Abstract
Introduction
Cancer clinical trials do not always represent the real-world cancer population, as older adults and marginalized racial groups are often underrepresented. This study assessed patterns of enrollment of underrepresented patients and how trial and site factors may influence enrollment.
Methods
This retrospective, pooled cross-sectional study used de-identified data from ECOG-ACRIN (EA)-sponsored breast cancer therapeutic clinical trials from 2002-2020. Patient- and trial-level data were extracted from EA trials and ClinicalTrials.gov. Site-level data were from de-identified Landscape Assessment surveys voluntarily completed by National Cancer Institute Community Oncology Research Program sites. Outcomes included the proportions of underrepresented patients enrolled on a trial. Fractional regression models evaluated associations between trial-level factors and enrollment proportions of underrepresented patients using adjusted means and 95% confidence intervals (CI). Weighted Kappa (Kw) statistics and corresponding 95% CI estimated the level of agreement between patient populations served by sites versus enrolled on a trial.
Results
Of 9,015 patients enrolled across 12 trials, 18% were aged ≥65 years old, 12% were Black, 15% were Medicare beneficiaries, and 15% were rural residents (unadjusted enrollment). Adjusted mean proportion enrollment of underrepresented patients was similar to unadjusted results. Over half of trials were randomized and 92% of studies had two or more drugs in the protocol, yet these did not appear to influence mean enrollment of underrepresented groups. Moderate levels of agreement were found between Black patients served versus enrolled (Kw 0.64; 95% CI 0.50-0.78), and low or no agreement for Medicare beneficiaries and patients aged ≥65 (Kw 0.18, 0.05-0.31; Kw 0.02, -0.06-0.11; respectively).
Conclusions
EA-sponsored breast trials continue to enroll few individuals from underrepresented backgrounds. Trial design had a minimal impact on enrollment and patient populations sites served did not typically match the patients enrolled on trials. More research is needed to engage sites and test strategies for enrolling underrepresented patients.
Plain Language Summary
Cancer clinical trials do not always represent the real-world cancer population, as older adults and marginalized racial groups are often underrepresented in clinical trials. This study assessed patterns of enrollment of underrepresented patients and how trial and site factors may influence enrollment. This study used anonymous data from ECOG-ACRIN (EA)-sponsored breast cancer therapeutic clinical trials from 2002-2020. Patient- and trial-level data were extracted from EA trials and ClinicalTrials.gov. Site-level (i.e. hospital) data were from anonymous Landscape Assessment surveys voluntarily completed by National Cancer Institute Community Oncology Research Program sites. Outcomes included the proportions of underrepresented patients enrolled on a trial. Also, we compared the level of agreement between patient populations served by sites versus enrolled on a trial. Of 9,015 patients enrolled across 12 trials, 18% were aged ≥65 years old, 12% were Black, 15% were Medicare beneficiaries, and 15% were rural residents. These numbers are quite low. Most individuals who enrolled on trials were aged 46-64 (57%), White (82%), privately insured (72%), and were urban residents (85%). Trial design, specifically if a trial used randomization or multiple drugs, did not appear to influence enrollment of underrepresented groups. Moderate levels of agreement were found between Black patients served versus enrolled while low or no agreement for Medicare beneficiaries and patients aged ≥65. This means that patient populations sites served did not typically match the patients enrolled on trials. More research is needed to understand how sites enroll underpresented patients onto trials.
Keywords
Background
Therapeutic cancer clinical trials offer benefits to patients including access to novel modalities or treatments and being closely monitored by cancer providers to ensure safety. 1 However, in the United States (US), certain patient subgroups are underrepresented in trials and thus miss out on these benefits. Underrepresented patients in trials include older adults (i.e., ≥65 years of age), Black or African American (henceforth referred to as Black) patients, and rural residents.2,3 Reasons for this underrepresentation include individual-level factors, such as mistrust in the US healthcare system, access to care, ability to pay, and/or transportation barriers. 4 While addressing individual-level factors is important to overcoming underrepresentation, so too may be factors associated with trials and site characteristics. However, there is a dearth in knowledge of how both trial-level factors, such as eligibility criteria, and site-level factors, such as the locations of oncology practices and makeup of these practices, affect enrollment.5,6
Firstly, for a patient to enroll in a clinical trial, a trial must be available at the hospital where he/she is seeking care.7,8 Depending on the patient population that sites serve, sites may be unintentionally exacerbating underrepresentation of certain patient populations. For example, our previous study found that trial sites are often located in areas with lower levels of social vulnerability. 5 Furthermore, a study by Ivory and colleagues found that trial sites located in racially diverse areas were associated with enrolling more Black patients. 9 However, there is a lack of data on site-specific demographics and how this can influence clinical trial enrollment.
