Abstract
Brain oscillations are regarded as important for perception as they open and close time windows for neural spiking to enable the effective communication within and across brain regions. In the past, studies on perception primarily relied on the use of electrophysiological techniques for probing a correlative link between brain oscillations and perception. The emergence of noninvasive brain stimulation techniques such as transcranial alternating current stimulation (tACS) provides the possibility to study the causal contribution of specific oscillatory frequencies to perception. Here, we review the studies on visual, auditory, and somatosensory perception that employed tACS to probe the causality of brain oscillations for perception. The current literature is consistent with a causal role of alpha and gamma oscillations in parieto-occipital regions for visual perception and theta and gamma oscillations in auditory cortices for auditory perception. In addition, the sensory gating by alpha oscillations applies not only to the visual but also to the somatosensory domain. We conclude that albeit more refined perceptual paradigms and individualized stimulation practices remain to be systematically adopted, tACS is a promising tool for establishing a causal link between neural oscillations and perception.
Keywords
Introduction
Perception is a constructive process, which does not rely exclusively on the incoming sensory information but requires the interaction between this information and the internal state of the brain including predictions, thoughts, and beliefs (Long and Toppino 2004; Picard and Friston 2014).
Using techniques such as single cell, local field potential (LFP) and electro-/magnetoencephalography (EEG/MEG) recordings as well as functional magnetic resonance imaging (fMRI), neural correlates of perception have been identified in a wide range of brain regions and at different temporal scales. Particularly, in humans and monkeys, neuronal firing rates, amplitudes of LFP/EEG and fMRI blood oxygen level–dependent (BOLD) signals related to visual stimulus perception have been reported in primary sensory cortices as well as in higher-order areas such as frontoparietal regions (reviewed in Boly and others 2017; Leopold and Logothetis 1999; Storm and others 2017). A special role has been attributed to brain oscillations (Fig. 1), which are thought to be key mechanisms facilitating information binding and neuronal communication (Fries 2005). Accordingly, perception might be shaped by fluctuations in brain networks dynamics at different frequencies (Singer 2017).
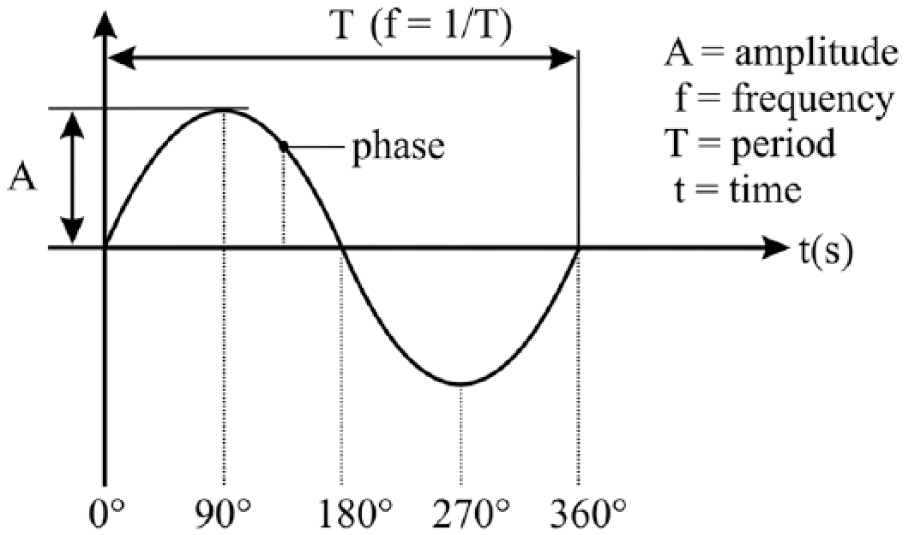
Oscillation parameters. Amplitude (A) = Maximum difference from the average value (strength of the signal); frequency (f) = number of cycles per second; phase = fraction of the oscillatory cycle at a given time point relative to the origin; period (T) = duration of one cycle.
Previous electrophysiological studies probed a correlative link between brain oscillations and perception. However, correlative links could represent epiphenomena of the percept. For instance, gamma-band synchrony previously related to conscious perception in different sensory modalities could represent the consequence or processes related to the report of the perceptual experience but not its cause (Aru and others 2012; Koch and others 2016; Tsuchiya and others 2015). Evidence for a causal link between brain oscillations and perception could be provided by externally manipulating the oscillatory activity in question while measuring the perceptual consequences. If the perceptual outcomes change (e.g., target visibility or number of perceptual switches) in response to this manipulation but not in a control condition (e.g., sham or stimulation at another frequency), then the oscillatory activity might causally contribute to perception.
Using non-invasive brain stimulation techniques, recent studies expanded correlational approaches to investigate the causal link between brain oscillations and perception. One of these techniques is transcranial alternating current stimulation (tACS), which is thought to interact with brain oscillations in a frequency-specific manner (Antal and Paulus 2013; Herrmann and others 2013; Vosskuhl and others 2018).
In the present article, we review the latest developments on the study of the causal contributions of specific oscillatory frequencies to perception by applying tACS. Note that we do not intend to exhaustively review the tACS literature on methodological issues or general effects on cognitive functioning, which has been done elsewhere (Antal and Paulus 2013; Herrmann and others 2013; Vosskuhl and others 2018). The review is structured into six sections, which include a short introduction on tACS techniques and its proposed neurophysiological effects, findings on perception separated by tACS frequency (theta, alpha, beta, and gamma, respectively, Table 1), and a section on limitations and future directions.
Summary of the Studies Using tACS to Modulate Perception.
