Abstract
Introduction
The Cancer Inflammation Prognostic Index (CIPI) is a composite biomarker that integrates carcinoembryonic antigen (CEA), neutrophil, and lymphocyte counts to reflect both tumor burden and systemic inflammation. Although CIPI has demonstrated prognostic value in early-stage and previously treated metastatic colorectal cancer, its role in untreated, de novo metastatic colon cancer remains unclear.
Methods
This multicenter, retrospective cohort study included 287 patients diagnosed with de novo metastatic colon cancer between 2010 and 2023. CIPI was calculated by multiplying serum CEA by the neutrophil-to-lymphocyte ratio, and the optimal cutoff value for overall survival (OS) was determined using receiver operating characteristic (ROC) curve analysis. Patients were stratified into low- and high-CIPI groups based on this threshold.
Results
The median follow-up period was 4.0 years, during which 202 patients (70.4%) died. The optimal CIPI cutoff value for OS was 86.09. Median OS was significantly shorter in the high-CIPI group compared with the low-CIPI group (36.0 vs 14.5 months, p < 0.001). In multivariable Cox regression analysis, a high CIPI remained an independent prognostic factor for OS (hazard ratio, 2.29; 95% confidence interval, 1.65–3.17; p < 0.001). In a separate multivariable model, elevated CEA was also independently associated with poorer overall survival.
Conclusion
CIPI independently predicts overall survival in patients with de novo metastatic colon cancer. Its simplicity, cost-effectiveness, and derivation from routine laboratory data make it a practical and promising tool for baseline risk stratification and individualized follow-up planning.
Plain Language Summary
People with metastatic colon cancer can have very different survival outcomes. Doctors need simple tools to help estimate prognosis when the disease is first diagnosed. In this study, we evaluated a blood-based score called the Cancer Inflammation Prognostic Index (CIPI). This score combines a tumor marker (CEA) with a measure of inflammation from routine blood tests. We found that patients with higher CIPI values had shorter survival. Even among patients who already had high CEA levels, CIPI could further distinguish differences in survival. Because CIPI is based on common and inexpensive blood tests, it may help doctors better assess risk and guide patient follow-up and supportive care. Further studies are needed to confirm these results.

Keywords
1. Introduction
Metastatic colon cancer remains a major global health challenge and continues to rank among the leading causes of cancer-related death worldwide.1-3 Despite notable improvements in systemic treatment options—including cytotoxic chemotherapy, targeted therapies, and immune checkpoint inhibitors—survival outcomes differ substantially between patients.1-3 This wide variability reflects not only tumor-intrinsic factors such as KRAS, BRAF, and microsatellite instability (MSI) status, but also host-related determinants including systemic inflammation, the immune microenvironment, comorbidities, and genetic background.2,4,5 In recent years, multiple molecular and immunologic biomarkers have been proposed to better capture this biological diversity, ranging from immune-related gene signatures to circulating tumor DNA (ctDNA) and various genomic classifiers.6-9 However, these tools often require advanced technology and specialized expertise, which limits their use in routine oncology practice. Consequently, there is growing interest in simple, inexpensive, and widely available biomarkers that can be readily applied in real-world clinical settings.
Carcinoembryonic antigen (CEA) is among the most routinely measured tumor markers in colon cancer and remains a useful indicator of tumor burden in clinical settings. However, its prognostic reliability is limited by several confounding influences, including smoking status, advancing age, and chronic inflammatory conditions.10-12 Furthermore, CEA mainly indicates tumor burden and does not sufficiently account for host-related factors like systemic inflammation and immune status, which are increasingly acknowledged as important determinants of outcome in metastatic disease. This limitation highlights the continuing need for a simple, reproducible, and biologically meaningful biomarker that can be seamlessly integrated into routine oncology practice.
In recent years, systemic inflammation has been recognized as a key determinant of cancer progression, therapeutic response, and overall prognosis.13-16 Evidence from both clinical and translational studies shows that inflammatory activity within the tumor microenvironment is reflected in peripheral blood parameters and circulating biomarkers.13-18 As a result, several inflammation-based indices—such as the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and prognostic nutritional index (PNI)—have been evaluated as practical prognostic tools in various solid tumors.18,19 However, these indices primarily reflect host inflammatory status and do not incorporate tumor burden, which may limit their ability to capture prognosis in metastatic disease fully.