Secondly, although it is known that strict and heterogeneous eligibility criteria (i.e., eligibility criteria are often not similar between trials)10-12 of US clinical trials affect patient enrollment, less is known about how eligible patients may choose to enroll or not enroll in a trial based on specific trial characteristics. 6 For example, certain patients may choose to not enroll on a randomized controlled trial due to the chance of not receiving the trial drug as some patients may see the trial drug as “better” than the standard-of-care.13-15 Furthermore, the complexity of a trial treatment regimen, such as the number of drugs and frequency of clinic visits, involved in a trial may affect enrollment decisions. Therefore, the objective of this study was to understand how trial characteristics and demographics of the patients served by the sites may affect the likelihood of underrepresented patients with breast cancer enrolling in breast cancer therapeutic clinical trials. All trials in our study were sponsored by the ECOG-ACRIN Cancer Research Group (ECOG-ACRIN or EA). EA is a scientific organization that partners with both academic and community cancer centers and hospitals to conduct cancer research, including clinical trials. 16
Methods
Study Design and Participants
This retrospective, pooled cross-sectional study used patient-, trial-, and site-level data. Patient-level data included de-identified breast cancer therapeutic clinical trials sponsored by ECOG-ACRIN (EA) that enrolled patients with cancer from 2002-2020 with cancer-related outcomes of interest. Included patients were selectively chosen based on having complete data on age, race and ethnicity, insurance status, and had a US zip code. All patient details have been de-identified. All trials enrolled US residents, while some enrolled residents of other countries. We only included US residents and kept the trials that enrolled both individuals in the US and other countries. Trial-level data were from an EA internal database and additional information were extracted from ClinicalTrials.gov via the National Clinical Identifier (NCT) number. Site-level data were from de-identified survey data from the Landscape Assessment (LA), in which National Cancer Institute Community Oncology Research Program (NCORP) sites provided information about their hospital and the patient populations they serve. Sites that are a part of NCORP were asked to voluntarily complete the 2022 LA survey separate from this study project. Of note, EA is a Research Base within NCORP, therefore NCORP designated sites or hospitals can participate in EA-sponsored clinical trials. De-identified site identification numbers were used to link datasets. This study was approved by the University of Alabama at Birmingham Institutional Review Board (IRB-300011306). Informed consent was not applicable to our study. The reporting of this study conforms to the STROBE guidelines. 17
Outcome: Proportion of Patients Enrolled on a Trial
Categorization of Patient Populations
Participant age at trial enrollment was categorized into ≤45, 46-65, and ≥65 years. Participant race and ethnicity was categorized as White, Black, and other (Hispanic, Asian, Native American, Indian, other race/ethnicity). Participant insurance status at time of enrollment was categorized into Medicaid (including individuals dual eligible), Medicare (including individuals with supplemental private insurance), none, private insurance, and other (military, Veterans Affairs[VA]). Medicaid eligibility varies by state but typically includes individuals who have low incomes. Medicare beneficiaries must be ≥65 years or have certain health conditions. Individuals are considered dual eligible if they are eligible for both Medicaid and Medicare. Military and Veterans Affairs insurance plans are typically for those who have served or are serving in the US miliary system but can include family members. Using patient zip code data, we categorized patients as rural or urban residents according to the Rural-Urban Commuting Area (RUCA) codes. 18 The enrollment or recruitment period was calculated as the difference between the last enrollment month from the first. Enrollment month was anchored to the first month a patient was enrolled according to each trial.
