Abstract
Bone metastases are a common and devastating complication of advanced solid tumors, significantly impairing patients’ quality of life and prognosis. Immune checkpoint inhibitor (ICI) plus bone-targeted therapies have emerged as a promising strategy to improve outcomes in patients with bone metastases. Denosumab, a fully human mAb targeting RANKL, can inhibit osteoclast-mediated bone resorption and prevent skeletal-related events. Beyond its direct bone remodeling activity, denosumab can also modulate the immune microenvironment, enhance anti-tumor immunity by reducing the release of immunosuppressive factors into the bone milieu, and improve vascular integrity within previously damaged bone tissue to facilitate immune cell infiltration into the metastatic niches. ICIs restore T-cell activity by overcoming tumor-induced immune suppression, thereby enabling more effective anti-tumor responses. Emerging evidence have suggested a potential synergy of denosumab plus ICIs, mechanistically by reinvigorating T-cell function and promoting maturation and antigen-presenting capacity of dendritic cells to further amplify adaptive immune response against metastatic bone tumors. The present narrative review summarizes the preclinical and clinical findings on this combination and discusses potential biomarkers and current challenges for the optimization of patient selection and therapeutic paradigm. Besides, we briefly overview current ongoing studies that investigate the potential antitumor synergy with denosumab plus ICIs in bone metastases.
Plain Language Summary
Bone metastases are a frequent and serious complication in patients with advanced solid cancers, often causing pain, fractures, and reduced quality of life. Combining immune checkpoint inhibitors (ICIs) with bone-targeted therapies has emerged as a potential approach to improve outcomes for these patients. Denosumab, an antibody that blocks RANKL, can prevent bone loss and skeletal-related events by reducing the activity of bone-resorbing cells. Beyond protecting bone, denosumab may also help the immune system fight cancer by improving the bone environment, reducing immunosuppressive factors, and supporting immune cell infiltration into metastatic sites. ICIs work by restoring the activity of T cells, helping the body mount stronger anti-tumor responses. Emerging evidence suggested that combining denosumab with ICIs could enhance the immune attack on cancer in the bone, potentially by boosting Tcell function and improving how immune cells recognize and respond to tumor antigens. This review summarizes the current preclinical and clinical evidence on this combination, discusses potential biomarkers and challenges, and ongoing studies aimed at optimizing treatment strategies in bone metastases.
Introduction
Bone metastases represent a common and severe complication in patients with advanced solid tumors, exhibiting a high incidence, especially in patients with breast cancer, prostate cancer, and lung cancer. 1 The metastatic disease to the bone significantly impacts the patient’s quality of life and prognosis. Under the substantial burden, current treatment strategies for bone metastases are multidisciplinary and tailored to address both tumor progression and skeletal complications. 2
The treatment of bone metastasis can be categorized into therapies targeting the tumors and those addressing tumor-induced bone diseases.
3
As for the tumor-targeting treatment, ICI has been approved as a standard treatment for various malignancies; however, its effectiveness in individuals with bone metastasis was limited by the unique microenvironment characteristics of bone metastases.
4
In the bone microenvironment, osteoblasts, osteoclasts, and tumor cells interact closely: osteoblasts can interact with tumor cells to activate the Wnt signaling pathway, abnormally promoting bone formation, while also producing factors such as IGF-1 and TGF-β to modulate local immunity (Figure 1).5,6 Tumor cells release factors including RANKL and M-CSF, which activate osteoclasts and induce osteolytic lesions.
7
In addition, the bone marrow is enriched with immunosuppressive cells, including Tregs (which preferentially migrate to bone marrow and have been proven as an important source of RANKL), myeloid-derived suppressor cells (MDSCs, which inhibit T cell activation and enhance Treg activity), tumor-associated macrophages (TAMs), and Th17 cells, collectively maintaining local immunosuppression.
