriction of splanchnic and lower limb blood vessels. (B) Mechanism of mitigating autonomic dysreflexia during visceral stimuli in the injured spinal cord at or above T6. The primary afferent inputs (gray) activate inhibitory interneurons (blue), inhibiting visceral stimuli and activating sympathetic efferent pathways (orange) to prevent vasoconstriction related to colonic visceral inputs.
Inhibition of Visceral/Noxious Sensory Transmission and Mitigation of Autonomic Dysreflexia
One preclinical study based on tSCS at T12-S3 vertebral levels showed mitigation of AD during colorectal distension in a rat model with C7-T1 and T4-T5 transection SCIs (Collins and DiCarlo 2002). Nineteen years later, we tested real-time tSCS at higher spinal levels, the T7 vertebral level, and showed the prevention of AD during colorectal distension in a rat model with T3 transection SCI (Sachdeva and others 2021). We also expanded the study to assess the long-term effect of tSCS in the same preclinical model, resulting in the sustained mitigation of AD, even without active tSCS.
Clinical work demonstrated the potential inhibitory effects of SCS on mitigating AD. An early case series reported the sustained effect of percutaneous eSCS at the lumbosacral spinal segments on regulating BP and reducing the frequency of uncontrollable hypertensive episodes in four of five individuals with thoracic and lumbar SCI (Richardson and others 1979). Forty-two years after this report, we presented a case study using noninvasive tSCS at T7-T8 spinal segments to prevent AD during digital anorectal stimulation in one individual with motor and sensory complete cervical SCI (Sachdeva and others 2021). Although there are considerable differences between these studies, including injury levels, types of SCS, stimulation sites, and outcome measurements, both clinical reports demonstrated that SCS potentially prevents hypertensive episodes in individuals with SCI.
These preclinical and clinical studies suggest a potential inhibitory mechanism of SCS for mitigating AD. First, SCS may reduce sympathetic outflow by inhibiting uncontrolled BP elevation (Collins and DiCarlo 2002; Sachdeva and others 2021). Second, SCS likely inhibits the sensory transmission to spinal neurons and SPNs. The gate control theory (Melzack and Wall 1965) explains the potential mechanism as follows: primary afferent inputs activate inhibitory interneurons to close the gate to the visceral and/or noxious afferent inputs of the small diameter fibers at the spinal cord level. There is evidence on the effect of SCS on increased GABA (Cui and others 1997) and glycine (Simpson and others 1993), potentially indicating the activation of inhibitory neurons in the spinal cord. The inhibition of the sympathetic outflow could result from blocking the spinal transmissions of visceral/noxious afferent impulses in the primary afferent neurons or from pre- and postsynaptic inhibition of SPNs via spinal interneurons (Figure 2B, spinal interneurons in blue).
Spinal Cord Neuromodulation for Gastrointestinal Function
Gastrointestinal function is controlled by a complex neural network. The majority of sympathetic circuits innervating the entire gastrointestinal tract arise from lower thoracic to upper lumbar segments (Browning and Travagli 2014). Parasympathetic innervations for the lower esophagus, stomach, small intestine, and proximal colon are from the vagus nerve, which is unaffected following SCI. Parasympathetic innervations for lower gastrointestinal tracts arise from the S2-S4 spinal segments. In addition, the enteric nervous system, located in the submucosa (Meissner plexus) and between the longitudinal and circular muscular layers (Auerbach plexus), directly controls peristalsis, mucosal secretion, and blood flow of the gut. Sympathetic and parasympathetic nerves indirectly innervate the smooth muscle cells through the enteric nervous system, which makes autonomic control of gastrointestinal function distinct from other organs and outputs from somatic inputs or SCS more difficult to predict.
The pudendal nerve (S2-S4) provides somatic control for the external anal sphincter. The internal anal sphincter is under reflex control from the enteric nervous system and the sacral spinal cord (S2-S4). Gastric, small intestinal, and colonic motility and anorectal contractions of nonsphincteric regions are reduced by the sympathetic nervous system and promoted by parasympathetic activation. Defecation is controlled by a number of reflexes: the myenteric defecation reflex; the parasympathetic defecation reflex, initiated by gastrointestinal stretch and distension; the rectoanal inhibitory reflex, within the enteric nervous system; and supraspinal inputs (Hou and Rabchevsky 2014). These autonomic and somatic neural circuits coordinate colonic transit, continence, and defecation.