The Cancer Inflammation Prognostic Index (CIPI), first proposed in 2021, integrates CEA and NLR to capture both systemic inflammation and tumor burden. 20 Initial studies demonstrated its prognostic significance in patients with metastatic colorectal cancer treated with regorafenib. 20 Subsequent studies have further validated its prognostic significance in the early stages of colorectal cancer.21,22 However, the prognostic role of CIPI in patients with de novo metastatic colon cancer receiving first-line systemic therapy has not yet been investigated.
This multicenter retrospective study aimed to evaluate the prognostic significance of CIPI in patients with de novo metastatic colon cancer and to assess its utility for baseline risk stratification with respect to overall survival prior to initiating first-line systemic therapy.
2. Materials and Methods
2.1. Study Design and Patient Selection
This multicenter, retrospective study included 287 patients who were diagnosed with de novo metastatic colon cancer between January 2010 and December 2023 at three tertiary oncology centers. Patients with rectal cancer, incomplete medical records, or missing laboratory data were excluded from the analysis. Those with acute infections, hematologic malignancies, or chronic inflammatory or autoimmune diseases that could potentially alter systemic inflammatory parameters were also excluded. Patients receiving systemic corticosteroids at the time of baseline blood sampling were excluded whenever this information was available in the medical records. The diagnosis of colon adenocarcinoma was confirmed histopathologically in all cases. Clinical, pathological, and laboratory data were collected from electronic medical records.
Given the retrospective design of the study, unrecorded use of corticosteroids or undocumented inflammatory conditions could not be excluded entirely and were therefore considered potential sources of residual confounding.
This study was designed and reported in accordance with the REMARK (Reporting Recommendations for Tumor Marker Prognostic Studies) guidelines. 23
The study protocol was approved by the Ethics Committee of Sancaktepe Şehit Prof. Dr. İlhan Varank Training and Research Hospital (approval no. 2024/253, date of approval: August 28, 2024) and was conducted in accordance with the Declaration of Helsinki (1975), as revised in 2024. Due to the retrospective nature of the study, informed consent was waived by the Ethics Committee.
2.2. Data Collection and Variables
Demographic, clinical, pathological, and laboratory data were obtained retrospectively from the electronic medical records of the participating centers. Baseline laboratory parameters were collected within two weeks before the initiation of first-line systemic chemotherapy, ensuring that all measurements reflected the pre-treatment status.
The CIPI was calculated using the following formula:
Additional clinical and pathological parameters included age, sex, body mass index (BMI), Eastern Cooperative Oncology Group Performance Status (ECOG-PS), tumor localization (right- or left-sided colon), histopathological subtype, KRAS, NRAS, and BRAF mutation, and MSI status. Metastatic sites, including liver, lung, peritoneal, and bone involvement, were recorded, along with the type of first-line chemotherapy regimens (FOLFOX, FOLFIRI, or other combinations) were also recorded.
2.3. Treatment and Follow-Up
All patients received first-line systemic chemotherapy according to institutional protocols, with or without targeted agents, based on physician discretion and national reimbursement policies. Dose modifications or delays were applied as clinically indicated.
Best treatment response was assessed radiologically and/or clinically according to the Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1.
Overall survival (OS) was defined as the primary study endpoint and was calculated as the time from the diagnosis of metastatic disease to death from any cause or last follow-up. The last follow-up date for survival analysis was December 31, 2023, and patients who were alive at that time were censored for OS analysis.