6
Then, hypoxia in the bone marrow can also induce upregulation of PD-1/PD-L1, further regulating the immune suppression.8,9 These features together constitute the immunosuppressive microenvironment of bone metastases, which makes ICI monotherapy of limited efficacy. Mechanism of denosumab and immune checkpoint inhibitor combination in the bone tumor microenvironment
As for the latter, the approaches to alleviate tumor-induced bone diseases mainly focus on anti-resorptive drugs. Among them, with advances in research on the role of the RANK-RANKL axis in the pathogenesis of bone metastasis, the anti-RANKL monoclonal antibody denosumab has been widely used in clinical practice to reduce the frequency of SREs in patients with advanced solid tumors and to increase their quality-adjusted life years. 10 In addition to its bone-protective effects, denosumab can also act as a key coordinator between bone biology and tumor immunology. 10 Specifically, in the bone metastasis tumor microenvironment, activated T cells—especially CD8+ T cells—can express RANKL and interact with dendritic cells (DCs), prolonging T-DC contact and thereby activating additional T cells. At the same time, this leads to elevated RANKL levels in the microenvironment, which act on osteoclasts and RANK+ myeloid cells, such as MDSCs, further exacerbating bone destruction and reinforcing immunosuppression. 11 Denosumab, by blocking the RANKL-RANK signaling axis, alleviates immunosuppressive states; additionally, RANKL inhibition can downregulate PD-1 expression on CD8+ T cells, further enhancing T-cell activity and antitumor effects. 12 By improving the immunosuppressive state of the bone metastatic microenvironment, denosumab provides more favorable conditions for ICIs to exert their effects, potentially enhancing overall antitumor efficacy. Overall, the combination of denosumab with ICIs may be an effective comprehensive treatment strategy for patients with bone metastasis, capable of treating the primary tumor, inhibiting bone destruction, and delaying the occurrence of SREs, finally improving prognosis and patients’ quality of life. The present narrative review focused on the preclinical advances and clinical evidence of potential synergistic effects with denosumab plus ICIs in bone metastases from solid tumors. Additionally, we also discussed key challenges and future directions in optimizing this therapeutic approach, providing insights into its potential application in clinical practice. This review is guided by the Scale for the Assessment of Narrative Review Articles SANRA. 13
Methods
A comprehensive literature search was conducted in PubMed, Web of Science, and Google Scholar to identify relevant studies on denosumab combined with ICIs in bone metastases from solid tumors. The search covered articles published from the database inception to June 2024. Keywords included “denosumab”, “immune checkpoint inhibitors”, “bone metastases”, and “RANKL” were used alone or in combination. Preclinical studies, prospective and retrospective clinical studies, relevant review articles, and conference abstracts reporting clinical or translational data were considered. Reference lists of included articles were also manually screened to identify additional relevant studies.
Treatments of Bone Metastases
Overview of Clinical Studies With Denosumab Plus ICIs in Bone Metastases From Solid Tumors Denosumab
Clinically, denosumab in combination with ICIs has mainly been explored for the treatment of bone metastases in melanoma and non-small cell lung cancer (NSCLC). Evidence from these studies generally indicates potential benefits of the combination therapy; however, improvements were not consistently statistically significant across all cohorts.
In retrospective studies of metastatic melanoma, denosumab combined with ICIs has demonstrated promising efficacy. For example, Angela et al. observed that patients receiving denosumab combined with PD-1 inhibitors (dual therapy) or nivolumab plus ipilimumab (triple therapy) achieved encouraging objective response rates (ORR, 50-54%) and progression-free survival (PFS, 6 months). 14 Similarly, Afzal et al. reported comparable efficacy, with patients receiving denosumab plus ICIs achieving an ORR of 54.5% and a median PFS of 4.2 months. 15 However, results from prospective studies have been highly heterogeneous. Gerhardt et al. conducted a prospective, multicenter clinical study involving 16 patients with unresectable stage IV melanoma and found that denosumab plus dual ICIs resulted in modest tumor responses and survival outcomes (ORR, 17.6%; PFS, 2.9 months). 16 In contrast, Moschos et al. observed an ORR of 48%, with a median PFS of 18.1 months and overall survival (OS) of 31.7 months. 17 This discrepancy may be attributable to differences in treatment sequencing, as Moschos et al. administered denosumab alone first and added ICIs on day 21 to form the combination regimen. Interestingly, Mabrut et al. observed in a retrospective study in patients with solid tumors that initiating ICIs prior to bone-targeted therapy was associated with numerically more favorable clinical outcomes compared with bone-targeted therapy prior to ICIs (PFS, 10.9 vs 9.1 months). 18 These findings suggest that the optimal treatment sequence remains to be further investigated.