Trial-Level Outcomes
For trial-level analyses, we calculated the proportion of clinical trial enrollment over time for the following patient populations: (1) older adults (aged ≥65 years); (2) Black patients; and (3) rural residents, as these are underrepresented patient populations. We calculated the proportion of Medicare beneficiaries for descriptive purposes as this is highly associated with patients ≥65. Well-represented patients included middle-aged adults, White patients, privately insured individuals, and urban residents.
Site-Level Outcomes
For the site-level analyses, we categorized proportion of clinical trial enrollment for the following patient populations: (1) Black patients; (2) Medicare beneficiaries; and (3) older adults (aged ≥65 years). We categorized enrollment proportions into the following categories: 0-5%, 6-10%, 11-30%, 31-49%, or ≥50%. We chose these response categories as they were necessary to be in the same format as the site-level exposures for our statistical analyses.
Exposures
Trial-Level Exposures
Trial-level variables of interest included randomization (randomized, non-randomized) and the number of drugs or biologicals in the experimental arm.
Site-Level Exposures
Sites reported the estimated proportion of new Black oncology patients, estimated percentage of Medicare coverage, and the estimated proportion of new breast cancer patients ≥65. All variables were categorized into 0-5%, 6-10%, 11-30%, 31-49%, ≥50%, or not reported. The “not reported” category was used for descriptive purposes only and not in the analysis. These response categories were created by the survey designers of the Landscape Assessment.
Additional Variables of Interest
Trial-Level Variables
Additional trial-level variables included site identification number, study status at time of data capture (active, not recruiting; completed; recruiting; terminated), recruitment time period, countries of enrollment (USA only, USA & international), number of sites that enrolled per trial, study phase (1,2; 2; 3), intervention model (parallel, single group), radiation as study procedure either in experimental or standard-of-care arm (yes, no), surgery as study procedure either in experimental or standard-of-care arm (yes, no), stage as eligibility criteria (1, 2, 3; 2, 3; 3, 4; 4), recurrence as eligibility criteria (yes, no), hormone status as eligibility criteria (any, positive, negative), and human epidermal growth factor receptor 2 (HER2) status as eligibility criteria (any, positive, negative).
Site-Level Variables
Site-level variables included number of medical oncology providers, total new breast cancer cases, critical access site (yes, no, not reported), type of ownership (hospital, clinic, or physician practice owned by a large regional/multi-state health system; independently owned [single hospital, small regional network, or independent clinic/physician practice]; and non-profit organization; publicly owned [state, county, city]; university owned, VA/military).
Statistical Analysis
Descriptive statistics were calculated using frequencies and percentages for categorical variables. For continuous variables we calculated medians and interquartile ranges (IQR). As site-level analysis used data from sites who completed the LA survey, we compared patient populations between the sites who did and did not complete the LA survey. Differences in characteristics for participants whose trial site did and did not complete the LA survey were calculated using measures of effect size such as Cohen’s d for numerical characteristics (i.e., the standardized mean difference; small: 0.2, medium: 0.5, large: 0.8) or Cramer’s V for cross tabulations. V of 0.1 is considered a small effect, 0.3 a medium effect, and 0.5 a large effect when comparing across two categories; 0.1 a small effect, 0.25 a medium effect, and 0.4 a large effect when comparing across more than two categories. 19
Trial-Level Analyses
Means, 95% confidence intervals (CI), and predicted proportions were estimated from repeated fractional regression models which evaluated the association between trial-level factors (randomization and number of drugs or biologics) and the proportion of enrollment for (1) patients aged ≥65, (2) Black patients, and (3) rural patients. All models contained the following variables unless the variable was the outcome of interest: proportion of patients aged ≥65 enrolled, proportion of Black patients enrolled, proportion of rural patients enrolled, randomization, number of drugs and biologics, cancer recurrence eligibility criteria, country of enrollment, and enrollment month. Due to the high level of collinearity between trial-level variables, we used a stepwise approach assessing variance inflation factors values (i.e., collinearity) and research from existing literature to determine which variables to include and focus on in our trial-level analyses. We used the proportion of patient populations enrolled monthly and therefore our models accounted for the repeated nature of the data.