Following SCI, parasympathetic supraspinal control of the proximal and middle gastrointestinal tract (via the vagus nerve) remains intact, while autonomic control of the distal colon is impaired due to loss of supraspinal control (Hou and Rabchevsky 2014). Prolonged gastric emptying time, delayed colonic transit time, and increased time needed for bowel management are common consequences of the disrupted autonomic control of the bowel following SCI (Krogh and others 2000). Impaired colonic motility and anal sphincter function after SCI result in constipation and fecal incontinence. Development of anal sphincter dyssynergia and impaired rectoanal reflexes also compromise voluntary defecation. Recent evidence suggests that SCS may increase anorectal sphincter pressure (Kreydin and others 2022) and shorten bowel management time (DiMarco and others 2021; Walter and others 2018) in individuals with SCI (Supplemental Table S3).
In preclinical SCI models, lumbosacral eSCS inhibited anorectal contractions, measured by anorectal manometry at 2 and 10 cm from the anal verge, but increased external anal sphincter EMG activity (Hoey and others 2021). Thoracolumbar eSCS also reduced rectal contractions at 2 cm from the anal verge while facilitating distal colon activity at 10 cm from the anal verge (Hoey and others 2022). These preclinical findings suggest that eSCS potentially promotes colonic motility and stool continence in rat SCI models. These studies indicate that SCS may activate autonomic activity and the enteric nervous system for colorectal function. However, there is no direct evidence of the association of SCS with the activation of autonomic efferent pathways for gastrointestinal function.
Furthermore, some clinical evidence supports the potential autonomic effects of SCS on bowel function after SCI. Two studies showed that real-time thoracolumbar eSCS decreased time for bowel management in individuals with complete SCI (DiMarco and others 2021; Walter and others 2018). Real-time lumbosacral tSCS also demonstrated decreased bowel management time and increased anorectal pressure in three individuals with SCI (Kreydin and others 2022). The strongest anorectal pressure responses to tSCS were at the level of the anal verge and puborectalis, indicating the somatic effects via the pudendal nerve in two participants with complete SCI. tSCS additionally induced the strongest anorectal pressure responses at 6 cm from the anal verge in one female participant with complete SCI.
Based on these preclinical and clinical data, the following potential mechanisms were proposed for the effect of SCS on gastrointestinal function. First, lumbosacral SCS may lead to activation of somatic circuits to promote contractions in the external anal sphincter and pelvic floor muscles (Hoey and others 2022; Kreydin and others 2022), which in turn could assist in stool continence (Figure 3, somatic pathway in blue). Second, SCS may lead to changes of distal colonic motility, potentially through the autonomic spinal circuits (Figure 3, sympathetic and parasympathetic efferent pathways in orange and purple, respectively) and the enteric nervous system (DiMarco and others 2021; Kreydin and others 2022; Walter and others 2018). Current evidence, however, cannot differentiate between whether SCS induces somatic activation of striated muscles or triggers autonomic activation (Hoey and others 2022; Kreydin and others 2022). To advance the understanding of SCS effects, the following should be tested in individuals with SCI before and after SCS: colonic transit time, colonic coordination for peristalsis, rectal contractions, rectal compliance, and anorectal resting and squeezing pressure. Last, abdominal muscle contractions during SCS may improve bowel function. Despite speculation, one study suggested the involvement of increased intra-abdominal pressure, which was based on the observation of increased airway pressure generated during eSCS (DiMarco and others 2021).