Clinical Studies With Denosumab Plus ICIs in Bone Metastases From Solid Tumors
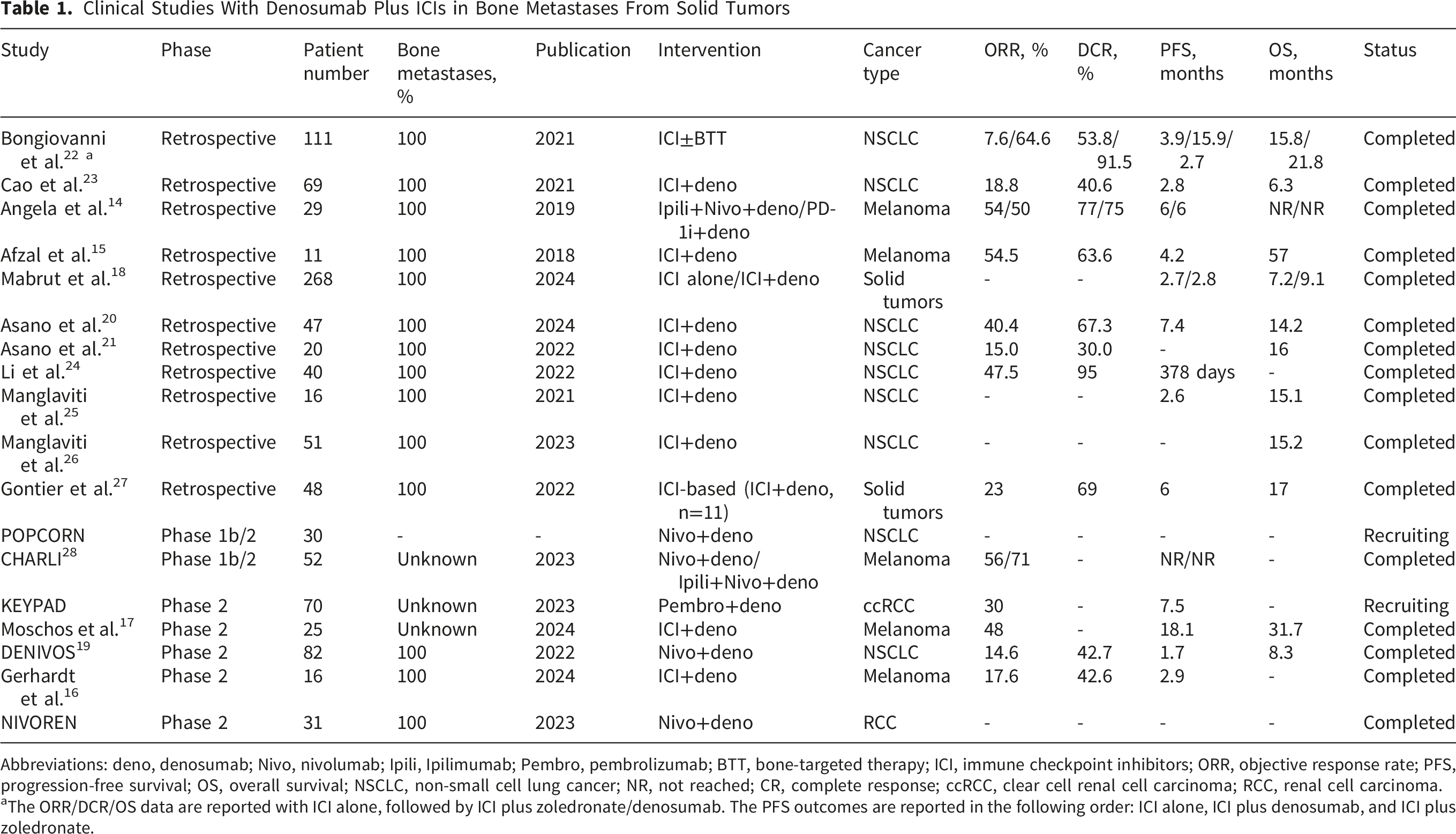
Abbreviations: deno, denosumab; Nivo, nivolumab; Ipili, Ipilimumab; Pembro, pembrolizumab; BTT, bone-targeted therapy; ICI, immune checkpoint inhibitors; ORR, objective response rate; PFS, progression-free survival; OS, overall survival; NSCLC, non-small cell lung cancer; NR, not reached; CR, complete response; ccRCC, clear cell renal cell carcinoma; RCC, renal cell carcinoma.
