Abstract
Introduction
Managing biopsy-confirmed cervical intraepithelial neoplasia grade 2 (CIN2) in women of reproductive age poses clinical challenges. Immediate treatment with large-loop excision of the transformation zone (LLETZ) is associated with a substantial risk of adverse obstetric outcomes. This study aimed to identify the risk factors for predicting CIN3+ lesions in reproductive-aged women with biopsy-diagnosed CIN2, to inform personalized management strategies that are particularly relevant to China’s evolving fertility policies.
Methods
This retrospective cohort study analyzed the data from a regional cervical lesion screening database. Reproductive-aged women (<45 years) with biopsy-confirmed CIN2 who underwent subsequent LLETZ between 2016-2024 were included in the study (n=516). Pathological upgrade was defined as CIN3+ in the LLETZ specimen. Univariate and multivariate logistic regression analyses identified independent risk factors for pathological upgrade.
Results
Following LLETZ, 18.4% (95/516) of the women had CIN3+ lesions, indicating biopsy underestimation. HPV 16 (56.3%) and HPV 52 (27.5%) were the most prevalent genotypes in CIN3+ and CIN2- groups, respectively. Multivariable analysis identified three independent predictors: liquid-based cytology (TCT) ≥HSIL (OR = 6.308; 95% CI: 2.390-16.650; P<0.001); specific HR-HPV genotypes: HPV 16 infection (OR = 2.372; 95% CI: 1.165-4.831; P=0.017) and HPV 33 infection (OR = 3.263; 95% CI: 1.035-10.292; P=0.044); endocervical curettage (ECC) ≥CIN2 (OR = 3.067; 95% CI: 1.474-6.384; P=0.003). Age did not increase the risk of developing CIN3+ lesions.
Conclusion
This risk-stratification model offers evidence-based guidance for optimizing individualized treatment decisions in clinical settings where fertility preservation is prioritized.
Plain Language Summary
What was the problem? Young women diagnosed with CIN2 face a difficult choice. Immediate surgery (like LLETZ) can reduce cancer risk but may increase future risks for pregnancy problems like preterm birth. However, some women with a CIN2 diagnosis actually have more severe cell changes (CIN3 or worse) hiding that need treatment. Doctors need a better way to tell who truly needs surgery right away and who might be safely monitored to protect future fertility. What did they find? Hidden Severe Changes: Surgery revealed that about 1 in 5 women (18.4%) diagnosed with CIN2 by biopsy actually had CIN3 or worse. Predicting Higher Risk: Three key factors made it much more likely that a woman with a CIN2 biopsy would have hidden worse changes (CIN3+): A recent TCT showing “≥HSIL” (a more serious abnormality). Having specific types of high-risk HPV infection: HPV type 16 or HPV type 33. A separate test sampling cells inside the cervical canal (ECC) showing at least CIN2. 3. Age Didn't Matter: Younger women weren't any less likely to have hidden CIN3+ than older women in this group. What did this study do? Researchers looked back at the records of 516 young women (under 45) in China who were diagnosed with CIN2 by biopsy and then had surgery (LLETZ). They wanted to see how many actually had worse changes (CIN3+) found during surgery and identify clues that could predict this before surgery. Why does this matter? This study gives doctors important clues to help young women with a CIN2 biopsy diagnosis. By checking the TCT result, specific HPV types (especially 16 and 33), and the endocervical sampling result, doctors can better identify women at high risk of having hidden severe changes who likely need surgery. Women at lower risk might be candidates for close monitoring instead of immediate surgery, helping to protect their chance.
