Abstract
Introduction
Cancer clinical trials represent the “gold standard” for advancing novel cancer therapies. Optimizing trial participation is critical to ensuring the generalizability of findings across patients, yet trial enrollment rates, particularly among minority and socioeconomically disadvantaged populations, remain suboptimal.
Methods
We conducted in-depth interviews with oncologists at a large academic medical center to explore their (1) attitudes and perceived barriers to offering clinical trials to minority and socioeconomically disadvantaged patients, and (2) recommendations for improving the enrollment of minority and socioeconomically disadvantaged patients in cancer clinical trials.
Results
Of 23 medical oncologists approached, 17 enrolled (74% response rate; mean age = 47; female = 42%; White = 67%). Content analysis revealed several barriers to enrollment: (1) ethical dilemmas; (2) ambivalence about trial risks and benefits; and (3) concern about patient well-being. Concerns about the legitimacy of informed consent, perceived lack of equipoise, and fear of personal bias influenced clinicians’ decisions to recommend trials during treatment discussions. Concerns about creating an imbalance between trial risks and benefits among patients with high-level needs, including patients with literacy, psychiatric, and other socioeconomic vulnerabilities, impacted clinicians’ enthusiasm to engage in trial discussions. Clinicians identified patient, provider, and system-level solutions to address challenges, including increasing patient and clinician support as well as involving external personnel to support trial enrollment.
Conclusion
Findings reveal multi-level barriers to offering cancer clinical trials to underrepresented patients. Targeted solutions, including system level changes to support clinicians, patient financial support, and implementation of clinical trial navigation programs were recommended to help reduce access barriers and increase enrollment of underrepresented patients into cancer clinical trials.
Introduction
Significant disparities exist in cancer care, with certain populations facing increased risk for poorer outcomes, including worse survival.1-3 Specifically, racial/ethnic minorities and socioeconomically disadvantaged populations bear a disproportionate burden of disease, demonstrating greater morbidity and lower 5-year survival. 4 Access to clinical trials represents a critical factor contributing to persistent disparities in cancer outcomes among these individuals.
Therapeutic clinical trials have helped to markedly improve cancer therapies. 5 A “gold standard” of clinical research, clinical trials are referenced for the approval of new treatments and are used to create treatment guidelines. 6 Findings from clinical trials are thus critical to informing the care of individuals with cancer however adequate representation of all patients is crucial to ensure the generalizability of trial findings. Despite national mandates, ethnic/racial minority and socioeconomically disadvantaged patients remain underrepresented.7,8 Recent estimates suggest that as few as 2-5% of adults with cancer participate in National Cancer Institute-sponsored trials,9,10 with older, racial/ethnic minority, and rural populations being least represented.11-13 This is despite evidence demonstrating similar levels of willingness to participate amongst racial/ethnic minority and white patients.14,15 Exclusion or under-representation of these vulnerable groups in cancer trials introduces bias in evidence-based treatment and challenges the applicability of effective treatments to all patients treated in real-world practice.
The past few decades of research have placed increasing attention on addressing barriers to trial enrollment amongst under-represented groups. While enrollment barriers are multifactorial, 16 clinician referral has been documented to strongly impact patients’ decision to participate in cancer clinical trials. 17 Studies report clinician reluctance with offering clinical trials to their patients as an option, yet few have examined the reasons underlying their perspectives. To address this need, we conducted in-depth interviews with medical oncologists at a large academic medical center to explore their attitudes barriers to offering clinical trials to minority and socioeconomically disadvantaged patients. We further explored their recommendations for improving the enrollment of these underrepresented patients in cancer clinical trials.