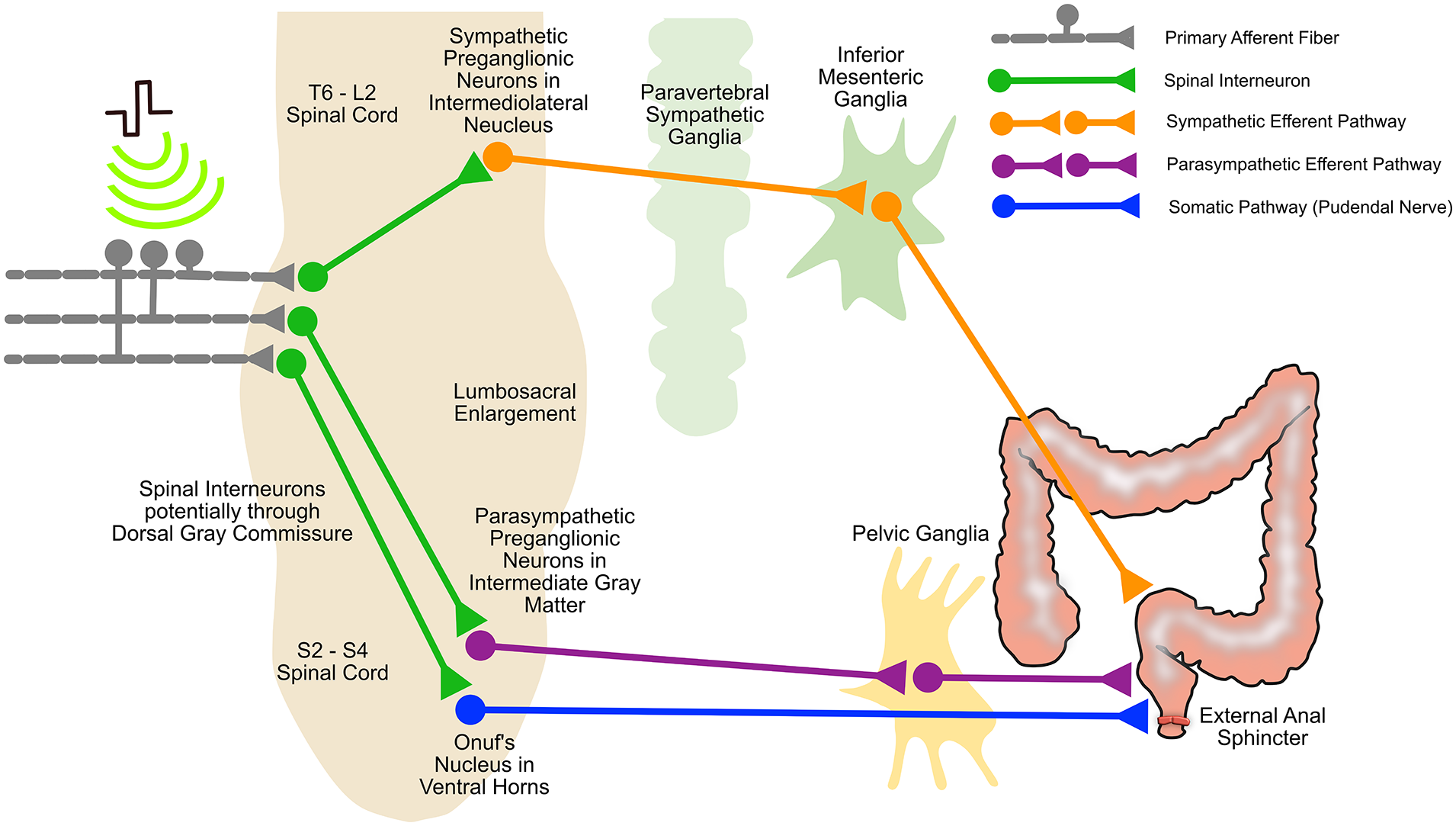
Putative mechanisms of SCS for gastrointestinal function. SCS activates primary afferents resulting in activation of spinal interneurons at the level of stimulation. Subsequent activation of somatic circuits involved in external anal sphincter control occurs through efferent output via pudendal nerve (blue). The activated spinal interneurons excite 1) the sympathetic efferent pathway to the internal anal sphincter for stool continence (orange), and 2) the parasympathetic efferent pathway, for colonic motility (purple), thereby affecting the enteric nervous system in the gut. SCS, spinal cord stimulation.
Spinal Cord Neuromodulation for Lower Urinary Tract Function
The coordinated activity of the bladder and urethra maintains LUT function, including low-pressure filling and voluntary voiding. The reciprocal activity is orchestrated by cortical and subcortical centers (i.e., Barrington nucleus) and spinal control distributed across autonomic and somatic pathways (Yoshimura and de Groat 1997). The detrusor and internal urethral sphincter receive sympathetic control from T10-L2 spinal segments and parasympathetic control from S2-S4 spinal segments. The sympathetic nervous system controls relaxation of the detrusor and contraction of the urethra for storage of urine. The parasympathetic nervous system induces contraction of the detrusor and relaxation of the urethra for micturition. The striated muscles of the urethra, composed of the external urethral sphincter (EUS), receives somatic control via the pudendal nerve (i.e., axons of Onuf nucleus at S2-S4). Voiding is under voluntary control based on afferent information from the bladder. Voiding is initiated by parasympathetic outflow and voluntary relaxation of EUS.