Keywords
Introduction
Persistent infection with high-risk human papillomavirus (HR-HPV) is etiologically linked to the development of high-grade squamous intraepithelial cervical lesions (HSIL) that may develop into cervical cancer if left untreated.1,2
Treatment for patients with HSIL includes excisional procedures, such as large loop excision of the transformation zone (LLETZ). LLETZ is a minimally invasive surgical technique that is frequently used in both diagnostic and therapeutic applications. It employs monopolar electrosurgery, enabling tissue excision in cut mode and achieving hemostasis through coagulation mode. 3 Appropriate application of LLETZ not only treats cervical intraepithelial neoplasia (CIN) by removing lesions but also allows histologic evaluation. 4 However, the risk of postoperative obstetric adverse outcomes may be observed after LLETZ, such as premature rupture of membranes, miscarriage, and premature delivery, as well as the delivery of low birth weight infants due to the conization of the cervix, which disrupts its anatomical structure to varying degrees.5-7
The American Society for Colposcopy and Cervical Pathology (ASCCP) recommends reporting HSIL as HSIL(CIN2) and HSIL(CIN3), emphasizing the differences between CIN2 and CIN3. 3 As reported, approximately 50% CIN2 cases naturally subside within 2 years, 32% persists, and only 18% progresses to more severe lesions. 8 For young women under 30 years of age, 60% CIN2 cases regress, 23% persist, and 11% progress, indicating the high rate of regression among reproductive-age women with CIN2. 9 Due to high regression rates of CIN2 and the increased risk of reproductive harm after LLETZ, active surveillance has been implemented in younger women with CIN2. 3
However, for biopsy-confirmed CIN2 cases that can be monitored without cervical conization, standardized management remains unclear. The 2019 ASCCP guidelines 3 recommend that for young women with a histologically confirmed diagnosis of CIN2 that is not otherwise specified, either treatment or observation for up to 2 years is acceptable, with combined colposcopy and cytology examinations performed at 6-month intervals. However, given the subjective nature of colposcopy and cervical cytology, the reliability of this approach is unclear. While CIN2 exhibits a high regression rate,8,9 CIN3 is considered a direct precursor of cervical cancer, and non-pregnant patients of any age should be treated. 3 In patients with biopsy-confirmed CIN2 lesions who opt for observation to preserve fertility without undergoing LLETZ, there remains a risk of an undetected underlying CIN3+ lesion. 2 Moreover, with the growing openness of the Chinese society and the introduction of the three-child policy, an increasing number of women of reproductive age are being diagnosed with cervical precancerous lesions. The demand for fertility preservation in women of childbearing age with cervical CIN2 is becoming increasingly urgent.
The aim of this retrospective study was to stratify the risk of reproductive-age women under 45 years of age with biopsy-confirmed CIN2, and identify those at high risk for CIN3 or worse after LLETZ. The findings can provide a basis for the implementation of precise and individualized management strategies for childbearing-age women diagnosed with CIN2 on biopsy.
Methods
Study Participants
This retrospective cohort study included participants selected from a regional cervical lesion screening database.10,11 From January 1, 2016, to December 12, 2024, women who underwent cervical biopsy with biopsy-confirmed CIN2 results were initially included. Participants who met the following criteria were excluded: (1) history of CIN2+; (2) history of LLETZ; or (3) previous vulvar or vaginal cancer. Only women aged <45 years who underwent LLETZ were eligible for inclusion in the study (Figure 1). Data were collected by searching the hospitals’ electronic medical records and databases to identify eligible patients. The reporting of this retrospective study conformed to the STROBE guidelines.
12
Flowchart of study population.
All protocols and informed consent procedures were reviewed and approved by our institutional ethics committee (Ethics Committee of Fujian Maternity and Child Health Hospital; Approval No: 2024KY272). All patient data were anonymized prior to analysis, and no personally identifiable information was retained in the research records. Given the retrospective and observational design of the study and the anonymized nature of the data, the requirement for written informed consent was waived.
Screening Protocols
Referral for colposcopy was mandated if any of the following criteria were met: 1) positive for HPV 16 or HPV 18; 2) any cytological abnormality accompanied by concurrent HR-HPV positivity; 3) persistence of an HR-HPV infection for one year or longer; or 4)any clinically suspicious cervical lesion detected during visual inspection. Basic pre-colposcopy information, including gravidity, parity, medication history, cervix-related lesions, and other malignancies, was recorded. Colposcopic examination included acetic acid and Lugol’s iodine application, with transformation zone (TZ) classification as per the Federation of Cervical Pathology and Colposcopy 2011. TZ type I is fully ectocervical and entirely visible. TZ type II is partially endocervical but fully visible with the aid of a speculum. TZ type III is partially or completely endocervical and not fully visible. Colposcopic diagnoses were categorized as normal, low-grade squamous intraepithelial lesion (LSIL), HSIL, or cancer. Colposcopy-directed biopsies were obtained from the most severe acetowhite/iodine-negative lesions. If the colposcopy revealed no lesions, a random 4-quadrant perforated biopsy was conducted. Endocervical curettage (ECC) was performed for TZ type II-III or cytologic discordance. All specimens were formalin fixed and topographically oriented for histopathological evaluation.
