Abstract
Background
Cervical cancer incidence and mortality in Estonia are among the highest in Europe, although the overall coverage with cervical cytology is high. This indicates potential issues with the quality of collection and/or laboratory evaluation of cervical cytology.
Objectives
The aim of the retrospective observational study was to assess the quality of cervical cytology specimen collection, evaluation, and reporting using laboratory reports in Estonia.
Methods
The study included women with a cervical cancer diagnosis in 2017−2018. Cervical cytology and histology reports for these women in 2007−2018 were obtained from ten laboratories. We described the quality of cytology specimen collection and reporting of cytology results. Multivariate logistic regression was used to calculate odds ratios (OR) with 95% confidence intervals (CI) to identify factors associated with NILM as the last cervical cytology result within 5 or 2 years before the cervical cancer diagnosis. Also, we calculated cytology-histology correlation (CHC).
Results
We identified 503 cytology and 100 histology reports from 138 women. The laboratories differed greatly regarding human resources, work capacity and volume. Differences between local and regional laboratories were observed in reporting specimen adequacy (P < .001). We found that local laboratories had 3 times higher odds (OR = 2.95, 95% CI: 1.05−8.33) of reporting normal results 2 years before cancer diagnosis than regional laboratories. According to the CHC, 58.9% of pairs were in agreement.
Conclusions
The study showed considerable heterogeneity and suboptimal performance of cervical cytology practices in Estonia, particularly at local laboratories. Efforts to improve laboratory quality assurance are crucial.
Keywords
Introduction
The high and stable cervical cancer incidence in Estonia has been a reason for concern for many years. 1 According to 2020 estimates, the incidence rate of cervical cancer in Estonia was the second highest in Europe reaching up to 18.8 per 100 000 women. 2 Despite the nationwide cervical cancer screening program since 2006 and good access to the healthcare providers, the annual screening coverage in the organised program has remained well under recommended 70%. In 2018, 25 445 (45.3%) invited women attended the screening, 3 but the total number of cervical cytology tests performed was 105 000, showing a high opportunistic screening prevalence, as usually 1 woman gives just 1 test. 4 Whereas it is known that opportunistic screening is not as efficient as organised screening, 5 the steadily high number of annual cervical cytology tests should have influenced cancer incidence.
The quality of collecting and evaluating cervical cytology or histology specimens plays a significant role in the early detection of cervical abnormalities and is the basis for further clinical management. 6 Furthermore, in the Estonian organized cervical cancer screening program, cervical cytology is used as a triage test after a positive human papillomavirus (HPV) test since 2021. 7 In case of a false-negative (or inadequate) cytology result, a possible precancer case will be missed, meaning that the high quality of management of cervical cytology samples remain crucial, regardless of the type of primary screening test for cervical cancer.
Except for the 2 audits that included laboratories providing the screening service in 2007 and 2013, the Estonian cervical cancer screening program, in general, has lacked national stakeholder level quality control and assurance. Furthermore, until the establishment of Estonian Cancer Screening Registry in 2015 no individual level screening history data was collected, which has omitted the basis of regular screening related statistics. However, screening history for previous years is available from pathology laboratories and medical history.
The aim of this study was to perform a retrospective assessment of the quality of cervical cytology specimen collection, evaluation, and reporting using laboratory reports from 2007 to 2018 for women diagnosed with cervical cancer in 2017−2018 in Estonia.
Material and Methods
Description of Taking, Assessing, and Reporting Cervical Cytology and Histology in Estonia
Until 2020, cervical cytology was the primary screening test within the Estonian cervical cancer screening program, offered every 5 years to women aged 30 to 55 in the specific birth cohort. However, cytology tests could have been and can be done at any time and are reimbursed for women insured by the Health Insurance Fund. Regardless of the setting of specimen collection, they should be taken, handled, and evaluated following the same procedural rules and standards, as stated in European guidelines for quality assurance in cervical cancer screening. 8
While cervical cytology can be taken by both gynaecologists and midwives, punch or excision biopsies can be taken only by a trained gynaecologist. According to the European guidelines, 8 the cytology must include samples from both ecto- and endocervix. After fixation, the specimen will be sent to the laboratory, where it will be handled, Papanicolaou stained, and assessed according to laboratory internal rules by the cytotechnologist or pathologist. In regional laboratories, cytotechnologists usually report only normal results, while the abnormal cytology tests are further referred to the residency trained pathologists. Cytotechnologists’ education and training are not nationally coordinated and rely currently on in-house training. Histology specimens are evaluated only by trained pathologists.