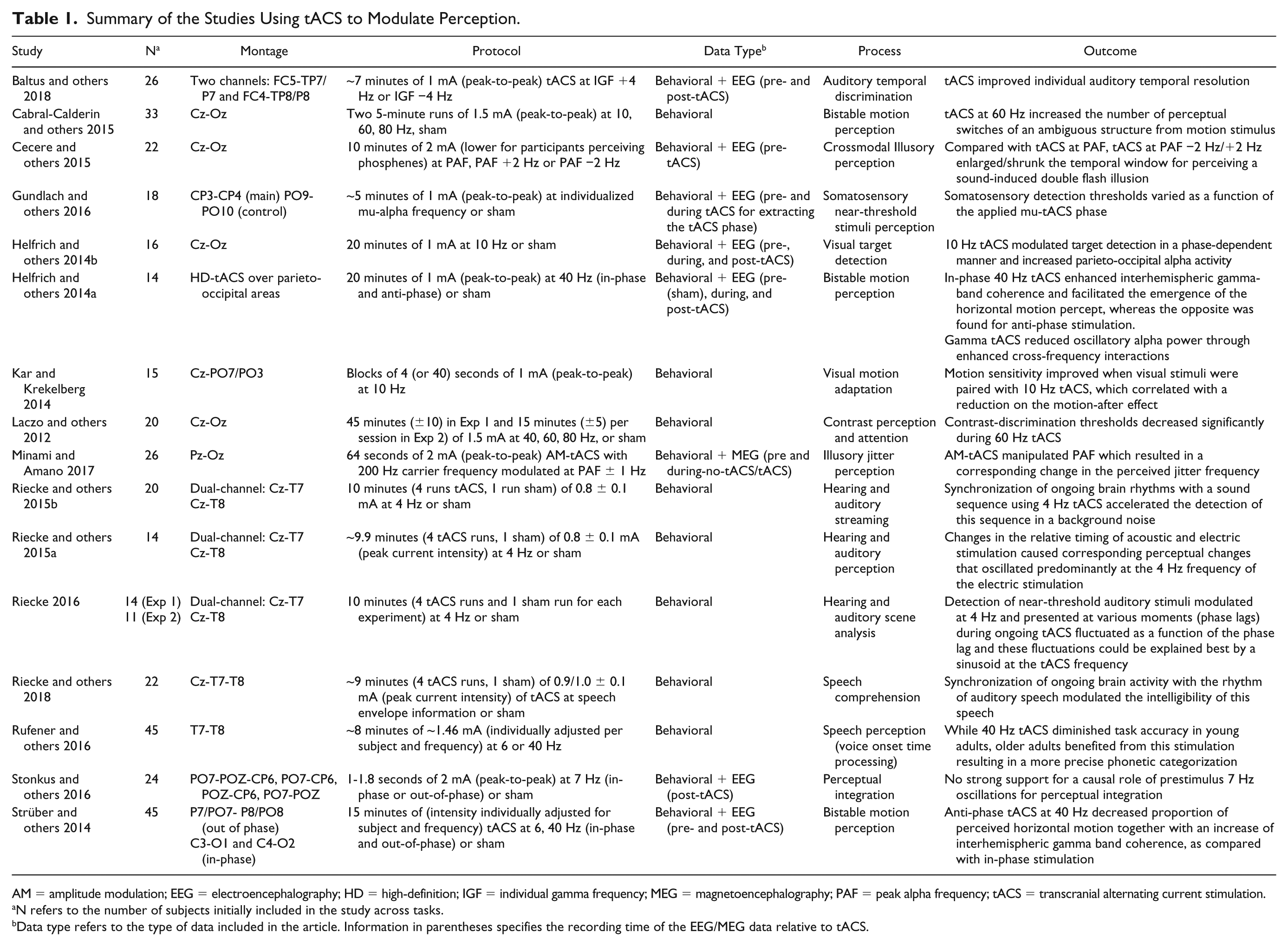
AM = amplitude modulation; EEG = electroencephalography; HD = high-definition; IGF = individual gamma frequency; MEG = magnetoencephalography; PAF = peak alpha frequency; tACS = transcranial alternating current stimulation.
N refers to the number of subjects initially included in the study across tasks.
Data type refers to the type of data included in the article. Information in parentheses specifies the recording time of the EEG/MEG data relative to tACS.
Modulating Brain Oscillations with tACS
tACS is a member of transcranial electrical stimulations techniques that also include transcranial direct current stimulation (tDCS) and transcranial random noise stimulation (tRNS) (Fig. 2A, Box 1). Typically, tACS involves a weak oscillatory electrical current (whose intensity fluctuates by switching polarity over time) applied over a task-relevant brain region at a relevant frequency (Antal and Paulus 2013; Herrmann and others 2013). This stimulation protocol is applied with the aim of synchronizing the intrinsic oscillations to the external force (Herrmann and others 2013; Reato and others 2013).
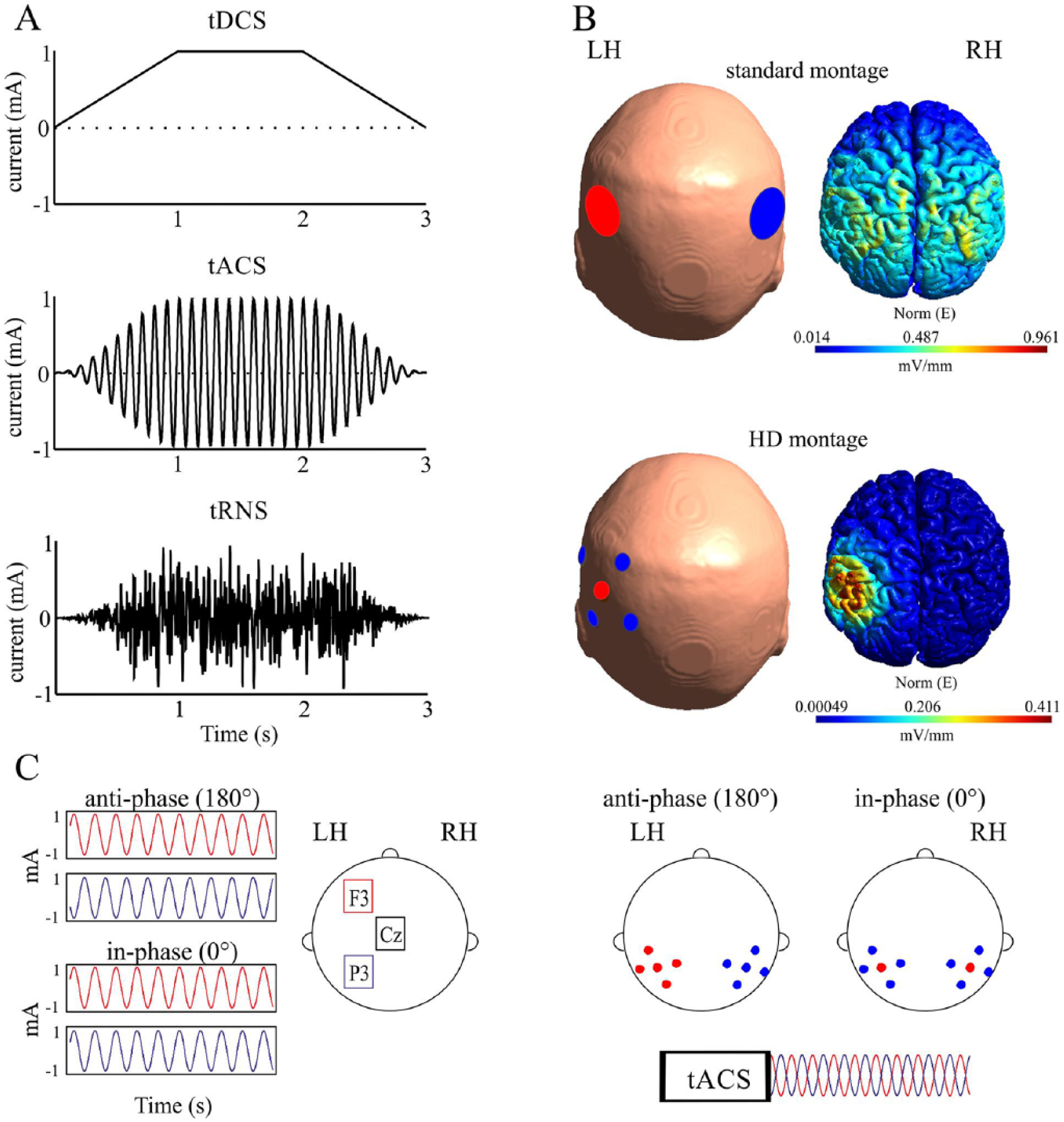
Stimulation protocols. (A) Each graph shows a cartoon representing the form of the applied stimulation for a duration of three seconds, with a fade in/fade out of 1 second; tDCS (upper graph), tACS (middle graph), and tRNS (lower graph). (B) Electrode positions and electric field distributions obtained from a finite element method simulation using Simnibs 2.0; standard montage (upper panel) and high-definition montage (bottom panel). (C) Protocols used for in-phase and anti-phase stimulation. The left panel shows the montage used, for example, in Polania and others (2012) for stimulating frontal and parietal areas with 0° (in-phase) or 180° (anti-phase) phase difference. Here in-phase stimulation is obtained by adding a third common returning electrode. The right panel shows a high-definition montage as used in Helfrich and others (2014a), where parietal areas are bilaterally stimulated with tACS in-phase or anti-phase between hemispheres. HD, high-definition; LH, left hemisphere; RH, right hemisphere; tACS, transcranial alternating current stimulation; tDCS, transcranial direct current stimulation; tRNS, transcranial random noise stimulation.
tACS and tDCS are the most popular modalities of transcranial electrical stimulation techniques.