For non-pregnant patients diagnosed with CIN2, LLETZ is the recommended treatment. Observation was permitted for young CIN2 patients who have a primary concern for fertility over the risk of disease progression, provided that the TZ and lesions are fully visible during colposcopy. Ablation therapy was permitted in selected cases: 1) a satisfactory colposcopy with full visualization of the TZ and lesions; 2) a ECC histology result less than CIN2; 3) all lesions fall within the treatable range. All LLETZ procedures were performed under colposcopic visualization using a Valleylab Force FX system (40 W cut/60 W coag). The TZ was excised en bloc with 3 mm lateral and deep margins. The specimens were topographically oriented for histopathological evaluation of the margins.
Polymerase Chain Reaction-Reverse Dot Blot (PCR-RDB) HR-HPV DNA Test
PCR-RDB for HR-HPV testing was used for the detection of HR-HPV DNA, which included 16 genotypes (HPV-16, -18, -31, -33, -35, -39, -45, -51, -52, -56, -58, -59, -66, -68, -73 and -82), in cervical exfoliated cells (Yaneng® Biosciences, ShenZhen, China). The test procedures were performed strictly according to the manufacturer’s instructions. 10 Participants with multiple detectable HR-HPV genotypes were categorized as having multiple infections. All detected genotypes were included in the individual genotype analysis, and multiple infections were analyzed as separate variables in the risk stratification model.
Liquid-Based Cytology
An auto-imaging system (Hologic, Inc., San Diego, CA, USA) was used for cytology tests. Cytological specimens were collected and assessed independently by two pathologists with adequate certification. If a different diagnosis was made, the samples were reviewed again until a consensus diagnosis was reached. The cytological results were classified according to the Bethesda system. 13
Histopathology
The specimens were fixed in 10% formalin and embedded in paraffin. Tissue sections of 4 μm thickness were stained with hematoxylin and eosin routinely, and then evaluated by an experienced pathologist according to the 2020 World Health Organization Classification of Tumors of the Female Genital Tract and the CIN system. 14 Any discrepancy was discussed and resolved by a second histological examination until consensus was reached.
Statistical Analysis
The means and standard deviations (SD) of the classified variables were calculated. Values and percentages were calculated. Bar charts were plotted using GraphPad Prism v9.4.1. Pathological upgrade was defined as the occurrence of CIN3+ lesions after LLETZ in women with biopsy-confirmed CIN2. Univariate and multivariate logistic regression were used to identify independent prognostic factors related to pathological upgrade in women under 45 years of age with biopsy-confirmed CIN2. Odds ratios (OR) and 95% CI were also calculated. All the statistical analyses were performed using SPSS (Version 28.0.1.1) (IBM Corporation, Armonk, NY, USA). Differences were considered statistically significant at P< 0.05.
The missing values for each variable are listed in Supplemental (Table S1). Multiple imputation (MI) techniques were employed to handle missing data using SPSS, and the reliability of the interpolated results was evaluated. The imputation model incorporates all variables involved in the subsequent substantive analysis. The parameter estimates and their standard errors from these 10 analyses were then combined into a single set of results using Rubin’s rules, which accounted for both within-imputation and between-imputation variability.