The cervical cytology and histology results will be described in the laboratory report, which will be shared with the referred midwife or gynaecologist, who is responsible for informing the women and managing the patient in case of an abnormal result. The template for reports is dependent on the software the laboratory is using.
Quality recommendations for laboratories that are evaluating cervical cytology have been introduced; however, since there is no implemented monitoring system, or penalties for not fulfilling those recommendations, the laboratory quality has remained on everyone’s good will and trust.
Since 2006, cervical cytology should have been reported in the Bethesda System (TBS); 9 however, it is known that both TBS and Papanicolaou classification systems have been in use over time. For histology, Tavassoli et al 2003 were replaced by Kurman et al 2014 classification 10 in 2014.
Until 2015, the cervical cytology and histology results were documented only in a laboratory report and in medical records. The first will remain in the laboratory, and the latter is accessible only to a specific healthcare provider. In 2015, the Estonian Cancer Screening Registry was established which collects data on all primary and follow-up screening tests. However, it is limited to women who belong to the screened birth cohort within an organised screening program in a particular year, excluding all other tests. Since 2018, cervical cytology and histology reports are also available from central Health Information System, however, the data quality has not been evaluated.
Study Design and Data Sources
In this nationwide retrospective observational study, we included 319 women who were, according to the Estonian Cancer Registry, diagnosed with cervical cancer (C53) or cervical carcinoma in situ (D06) in the years 2017 and 2018. Estonian Cancer Registry is a nationwide population-based cancer registry with data available since 1968, and as a result of compulsory cancer case reporting, the registry has high data validity and quality. 11 We used each woman’s unique personal identification code to search laboratory reports from ten known pathology laboratories covering the whole Estonia that assessed cervical cytology and histology samples between 2007 to 2018. We excluded all reports within 6 months before cancer diagnosis since we considered these tests to be already diagnostic. At least 1 cytology report meeting these criteria was obtained for 138 women; the total number of reports was 503.
Data Collection From Laboratory Reports
From each collected report, we extracted the following information: specimen collection date; specimen type (cytology or histology); qualification of sampler (gynaecologist or midwife); laboratory type where the specimen was evaluated; specimen evaluation date; qualification of specimen evaluator (pathologists or cytotechnologist); the final diagnosis according to the TBS; and presence of additional comments on specimen quality, or suggestions for follow-up. Diagnoses of PAP I/PAP II, PAP III, PAP IV, and PAP V were translated to the TBS as negative for intraepithelial lesions or malignancy (NILM), low-grade squamous intraepithelial lesion (LSIL), high-grade squamous intraepithelial lesion (HSIL), and cancer, respectively. Due to several PAP III and PAP IV diagnosis translation possibilities, we chose the most severe corresponding diagnosis in the TBS to avoid underestimations.
To evaluate the aspects of cytology specimen collection, we recorded how pathologists or cytotechnologists reported specimen adequacy (both ecto- and endocervical specimen was adequate; only 1 specimen was adequate/adequate but limited; both specimens were inadequate for evaluation; adequacy was not reported), and whether the squamous and glandular cells were present in the specimen (both squamous and glandular cells present; only squamous cells present; only glandular cells present; both squamous and glandular cells were absent).
To assess how well cytology specimens were described in reports, we recorded whether the presence of both squamous and glandular cells was noted or not. Also, we recorded additional comments related to the quality of the specimen or suggestions for further follow-up and in which cytology classification system was used.
To assess the correlation between cytology and follow-up histology results (CHC), we identified all cytology tests, followed by histology within the next 6 months. We assessed CHC on a grade-to-grade basis using the Gupta et al 12 suggested discrepancy assessment grid.
In addition, we asked each laboratory for self-reported information regarding their yearly number of cytology and histology tests; the number of pathologists and cytotechnologists involved in cytology evaluation daily; the number of pathologists who are involved with histology assessment; the number of healthcare providers using the laboratory to assess cervical cytology tests and/or biopsies; and presence of regular feedback routine between the laboratory and health-care providers (yes/no). We asked for the information reflecting the situation before the global pandemic (or the last working year) since the healthcare services capacity was generally reduced in 2019−2021. The laboratories were grouped into local and regional based on their affiliation, number of annual cytology tests, and human resources capacity. Among ten included laboratories, 5 were classified as regional and 5 as local laboratories.