Transcranial alternating current stimulation (tACS, left) and transcranial direct current stimulation (tDCS, right) are two of the most popular non-invasive brain stimulation techniques. Both modalities are reported to be safe and painless and involve a weak (typically ≤2 mA) electric current applied transcranially to the brain via two or more electrodes attached to the scalp.
In the case of tDCS, the applied electric current (anodal or cathodal) remains constant (polarity specific) at each electrode. tDCS is most frequently used to modulate cortical excitability in a polarity specific manner, that is, increased excitability has been reported with anodal stimulation and decreased excitability with cathodal stimulation (for a review, see Nitsche and others 2008).
During tACS, an alternating electric current is applied. Contrary to tDCS, here the electrical current switches polarity over time commonly with a sinusoidal wave. tACS is frequently applied with the aim of synchronizing the intrinsic oscillations to the external force and to evaluate behavioral changes associated with this manipulation. Using tACS, important parameters of brain oscillations can be manipulated by modulating the stimulation amplitude, frequency, and relative phase (for a review on tACS mechanisms, see Herrmann and others 2013).
The standard stimulation montage involves two large (about 3-7 cm diameter or length) scalp electrodes attached to the participants’ head with electrode paste (Fig. 2B). More advanced setups include high-definition montages involving more than two electrodes (positioned over a relevant area to induce more focal electric fields—Fig. 2B—or placed over several brain regions for network stimulation; Datta and others 2012; Ruffini and others 2014) and individualized electrodes where the electrode shape is adapted to the individuals’ head anatomy (Cancelli and others 2015).
When using tACS, the main oscillation parameters such as frequency, amplitude, and phase (Fig. 1) can potentially be manipulated. Regarding the frequency (number of cycles per second), tACS can be applied at a wide range, from very low to high frequencies (ranging from 0.75 Hz to 200 kHz) (Antal and Paulus 2013). Current strengths during tACS are typically ≤2 mA to avoid significant side effects, and sometimes are adjusted to the participant’s threshold for perceiving the stimulation. In terms of phase (fraction of the oscillatory cycle at a given time point relative to the origin), tACS can be applied varying its phase lag relative to a given stimulus or ongoing neural activity (e.g., Riecke and others 2015a), or manipulating the phase difference between electrodes, and thus between brain regions (i.e., in-phase, 0° difference; anti-phase, 180° difference; Fig. 2C; e.g., Helfrich and others 2014a). Using different combinations of frequency and phase with the appropriate electrode montage, tACS can be tuned to test the causality of brain oscillations for perception while considering the relevance of individual peak frequencies, stimulus-brain synchrony, and inter-area or inter-hemispheric coherence.
The effects of tACS on brain activity have shown to be dependent on the stimulation parameters and current oscillatory brain state (Cabral-Calderin and others 2016a; Cabral-Calderin and others 2016b; Feurra and others 2013) and have been reported to influence a wide range of brain functions including motor performance (Feurra and others 2013; Pollok and others 2015), intelligence (Santarnecchi and others 2013), memory (Alekseichuk and others 2016; Polania and others 2012), somatosensory (Feurra and others 2011b), and visual perception (Cabral-Calderin and others 2015; Helfrich and others 2014a; Kar and Krekelberg 2014; Laczo and others 2012; Strüber and others 2014).
Two main mechanisms have been proposed to explain tACS effects on perception and behavior, including (a) frequency-specific entrainment, that is, phase alignment of endogenous brain oscillations to externally applied oscillating electrical currents (Herrmann and others 2013; Reato and others 2013) and (b) modulation of spike-timing dependent plasticity (STDP) (Vossen and others 2015; Zaehle and others 2010). Frequency-specific entrainment can explain online tACS effects, for instance in cases wherein neural firing has been shown to synchronize to externally applied electric fields (Frohlich and McCormick 2010; Ozen and others 2010). Modulation of STDP has been used to explain tACS aftereffects (Vossen and others 2015). In addition, several non-neuronal mechanisms mediated by astrocytes, glia, and perivascular elements (Monai and others 2016) and afferent nerve stimulation (Fertonani and others 2015) have been suggested as well.
The entrainment of brain oscillations by tACS has been partially supported by electrophysiological studies at the single or multi-unit level, in cell cultures or animal models (for a review, see Reato and others 2013), and at the whole brain level in human subjects by means of EEG or MEG (Helfrich and others 2014a; Helfrich and others 2014b; Neuling and others 2015). The evaluation of online tACS effects in human neural activity is challenging because of the strong artifacts induced by tACS in the EEG/MEG signals. Most human EEG/MEG studies applied alpha-tACS (e.g., at 10 Hz or at peak alpha frequency) and results are not always in agreement. While some studies reported increased parieto-occipital EEG alpha activity during (Helfrich and others 2014b) and after alpha-tACS (Kasten and others 2016; Neuling and others 2013; Zaehle and others 2010), more recent studies did not find alpha-tACS aftereffects (Fekete and others 2018; Stecher and Herrmann 2018). This latter lack of effect was attributed to several confounds such as the mismatch between tACS frequency and individual alpha peak as well as individual variability in alpha power (Stecher and Herrmann 2018).
Frequency- and task-dependent effects of tACS have also been reported with fMRI (Alekseichuk and others 2015; Cabral-Calderin and others 2016a; Cabral-Calderin and others 2016b; Chai and others 2018; Vosskuhl and others 2015; Williams and others 2017), which offers advantage over EEG/MEG due to its lower susceptibility to tACS artifacts (Antal and others 2014; Cabral-Calderin and others 2016a). However, because of the low temporal resolution of fMRI and the fact that BOLD signals are only an indirect measure of brain activity, conclusions about tACS modulation of oscillatory activity when using fMRI can only be inferred.
In brief, despite technical limitations, tACS is a promising technique for testing the causality of brain oscillations for perception—and brain function in general—in a non-invasive manner. In the following sections, we will discuss the tACS findings suggesting that perception is not only supported but also influenced by oscillatory dynamics at different temporal scales.
Theta-tACS Modulates Auditory but Not Visual Perception
In the visual domain, previous electrophysiological studies suggested that the phase of a prestimulus 7-Hz oscillation predicts the integration of visual targets in a contour detection task (Hanslmayr and others 2013). However, tACS studies do not support a causal effect of theta oscillations for visual perception. For example, a recent study applied tACS at 7 Hz over left occipital and/or right parietal regions to evaluate whether this manipulation affects performance in a perceptual integration task, where subjects had to indicate whether they perceived a contour stimulus or not (Stonkus and others 2016). The authors observed that in-phase stimulation of occipital and parietal regions improved contour detection compared with sham; however, performance did not differ from anti-phase stimulation, suggesting that prestimulus interareal theta phase between occipitoparietal regions might not be as relevant for perceptual integration. Overall, the results of this study did not strongly support a causal role of prestimulus 7-Hz oscillations for perceptual integration, although the authors did report modulation of the oscillatory activity at 7 Hz due to tACS measured by EEG. A previous study also failed to report a significant effect of theta-tACS (at 6 Hz) on (bistable) visual perception, where subjects had to report the perception of an apparent motion stimulus (Strüber and others 2014); see the section on gamma oscillations.