Results
Baseline Characteristics
Demographic and Clinical Features of Included Participants (n=516)
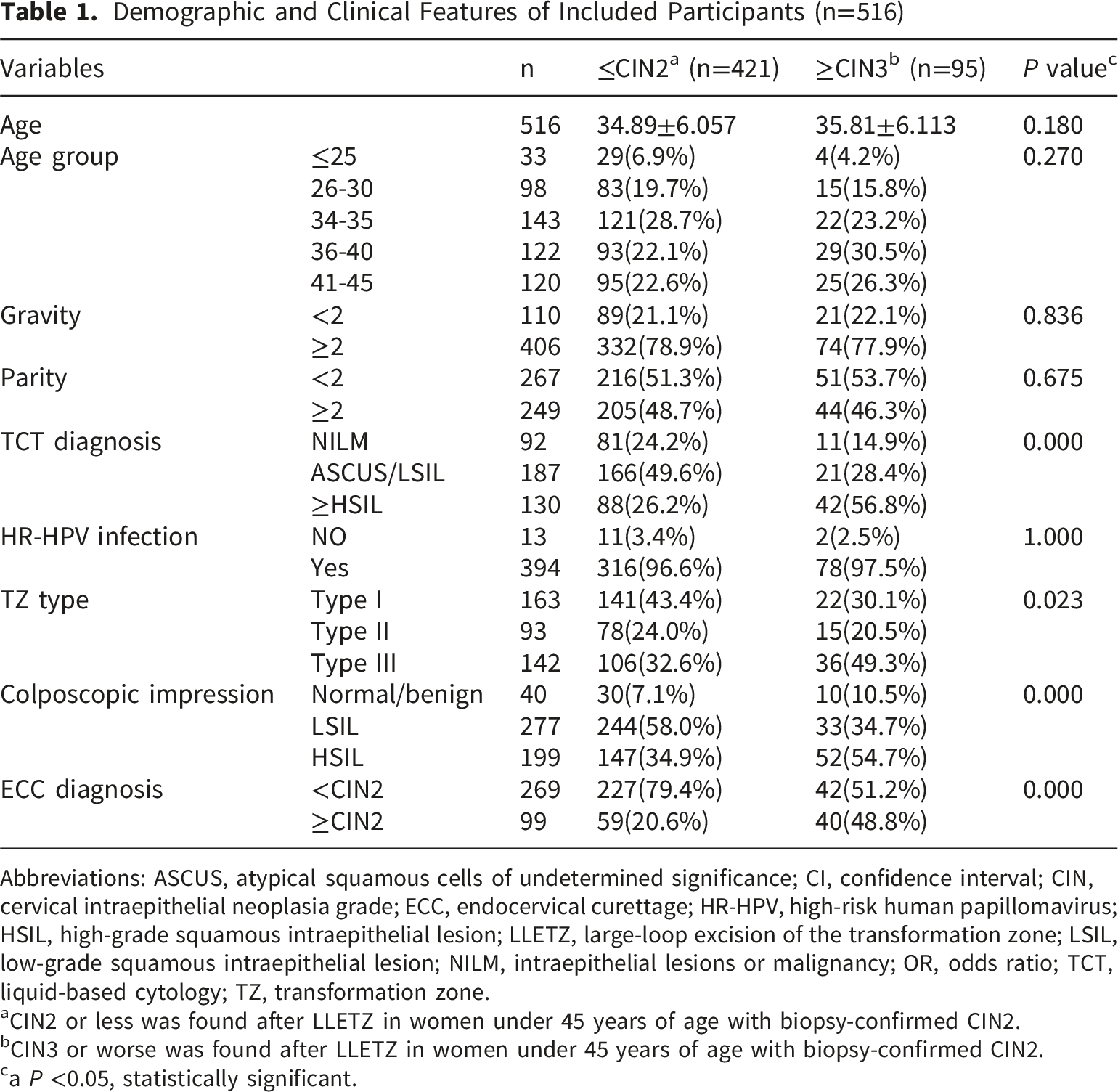
Abbreviations: ASCUS, atypical squamous cells of undetermined significance; CI, confidence interval; CIN, cervical intraepithelial neoplasia grade; ECC, endocervical curettage; HR-HPV, high-risk human papillomavirus; HSIL, high-grade squamous intraepithelial lesion; LLETZ, large-loop excision of the transformation zone; LSIL, low-grade squamous intraepithelial lesion; NILM, intraepithelial lesions or malignancy; OR, odds ratio; TCT, liquid-based cytology; TZ, transformation zone.
aCIN2 or less was found after LLETZ in women under 45 years of age with biopsy-confirmed CIN2.
bCIN3 or worse was found after LLETZ in women under 45 years of age with biopsy-confirmed CIN2.
ca P <0.05, statistically significant.