All the data is handled carefully with the highest respect to ensure that any person’s identity may not be ascertained in any way in this or any other publication.
Statistical Analysis
This population-based study includes all the women with a confirmed cervical cancer diagnosis to evaluate the quality of taking and assessing cervical cytology. Data is collected from all Estonia pathology laboratories, giving complete nationwide coverage. Hence, no sample size calculations before the study were conducted.
Since there were no major changes in health care settings or legislation, the study period was divided into 2 6-year periods to study changes over time: (1) 2007−2012 and (2) 2013−2018. We used cross-tabulations and percentages to describe the distribution between the groups. The chi-square test and Fisher’s exact test were used to distinguish statistical differences between the groups.
Cytology results reported as NILM less than 2 years before a confirmed cancer diagnosis are considered a false negative and indicate laboratory work quality issues. 13 We used univariate and multivariate logistic regression and calculated the odds ratio (OR) with 95% confidence interval (CI) to evaluate the association between NILM as the last cervical cytology result within 5 or 2 years before the cervical cancer diagnosis and the following factors related to the last cytology test: woman’s age at the time of specimen collection, the qualification of specimen evaluator, and laboratory type. All these factors showed significant association in univariate analyses and were therefore included in the final model.
The reporting of this study conforms to STROBE guidelines. 14
Ethics
The study protocol was approved by the Research Ethics Committee of the National Institute for Health Development (decision no. 632, date 26.01.2021).
Results
Characteristics of laboratories evaluating cervical cytology and histology, Estonia 2007−2018.
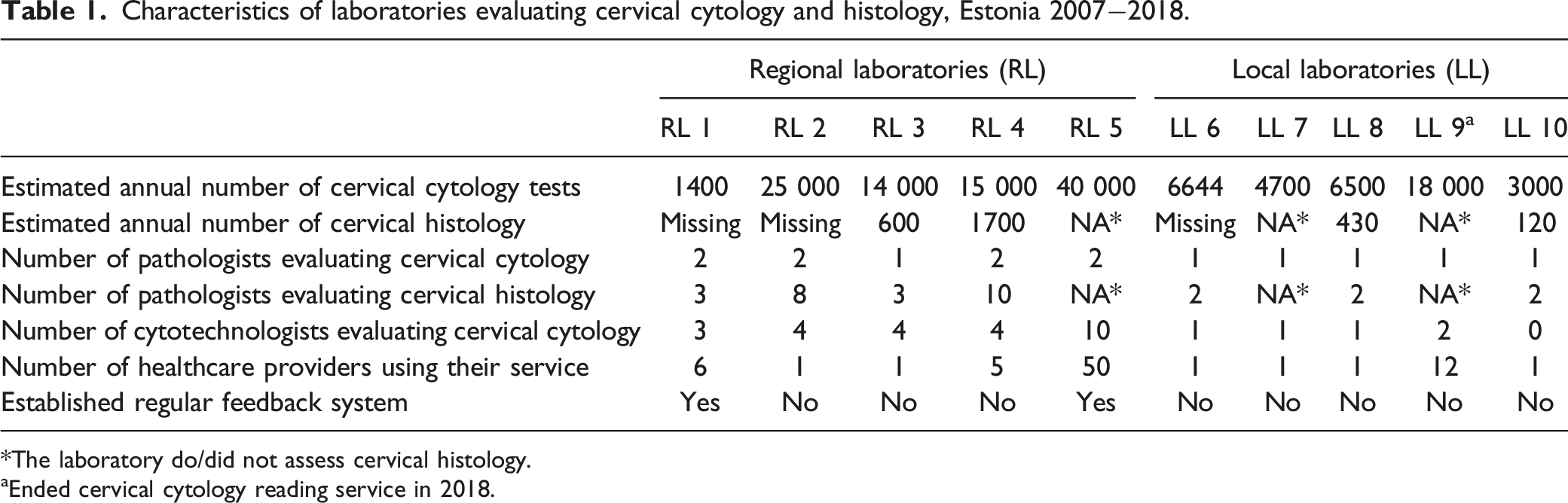
*The laboratory do/did not assess cervical histology.
aEnded cervical cytology reading service in 2018.