2.4. Statistical Analysis
Statistical analyses were carried out using IBM SPSS Statistics, version 25.0 (IBM Corp., Armonk, NY, USA). Continuous variables were reported as mean ± standard deviation (SD) or median (interquartile range [IQR]), and categorical variables as frequencies and percentages. Differences between groups were examined with the χ2 or Fisher’s exact test for categorical data and the Student’s t-test or Mann–Whitney U test for continuous data, as appropriate. Receiver-operating-characteristic (ROC) analysis was used to identify the optimal CIPI threshold for predicting OS; the area under the curve (AUC) with 95% confidence intervals (CIs) was recorded. The cutoff value was identified using the Youden index. Patients were subsequently categorized into low- and high-CIPI groups according to this cutoff. The association between categorical baseline CEA levels and CIPI status was evaluated using the chi-square test. As no patients with low CEA levels exhibited high CIPI, subsequent survival analyses were restricted to patients with elevated CEA levels, stratified according to CIPI status to assess the incremental prognostic value of CIPI beyond CEA. Overall survival estimated by the Kaplan–Meier method and compared using the log-rank test. Because CIPI incorporates CEA as one of its components, multivariable Cox regression models were constructed separately to avoid collinearity: Model A included CIPI, while Model B included CEA. Variables with a p value < 0.10 in univariable analyses—or considered clinically relevant from prior literature—were entered into a multivariable Cox proportional-hazards model to determine independent predictors of OS. Before model construction, potential multicollinearity was evaluated by examining correlation patterns and considering the conceptual overlap between composite inflammatory indices and their individual components. To ensure a parsimonious and interpretable model, composite indices and their constituent variables were not included simultaneously in multivariable analyses. All statistical tests were two-sided, and p < 0.05 was regarded as statistically significant. As this was a retrospective study that included all eligible patients within the study period, a formal sample size calculation was not conducted.
3. Results
A total of 537 patient files with metastatic colon cancer diagnosed between 2010 and 2023 were retrospectively reviewed. After excluding patients who were not metastatic at diagnosis and those without available CEA, complete blood count, or albumin data, 287 patients met the inclusion criteria and were included in the final analysis.
The Relationships Between the CIPI and Clinicopathological Characteristics of patients

N = Number of patients; BMI = Body Mass Index; ECOG-PS = Eastern Cooperative Oncology Group Performance Status; MSI = Microsatellite Instability; FOLFIRI = 5-Fluorouracil, Leucovorin, and Irinotecan; FOLFOX = 5-Fluorouracil, Leucovorin, and Oxaliplatin; CR = Complete Response; PR = Partial Response; SD = Stable Disease; PD = Progressive Disease.
Molecular analysis identified RAS mutations in 39.0% of patients (106 KRAS and 6 NRAS) and BRAF mutations in 1.7%. Regarding systemic therapy, FOLFOX was the most commonly administered first-line regimen (55.6%), followed by XELOX or capecitabine monotherapy (26.9%) and FOLFIRI (17.5%). In addition to chemotherapy, 48.0% of patients received anti-VEGF therapy, and 21.3% received anti-EGFR monoclonal antibodies, administered exclusively to KRAS/NRAS wild-type, left-sided tumors in accordance with treatment guidelines.
When patients were stratified according to CIPI, significant differences were found only in ECOG-PS (p < 0.001) and the presence of liver metastases (p = 0.020). No other baseline demographic, clinical, or molecular characteristics differed significantly between the low- and high-CIPI groups (Table 1).
3.1. CIPI Cutoff Value
ROC curve analysis was performed to determine the optimal CIPI cutoff value for predicting OS. The cutoff was identified as 86.09, corresponding to the point that maximized the Youden index. Patients were then classified into low- and high-CIPI groups according to this threshold.
Predictive Values of Various Inflammation-Based Indices for Overall Survival

AUC = Area Under the Curve; 95% CI = 95% Confidence Interval; NLR = Neutrophil-to-Lymphocyte Ratio; CIPI = Cancer Inflammation Prognostic Index; LMR = Lymphocyte-to-Monocyte Ratio; PLR = Platelet-to-Lymphocyte Ratio; PNI = Prognostic Nutritional Index.