Prevalence of HR-HPV Genotypes
Figure 2 shows the incidence rates of the different HPV genotypes among women aged <45 years with biopsy-confirmed CIN2. The HR-HPV infection rate was 96.8% (394/407) (Figure 2A). The HR-HPV infection rate was slightly higher in women with CIN3+ lesions after LLETZ (78/80, 97.5%) compared to women with CIN2- lesions after LLETZ (316/327, 96.6%). In women with CIN3+ lesions after LLETZ, HPV 16 was the most prevalent and was detected in 45 women (45/80, 56.3%), followed by HPV 52 (12/80, 15.0%), HPV 33 (11/80, 13.8%), HPV 58 (11/80, 13.8%) and HPV 18 (8/80, 10.0%) (Figure 2C). In women with CIN2- lesions after LLETZ, HPV 16 was still the most prevalent genotype (131/327, 40.3%), followed by HPV 52 (89/327, 27.5%), HPV 58 (52/327, 16.0%), HPV 18 (35/327, 10.8%) and HPV 51 (24/327, 7.4%) (Figure 2B). Prevalence of HR-HPV genotypes.
Distribution of TCT Results
Among women with CIN3+ lesions after LLETZ, the majority had TCT results ≥HSIL (42/74, 56.8%), 21 women (28.4%) had TCT of atypical squamous cells of undetermined significance (ASCUS) and LSIL, and only 11 women (14.9%) were diagnosed with TCT of intraepithelial lesions or malignancy (NILM) (Figure 3C). In contrast, the prevalence of the presence of TCT results ≥HSIL dropped to 26.2% (88/335) in women with CIN2 lesions after LLETZ. TCT results for NILM were more frequently observed in women with CIN2 lesions (216/335, 51.3%) (Figure 3B). Distribution of TCT results.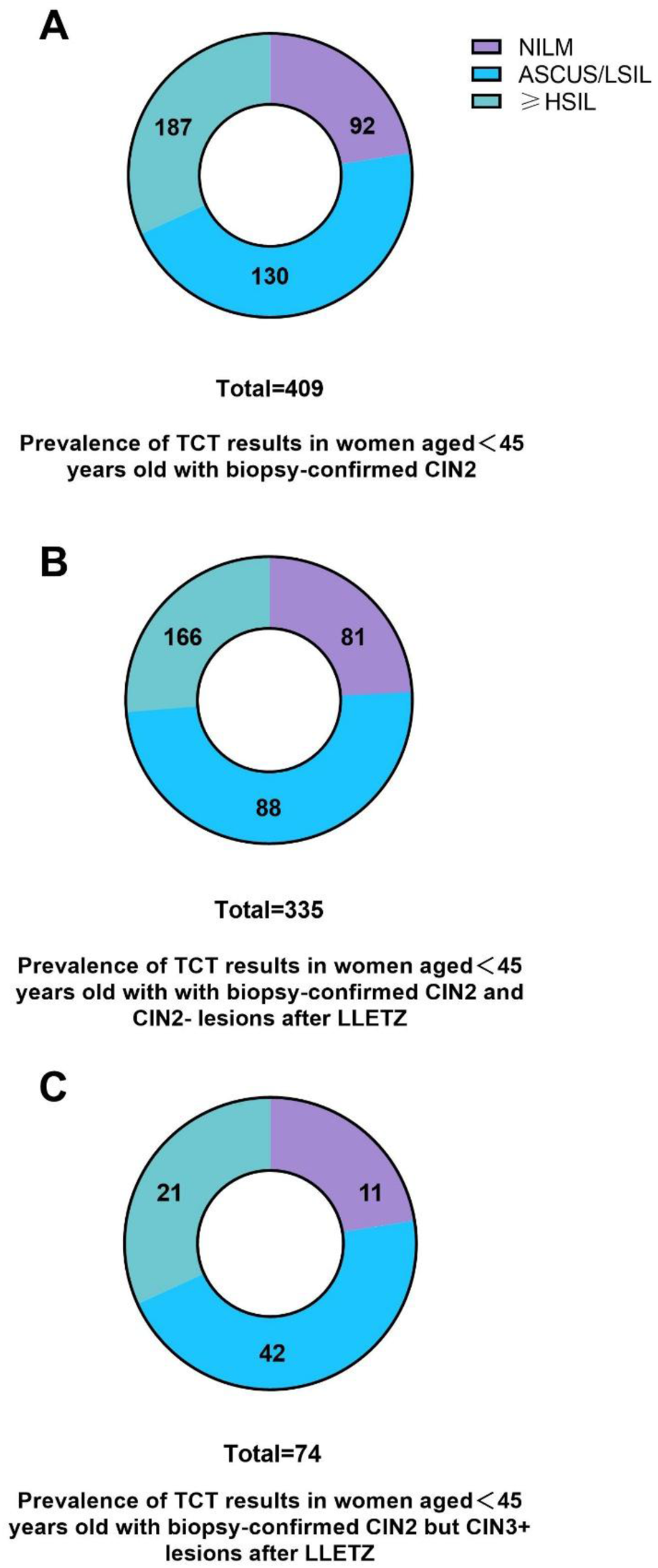
Risk of Pathological Upgrade after LLETZ
Univariable and Multivariable Analysis of Risk of Pathological Upgrade After LLETZ in Childbearing-Age Women Under 45 Years Old With Biopsy-Confirmed CIN2 (n=516)

Abbreviations: ASCUS, atypical squamous cells of undetermined significance; CI, confidence interval; CIN, cervical intraepithelial neoplasia grade; ECC, endocervical curettage; HR-HPV, high-risk human papillomavirus; HSIL, high-grade squamous intraepithelial lesion; LLETZ, large-loop excision of the transformation zone; LSIL, low-grade squamous intraepithelial lesion; NILM, intraepithelial lesions or malignancy; OR, odds ratio; TCT, liquid-based cytology; TZ, transformation zone.