Materials and Methods
We conducted this study at the Massachusetts General Hospital (MGH), Boston. The MGH has a robust clinical trial program with an annual enrollment rate of approximately 1000-2000 patients to cancer clinical trials. Among these, approximately 3% of enrolled patients identify as Black or African American, and about 4% identify as Hispanic or LatinX. This reflects the general patient population seen at the cancer center. Study procedures were approved by the Mass General Brigham Institutional Review Board (IRB) (Partners Healthcare) (reference number: 2016P000871/PHS). Additional study information can be found in the STROBE document. 18
To inform the development of a semi-structured interview guide, we reviewed the literature on barriers to clinical trial enrollment to identify areas for further exploration. We refined the interview guide with our co-investigative team, consisting of medical oncologists, clinical psychologists, behavioral scientists, and health services researchers. We cognitively tested and refined the interview guide with five medical oncologists to ensure questions accurately explored domains of interest. The final interview guide included the following domains: (1) attitudes and barriers to offering clinical trials to underrepresented patients, and (2) recommendations for improving the enrollment of underrepresented patients into clinical trials. Although our overarching objective was to explore practices related to the enrollment of underrepresented populations, our questions purposively probed for patient characteristics that made it more challenging for clinicians to offer a clinical trial in efforts to encourage openness. Additional probes were used to gain more in-depth information about why it was challenging to offer trials to these patients.
We identified participants from a centralized cancer center listserv that detailed the number of patients enrolled per medical oncologist over the prior year. Utilizing a stratified sampling approach based on enrollment rate (i.e., high enrollers: ≥10 vs low enrollers: <10 patients), we randomly selected a subset of “high” and “low” enrolling clinicians and invited them via email to participate in an individual, 30-minute interview.
The lead investigator (GP) consented (written consent was obtained) and conducted interviews with participants in-person or by phone from 2016-2017. GP is a health psychologist trained in qualitative interviewing and coding with no relationship to the participants but does interact with patients in the Cancer Center. Participants also completed a 5-minute questionnaire to provide sociodemographic data and clinical practice information. All participants were offered a $5 gift card for the hospital’s coffee shop.
Interviews were audio recorded, professionally transcribed using a HIPAA-compliant transcription service (transcribeme.com), and analyzed in an iterative process using the Framework method. 19 The research team was comprised of 3 researchers (GP, AB, SC) who were trained in qualitative methods. An expert in qualitative methodology oversaw the qualitative analysis (EP). One researcher (GP) first reviewed each transcript for quality and consistency, developing an initial coding framework inductively using open coding. Two researchers (SC, AB) separately reviewed each transcript to familiarize themselves with the data and to refine the preliminary coding framework based on emergent themes and subthemes. Codes were guided by the raw data and interview guide. Transcripts were read and re-read, providing flexibility to test the codes and clarify the coding framework as the reviews progressed. The final framework was entered into NVivo12 qualitative analysis software; 20 two investigators (SC, AB) independently coded each interview. Coders examined discrepant, unexpected or unclear data with GP and EP until resolution. Kappa coefficients were generated to ensure reliability (Kappa>0.80); discrepancies were resolved through discussion with the lead investigator and via comparison to raw data. We achieved a final agreement rating of Kappa = .94. With our sample size of 17 clinicians, we were able to achieve thematic saturation. 21
Results
Characteristics of Clinicians (n = 17).

Interview Findings: Categories, Themes, Subthemes, and Supporting Quotations.