Cervical Cytology
Aspects related to the reported quality of cervical cytology specimen collection, Estonia 2007−2018.

*P-value calculation is excluding the reports without noted adequacy (N = 198).
Aspects related to the quality of reporting cervical cytology results, Estonia 2007−2018.

NILM = negative for intraepithelial lesions or malignancy; ASCUS = atypical squamous cells of undetermined significance; AGUS = atypical glandular cells; LSIL = low-grade squamous intraepithelial lesion; ASC-H = atypical squamous cells cannot rule out HSIL; HSIL = high-grade squamous intraepithelial lesion; TBS = The Bethesda System.
The proportion of adequately taken tests was comparable between gynaecologists and midwives, 47.7% vs 41.9%, respectively (P = .61) (Table 2). Half of the assessed samples (47.3%) had both ecto- and endocervical cells, 12.1% had only either cells or the adequacy was limited, and 1.2% of the samples were inadequate for evaluation (Table 2). Adequacy was not reported for nearly half of the specimens in 2007-2012 and for about a quarter of the specimens in 2013-2018. The proportion of adequately taken cytology samples increased from 37.4% to 62.6% (P < .001). 74.5% of the samples were reported to have the presence of both squamous and glandular cells, and 24.4% only presence of squamous cells. This distribution was similar across the profession of the sampler (P = 1.00) and time periods (P = .40) (Table 2).
The majority (90.5%) of cytology reports noted the presence or absence of squamous and glandular cells, and no differences were observed by the evaluator qualification (P = .87) nor by the laboratory type (P = .58). We noticed an improvement over time (P = .01), and in 2013−2018, 94.4% of reports noted the presence or absence of both cell types (Table 3).
Factors associated with NILM as the last cervical cytology result within 5 or 2 years before the cervical cancer diagnosis, Estonia 2007−2018.

OR = odds ratio; CI = confidence interval.
*Adjusted for all variables in the table. Statistically significant results (<.05) are marked in bold.
Cervical Histology
We identified 100 cervical histology reports; the majority of which were analysed in regional laboratories (92.0%), in 2013−2018 (68.0%), and resulted in other diagnoses, not related to cervical intraepithelial neoplasia or glandular changes (usually cervicitis) (40.0%) (data not shown). We identified 73 cytology tests followed by cervical histology within 6 months, and among them, 58.9% were in agreement according to the Gupta et al criteria (Figure 1). Agreement between cervical cytology and histology results, Estonia 2007−2018.
Discussion
This is the first study ever to assess the quality of specimen collection and reporting of cervical cytology and histology in Estonia. We found that within 10 years prior to the diagnosis of cervical cancer or carcinoma in situ, NILM and LSIL cases were more often diagnosed in local laboratories than in regional laboratories. This resulted in almost 3 times higher odds of NILM diagnosis as the last result within a short period before cancer diagnosis for women whose cytology was evaluated in local laboratories compared to regional laboratories.
The described difference between the laboratories in terms of workforce and lower number of annually processed cytology tests most likely contributes to the suboptimal quality of specimen assessment in local laboratories. Given that cytology reading is a subjective matter that needs constant exposure, local laboratories are not reaching the optimal volume, which according to a recent study would be 9000 slides per year 13 and according to the European guidelines for quality assurance in cervical cancer screening is 15 000 slides per year. 8 Currently, over half of the laboratories in Estonia fall under these suggested thresholds.
Like in many other countries, 15 the number of pathologists is decreasing due to the retirement and/or low popularity of this field, which makes it rational to delegate reporting normal cytology tests to trained cytotechnologists. The importance of high standard training and education for cytotechnologists has been recently raised by the European Advisory Committee of Cytotechnology and the European Federation of Cytology Societies. 16 Due to known variations in qualification, methods, quality, level of education and the profession recognition, and the shift from conventional cervical cytology to HPV screening, it is crucial to harmonize the educational programs for cytotechnologists in Europe. Estonia would benefit from this proposal since, until now, the main training for cervical cytology reading will take place in the laboratories on an individual level. Though the trainee must have a degree in biomedical science, the one-on-one base training is not sustainable.
In 2021, Estonia introduced HPV test as a primary screening test, followed by the liquid-based cytology (LBC) triage test. 7 Since the LBC and conventional cytology reading differ, 17 special training is required, which is again up to laboratories and their willingness.