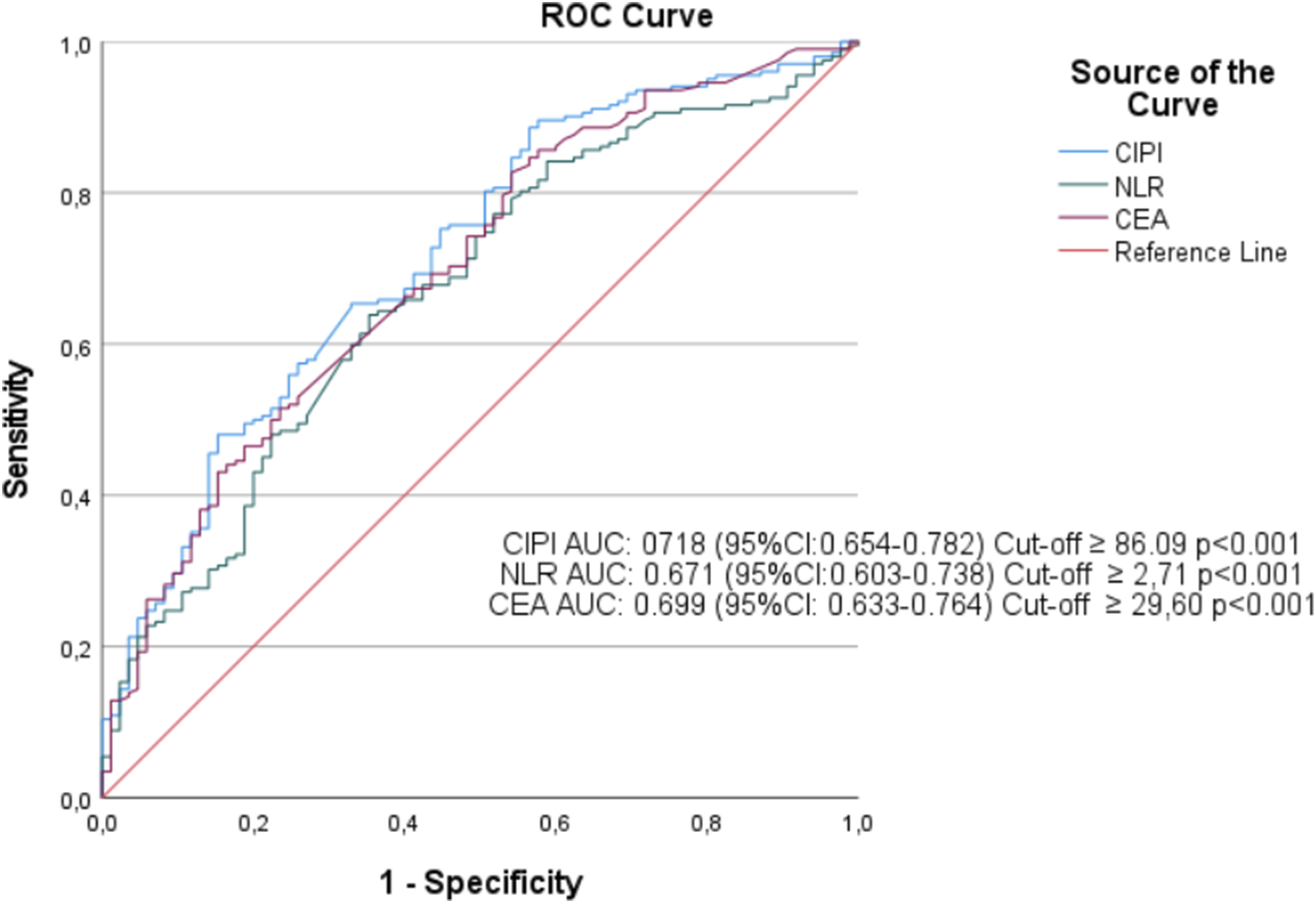
Receiver operating characteristic (ROC) curves showing the prognostic performance of the Cancer Inflammation Prognostic Index (CIPI), neutrophil-to-lymphocyte ratio (NLR), and carcinoembryonic antigen (CEA) for overall survival. The area under the curve (AUC) for CIPI was 0.718 (95% CI: 0.654–0.782), which was higher than that for NLR (AUC: 0.671; 95% CI: 0.603–0.738) and CEA (AUC: 0.699; 95% CI: 0.633–0.764). The optimal cutoff value for CIPI was determined as 86.09 using the Youden index
3.2. CIPI, CEA, and Overall Survival
The median follow-up time was 4 years (range, 1.02–11.41), during which 202 of the 287 patients (70.4%) died. Patients were stratified into high- and low-CIPI groups according to the cutoff value determined by ROC analysis. The Kaplan–Meier survival curves for OS, including numbers at risk, are shown in Figure 2A. (A) Kaplan–Meier curves for overall survival (OS) stratified by the Cancer Inflammation Prognostic Index (CIPI). Low CIPI (<86.09) was associated with longer survival than high CIPI (≥86.09) (36.0 vs. 14.5 months; p < 0.001). (B) OS according to baseline carcinoembryonic antigen (CEA) levels. Patients with low CEA (<5 ng/mL) had better survival than those with elevated CEA (≥5 ng/mL) (35.0 vs. 16.0 months; p < 0.001). (C) OS among patients with elevated CEA (≥5 ng/mL) stratified by CIPI. High CIPI remained associated with worse survival (15.0 vs. 19.0 months; p = 0.016)
Median OS was significantly longer in the low-CIPI group compared with the high-CIPI group (36.0 vs. 14.5 months; p < 0.001, log-rank test; Figure 2A). Consistently, in univariable Cox proportional hazards analysis, high baseline CIPI was associated with a significantly increased risk of mortality (HR = 2.29, 95% CI = 1.66–3.17), confirming the strong prognostic impact of elevated baseline CIPI. This nearly two-fold difference in survival highlights the potential utility of CIPI for baseline risk stratification in de novo metastatic colon cancer.