As shown in Table 2, independent factors associated with the diagnosis of CIN3+ lesions after LLETZ were identified using multiple logistic regression analysis. It is demonstrated that the presence of TCT diagnosis ≥HSIL is most strongly associated with an increased likelihood of potential CIN2+ lesions (OR, 6.308; 95% CI 2.390-16.650, P<0.001). Regarding specific HPV genotypes, HPV 33 infection was still associated with an increased chance of confirmed pathological upgrade after LLETZ, which presented a higher risk (OR, 3.263; 95% CI 1.035-10.292, P=0.044) than HPV 16 infection (OR, 2.372; 95% CI 1.165-4.831, P=0.017). ECC diagnosis ≥CIN2 also remained an independent diagnostic predictor of CIN3+ lesions after LLETZ, with the OR of 3.067(95% CI, 1.474-6.384, P=0.003). However, TZ type was not independently associated with the risk of pathological upgrade after LLETZ.
To address missing data, which were determined to be missing at random, a multiple logistic regression analysis was conducted after MI. The outcomes of the pooled analysis from 10 imputed datasets are provided in the Supplemental (Table S2).The results of the MI analysis were entirely consistent with those of the complete-case analysis, showing similar effect sizes and unchanged conclusions for our primary outcomes (Figure 4). Among them, TCT ≥HSIL (OR = 3.552; 95% CI: 1.350-9.343; P = 0.011); specific HR-HPV genotypes: HPV 16 infection (OR = 2.802; 95% CI: 1.100-7.135; P = 0.032) and HPV 33 infection (OR = 6.643; 95% CI: 1.574-28.039;P = 0.013); and ECC diagnosis ≥CIN2 (OR = 2.960; 95% CI: 1.590-5.51; P = 0.001) remained statistical significant. The reliability analysis of the imputed values for the primary predictor was conducted to assess the consistency and accuracy of the estimated data (Supplemental Table S2). Multivariable analysis of risk of pathological upgrade after LLETZ in childbearing-age women under 45 years old with biopsy-confirmed CIN2 (n=516). Original data analysis (circle); data analysis after MI (square).
Discussion
The management of CIN2 with immediate LLETZ has been a subject of debate in recent years, given the high rates of spontaneous regression. The 2019 ASCCP guidelines clearly state that for patients with fertility concerns, if follow-up conditions are met and both the cervical squamocolumnar junction and lesions are fully visible, CIN2 can be managed through surveillance rather than immediate intervention. However, a recent study by Lycke et al. demonstrated that active surveillance for CIN2, which involves leaving the lesion untreated, is associated with an increased long-term risk of cervical cancer compared to immediate LLETZ. 8 Placing excessive emphasis on fertility preservation may result in delayed diagnosis and treatment for some patients with CIN.