aThe ORR/DCR/OS data are reported with ICI alone, followed by ICI plus zoledronate/denosumab. The PFS outcomes are reported in the following order: ICI alone, ICI plus denosumab, and ICI plus zoledronate.
Overall, these studies provide signals suggestive of a potential synergistic effect between denosumab and ICIs, indicating that combination therapy may offer clinical benefits for patients with bone metastases. However, both retrospective and prospective studies have notable limitations, including small sample sizes, patient heterogeneity, differences in ICI regimens and treatment duration, and the lack of control groups in most studies. Therefore, these findings should be considered hypothesis-generating, and the current evidence is of moderate-to-low quality. The causal relationship of the potential synergy between denosumab and ICIs remains uncertain and requires validation in prospective, large-scale studies. In addition, the impact of treatment sequencing on the synergistic effect warrants further investigation. It is also unclear whether the observed benefits are primarily due to direct anti-tumor effects or mediated through the bone microenvironment. Finally, the question of whether denosumab is used as a concomitant therapy or one of the main therapeutic components should also be further answered in the future.
Challenges and Prospects
The combination of ICIs and denosumab offers a theoretical benefit and shows promise for the treatment of bone metastases, but faces challenges and uncertainties that require further investigation. These issues include resistance mechanisms arising from the unique characteristics of the bone microenvironment and the irAEs requiring careful consideration and management in clinical practice.
Toxicity Management of irAEs
Patients who received ICIs often experienced irAEs accompanied by the regression of the primary tumors. These irAEs are typically caused by inflammation and the expansion of immune cells and include common manifestations with dermatologic, gastrointestinal, pulmonary, and endocrine toxicities. Despite the broad spectrum of these toxicities, most issues can be managed with close monitoring and symptomatic treatment without necessitating discontinuation of ICI therapy. 4 Recent studies indicated that patients who have previously or concurrently received denosumab treatment alongside anti-PD-1 therapy experience a numerically higher rate of grade 3 or higher irAEs compared to those receiving PD-1 inhibitors alone (21.4% vs. 11.6%). 29 Mechanistically, preclinical studies in mouse models have supported a potential role of anti-RANKL therapy in enhancing ICI-induced immune toxicity, including increased endocrine irAEs and modulation of Treg populations. 29 Additionally, recent case reports have revealed several uncommon irAEs during combination, such as multi-organ toxicities and granulomatous reactions. Although investigators primarily attribute these irAEs to immunotherapy, the complex interactions between the ICIs and denosumab make it difficult to entirely rule out contributions from bone-targeted therapy. Clinicians often prescribed bone-targeted agents as concomitant therapy to prevent SREs; however, denosumab was associated with hypocalcemia, and the inflammatory response induced by immunotherapy may exacerbate this condition. 14 Therefore, careful monitoring of calcium levels and proactive management of hypocalcemia are critical components of care for these patients.
Beyond the commonly observed ICI-induced toxicities, patients may also develop rheumatic and musculoskeletal adverse events (AEs), including arthritis and rheumatoid arthritis.30,31 Moreover, there is a significant increase in bone loss and skeletal complications following ICI therapy, including vertebral compression fractures, femoral neck fractures, vertebral and thoracic fractures, as well as pathological and osteoporotic fractures. A multidisciplinary approach—incorporating oncologists, endocrinologists, and pain management specialists—is essential for effectively managing such toxicities while minimizing their impacts on bone health.