Kaplan–Meier analysis according to baseline CEA levels also demonstrated a significant survival difference. Patients with low CEA (<5 ng/mL) had longer OS than those with elevated CEA (≥5 ng/mL) (35.0 vs. 16.0 months; p < 0.001, log-rank test; Figure 2B).
Chi-square analysis demonstrated a strong association between baseline CEA categories and CIPI status (p < 0.001). Notably, all patients with low CEA levels were classified in the low-CIPI group (n = 68), with no patients exhibiting low CEA and high CIPI. Among patients with elevated CEA levels, 161 patients had high CIPI, while 58 patients were classified as having low CIPI. Subsequent survival analysis was restricted to patients with elevated CEA levels and performed according to CIPI status. Overall survival differed significantly between these two groups, with patients exhibiting high CIPI demonstrating worse outcomes compared to those with low CIPI (median overall survival: 15 vs. 21 months; log-rank χ2 = 5.766, p = 0.016; Figure 2C).
3.3. Multivariable Cox Regression Analysis
Multivariable Cox proportional hazards modeling was performed to identify independent prognostic factors for OS. Variables with a p-value < 0.10 in univariable analyses and those deemed clinically relevant were included in the model. To avoid overfitting and collinearity, a parsimonious modeling strategy was applied. The proportional hazards assumption was assessed for all covariates.
In Model A (the CIPI model), high CIPI remained an independent predictor of shorter OS (HR, 2.29; 95% CI, 1.66–3.17; p < 0.001). Age >65 years was also independently associated with inferior survival outcomes (HR = 1.43, 95% CI = 1.03–1.98; p = 0.036). Regarding performance status, ECOG-PS 2 was significantly associated with a higher mortality risk compared to ECOG-PS 0 (HR = 1.73, 95% CI = 1.10–2.72; p = 0.017), while ECOG-PS 1 did not have independent prognostic significance.
In Model B (the CEA model), elevated CEA (≥5 ng/mL) was identified as an independent adverse prognostic factor (HR, 3.12; 95% CI, 1.70–5.73; p < 0.001). In this model, age >65 years was not independently associated with OS (HR, 1.05; 95% CI, 0.58–1.92; p = 0.871).For performance status, ECOG-PS 2 remained a strong independent predictor of mortality (HR, 5.20; 95% CI, 1.65–16.40; p = 0.005), whereas ECOG-PS 1 was not statistically significant.
Kaplan–Meier Survival Analysis of Overall Survival According to Baseline Clinicopathological Characteristics

BMI = Body Mass Index; FOLFIRI = 5-Fluorouracil, Leucovorin, and Irinotecan; FOLFOX = 5-Fluorouracil, Leucovorin, and Oxaliplatin; CEA: Carcinoembryonic Antigen; CIPI = Cancer Inflammation Prognostic Index; NLR = Neutrophil-to-Lymphocyte Ratio; LMR = Lymphocyte-to-Monocyte Ratio; PLR = Platelet-to-Lymphocyte Ratio; PNI = Prognostic Nutritional Index; HR = Hazard Ratio; Ref = Reference Category.
Bold values indicate statistically significant results (p < 0.05).