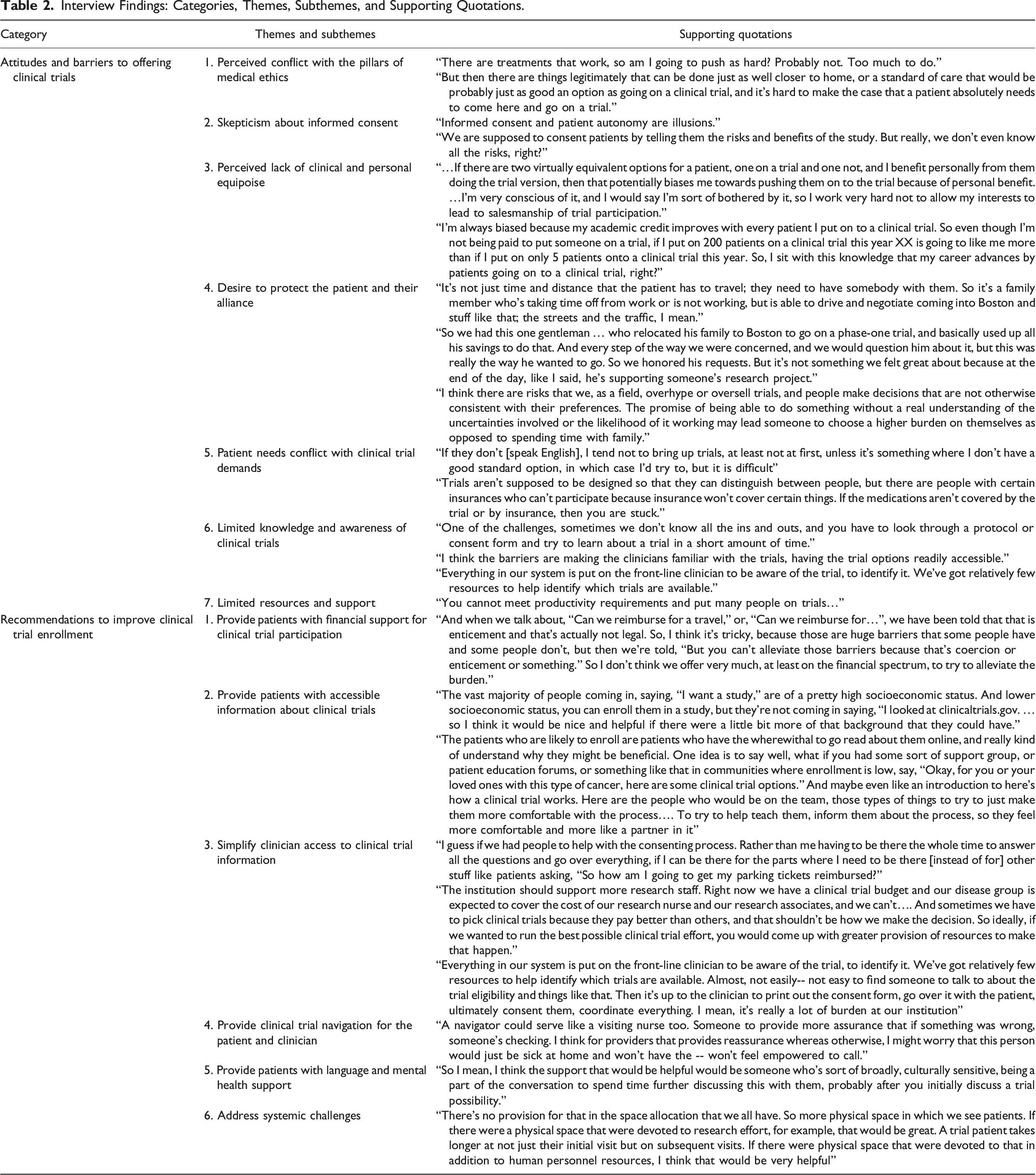
Attitudes and Barriers to Offering Cancer Clinical Trials
1. Perceived conflict with the pillars of medical ethics. While clinicians viewed clinical trials favorably, many experienced ambivalence when considering clinical trials for their patients, citing multiple ethical conflicts. Several shared concerns about exposing their patients to potentially harmful, costly procedures and treatment delays, especially when the outcome of clinical trials was uncertain. Given this, clinicians questioned the net benefit of offering clinical trials in lieu of standard care. Many worried that discussions added confusion to an already complex conversation about treatment. These concerns were heightened in situations where patients presented with certain vulnerabilities, such as limited health knowledge or limited English proficiency. Altogether, these practices were deemed to disadvantage patients’ overall safety and health, which conflicted with their intentions to optimize outcomes. 2. Skepticism about informed consent. Many clinicians questioned the parameters of informed consent. Specifically, several participants questioned patients’ capacity to make informed treatment decisions when clinicians themselves lacked understanding of clinical trial procedures and felt uninformed of the full scope of risks potentially incurred with clinical trial therapies. One interviewee described the process as “under-informed consent by a biased person.” Given these concerns, clinicians worried that advocating for clinical trial participation risked jeopardizing their patients’ autonomy. Many thus reportedly avoided clinical trial discussion or endeavored to discuss clinical trials without overselling. 3. Perceived lack of clinical and personal equipoise. Clinicians’ concerns were magnified by skepticism around the concept of equipoise. Indeed, few considered clinical trial therapies to be co-equal clinical options due to unknown side effects and toxicities. Several clinicians thus considered clinical trials as an alternative to care only after exhausting standard therapy options. At times when clinical equipoise existed, many voiced caution about offering clinical trials due to secondary benefits related to academic promotion, salaries, and social accolade. This prompted clinicians to question their judgement when making clinical trial recommendations. Several reportedly approached clinical trial discussions with neutral language to avoid influencing patients’ decisions. For others, concerns about productivity gaps (arising from extended encounters occurring with clinical trial discussion) posed a barrier to clinical trial discussion. 4. Desire to protect the patient and their alliance. Clinicians’ connectedness to their patients increased concerns about asking for “more” by offering clinical trials as a treatment option. Knowing that patients relied on them to make treatment decisions led some clinicians to worry about influencing patients’ decision to enroll. On those occasions wherein the trade-off between clinical trial enrollment and standard therapy was deemed unreasonable, either due to time constraints or perceived patient burden, clinicians either avoided clinical trial discussions altogether, tempered their enthusiasm when offering clinical trials as an option, or minimally shared clinical trial information to protect their patients. Some worried about disrupting the continuity of care, suggesting that patients may feel abandoned if care is transitioned to a clinical trial physician. Wanting to establish and/or maintain patient trust was a recurring theme, with some clinicians describing concerns about fueling medical mistrust if clinical trials were introduced early in the relationship. 