Following SCI, loss of bulbospinal control results in the disrupted central control of spinal reflexes, leading to an overactive bladder body and neck. The impaired spinal reflexes cause the uncoordinated activation of autonomic and somatic neurons, termed detrusor sphincter dyssynergia (de Groat and Yoshimura 2006). The disrupted ascending pathways impair the awareness of sensation from the bladder, such as the need for voiding. Detrusor overactivity causes incontinence, whereas increased urethral muscle tone and the inability to initiate voiding impair urine elimination. These dysfunctions lead to significant postvoid residual volume, high intravesical pressure, and vesicoureteral-renal reflux. Consequently, impaired LUT function increases the incidence of urinary tract infections and impaired renal function, such as hydronephrosis and kidney failure (Panicker and others 2015). Excitingly, recent studies report that SCS potentially mitigates LUT dysfunction after SCI (Supplemental Table S4).
Preclinical studies demonstrate that SCS promoted control of the detrusor and EUS for LUT function. In rat models of T8 transection SCI, thoracolumbar eSCS induced increased detrusor pressure as measured by cystometry (Hoey and others 2022). In the same rat models, thoracolumbar eSCS simultaneously inhibited EUS EMG activity, thereby promoting voiding function (Abud and others 2015; Sysoev and others 2020). Conversely, lumbosacral eSCS evoked tonic EUS EMG activity facilitating storage of urine in rat models of T9 transection SCI (Hoey and others 2021). Last, lumbar tSCS improved storage of urine and voiding function in mouse models of T13 contusion SCI (Ahmed 2017).
Several clinical studies demonstrated the effects of SCS on the autonomic and somatic control of LUT function. In an individual with cervical complete SCI, lower lumbar and sacral eSCS increased detrusor pressure and EUS/pelvic floor muscle EMG activity, but upper lumbar eSCS had no effect (Walter and others 2018). Another case series showed that lower lumbar and sacral eSCS promoted greater voiding efficiency when compared with upper lumbar eSCS in individuals with SCI even though the stimulation was not optimized for LUT function (Herrity and others 2018). More recently, a mapping study showed that eSCS at 60 to 85 Hz over the upper lumbar or midlumbar spinal segments, where sympathetic circuits for LUT function reside, promoted bladder compliance in four of five participants with motor-complete SCI (Herrity and others 2022). Yet, eSCS at 20 to 30 Hz over the lower lumbar and sacral spinal segments, where parasympathetic circuits reside, was the most efficient for initiating voiding in four of five participants (Herrity and others 2022). The locations and burst frequency of eSCS potentially changed the outputs of LUT function.
Meanwhile, in seven individuals with SCI, upper lumbar tSCS demonstrated activation of the detrusor with minimal urethral and abdominal muscle activations (Gad and others 2018). Upper lumbar tSCS at 1-Hz burst frequency promoted voiding efficiency for micturition. Interestingly, upper lumbar tSCS at 30-Hz burst frequency reduced detrusor overactivity and increased bladder compliance for storage in the same individuals (Gad and others 2018). In this study, the burst frequency of tSCS at the same electrode placements could modulate LUT function. In five individuals with cervical and thoracic SCI, tSCS applied to the entire lumbar and sacral regions at 30-Hz improved bladder capacity for storage and voiding efficiency for micturition, with less detrusor sphincter dyssynergia (Kreydin and others 2020).
The findings of these preclinical and clinical studies suggest the following potential mechanisms for the effect of SCS on LUT function. First, eSCS at lower lumbar and sacral spinal segments may lead to activation of the sacral somatic pathways to promote tonic contractions of sphincter and pelvic floor muscles for storage of urine (Figure 4, somatic pathway in blue; Gad and others 2018; Herrity and others 2022; Walter and others 2018). Second, eSCS at the upper and midlumbar spinal segments and tSCS at the upper lumbar spinal segments may also activate sympathetic efferent pathways to reduce detrusor overactivity (Ahmed 2017; Gad and others 2018; Herrity and others 2022; Kreydin and others 2020). SCS at the upper and midlumbar spinal segments potentially activates sympathetic efferent pathways to relax the detrusor (Figure 4, sympathetic efferent pathway in orange). Last, based on findings that eSCS at lower lumbar and sacral spinal segments and tSCS at upper lumbar spinal segments increased detrusor pressure and promoted voiding efficiency, SCS potentially leads to the activation of parasympathetic efferent pathways at S2-S4 for voiding (Gad and others 2018; Herrity and others 2022; Herrity and others 2018; Hoey and others 2022; Kreydin and others 2020; Walter and others 2018).