In addition to visual perception, electrophysiological studies with EEG/MEG also linked theta oscillations (4-8 Hz) to auditory processes such as sound segregation and detection, auditory selective attention, and speech comprehension (Kerlin and others 2010; Luo and Poeppel 2007; Toth and others 2016). The available tACS evidence supports this role suggesting that the phase of theta oscillations causally modulates hearing and auditory scene analysis (Riecke and others 2015a; Riecke and others 2015b; Riecke and others 2018).
In a set of experiments, Riecke and colleagues varied the phase lag between tACS in the theta frequency band (4 Hz) and auditory stimulation at the theta frequency and evaluated how this manipulation impacts auditory perception throughout different stimulus conditions (Riecke 2016; Riecke and others 2015a; Riecke and others 2015b). In one of the experiments (Riecke and others 2015b), listeners detected rhythmic streams of various musical chords fluctuating at 4 Hz and masked by background sounds. Simultaneously, 4 Hz tACS was applied bilaterally over the auditory cortex to entrain ongoing theta oscillations. The delay between tACS and the auditory streams was cyclically varied at phase lags between 30° and 330° (Fig. 3A), hypothesizing that, if stream-brain phase entrainment is contributing to stream perception, such a manipulation should lead to cyclical fluctuations in the perceptual buildup of streaming. On average, subjects performed better when streams preceded tACS by a stimulus phase lag of 16° ± 23°. Assuming that tACS influenced behavior via neuronal oscillatory phase entrainment, the authors associated stream-brain synchrony with the listeners best stimulus lag. The waveform resulting from the alignment to the subject’s best lag exhibited the expected cyclical fluctuations in the perceptual buildup, that is, the buildup was relatively brief at the hypothesized strong stream-brain synchronies and it lasted relatively longer at the hypothesized weak stream-brain synchronies, compared to sham (Fig. 3B).
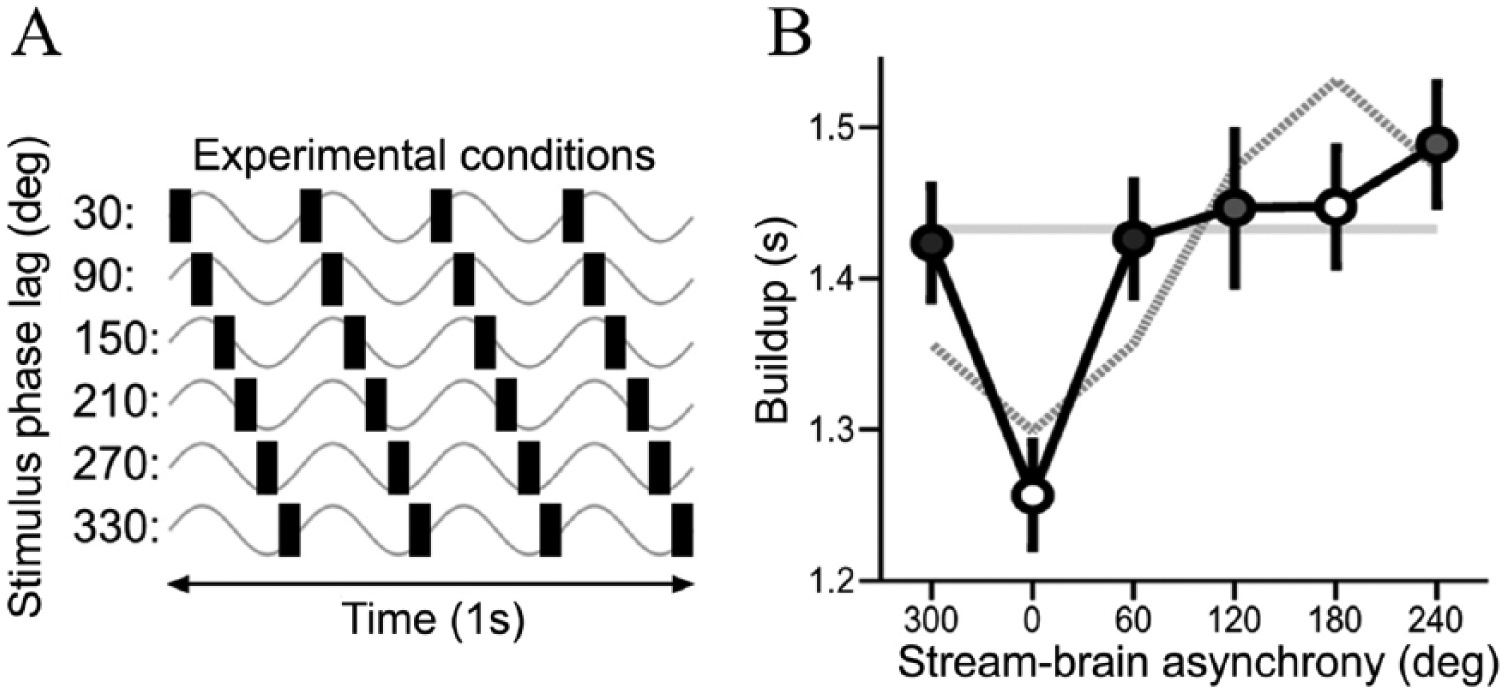
Effect of theta-tACS on auditory streaming. (A) Experimental conditions. Each condition is characterized by a different phase lag between the stream (black bars) and tACS (gray waveforms). (B) Group data (mean ± SEM across listeners) showing the buildup as a function of presumed stream-brain synchrony after subject’s best-lag alignment to 0° (black), fitted 4-Hz sinusoids (gray) and average buildup under sham stimulation (baseline; horizontal line). Note the expected cyclical fluctuations in the perceptual buildup. SEM, standard error of the mean; tACS, transcranial alternating current stimulation. (A) and (B) Adapted with permission from Riecke and others (2015b)
Using a similar approach, the authors varied the delay between near-threshold 4-Hz click trains and 4-Hz tACS applied over auditory cortices (Riecke and others 2015a). As for stream buildup, it was observed that the detection of near-threshold low-level auditory stimuli also fluctuates as a function of the phase lag between the stimulus and the tACS, that is, performance was better at phase angles during the hypothesized positive half of the 4-Hz cycle than at phase angles during the negative half-cycle (Riecke 2016; Riecke and others 2015a). In another study, tACS applied at the frequency of the speech envelope modulated the intelligibility of speech in both a cocktail party–like scenario and a listening situation devoid of aural speech-amplitude envelope input (Riecke and others 2018). Note that none of these studies showing a significant effect of 4-Hz tACS on auditory perception measured neural oscillations per se. Contrary to a causal role of theta oscillations for speech perception, a recent study reported no effect of 6-Hz tACS on voice onset time categorization (Rufener and others 2016); see the section on gamma oscillations.
In general, while tACS studies do not support a causal role of theta oscillations for visual perception they suggest that stimulus-brain synchrony in the theta frequency range is not a byproduct, but could be causally involved in auditory perception, including streaming buildup, near-threshold stimulus detection, and speech intelligibility. However, since the latter studies applied tACS exclusively at 4 Hz and no concurrent measure of brain activity was recorded, the specific relevance of 4-Hz oscillatory activity for auditory perception should be taken with caution.