Multivariable Cox Proportional Hazards Regression Analyses for Overall Survival: Model a Including CIPI and Model B Including CEA
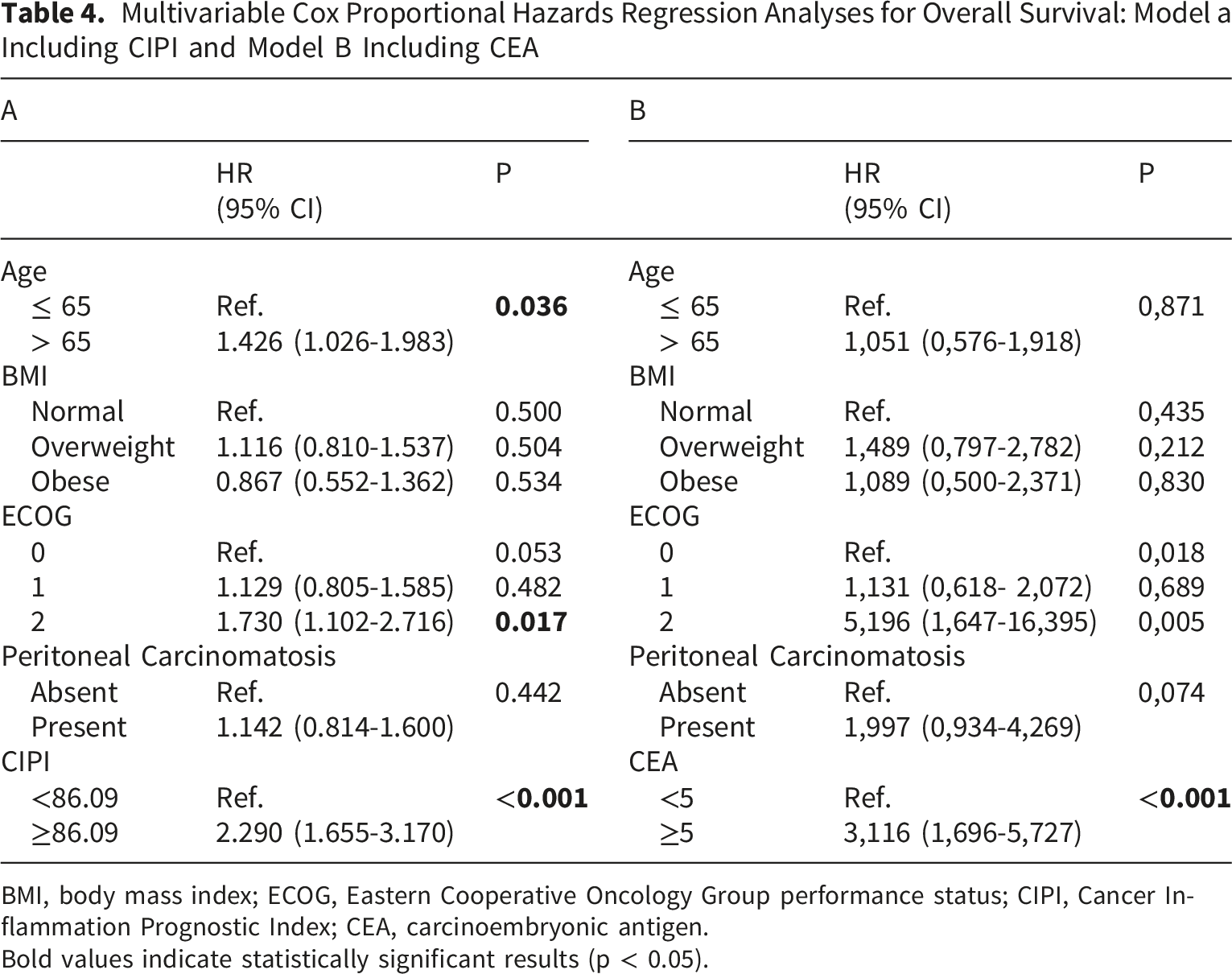
BMI, body mass index; ECOG, Eastern Cooperative Oncology Group performance status; CIPI, Cancer Inflammation Prognostic Index; CEA, carcinoembryonic antigen.
Bold values indicate statistically significant results (p < 0.05).