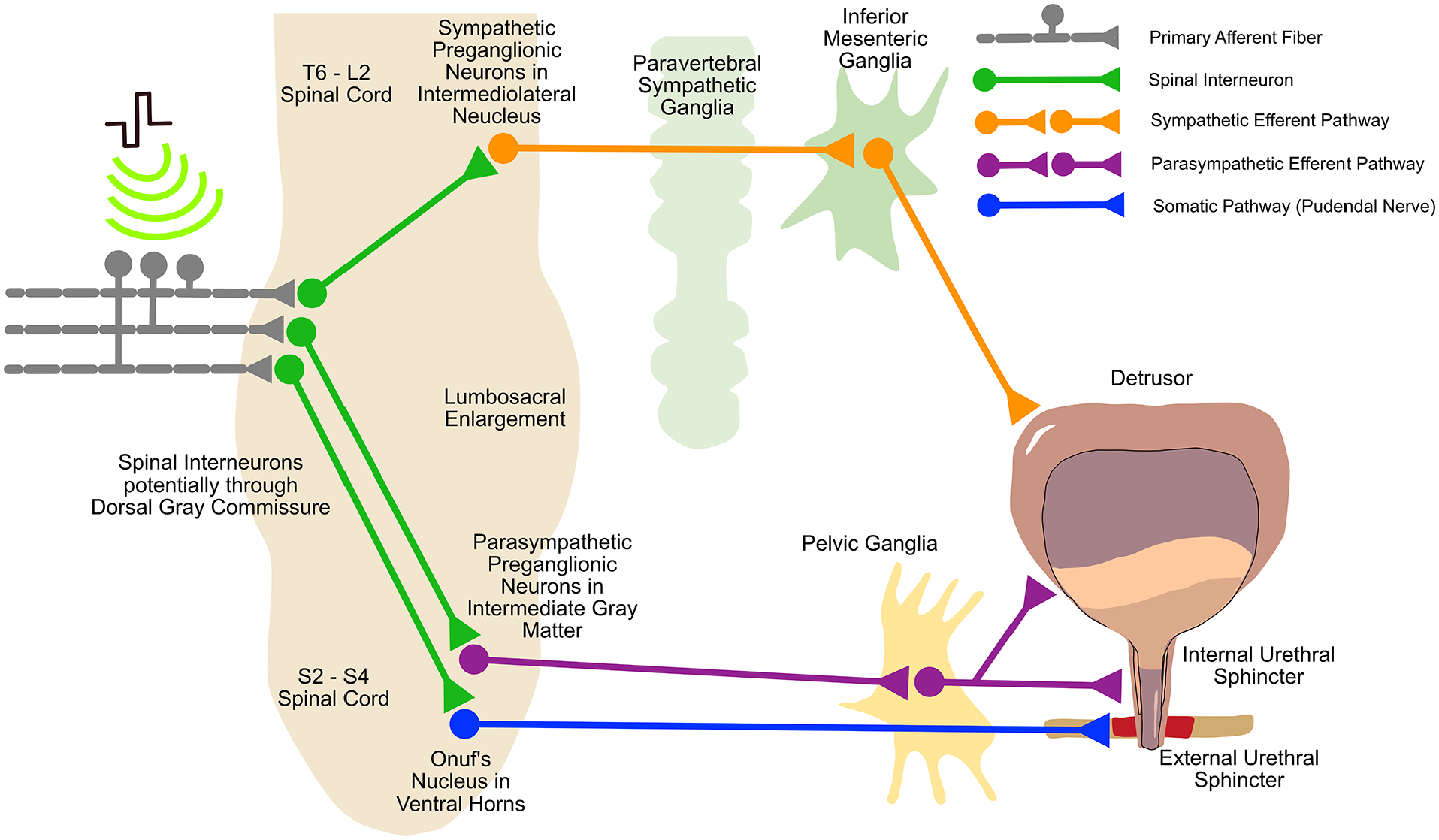
Putative mechanisms of SCS for lower urinary tract function. SCS activates primary afferents (gray), resulting in activation of spinal interneurons (green) at the level of stimulation. These result in activation of somatic circuits involved in external urethral sphincter control (efferent output via pudendal nerve; blue). The activated spinal interneurons also activate the sympathetic efferent pathway for storage of urine (orange) and the parasympathetic efferent pathway for urinary voiding (purple). SCS, spinal cord stimulation.