Perception Is Temporally Shaped by Alpha Oscillations
Previous EEG/MEG studies showed that the speed, phase, and power of alpha oscillations (~7-12 Hz) are reliable predictors of visual perception (Busch and others 2009; Mathewson and others 2009; Mazaheri and others 2014; Samaha and Postle 2015). For example, faster alpha frequencies between and within subjects have been associated to lower thresholds for perceptually fusing two independent flashes into one, suggesting that the exact alpha frequency is an indicator of the temporal resolution of perception (Samaha and Postle 2015). Conversely, it has been shown that visual detection and discrimination are worst when visual stimuli are presented during the trough of the alpha cycle (Mathewson and others 2009), or when alpha power is high (van Dijk and others 2008). In general, it has been proposed that alpha oscillations gate sensory information by pulsed inhibition of ongoing neural activity. This “pulsed inhibition” concept suggests that alpha oscillations create windows in which the processing of sensory stimuli is attenuated or facilitated, through alternating microstates of inhibition and excitation (Klimesch and others 2007; Mathewson and others 2011).
Regarding the exact frequency of alpha oscillations, Minami et al (Minami and Amano 2017) showed that individual variations in the peak alpha frequency (PAF) measured with MEG correlate with the frequency of illusory jitter perception in a motion-induced spatial conflict task. Subjects judged whether a physical jitter (at seven possible frequencies from 5.5 to 15 Hz) presented in the lower visual field was faster than an illusory jitter (here moving borders defined by color contrast are perceived as jittering when placed near moving borders defined by luminance contrast) presented in the upper visual field (Fig. 4A). The results showed that the perceived illusory jitter frequency was correlated with the individual PAF (Fig. 4B, but not the power) during the illusory jitter perception and the PAF from a separate session during eyes-open and eyes-closed resting-state conditions. Moreover, stimulation over parieto-occipital areas with tACS at PAF + 1 Hz increased PAF and the perceived frequency of the illusory jitter while the opposite was found for tACS at PAF − 1 Hz, both compared to non-stimulation (Fig. 4C).
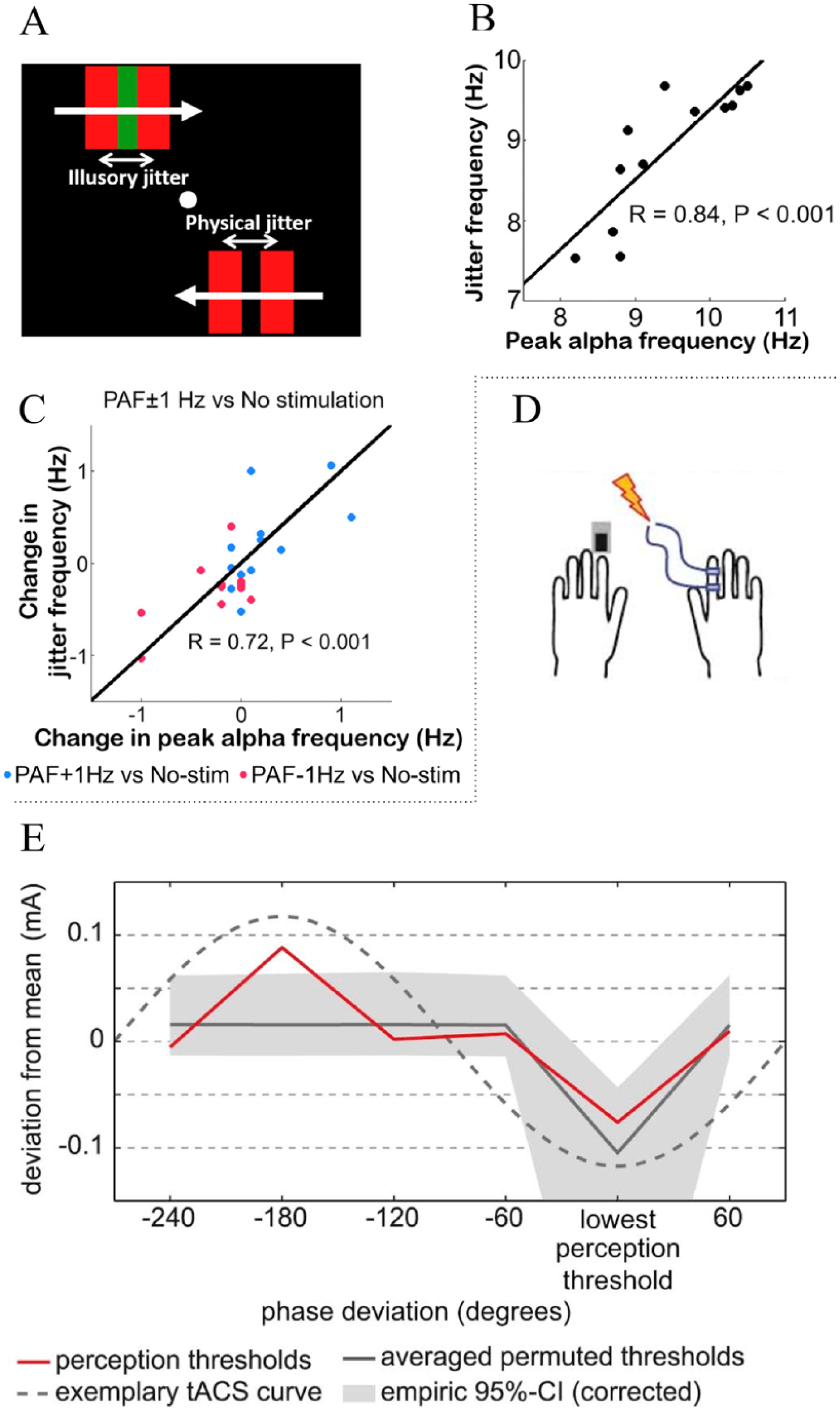
Effects of alpha-tACS on visual and somatosensory perception. (A) Visual stimulus. Subjects judged whether a physical jitter presented in the lower visual field was faster than an illusory jitter presented in the upper visual field. The green bar in the upper visual field was isoluminant with the surrounding red square, which created an illusory jitter perception of the green bar. B) Correlation between PAF measured with MEG during the eyes-closed resting condition and the perceived jitter frequency for all participants. (C) Correlation between the change in the MEG PAF and the perceived jitter frequency in the tACS at PAF + 1 Hz (blue) and PAF − 1 Hz (red) conditions relative to the no-stimulation condition. Dots represent individual data. Note that tACS at PAF + 1 Hz increased PAF and perceived jitter frequency, while tACS at PAF − 1 Hz decreased PAF and perceived jitter frequency. Moreover, the strong correlation suggests that modulation of PAF is associated with modulation of illusory jitter frequency. MEG, magnetoencephalography; PAF, peak alpha frequency; tACS, transcranial alternating current stimulation. (A)-(C) adapted with permission from Minami and Amano (2017). (D) Somatosensory stimulus. Participants reported the detection of somatosensory near-threshold stimuli applied to the index finger. (E) Perceptual thresholds varied as a function of the tACS phase. The figure shows the mean phase-aligned deviations from average perception thresholds for stimuli presented within six different bins of the mu-alpha tACS cycle (red), the 95% confidence interval for the empiric null-distribution derived from a permutation test Bonferroni-corrected for multiple comparisons (shaded gray areas) and the exemplary tACS curve (dashed lines). (D)-(E) adapted with permission from Gundlach and others (2016).