4. Discussion
The survival rates for metastatic colon cancer are still quite variable, despite significant advancements in systemic treatment. This highlights the critical need for trustworthy, easily available, and biologically meaningful prognostic markers that can be integrated into routine clinical practice. In this context, CIPI emerges as a promising biomarker that integrates tumor burden and systemic inflammation into a single, easily measurable parameter. Our study provides novel data in a de novo metastatic setting, demonstrating that CIPI is an independent prognostic marker for overall survival and may aid in baseline risk stratification before first-line systemic therapy.
There is substantial evidence linking systemic inflammation to cancer progression and survival in colon cancer. 24 The protective effect of nonsteroidal anti-inflammatory drugs against colorectal carcinogenesis further supports this biological rationale. 25 Peripheral blood components, particularly neutrophils and lymphocytes, reflect the dynamic balance between tumor-promoting inflammation and anti-tumor immune responses.18,24 Neutrophils promote tumor growth by releasing pro-inflammatory cytokines (e.g., IL-1, IL-6) and angiogenic factors such as vascular endothelial growth factor (VEGF), whereas lymphocytes exert cytotoxic and immunoregulatory effects that suppress tumor proliferation and invasion. 24 Elevated CEA levels have also been associated with enhanced tumor angiogenesis and increased tumor burden in colon cancer.11,12 In this context, CIPI integrates complementary biological dimensions by combining CEA, which reflects tumor burden, with the NLR, a marker of systemic inflammation and immune status. The prognostic significance of CIPI may therefore derive from its ability to simultaneously represent tumor biology and host inflammatory status. This integrated biological framework may partly explain the strong association between high baseline CIPI values and poor overall survival observed in our cohort. Future prospective studies are warranted to determine whether CIPI can aid in risk stratification and surveillance strategies for patients with metastatic colon cancer.
Recent studies have increasingly highlighted the prognostic importance of CIPI across different stages of colorectal cancer. The index was first described in 2021 in a cohort of heavily pretreated metastatic colorectal cancer patients receiving regorafenib, where it was identified as an independent prognostic factor for OS. 20 Subsequent investigations have demonstrated that the clinical relevance of CIPI extends beyond metastatic disease, maintaining prognostic value even in non-metastatic colorectal cancer. In patients with stage I–III colorectal cancer, higher preoperative CIPI levels have been shown to independently predict poorer relapse-free and OS.21,22 Moreover, combining CIPI with other inflammation- and nutrition-based indices, such as the platelet-to-albumin ratio, has been reported to further enhance its predictive accuracy for survival outcomes. 26 Elevated CIPI levels have also been associated with more advanced tumor stages, underscoring its biological link to tumor aggressiveness and systemic inflammation. 27 The optimal CIPI cutoff found in our cohort is different from those reported in previous studies, likely due to differences in disease stage, treatment setting, and patient characteristics, highlighting the context-dependent nature of inflammation-based prognostic indices.20-22 In line with these findings, our study demonstrates that CIPI retains independent prognostic value when assessed at baseline in patients with de novo metastatic colon cancer, prior to the initiation of first-line systemic therapy, even after accounting for established clinical factors such as performance status and metastatic pattern. This extends its clinical applicability to this distinct and understudied setting.
Inflammation-based biomarkers such as NLR, PNI, SII, and PIV have been widely investigated for their prognostic relevance across various solid tumors, including colorectal cancer, and remain an active area of research.28-31 However, the existing literature suggests that the prognostic value of these indices is not entirely consistent; while some studies report significant associations with survival outcomes, others do not demonstrate a clear prognostic effect.28,32-34 This heterogeneity may be attributable to differences in patient populations, disease stage, treatment setting, and statistical methodology. Similarly, the prognostic role of CEA in colorectal cancer has been well established, and in our cohort, elevated CEA was also independently associated with poorer overall survival when evaluated in a separate multivariable model. This finding is consistent with its recognized function as a surrogate marker of tumor burden.10-12 Nevertheless, several inherent limitations should be acknowledged when interpreting CEA as a prognostic marker. CEA levels may be influenced by non–tumor-related factors such as smoking status, advancing age, and chronic inflammatory conditions, which can limit its ability to fully reflect tumor biology.10-12 Moreover, CEA primarily represents tumor burden and does not adequately capture host-related factors such as systemic inflammation and immune status, which are increasingly recognized as key determinants of outcome in metastatic disease. Because CIPI incorporates CEA together with an inflammation-based parameter, it provides a more integrative representation of both tumor-related and host-related biological processes. Importantly, our findings show that even among patients with similarly elevated CEA levels, CIPI was able to provide additional prognostic discrimination, with significantly worse survival observed in those with high CIPI. These results suggest that CIPI should not be viewed as a substitute for CEA, but rather as a complementary tool that may enhance prognostic stratification beyond tumor burden alone.