5. Patient needs conflict with clinical trial demands. Nearly all clinicians agreed on a few patient characteristics that made them question offering clinical trials to their patients. In effect, clinicians expressed greater ambivalence when presented with patients perceived as having higher-level needs that conflicted with clinical trial demands. This included patients who faced higher travel burden, greater financial concerns, poor health literacy, limited cognitive capacity, greater mental health concerns, limited English proficiency (LEP), and poor social support. Specifically, clinicians worried that patients with access challenges, such as those who lived far or who were financially insecure, endured greater financial hardship and fewer benefits if enrolled in clinical trials. Concerns about patients’ literacy, cognitive capacity, and mental health challenged clinicians’ perceptions of patients’ self-agency and autonomy; these characteristics purportedly interfered with patients’ ability to comprehend the subtle nuances of clinical trials (“do they really know what they are getting themselves into”). These concerns further fueled clinician worries about letting their own personal biases/gains influence their decision to discuss clinical trials. Compelled to protect these patients, clinicians often decided to forego clinical trial discussion. 6. Limited knowledge and awareness of clinical trials. Clinicians reported feeling overwhelmed by the revolving landscape of clinical trials. Several struggled with knowing which clinical trials were available, open, and applicable; many also felt ill-equipped to review potential clinical trials due to lack of knowledge about the logistics or scientific underpinnings of the therapies under investigation. Lacking time to sift through studies, these reservations further deterred clinical trial discussion. 7. Limited resources and support. Time demands in the context of limited resources were a prominent theme. Clinicians commonly reported balancing the need to negotiate their time between clinical trial accrual, engaging in clinical trials, and meeting their clinical demands given a lack of support staff. Post-enrollment documentation, regulatory and administrative demands further discouraged patient enrollment; this practice did not fit into the time allotted for a clinical visit, and clinicians experienced too many systemic obstacles to scheduling longer appointments to accommodate clinical trial discussions. Time constraints were particularly magnified by patients who presented with high-level needs, such as those with visual/auditory impairments or limited English proficiency. If time was limited, needs were high, and resources were low, clinicians reportedly avoided clinical trial discussion altogether. One participant mentioned the experience of burnout in contributing to their reluctance to enroll.