In a series of experiments, Kar and Krekelberg showed that 10-Hz tACS targeting the human visual motion area (hMT+) improved motion sensitivity by attenuating motion adaptation (Kar and Krekelberg 2014). Subjects had to report the perceived global motion direction of a dynamic random dot kinematogram (RDK) that was presented with different coherence levels ranging from all dots moving downward to all dots moving upward. In the first experiment, 10-Hz tACS targeting the left hMT+ (electrodes PO7-PO3 in the 10-20 system) was applied simultaneous to the stimulus presentation. Compared with non-stimulation, motion discrimination sensitivity improved in the tACS condition when stimulation was contralateral to the stimulus. In the second experiment, tACS was applied simultaneous with an adapter stimulus that was presented before the test stimulus. In such adaptation paradigms, the exposure to the upward-moving stimulus (adapter) results in a perceptual motion aftereffect where a subsequent stationary or random motion stimulus is perceived as moving in the opposite, that is, downward direction. This motion aftereffect was reduced due to the contralateral application of tACS, and this effect was more pronounced in subjects exhibiting larger motion aftereffects in the non-stimulation condition. Additional experiments showed that tACS applied either in the prestimulus phase or during the recovery from adaptation did not have consistent effects on motion perception, suggesting that tACS affected the induction of adaptation (Kar and Krekelberg 2014). Although no concurrent measures of brain oscillations were performed in this study, a subsequent intracranial recording study from the same laboratory in monkeys in the motion-sensitive area MT showed that tACS at 10 Hz resulted in a broadband power increase and reduced spike-frequency adaptation of neurons, lending further support of the effectiveness of tACS (Kar and others 2017). Apart from the peak frequency and power, also the phase of alpha tACS has been reported as critical for stimulus detection. For example, 10Hz tACS over the occipital cortex has been shown to increase parieto-occipital EEG alpha activity and to modulate the detection of a visual target in a phase-dependent manner (Helfrich and others 2014b).
The role of alpha oscillations for perception has been reported not only in the visual domain but also in other sensory modalities. Stimulation with tACS at PAF−2Hz/+2Hz has been shown to enlarge/shrink the temporal window for perceiving a sound-induced double flash illusion (wherein two beeps presented within ~100 ms together with one flash induced the perception of a second illusory flash) compared with tACS at PAF, suggesting that alpha oscillations also promote audio-visual interactions (Cecere and others 2015). No electrophysiological data were collected during tACS to assess its effect on the oscillatory dynamics.
In the somatosensory domain, Gundlach and colleagues applied tACS (or sham) at the individual mu-alpha frequency over primary somatosensory cortex, while participants reported the detection of somatosensory near-threshold stimuli applied to the index finger (Fig. 4D) (Gundlach and others 2016). Overall, tACS did not modulate the mean perceptual thresholds. However, when analyzing the phase lag between tACS and the somatosensory stimuli, it was observed that perceptual thresholds varied as a function of the tACS phase (Fig. 4E). This result indicates that individual mu-alpha frequency tACS affects somatosensory perception by inducing phase-dependent periods of neural excitation and inhibition. No information about tACS effects on brain oscillations was provided.
Overall, tACS studies at the alpha frequency range suggest that alpha oscillations might be causally related to perception in different sensory modalities. The effects of alpha-tACS agree with the pulsed inhibition framework suggesting that alpha oscillations temporally shape perception by gating sensory information due to oscillatory modulation of excitability states. In addition, the results suggest that alpha-tACS affects perception not by the addition of neural noise to the brain—which should have disturbed the performance of the motion discrimination task in (Kar and Krekelberg 2014)—but by the modulation of plastic changes related to neural adaptation.
Are Beta Oscillations Limited to Sensorimotor Performance?
Beta oscillations (∼13-30 Hz) have been associated with sensorimotor performance, working memory, decision making, spatial attention and perceptual grouping (Donner and Siegel 2011; Engel and Fries 2010; Meindertsma and others 2017; Spitzer and Haegens 2017; Wilke and others 2009; Wrobel 2000). At the same time, only scarce information is available on the impact of beta-tACS on perceptual tasks. Several tACS studies investigated entrainment of motor populations at beta frequencies (Feurra and others 2011a; Feurra and others 2013; Guerra and others 2016; Pollok and others 2015; Schilberg and others 2018) and showed that beta-tACS over primary motor cortex modulates motor learning (Pollok and others 2015) and sensorimotor integration (Guerra and others 2016). Beta-tACS has also been related to phosphene and cutaneous sensations (Kanai and others 2008; Turi and others 2013). A recent study showed that tACS applied at 20 Hz in anti-phase over the right parietal and left frontal cortex improved performance in an attentional blink task (Yaple and Vakhrushev 2018) and another study showed that tACS at 20-Hz over the left prefrontal cortex increased risky decision making (Yaple and others 2017). Given the scarce number of studies explicitly reporting on effects of beta-tACS on reported stimulus perception, it is currently difficult to either reject or establish a causal link between the two.
Gamma Rhythms Influence Perceptual Resolution and Binding
Gamma oscillations in neural circuits have long been hypothesized as a mechanism to facilitate the transient integration of distributed neuronal ensembles enabling cognitive functions such as attention and perception in different sensory modalities (Fries 2009; Singer 1999; Tallon-Baudry 2009). In the visual domain, electrophysiological studies have shown that gamma oscillations are related to sensory processing, feature binding, perceptual switching of bistable stimuli, visual suppression and perceptual decision making (Başar-Eroglu and others 1996; Castelhano and others 2014; Honkanen and others 2015; Panagiotaropoulos and others 2012; Wilke and others 2006). Laczó and colleagues evaluated potential changes in contrast perception and spatial attention induced by tACS, applied over the occipital cortex at three different gamma frequencies, that is, 40, 60, and 80 Hz (Laczo and others 2012). While no significant modulation of spatial attention was observed, a selective modulation of contrast discrimination thresholds was found with 60Hz tACS (Laczo and others 2012). No additional measures of brain oscillations during tACS were performed.
Regarding visual awareness and perceptual switching, several studies combined tACS with bistable perception paradigms. In a first study, Strüber and colleagues stimulated occipitoparietal cortices with tACS in or out of phase across hemispheres, while participants observed a stroboscopic alternating motion (SAM) stimulus across the two visual hemifields, in which two moving dots are perceived to alternate between horizontal or vertical positions (Fig. 5A) (Strüber and others 2014). In this paradigm, perception of the horizontal motion requires the integration of information across both hemifields, which has been related to increased interhemispheric gamma band coherence compared to the perception of vertical motion (Helfrich and others 2014a; Rose and Buchel 2005). Accordingly, the authors reported that anti-phase (across hemispheres) 40-Hz tACS decreased the proportion of horizontal motion perception, although no corresponding increase in horizontal motion perception was observed when in-phase stimulation was applied (Strüber and others 2014). The online effects of tACS on brain oscillations were not evaluated due to tACS-induced artifact in the EEG signal. Nevertheless, it was observed that anti-phase stimulation resulted in increased interhemispheric coherence after stimulation. In a follow-up study, Helfrich and colleagues used a high-density montage also with the aim of stimulating both parieto-occipital regions with 40Hz tACS in or out of phase across hemispheres (Fig. 5B) (Helfrich and others 2014a). The authors found that these stimulation conditions induced opposite effects on the perception of the SAM stimulus (Fig. 5A), that is, in-phase stimulation increased while anti-phase tACS decreased the horizontal motion perception (Fig. 5C) (Helfrich and others 2014a). It was also observed that interhemispheric gamma band coherence measured with EEG was always higher for the horizontal than for the vertical percept, regardless of the tACS stimulation condition (in-phase or anti-phase). Nevertheless, in-phase tACS increased interhemispheric gamma-band synchronization over parieto-occipital cortex while anti-phase stimulation decreased it (Fig. 5D). This coherence change during stimulation was positively correlated with the perceptual changes. While no concurrent power changes were observed in the gamma range, 40-Hz tACS decreased alpha power in parieto-occipital areas as compared with sham, regardless of the stimulation condition (in-phase or anti-phase). These results support the antagonist role of gamma and alpha oscillations and suggest that cross-frequency coupling is causally linked to perceptual binding and conscious perception (Canolty and Knight 2010).