Site-Level Analyses
Furthermore, to understand concordance between Black patients, Medicare beneficiaries, and older adults that sites reported they served versus who they enrolled into a trial, we ran agreement analyses with weighted Kappa statistics and Mantel-Hanzel Paired tests 20 for ordinal variables (row-means differ). Weighted Kappa estimates the level of agreement between two ordinal variables that have identical categories. The weighted Kappa statistic is interpreted as follows: 0-0.20 no, 0.21-0.39 minimal, 0.40-0.59 weak, 0.60-0.79 moderate, 0.80-0.90 strong, and >0.90 almost perfect agreement. Any negative values represent the level of disagreement. The null hypothesis for Mantel-Hanzel Paired tests is that that there is no difference in the distribution of paired ordinal data tabulations; meaning there is no difference between who sites served versus who they enrolled on a trial. The statistical significance level was set to 0.05. Analyses were performed using SAS© software, version 9.4 (SAS Institute, Cary, NC).
Results
Patient, Trial, and Site Characteristics
Patient-Level Characteristics
Patient Demographics and Clinical Characteristics of Overall Sample (N=9015), Patients Whose Site Completed the Landscape Assessment Survey (n=1186), or did not Complete the Survey (n=7829)

IQR=interquartile range. Other racial group include Hispanic, Asian, Native American, Indian, and other. Other insurance types includes military and Veteran Affairs.
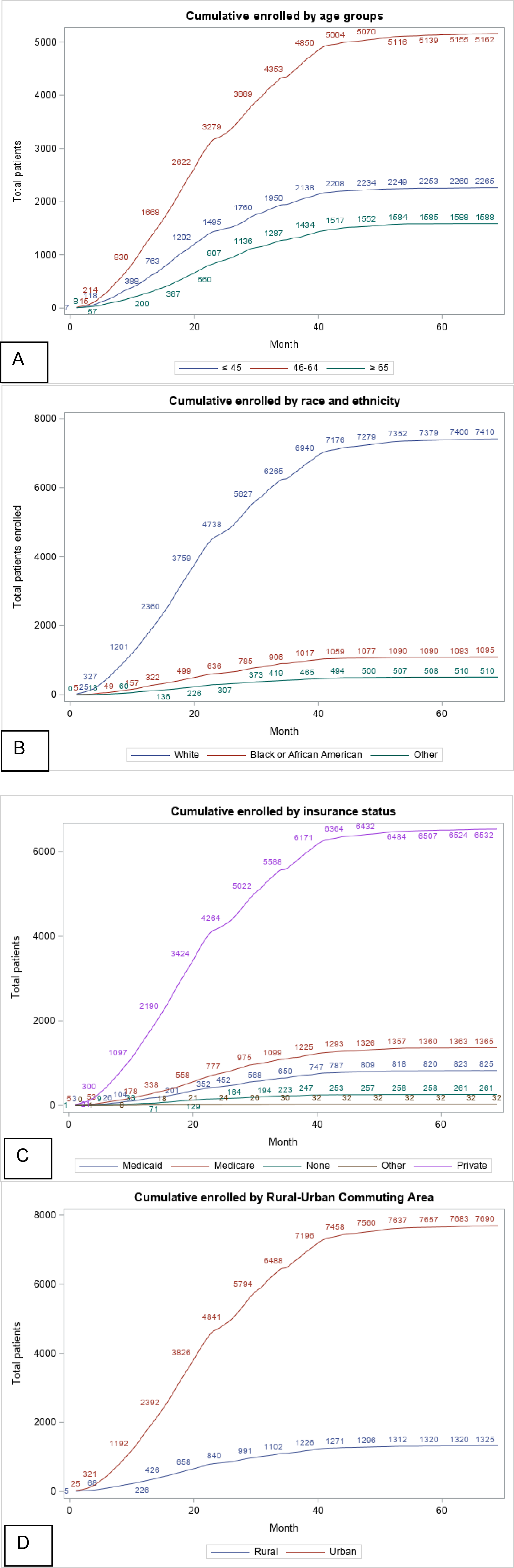
Cumulative enrollment of patients enrolled in 12 trials across recruitment months by (A) age groups; (B) race and ethnicity; (C) insurance status; (D) Rural-urban commuting areas
Trial-Level Characteristics
Overall Trial Characteristics (N=12)

IQR=interquartile range; HER2=human epidermal growth factor receptor 2.