First proposed in 2021 and initially validated in metastatic colorectal cancer patients treated with regorafenib, 20 CIPI has since demonstrated consistent prognostic value across disease stages. Subsequent studies in non-metastatic colorectal cancer cohorts published in 2022 and 2023,21,22 confirmed that higher CIPI values were associated with poorer overall and recurrence-free survival, supporting its broader applicability beyond metastatic settings. To our knowledge, this study provides initial evidence that CIPI serves as an independent prognostic factor for overall survival in patients with de novo metastatic colon cancer receiving first-line treatment. These findings further underscore the relevance of inflammation-based markers in prognostic stratification of metastatic disease.
Our study also possesses several strengths, including a focus on de novo metastatic colorectal cancer—a population rarely examined in previous literature—and the use of a relatively homogeneous cohort with comprehensive evaluation of systemic inflammatory indices. Additionally, the multicenter design increases the representativeness of the study population and enhances the generalizability of our findings within real-world clinical practice. Nonetheless, certain limitations should be acknowledged. The retrospective design and absence of a predetermined sample size calculation may limit the statistical power of this study and should be taken into account when interpreting the results. Additionally, heterogeneity in treatment regimens over time may have introduced selection and temporal biases. The lack of an external validation cohort also limits the generalizability of our findings. Although patients receiving systemic corticosteroids and those with active infections, hematologic disorders, or chronic inflammatory diseases were excluded at baseline, unrecorded corticosteroid use or subclinical infectious or inflammatory conditions could not be completely ruled out due to the retrospective nature of the study, which may be a potential source of residual confounding. While ECOG-PS differed between CIPI groups, adjustment for ECOG-PS in multivariable analyses did not eliminate the independent prognostic association between CIPI and overall survival. Additionally, this study was designed with overall survival as the primary endpoint; therefore, progression-free survival and objective response rate were not evaluated. As treatment strategies were not compared, CIPI should be regarded as a prognostic rather than a predictive marker. Although CIPI is a prognostic rather than a predictive marker, a high CIPI can help identify patients at greater risk who may benefit from closer clinical monitoring, early response evaluation, and timely supportive care. Future multicenter, prospective studies are warranted to validate these observations and to establish standardized CIPI cutoff thresholds across clinical settings. Despite these limitations, the present study adds meaningful evidence supporting CIPI as a simple, cost-effective, and clinically relevant biomarker for baseline risk stratification in metastatic colon cancer.
5. Conclusions
The Cancer Inflammation Prognostic Index (CIPI) is a practical inflammation-based biomarker that integrates tumor burden and systemic inflammatory status and shows independent prognostic value in patients with de novo metastatic colon cancer. Higher baseline CIPI levels were associated with poorer overall survival, suggesting that this index may support baseline risk stratification in this patient population. The multicenter design of the present study enhances the representativeness of the findings and their relevance to real-world clinical practice. Given the observational nature of the study, these results should be interpreted with caution, and further large-scale prospective studies are warranted to validate the prognostic role of CIPI and to clarify its potential clinical implications.
Footnotes
Acknowledgments
The authors sincerely thank the medical and administrative staff at all participating oncology centers for their valuable assistance in data collection and patient follow-up. The authors also express their appreciation to all colleagues who contributed to the coordination and management of this study. Artificial intelligence–assisted tools were used during the revision process exclusively for language editing and to improve the clarity and readability of the manuscript. No artificial intelligence tools were used for data analysis, data interpretation, or the generation of scientific results. The authors take full responsibility for the content of the manuscript.
Ethical Considerations
This study was approved by the Ethics Committee of Sancaktepe Şehit Prof. Dr. İlhan Varank Training and Research Hospital (approval no. 2024/253, date of approval: August 28, 2024).
Consent to Participate
Due to the retrospective design, the requirement for informed consent was waived by the Ethics Committee.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are not publicly available due to patient privacy restrictions but are available from the corresponding author upon reasonable request and with institutional approval.