CHC assessment showed that in 58.9% of all correlating pairs, the cytology results agreed with histological diagnosis and 24.6% pairs had minor under- or overcall. While the major undercall percentage was low (1.4%), 15.1% of cases had major overcall where cytology results ASC-H and HSIL were followed by a histological diagnosis of NILM or chronic cervicitis. It is difficult to say if the found disagreement was related to either pathologist (as they assess all the abnormal cytology test) or gynaecologists (as almost all the tests were taken by them) work. Results from similar studies vary, and the reported agreement rate ranges from 58% in Russia 18 to 73% in the US. 12 While undercall is more dangerous from the health perspective, we cannot underestimate the damage of overcall since it causes increased anxiety for patients and adds health care costs due to unneeded additional investigations. 19
In Estonia, both gynaecologists and midwives can take conventional cytology or LBC samples. Interestingly, we found that the midwives collected less than 7% of all cytology specimens. We could argue that this is due to our specific study cohort, who needed specialist care, but more likely is it a historical convention. While national and international recommendations 8 advise midwives or nurses to do regular screening related procedures, advising, and consulting, many women still want to see the doctor. In terms of both human and financial resources, this finding should be further discussed at the stakeholder level.
In addition, our study revealed that only 2 laboratories out of ten have regular feedback system with the physicians in place. While some other laboratories claimed that they contact physician when it is needed, the literature is highlighting the benefits of regular contact to improve the quality of taken samples. 8
This study benefits from the data collected right from the source. We extracted information directly from the reports, which resulted in a complete dataset and reduced mis-classification errors. The study period expands over 10 years, allowing us to evaluate the whole cytology history for each woman, and see the changes in laboratory practices over time. Indeed, we missed some laboratory reports, however, they were mainly from the early years (data not shown) and should not have impacted our results. Further studies including rereading and -evaluation of cytology slides are warranted to provide more in-depth information on the quality of cervical cytology.
This study revealed several shortcomings in the Estonian health care system. First, the absence of centralized data on all cytology and histology reports has inhibited systematic and regular program evaluations. The lack of data and initiative has led to a situation where, until this day, no stakeholder level quality assessment (besides annual screening coverage calculations) has been enforced. Estonia has had national, international, and academic society-based recommendations that have not been binding, however, they have had not showed enough hoped positive effect on the changes in laboratory work management. For health insured women, and since 2021 for all the women in screening cohort, the Estonian Health Insurance Fund reimbursed all the performed cytology tests, regardless in which laboratory the test is assessed. Therefore, we see that the only way how to improve the quality is to introduce national law together with monitoring body which assesses and monitors the quality of work in pathology labs. This non-existent quality assurance has commanded a situation where women’s health and future depend on the laboratory type where their sample is assessed. Second, we noted the underuse of cytotechnologists and midwives. Both types of specialists would increase accessibility and quality of screening program while decreasing general healthcare costs. Lastly, reducing number of laboratories would allow to optimise workload, human resources and knowledge and thus increase the quality.
Conclusion
This study provides Estonia with an important landmark and points out the weaknesses in current cervical cancer prevention that need to be addressed, such as heterogeneity in terms of laboratory type, cytology reading capacity and volume, lack of standardised pathology reports and the need for regular quality assessment of services. In order to prevent avoidable cervical cancer diagnoses, quality assurance must be enforced on national legislative level. The volume of annual cervical cytology tests in Estonia suggest 1 or 2 reference laboratories is sufficient.
Abbreviations
atypical squamous cells cannot rule out HSIL
atypical glandular cells
atypical squamous cells of undetermined significance
cervical intraepithelial neoplasia grade 1/2/3
cytology histology correlation
confidence interval
human papillomavirus
high-grade squamous intraepithelial lesion
liquid-based cytology
low-grade squamous intraepithelial lesion
negative for intraepithelial lesions or malignancy
odds ratio
the Bethesda System
Footnotes
Acknowledgments
The authors would like to thank all the laboratories for their cooperation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Mobilitas Pluss (Project number MOBJD579) and by the Estonian Research Council (Grant No. PRG722). The funding bodies had no role in the study design, data collection, data analysis, interpretation of data, writing of the report, or the decision to submit the paper for publication.
Ethical Approval
The study protocol was approved by the Research Ethics Committee of the National Institute for Health Development (decision no. 632, date 26.01.2021).