Site-Level Characteristics
Overall Self-Reported Site Characteristics From the 2022 Landscape Assessment Survey (N=115)

IQR=interquartile range; VA=Veteran’s Affair.
Trial-Level Analyses: Estimated Proportion Enrollment Numbers for Underrepresented Patient Populations
Model-Estimated Proportions of Enrollment and 95% Confidence Intervals for Patients Aged ≥65, Black, and Rural by Trial Characteristics (N=435)

All models contained the following variables, unless the variable was the outcome variable of interest: proportion of patients aged ≥65 enrolled, proportion of Black or African American patients enrolled, proportion of rural patients enrolled, randomization, number of drugs and biologics, cancer recurrence eligibility criteria, country of enrollment, and enrollment month.
CI=confidence intervals.

Predicted proportion of patient populations enrolled throughout the studies’ enrollment time period for (A) patients aged ≥65; (B) Black or African American patients; (C) rural patients (N=435)
Site-Level Analyses: Comparing Patient Populations Served versus Enrolled on a Breast Cancer Clinical Trial
Estimated Weighted Kappa Statistics and Mantel-Hanzel Paired Test Assessing Level of Agreement Between Site-Reported Patient Population Served and Patients Populations Enrolled on Trial
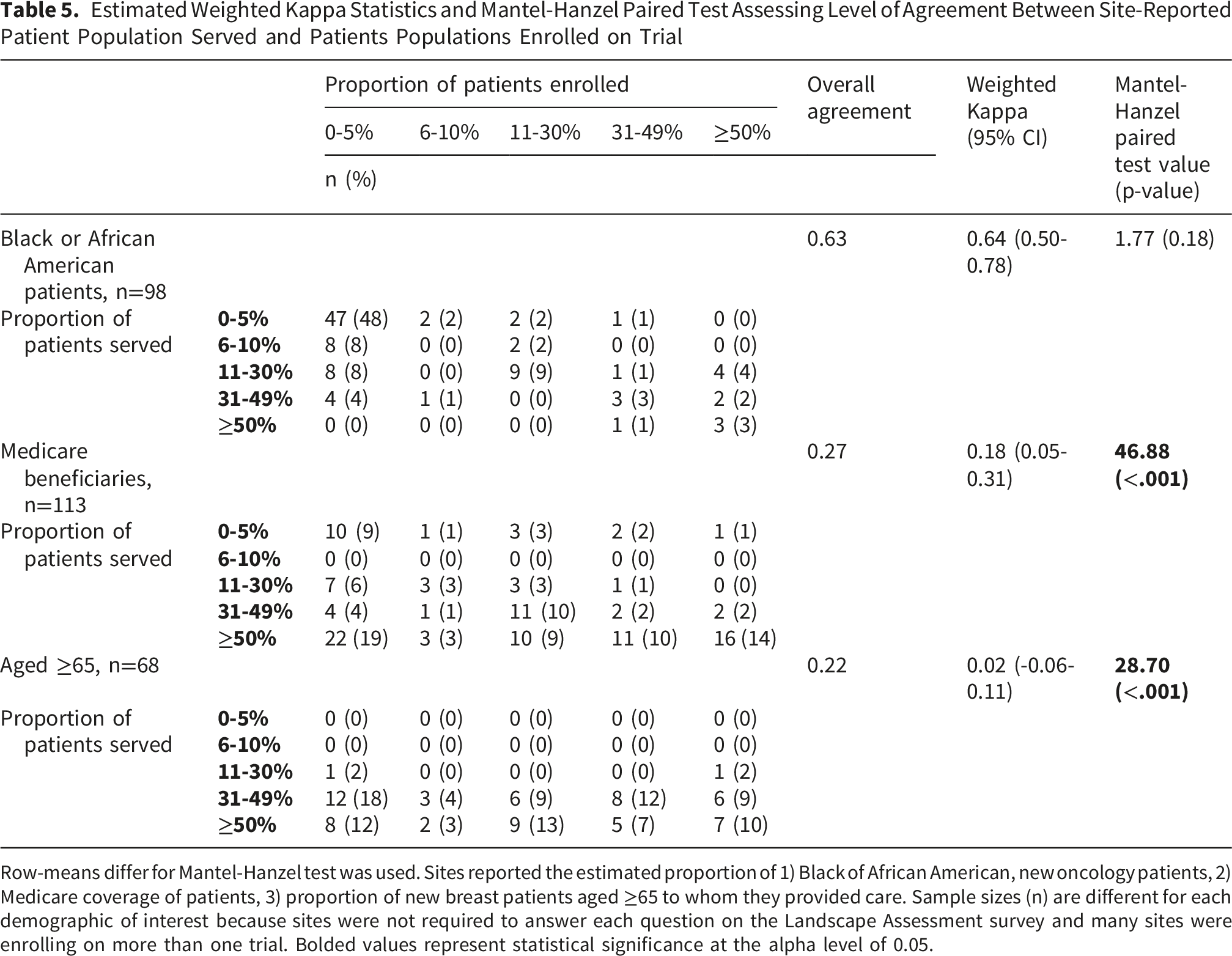
Row-means differ for Mantel-Hanzel test was used. Sites reported the estimated proportion of 1) Black of African American, new oncology patients, 2) Medicare coverage of patients, 3) proportion of new breast patients aged ≥65 to whom they provided care. Sample sizes (n) are different for each demographic of interest because sites were not required to answer each question on the Landscape Assessment survey and many sites were enrolling on more than one trial. Bolded values represent statistical significance at the alpha level of 0.05.
Discussion
This paper assessed enrollment patterns among underrepresented patient populations in the United States enrolled in ECOG-ACRIN-sponsored breast cancer therapeutic clinical trials. We found that proportions of enrollment differed from the reported percentage of patients served at trial sites for Medicare beneficiaries and patients aged ≥65. However, we found moderate levels of agreement between Black patients served versus enrolled at trial sites. Furthermore, our study found that few sites were enrolling more Black patients above the percentages they reported to serve. More research is needed to understand enrollment processes that are implemented at these sites in order to adapt them to other trial sites. These adaptations could improve clinical trial enrollment among Black patients.