Recommendations to Improve Clinical Trial Enrollment
While clinicians voiced many clinical trial concerns, they offered several systems, patient and clinician-level solutions to overcome observed challenges. 1. Provide patients with financial support for clinical trial participation. Clinicians underscored the need to minimize patient burden to offset perceived risks. Many reported that patients should receive resources to offset monetary costs associated with clinical trial participation, such as mileage reimbursement, parking/cab vouchers or travel accommodations. Some clinicians worried financial remuneration could also be misperceived as coercive. 2. Provide patients with accessible information about clinical trials. Participants stressed the need to educate patients about clinical trials. Several recommended providing patients with clinical trial information prior to a clinical encounter to prepare them to engage in an informed discussion, reduce concerns about time burden and physician bias, and lessen the confusion associated with lengthy consent forms. Some suggested providing language-concordant material, using videos, or creating patient education forums to demystify the clinical trial process, share knowledge about why clinical trials are done and how they function, and encourage questions. 3. Simplify clinician access to trial information. Participants sought information to increase their awareness of available clinical trials and support their efforts to accrue patients. Specifically, they desired a regularly updated database that pulls applicable clinical trials based on patients’ clinical profile and that summarizes clinical trial information, eligibility criteria, and available slots to streamline the process. 4. Provide clinical trial navigator for the patient and clinician. Nearly all participants alluded to the benefits of including an impartial individual that blends the role of navigator, clinical trialist, and medical interpreter. Essentially, clinicians sought opportunities to create distance from the clinical trial enrollment process. Specifically, they described wanting an individual to identify appropriate clinical trials, review clinical trial information with prospective patients, orient the clinician to the clinical trial, and connect patients with essential resources once enrolled. This person would ideally have a general understanding of oncology and clinical trials while also maintaining understanding of patients’ cultural needs. 5. Provide patients with language and mental health support. Clinicians suggested increasing the availability of in-person interpreters given the difficulties with using virtual interpreters. A few recommended integrating psychiatric consultation services or social workers to assist with care coordination when presented with patients with mental health needs. 6. Address systemic challenges. Clinicians called for system-level changes to facilitate clinical trial participation, including scheduling changes to permit lengthier visits, providing space, and addressing productivity gaps would help overcome common obstacles to enrolling patients.
Discussion
Our findings extend the current body of research by highlighting perceived ethical dilemmas as a critical barrier to the enrollment of underrepresented patients into cancer clinical trials. A key finding was the apparent moral distress and uncertainty clinicians experienced when considering clinical trials for patients they perceived as vulnerable. Concerns about the legitimacy of informed consent, perceived lack of clinical equipoise, and fear of personal bias due to conflicting interests (e.g., academic advancement) influenced their decision to present clinical trials as an option during treatment discussions. Of note, studies suggest that individuals with limited English proficiency, educational resources, or other characteristics that necessitate medical decision-making support may be particularly impacted by oncologist preferences to make decisions on their patient’s behalf.10-12,22-24 In this study, concerns about patient safety were heightened if clinicians lacked an understanding of available clinical trials, believed referrals gained them secondary benefits, or if they had concerns about their patients’ decisional capacity due to identified vulnerabilities. Ultimately, clinicians’ desire to protect their patients outweighed their perceived duty to promote patient autonomy in decision-making. These findings are noteworthy, as perceived ethical dilemmas can increase clinicians’ risk for chronic distress and burnout, 24 which can have negative downstream effects on patient care.