Despite the reported therapeutic effects of SCS on LUT function following SCI, some case reports suggest that SCS could adversely affect LUT function. Urinary incontinence could be caused by eSCS due to reduced bladder compliance (Beck and others 2021). Therefore, the effect of SCS on LUT function is still not fully understood. Last, there are no mechanistic studies reporting direct evidence on SCS-induced activation of spinal interneurons, preganglionic neurons, and postganglionic neurons associated with LUT function (illustrated in Figure 4).
Discussion
There is a growing body of evidence for the impact of SCS on recovery of autonomic dysfunctions. However, the mechanistic evidence underlying SCS-mediated functional changes in the autonomic spinal circuits is still limited. Thus, this review attempts to derive logical hypotheses based on currently available evidence and highlights the need for concrete mechanistic evidence to support these hypotheses. The current limitations of our understanding of the mechanism underlying SCS for autonomic recovery include a lack of direct evidence for 1) the activation of SPNs for mitigating OH by SCS and 2) the activation of inhibitory interneurons or the decrease of sensory transmission for inhibiting AD.
To fill the gap that was addressed in this review, we propose to employ noteworthy advanced scientific methods to dissect the mechanisms. For preclinical mechanistic studies, we can use neural activity markers such as cFOS protein expression, fast and targeted control of specific neurons such as calcium and photon imaging, and optogenetic strategies. It is possible to further investigate the downstream effect of SCS on the modulation of sympathetic activity following SCI through these methods. In clinical studies, we need detailed investigation for autonomic peripheral nerve activity via muscle sympathetic nerve activity recordings and plasma catecholamine concentration. Detailed bowel and bladder assessments—such as colonic motility, anorectal sphincter function, detrusor function with SCS via clinically approved wireless motility capsules, high-resolution anorectal manometry, and video urodynamics—can provide physiologic evidence on the underlying mechanisms of SCS. Based on the accumulation of clinical evidence, future clinical studies may demand a carefully randomized controlled clinical trial design including blinding, a targeted population, sufficient power, and consistent assessments with urodynamics. This approach will allow us to test how the real-time SCS and long-term SCS affect neural structures in chronic SCI, which will lead to the validation of potential mechanisms and the advancement of this promising intervention.
Conclusion
The primary benefits of SCS on autonomic functions likely occur via activation of somatoautonomic reflexes. Stimulation directly activates primary afferents and in turn potentially excites spinal cord interneurons and pre- and postganglionic autonomic neurons. Long-term SCS promotes neuroplastic changes in the spinal cord and afferent fibers. More evidence is needed to identify the underlying mechanisms of SCS for restoring vital but invisible autonomic functions. Ultimately, insights from future mechanistic studies and pilot clinical evidence could lead to well-designed multicenter clinical trials. This pathway can accelerate regulatory approval of this promising treatment for autonomic recovery and improve the quality of life of people with SCI.
Supplemental Material
sj-pdf-1-nro-10.1177_10738584221145570 – Supplemental material for Mapping the Iceberg of Autonomic Recovery: Mechanistic Underpinnings of Neuromodulation following Spinal Cord Injury
Supplemental material, sj-pdf-1-nro-10.1177_10738584221145570 for Mapping the Iceberg of Autonomic Recovery: Mechanistic Underpinnings of Neuromodulation following Spinal Cord Injury by Soshi Samejima, Claire Shackleton, Tiev Miller, Chet T. Moritz, Thomas M. Kessler, Klaus Krogh, Rahul Sachdeva and Andrei V. Krassioukov in The Neuroscientist
Footnotes
Author Contributions
S.S., R.S., and A.V.K. contributed to conceptualizing the organizational structure, content, and scope of the manuscript. S.S. and A.V.K. were primarily responsible for writing, figures, and the manuscript. C.T.M, T.M.K., and K.K. provided expert feedback for the content. All authors contributed to editing the entire document and approved the final document.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A.V.K. held the endowed chair in rehabilitation medicine, University of British Columbia, and his laboratory was supported by funds from the Canadian Institute for Health Research, Canadian Foundation for Innovation and BC Knowledge Development Fund, International Spinal Research Trust, Rick Hansen Foundation, PRAXIS Spinal Cord Institute, Wings for Life Research Foundation, and US Department of Defense. S.S. was supported by a Paralyzed Veterans of America Fellowship and by the Wings for Life Spinal Cord Research Foundation. C.S. was supported by a Paralyzed Veterans of America Fellowship. T.M. was supported by a Michael Smith Health Research BC Fellowship. R.S. was supported by the Wings for Life Spinal Cord Research Foundation and the US Department of Defense.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