We also found most trial sites served large percentages of older patients; however, they did not enroll equal or similar proportions of older patients on studies. Older adults, when compared to younger patients, may be more likely to not participate in trials due to being ineligible. More often older adults have multiple comorbidities, which may result in being excluded from a trial,21,22 even though half of patients with cancer are diagnosed as older adults. 23 In December 2025, the Food and Drug Administration (FDA) released updated guidance regarding ways to enhance participation in trials with a focus on broadening eligibility criteria. 24 They highlight that trialists should enroll patients who are representative of the cancer population who will receive the drug in practice without increasing the risk of adverse events, as others have noted as well. 25 This is especially important as investigational drugs or regimens, once approved, will be given to individuals who did not participate in clinical trials, which can lead to a knowledge gap in how to treat this patient population.
We also found that enrollment patterns differed between patient populations; our findings show that sites typically enroll middle-aged, White, privately insured, and urban dwelling patients at a faster rate than patients considered underrepresented. Additionally, we found that the median number of patients per trial per site was 2 patients, so it is likely that sites are focusing first on enrolling patients deemed “easy to enroll” (i.e., well-represented patients). For example, a systematic review by Howerton and colleagues found that a barrier to enrolling patients onto trials was providers’ attitudes, specifically their views on which patients would adhere to the study protocol. 26 This negative perspective among providers (i.e., a form of implicit bias) can negatively affect enrollment of underrepresented patients.27-29 However, a dermatological clinical trial found that by promoting cultural competency and including sites with diverse investigators, they increased recruitment timing and the enrollment of patients of color compared to historical trials conducted by the same study team. 30 Therefore, implicit bias and cultural competency trainings, along with intentionality in enrolling diverse patients, are needed, especially among sites that serve many diverse patients.