While ethical dilemmas are common in healthcare,25,26 their impact on clinicians’ decision-making process and equity in clinical trial access is less well understood. This challenge of balancing professional values of patient autonomy with clinical beneficence for vulnerable patients has been well-documented in other contexts; for example, oncologist comfort with making decisions on behalf of late-state cancer patients has been reported, and it was associated with greater medical utilization at end of life. 27 It’s important to understand these ethical dilemmas in the context of increasing demands for clinicians to maintain productive engagement with research to ensure their own professional advancement. 28 Clinicians working in academic medical centers must constantly balance responsibilities to patients, research collaborators, and trainees and often receive financial and professional advancement incentives to maintain a high level of productivity across multiple domains (e.g., securing grant funding, publishing, responding to specialized patient referrals).29,30 Though providers are not directly compensated for each individual patient who enrolls in a clinical trial, they may receive indirect professional benefits from rapid clinical trial recruitment rates, data collection, and publishing. Due to mounting pressures and increasing clinical caseloads, the literature suggests that oncologists may struggle from compassion fatigue when engaging in complex decision-making processes. 31 This may impact clinical decisions, patient care and patient engagement.
Clinical Implications
It may be helpful to place these findings in the context of the ethical literature. Emanuel and colleagues offer a universal framework for evaluating the ethics of clinical research. 32 They assert that 7 key principles, which include informed consent, favorable risk-benefit ratios, and scientific validity, must be met for research trials to be considered ethical. While existing cancer clinical trials may objectively meet these standards, our findings suggest that clinicians’ interpretation of these principles may vary based on the clinical context and patients’ circumstances. For instance, the principle of clinical equipoise may be impacted by clinicians’ evaluation of the risks and benefits of clinical trials. Specifically, patients enrolled in clinical trials may incur added costs associated with attending extra appointments or procedures for data collection—elements that are not part of standard care. These added costs may be perceived as greater or more prohibitive for patients who are already financially disadvantaged, leading to a perceived imbalance in the harm-benefit ratio for these patients that could be ethically offset by higher remuneration. 33 Indeed, Emanuel and colleagues acknowledge that context and culture may inherently influence the way these principles are realized in the clinical trial environment. These findings suggest that clinicians’ perceived ethical dilemmas may be reduced by updating this ethical framework to offer guidelines that reflect the diversity of patient needs and vulnerabilities.
Our findings suggest that interventions aimed at enhancing clinical trial accrual for all patients, particularly underrepresented populations, should also consider implementing practices to support the clinician. This can include providing resources to reduce burnout, such as resiliency training. It can also include training to recognize and respond to perceived ethical conflicts, which can be accomplished by integrating individual ethical consultation services into clinical trial protocols or by establishing monthly clinical trial supervision meetings mediated by ethical consultants. 25 Alternatively, hospital systems can opt to create structures for clinician incentives; for instance, clinicians can be rewarded for directing patients to a patient navigator to discuss clinical trial opportunities. 34 Indeed, our findings show that clinical prefer the inclusion of an objective navigator to help address potential patient burden, functioning as an intermediary who can initiate clinical trial discussion with patients. Programs such as patient navigation, access to a research nurse, 35 and other structured resources for increasing access to clinical trials information and decision-making resources (e.g., a mobile application listing all available clinical trials available at the patient’s medical center) may facilitate standardized systems for offering clinical trials to all patients. 36
Our findings confirm that oncology clinicians practicing in a large academic medical center value cancer clinical trials. However, consistent with existing studies,37,38 time constraints, heavy clinical loads, limited understanding of clinical trials, and lack of support staff posed obstacles to enrollment. These findings provide insight into why these concerns negatively impact equitable clinical trial access. Specifically, time constraints, heavy caseloads, and lack of support staff interfered with oncologists’ ability to become informed about clinical trials, raising concerns about their capacity to adequately inform their patient, address patient/clinician concerns, and guide their patient in making informed decisions about clinical trials in the context of their care. Expanding information and access to clinical trial resources, such as ct.gov, or clinical trial databases specific to the medical center, can increase clinicians’ understanding and awareness of trials. Clinicians may also benefit from access to professional trainings or educational resources focused on communication and decision-making about clinical trials enrollment, which have elsewhere strengthened academic clinicians’ knowledge about clinical trials and beliefs that participation is beneficial for cancer patients. 39 In behavioral healthcare, structured resources for shared decision-making have also enhanced perceived quality of care for multicultural patients, particularly when clinicians treat patients who speak a different primary language than their own. 40