Our study revealed that biopsies underestimated the presence of CIN3+ in 18.4% of cases compared to LLETZ diagnosis. This finding is consistent with that of Gustafson et al., 15 who reported a 17.7% discrepancy between women who underwent both biopsy and LLETZ. Identifying high-risk groups for underlying CIN3+ lesions and timely intervention are crucial for preserving fertility while ensuring timely treatment of CIN3+ lesions in women of childbearing age. Given that patients with CIN2 aged over 45 are typically in the perimenopausal stage and do not have fertility requirements, evaluating the risk of conservative treatment is less applicable. Consequently, this study focused exclusively on women aged <45 years.
In order to facilitate the long-term and balanced development of the population, China liberalized the “three-child” policy in May 2021. Consequently, the number of older pregnant women with fertility intentions has steadily increased. A study reported that in young women with CIN2 under the age of 30 years, 60% of the disease regressed, 23% persisted, and only 11% progressed to more severe lesions. 16 Clinically, older women with CIN2 lesions are frequently referred for cervical conization due to the higher likelihood of harboring more severe lesions. In the present study, the incidence of CIN3+ lesions was higher among women aged 36-40 years than in those of other age groups. These findings are consistent with those of a previous cross-sectional study conducted in the Yangtze River Delta region (China), which reported that the prevalence of CIN3 peaked at 35-39 years. 17 However, our study also revealed that age does not increase the risk of pathological upgrade following LLETZ. Consequently, for women with reproductive intentions, in the absence of other combined risk factors, clinical decision-making should not consider patient age as a reference criterion for determining whether to perform LLETZ, thereby aligning with the recommendations outlined in the guidelines. 3 This provides evidence to support the preservation of fertility in women undergoing biopsy for CIN2 in contemporary Chinese society.
The included outpatients primarily comprised individuals with abnormal cervical cancer screening results, which led to a relatively high HPV positivity rate. Specifically, the positivity rate was 96.8% in patients with biopsy-confirmed CIN2 lesions. In the subgroup analysis, HPV 16 was identified as the most prevalent genotype, followed by HPV 52, 33, 58, and 18, among women with CIN3+ lesions post-LLETZ treatment. Among women with CIN2- lesions following LLETZ, HPV 16 remained the predominant genotype, succeeded by HPV 52, 58, 18, and 51. After conducting risk analysis, HPV 16 and 33 infections were identified as independent risk factors for pathological upgrading following LLETZ. This finding aligns with prior studies, which have demonstrated that HPV 16 is the most aggressive HR-HPV genotype associated with the development of cervical precancerous lesions and malignant cancer. 18 The literature further indicates that the prevalence of HPV 33 increases with the severity of cervical lesion grades. 19 Our study highlights the importance of monitoring HPV 33 infections in Chinese women. For patients with biopsy-confirmed CIN2 who test positive for HPV 16 or 33, careful consideration should be given to selecting an appropriate follow-up strategy. However, the present study demonstrated that the prevalence of HPV 18 was only 10% among patients with CIN3+, which is consistent with other Chinese studies 20 but differs from international data.21,22 The low incidence of HPV 45 was also similar to that reported in most domestic studies. 20 Although HPV 18 and 45 exhibited a relatively lower prevalence in the current analysis, these types have been reported to be associated with glandular lesions in the endocervical canal,23,24 which remains clinically significant.
Our findings suggested that women with TCT diagnosis ≥HSIL were at increased risk of having disease missed in biopsy, which was consistent with the results of other studies. Studies have demonstrated that among patients with cytology HSIL who receive immediate treatment, 55.4% are diagnosed with CIN3+. 25 Data from KPNC reveal that the immediate risk of CIN3+ for patients with cytology HSIL and concurrent HPV 16 positivity is 60%, the risk of CIN2+ is 77%, and the immediate cancer risk is 8.1%.26,27 Therefore, when TCT diagnosis≥HSIL is identified in biopsy-confirmed CIN2 women, clinicians should adopt a more proactive treatment strategy.