Additionally, we found that aspects of the trials did not appear to affect enrollment of patients who are underrepresented in trials, specifically randomization and number of drugs. One reason for this may be that patients who enrolled on trials were already willing to participate compared to patients who declined participation. Therefore, trials should be conducted on what is scientifically most interesting and utilize complex trial designs if necessary as it does not deter patients from participating. In fact, underrepresented patients may be drawn to certain aspects of trials. For example, Unger and colleagues found that diverse patient populations are drawn to federally-sponsored trials for the research questions these studies ask and the various modalities utilized to answer these questions. 31 Furthermore, to enroll diverse patient populations, trials must first be available at diverse sites. Having more diverse trial sites that offer cancer clinical trials increases quality care for all patients both partaking in the trial and future patients with cancer. 32 Buse and colleagues developed “A Framework for Assessing for Clinical Trial Site Readiness,” which includes the following domains: research team, infrastructure, study management, data collection and management, quality oversight, and ethics and safety. 33 Buse and colleagues advocate for the need to include and engage with diverse patient populations among each domain. However, most importantly noted in this readiness framework is that, for sites to begin offering trials, they must have the resources and infrastructure to do so, including providers & staff, time, training, and budget.34-36 Therefore, systematic changes are needed to increase clinical trial availability at sites, especially sites that serve diverse and underrepresented patient populations.
This paper contains several limitations. Screening data were unavailable therefore, we were unable to ascertain the true denominator of eligible patients, patients offered the trial, and patients who declined enrollment. It is important to know the patient demographics at these various clinical trial process steps to understand where underrepresented patient populations are dropping off and therefore develop tailored interventions. Furthermore, without screening data we were unable to determine reasons patients declined enrollment as trial aspects could have been one of the reasons. Additionally, while the Landscape Assessment data was completed in 2022, we assumed that the proportion of patient populations served at sites were consistent throughout time. Furthermore, as this study included patients who enrolled onto ECOG-ACRIN-sponsored breast cancer studies, it may not be generalizable to other cancer types or other breast cancer studies. Furthermore, the Landscape Assessment survey included only a subset of trial sites and therefore may not be generalizable to other trial sites. Finally, as we did not perform a sample size calculation, we acknowledge that the limited number of patients in our study may affect the statistical significance of our results as studies with small sample sizes typically are underpowered to show any statistical significance.
Conclusions
Using patient-, trial, and site-level data, we found that breast cancer clinical trials sponsored by ECOG-ACRIN enrolled small numbers of underrepresented patient populations. We also found that randomization and number of drugs involved in a trial appeared to have minimal impact on enrollment for underrepresented patients. Finally, sites that offer trials to their patients often do not enroll consistently with the patients they serve. Understanding more about how sites enroll underrepresented patient populations, especially those sites that serve and/or enroll large numbers of underrepresented patients, is important to increasing enrollment of diverse patients. Furthermore, more research is needed on how best to utilize intentional site-level strategies to enroll diverse patients.
Footnotes
Acknowledgements
We would like to acknowledge JoRean Sicks and Seth Bittig with their assistance in providing the Landscape Assessment survey data.
Ethical Considerations
This study was approved by the University of Alabama at Birmingham Institutional Review Board (IRB-300011306) on June 12, 2023.
Author Contributions
Study concept/design: GR, CPW, NEC.
Provision of study material or patients: FZ, JWL
Data collection/assembly: FZ.
Data analysis and interpretation: NEC, CPW, AA, GR.
Manuscript writing: All authors.
Final approval of manuscript: All authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was conducted by the ECOG-ACRIN Cancer Research Group (supported by the National Cancer Institute of the National Institutes of Health: UG1CA189828) and the O’Neal Comprehensive Cancer Center at the University of Alabama at Birmingham. Dr. Caston is supported by the National Cancer Institute’s National Research Service Award sponsored by the Lineberger Comprehensive Cancer Center at the University of North Carolina at Chapel Hill (T32 CA116339).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available through ECOG-ACRIN. Data requests must be made to ECOG-ACRIN.
Disclaimer
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Prior Presentations
Previously presented at the ECOG-ACRIN Fall Group Meeting 2024.