As in other studies,41,42 clinical trial logistics were a common barrier to clinical trial discussions. In this study, this obstacle emerged when clinicians believed clinical trial logistics conflicted with their commitment to reduce harm and ensure patient well-being. Participants expressed uncertainty about the risk-benefit ratio associated with directing patients toward experimental treatments. Concerns were amplified when clinicians were not directly involved as study stakeholders or had limited understanding of clinical trial procedures. To address concerns about risks and patient safety, particularly among patient with medical or resource-related vulnerabilities, increased communication between clinical and research teams through each phase of the research is warranted. 43 System-level changes, such as the use of clinical trial navigators, may help bridge existing gaps between clinical and research teams by reducing the burden on clinicians to remain informed about relevant clinical trials.16,44
Study Limitations
Limitations to the present research warrant discussion. First, participants reported spending at least half of their time providing clinical care with some time engaged in research; as such, the concerns identified may be most representative of oncologists working in an academic medical center. Also, it is possible that attitudes towards clinical trials may differ amongst clinicians who receive more training in clinical trial research during fellowship training. Relatedly, our sample was comprised of predominantly White, English-speaking clinicians; although this reflects the population of medical oncologists at our center, our findings may not represent the perceptions of clinicians from more diverse backgrounds. Third, it is possible that clinician responses were influenced by perceptions of social desirability bias despite the team’s efforts to present questions in a sensitive manner. While the interviewer had no relationship with the study participants, future studies would benefit from having an external party lead the interviewer to reduce potential bias. Finally, we designed interview questions to capture clinician experiences with enrolling patients into a broad range of clinical trials. Future research may examine specific concerns about clinical trial enrollment depending on cancer type and other nuanced clinical trial considerations.
Conclusions
In summary, our findings suggest that oncology clinicians value cancer clinical trials in advancing cancer care however ethical considerations play an important role in motivating their decision to discuss clinical trials with their patients. While personal, trial-specific and systemic obstacles to clinical trial enrollment are commonly communicated barriers, it is the intricate interplay between these factors and patient vulnerabilities that creates an ethical dilemma. Clinician ambivalence arises when presented with patients considered at risk for incurring more harm or burden with clinical trial participation. These concerns are heightened if clinicians perceive themselves as gaining secondary benefits when making clinical trial referrals, especially if they have limited time to ensure safety. To address these ethical dilemmas, clinicians report carefully monitoring their own capacity for making unbiased decisions around offering clinical trials. These findings underscore the need for clinical trial systems that integrate informational support with clinician navigation to increase the enrollment of underrepresented patients into cancer clinical trials. Incorporating multi-level decision-making support for oncologists will enhance cancer centers’ preparedness to equitably offer clinical research participation to patients who may not otherwise have access. This work informs critical next steps for tackling pervasive disparities in clinical trial enrollment and medical innovation, helping to ensure maximal benefit in the contributions made by study participants and investigators.
Footnotes
Acknowledgement
Sydney Crute and Kit Quain assisted in carrying out the study. Permission and approval have been received from everyone who contributed to the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the study was offered by the Lazarex Cancer Foundation. Dr. Giselle Perez is funded by the National Cancer Institute (K07CA211955). Dr. Elyse R. Park is supported by the National Cancer Institute at the National Institutes of Health (K24CA197382, ERP). Dr Lucy Finkelstein-Fox is supported by the National Institutes of Health (T32CA092203)
Ethical Approval
Ethical approval to report this case was obtained in 2016 from the Massachusetts General Hospital’s institution review board (Partners Healthcare) in Boston, MA (reference number: 2016P000871/PHS).
Statement of Human Rights
All procedures in this study were conducted in accordance with Massachusetts General Hospital’s IRB (2016P000871/PHS) approved protocol.
Statement of Informed Consent
Written informed consent was obtained from the participants for their anonymized information to be published in this article.