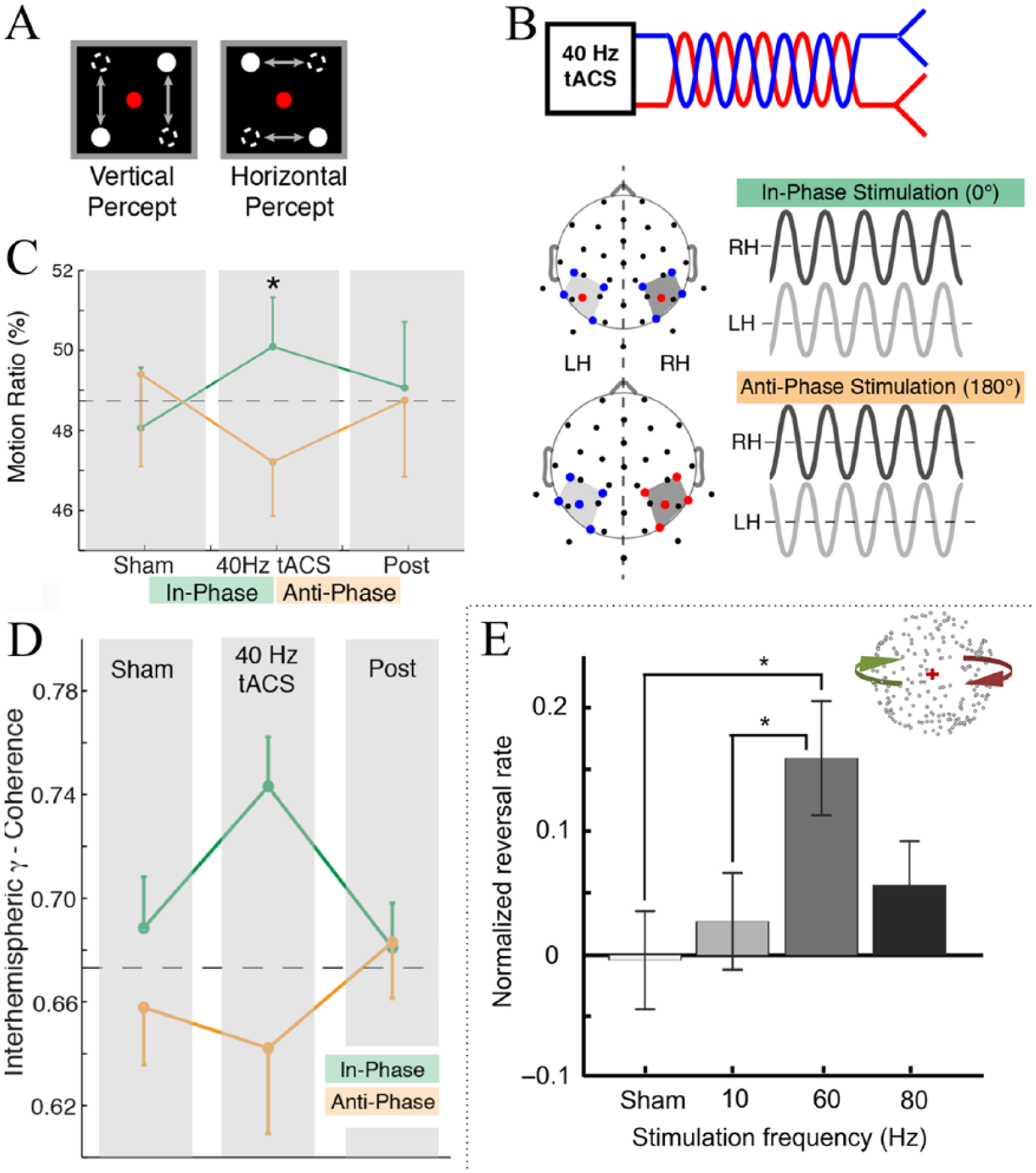
Effects of gamma-tACS on bistable perception. (A) Stroboscopic alternating motion stimulus. Two moving dots are perceived to alternate between horizontal or vertical positions. (B) tACS setup. The output signals of the tACS-stimulator were split and fed into 10 Ag/AgCl electrodes positioned on the cortex to create an in-phase (0° phase difference between hemispheres, green) and anti-phase (180° phase difference, orange) setup. Red and blue lines/dots depict the connection to the respective stimulator channels. (C) Behavioral results. In-phase stimulation (green) increased while anti-phase tACS (orange) decreased the horizontal motion perception (motion ratio = time horizontal/total time). The dashed black line depicts the average sham baseline and the star indicates the significant difference as revealed by a two-way repeated-measures ANOVA with planned contrasts. (D) Changes in interhemispheric gamma-band coherence changes over time. The black dashed line indicated the mean sham value. ANOVA, analysis of variance; tACS, transcranial alternating current stimulation. (A)-(D) adapted with permission from (Helfrich and others 2014a). (E) The structure from motion (SfM) stimulus is illustrated at the right of the panel. A rotating sphere is either perceived as rotating clockwise or counterclockwise. The graph shows the effect of tACS on the perception of the SFM stimulus. While no significant effects of 10- and 80-Hz tACS were observed, 60-Hz tACS increased the number of spontaneous perceptual reversals of the stimulus (normalized reversals rate = (reversals rate tACS-on − reversals rate tACS-off baseline)/(reversals rate tACS-on + reversals rate tACS-off baseline)). Error bars indicate SEM (standard error of the mean) across 22 participants. *P < 0.05. (E) adapted with permission from Cabral-Calderin and others (2015).
We previously showed that gamma-tACS increased perceptual reversal rates of a bistable structure from motion stimulus, where a rotating sphere is either perceived as rotating clockwise or counterclockwise (Fig. 5E). Simultaneously with this task, tACS was applied over the occipital cortex at sham, 10, 60, or 80 Hz. While no significant effects of 10- and 80-Hz tACS were observed, 60-Hz tACS increased the number of spontaneous perceptual reversals of the stimulus (Fig. 5E; Cabral-Calderin and others 2015), suggesting that gamma oscillations causally contribute to resolving perceptual ambiguity. Nevertheless, since no measures of brain oscillations were performed, the relevance of the particular frequency should be taken with caution.