ECC can be used to obtain sample tissue from the cervical canal for cervical histological evaluation.28,29 According to a Swedish study, the collection of random biopsies and ECC was identified as a valuable clinical tool for detecting CIN2+ in older women when visible lesions were absent. 30 Several studies have demonstrated that ECC in high-risk populations can significantly enhance the detection rate of cervical lesions.31-33 Our findings also suggest that caution should be exercised when an ECC diagnosis reveals CIN2+ lesions in biopsy-confirmed CIN2 cases, as these women have a 3.067-fold increased risk of harboring potential CIN3+ lesions following LLETZ treatment. These results underscore the critical role of ECC in guiding clinical decision-making for women with CIN2. Notably, our study found that 51.2% CIN2 patients (42/82) whose LLETZ pathology was upgraded to ≥CIN3 had a preceding ECC result of <CIN2. Wang et al. 34 similarly reported that 28.1% of patients ultimately diagnosed with high-grade pathology had pre-operative ECC results that were either benign or indicated only LSIL. The higher false-negative rate of ECC in our study is primarily attributed to the blind sampling nature of ECC, particularly in reproductive-age women where high-grade lesions can be focal and in the early stage. Given this false-negative rate, clinical decision-making of ECC <CIN2 patients should be guided by a comprehensive evaluation that incorporates the patient’s history and current HR-HPV and cytology results.
This study has several strengths. First, in the context of China’s evolving “three-child” policy and shifting demographic trends, this study presents the evidence-based risk stratification for managing CIN2 in reproductive-age women. This approach seeks to address the critical balance between oncological safety and fertility preservation. Second, we highlight that the predictive value of HPV infection, TCT results, and ECC pathology for pathological progression following LLETZ among reproductive-age women with biopsy-confirmed CIN2, is easily assessed and may support clinical decision-making regarding the use of LLETZ in reproductive-age women with CIN2 lesions.
This study has some limitations. The most notable limitations include its retrospective design and relatively small sample size derived from a single center, which may compromise the statistical power of the study. Besides, these findings should be interpreted in the context of secondary and tertiary hospitals in China. Caution is advised when generalizing the results to populations in primary care settings or with different screening protocols. Finally, owing to the lack of clinical guidelines, women were managed differently between hospitals and within each hospital, depending on the colposcopist. This introduces the risk of selection bias and makes it difficult to compare the data.
Conclusion
In this population-based study of 516 women under 45 years with biopsy-confirmed CIN2, the absolute risk of CIN3 or worse lesions was low (18.4%) post-LLETZ. Age did not increase the risk of developing CIN3+ lesions. TCT diagnosis (HSIL), HPV infections (HPV 16, and 33 infections) and ECC diagnosis (≥CIN2) were independent factors of pathological upgrade after LLETZ. Women without these risk factors may be suitable candidates for fertility-preserving conservative management and active surveillance. Under China’s current social fertility policy, the findings of this study may serve as a valuable reference for clinical counseling and triage strategies for women with CIN2 who are seeking fertility options.
Supplemental Material
Supplemental Material - Risk Stratification for Underlying Cervical Intraepithelial Neoplasia Grade (CIN)3+ in Reproductive-Age Women With Biopsy-Confirmed CIN2: Implications for Fertility-Preserving Management
Supplemental Material for Risk Stratification for Underlying Cervical Intraepithelial Neoplasia Grade (CIN)3+ in Reproductive-Age Women With Biopsy-Confirmed CIN2: Implications for Fertility-Preserving Management by Hangjing Gao, Qiao Gao, Lifang Xue, Hao Lin, Diling Pan, Xiqi Huang, Huifeng Xue, Zhenhong Wang, Xiaoqi Sun in Cancer Control
Footnotes
Acknowledgment
The authors thank all the participants for their great efforts.
Ethical Considerations
This study complied with the Declaration of Helsinki and was approved by the Ethics Committee of Fujian Maternity and Child Health Hospital (2024KY272) on December 12, 2024. Informed consent was waived by the Ethics Committee of Fujian Maternity and Child Health Hospital owing to the retrospective nature of the study and the fact that the analysis used anonymous clinical data.
Author Contributions
Conception and design: HJG, QG, XQS.
Collection and assembly of data: HJG, QG, LFX, ZHW, XQS.
Data analysis and interpretation: HJG, QG, ZHW, XQS.
Manuscript writing: All authors.
Final approval of manuscript: All authors.
Funding
No funding was received for conducting this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due to restrictions on medical records and patient privacy, but are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