Apart from the visual domain, gamma oscillations have also been related to auditory perception. It has been shown that the temporal resolution of auditory processing is linked to the peak gamma frequency in the auditory cortex, that is, individuals with a higher gamma peak frequency tend to have better temporal resolution (Baltus and Herrmann 2015). Building on this finding, a recent study applied tACS over auditory cortices at the individual gamma frequency ± 4 Hz (IGF, median = 49 Hz) to evaluate the effect of increasing or decreasing IGF on temporal resolution (Baltus and others 2018). The results showed that subjects receiving tACS above the IGF improved performance compared with subjects receiving tACS below their IGF, suggesting that gamma band activity is causally related to temporal resolution in the auditory domain. The authors also reported significant relative changes of auditory steady state response amplitudes in the EEG signal after tACS, nevertheless, this effect was not group specific.
Gamma oscillations have also been related to speech perception (Rufener and others 2016), where the incoming auditory information appears to be parsed into units at the phonematic scale via gamma oscillations. Interestingly, 40-Hz tACS over temporal areas affected speech perception (measured by voice onset time [VOT] categorization) differently in young and older adults. While 40-Hz tACS increased VOT categorization precision in older adults the opposite was observed for younger adults. No significant effect was observed for the control condition with 6-Hz tACS. Results from this study support the relevance of gamma oscillations for the processing of temporal acoustic features such as phonemes (Rufener and others 2016). These results were also interpreted as evidence for the existence of an inverted U-shape relationship between auditory gamma activity and phoneme processing, which predicts enhancement of temporal resolution and encoding by tACS when applied in a hypofunctioning system (i.e., older adults) but the opposite when applied in a normally functional system (Rufener and others 2016). No measures of brain oscillations were performed.
Taken together, the studies with gamma-tACS suggest that gamma oscillations are causally related to perceptual binding and switching during visual stimulation and to the temporal resolution of auditory processing. Since different gamma frequencies have been linked to bistable perception (40 Hz in Helfrich and others 2014a; Strüber and others 2014, and 60 Hz in Cabral-Calderin and others 2015), one could speculate that the specific frequency within the gamma band relevant for perception depends on the type of sensory stimulus.
Limitations and Future Directions
A note of caution before interpreting the “causal” effects of tACS on conscious perception is the fact that one cannot exclude that third factors contributed to or mediated the measured effects. For example, tACS might also exert some of its effects by activating peripheral nerves in the scalp such as trigeminal nerve branches or the retina (Liu and others 2018; Schutter and Hortensius 2010) that in turn mediate its effects on conscious perception. Likewise, a negative finding with tACS does not rule out that a certain frequency has functional impact when applied over a given brain region since the current that reaches the brain heavily depends on several individual anatomical factors such as skull thickness, musculature, orientation of neurons in sulci and gyri with regard to the stimulating electrodes together with context-related factors such as the behavioral and intrinsic oscillatory state (Liu and others 2018).
Most tACS studies included only visual or auditory stimuli, making it difficult to generalize findings across sensory modalities. Motor responses are normally used for accessing the contents of perception, which could lead to confounds such as attention, response selection, and motor preparation (Tsuchiya and others 2015). For example, spatial attention is known to increase perceptual reversals of bistable figures (Slotnick and Yantis 2005) and the probability of perceptual suppression (Scholvinck and Rees 2009). Thus, if tACS over a given brain regions is affecting visual attention, it could be also the real factor behind the perceptual changes. Future studies could use more refined perceptual paradigms where subjective and objective measures of report are combined.
Since tACS is often applied using one protocol for all subjects disregarding individual anatomy or electrophysiological features, interpretations about the relevant frequency and electrode configuration should be made with caution. Moreover, several studies tested only one tACS frequency which was contrasted to a sham condition. When no additional control frequencies are used, observed effects might be driven by a general effect of the electrical current applied on the scalp but not due to a causal role of the frequency in question. Future studies might also benefit from adjusting the stimulation to the ongoing oscillatory activity (close-loop stimulation). Closed-loop tACS stimulation that takes the phase of the ongoing activity into account might also be important since larger frequency and phase offsets can lead to a breakdown of the oscillation, potentially rendering the tACS effects unreliable. Another aspect to consider when interpreting tACS results refers to the optimal stimulation length. The perceptual studies previously discussed applied tACS with different durations from only a few seconds (Kar and Krekelberg 2014; Stonkus and others 2016) to 20 minutes or more (Helfrich and others 2014a; Laczo and others 2012). It is not clear what could be the optimal duration to be able to affect perceptual processes when using tACS, albeit the studies with positive effects suggest that several minutes might be preferable (Strüber and others 2015). Only a few studies used computational modeling to simulate the electric field induced by tACS. Electric field simulations can help planning more effective stimulation protocols (e.g., regarding individualized stimulation sites and current strength (Baltus and others 2018; Windhoff and others 2013)). Concurrent measures of oscillatory brain activity are not always used, and conclusions are mostly made under the assumption that tACS did entrain oscillations at the stimulation frequency, which is not necessarily the case (see Ali and others 2013; Reato and others 2010). Without concurrent electrophysiological measures, conclusions about the relevance of the power, phase, or synchrony of a given rhythm remain speculative. Although the online combination of tACS and electrophysiological measures is very challenging due to tACS-induced artifacts, recent studies use advance methods for artifact removal which could be more frequently used (Neuling and others 2017; Noury and Siegel 2017, 2018).
In addition to the scientific knowledge, results from tACS studies on perception have clinical implications. For example, the findings that speech-brain entrainment is critical for auditory speech comprehension suggest that tACS carrying speech-envelope information could be used to improve speech comprehension (Riecke and others 2018). In addition, the improvement of temporal resolution by tACS offers the possibility of using tACS for auditory rehabilitation.
Despite some positive findings, the field of tACS remains controversial and its effects on brain function are not always replicated. For instance, it was recently shown that low-frequency tACS (0.75, 1 Hz) does not entrain sleep rhythms (neither spindle activity during sleep nor theta activity during waking rest), as measured with intracranial recordings in humans (Lafon and others 2017). This lack of effect has been related to the attenuated electric fields reaching the brain, suggesting that at common intensities, tACS per se might be ineffective at entraining oscillatory activity. The existence of robust tACS aftereffects that outlast the stimulation also remains a debated issue, which is particularly relevant for development of therapeutic tACS applications. Several studies measuring EEG in humans reported frequency-specific enhancements after tACS up to 20 minutes (Helfrich and others 2014a; Kasten and others 2016; Neuling and others 2013; Strüber and others 2015), while invasive recordings in animals typically reported no such after-effects (Reato and others 2013). Differing stimulation durations or task-related brain states might be underlying such discrepancies (Kar 2015; Strüber and others 2015) and should be systematically varied in studies investigating the neurophysiological effects of tACS.
Future research might profit from combinations of optimized tACS protocols with close-loop stimulation, electrophysiological measures, computer modeling to obtain more individualized stimulation practices and double-blind designs. The use of these methods and their adequate report are critical for ensuring the reproducibility and reliability across brain stimulation studies (Bikson and others 2017), which are central to their adequate research and therapeutic applications. Such approaches combined with more refined perceptual paradigms will be of great value to probe the real neural correlates of perception.
Conclusions
In summary, although more refined perceptual paradigms and individualized stimulation practices remain to be systematically adopted, tACS is a promising tool for establishing a causal link between neural oscillations and perception. The current evidence derived from electrophysiological and tACS studies suggests a causal role of theta and gamma oscillations in auditory cortices for auditory processing and alpha and gamma oscillations in parieto-occipital regions for visual perception. In addition, the sensory gating by alpha oscillations applies not only to the visual but also to the somatosensory domain.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Herman and Lilly Schilling Foundation (to M.W.).