Recently, osteonecrosis of the jaw (ONJ), a well-known and serious AE initially associated with bone-targeted therapies, has been identified as a new irAE in patients receiving ICIs, with or without bone-targeted agents such as denosumab. Patients typically present with recurrent jaw infections, accompanied by soft tissue swelling and pain, which can progress to jaw necrosis and bone exposure, and pathological fractures in severe cases. Moreover, these complications often necessitate the discontinuation or modification of immunotherapy, potentially compromising treatment efficacy and negatively impacting prognosis.14,18,32 This underscores the need for heightened vigilance regarding ONJ as an unforeseen irAE. A comprehensive oral examination is recommended both before and during treatment to facilitate early diagnosis and optimal patient management.
In addition to the aforementioned AEs, case reports have also identified systemic bone loss and/or focal bone lesions in patients receiving α-PD-1 or α-PD-1/α-CTLA-4 therapy. Although the underlying mechanisms remain poorly understood, these effects may be linked to disrupted bone remodeling in a pro-inflammatory state, because all reported cases demonstrated elevated levels of the bone resorption marker CTX. 30
Additionally, Álvaro et al. reported that denosumab might induce transient but severe neuropsychiatric symptoms, potentially through mechanisms involving RANKL blockade and subsequent immune-inflammatory alterations. 33 Therefore, caution and close monitoring are recommended, particularly in patients with preexisting neurobiological vulnerabilities, to ensure early detection and appropriate management of potential AEs.
Clinically Reported IrAEs Associated With Denosumab and ICIs in Bone Metastases

Abbreviations: irAEs, immune-related adverse events; ICIs, immune checkpoint inhibitors; CTX, C-telopeptide levels.
aSummarizes the irAEs reported so far in patients receiving denosumab plus ICIs; management strategies are based on ASCO guidelines. 34
bRare irAEs’ management strategies were the specific interventions applied to each patient.
Overcome Resistance
Resistant Mechanism of ICIs in Bone Metastases
One of the primary mechanisms of immune evasion in bone metastases was the ability of tumor cells to manipulate the bone microenvironment, creating an immunosuppressive milieu that inhibited effective immune responses. Currently, commonly used PD-1/PD-L1/CTLA-4 inhibitors function by blocking immune checkpoints, thereby activating T lymphocytes and promoting an anti-tumor immune response. However, the presence of MDSCs in the bone marrow has been shown to inhibit T cell activation and facilitate tumor progression. 6 In parallel, Tregs within the bone marrow further exacerbated this immunosuppressive environment by inhibiting immune cell function and contributing to poor therapeutic responses. 35 Persistent hypoxia in bone marrow metastases further enhanced immunosuppression by upregulating PD-L1 expression in tumor cells, dendritic cells, macrophages, and MDSCs, which not only restricted T cell activity but also increased the prevalence of immunosuppressive Tregs and TAMs, ultimately limiting the host’s immune response to tumor growth.4,35 Preclinical studies have explored the use of nanoparticles to alleviate hypoxia in bone metastases, reducing Treg levels and improving survival rates in mice models of established bone metastases. 35
Moreover, the inherent heterogeneity of bone metastases may also influence the immune landscape within the bone, leading to cancer-type-specific responses and resistance patterns to ICIs. 36 For instance, in osteoblastic lesions, immune cells were enriched with phosphorylated STAT3, components of the JAK-STAT pathway, and key immune checkpoints such as PD-L1, B7-H4, OX40L, and IDO-1, offering potential therapeutic targets for patients with bone metastases. By contrast, osteolytic lesions exhibited an increased presence of immune cells (including cytotoxic cells, macrophages, exhausted CD8+ T cells, CD45+ immune cells, neutrophils, and mast cells) rich in phosphorylated AKT and components of the PI3K-AKT pathway, which were associated with reduced bone mass. 36 Overall, these emerging insights into resistance mechanisms provide a foundation for future clinical studies aimed at improving the efficacy of immunotherapy in bone metastases.
Theoretical Breakthrough Points for the Combination Therapy
From a mechanistic perspective, denosumab targets and blocks the RANK-RANKL axis, disrupting the immunosuppressive network established within the bone metastatic microenvironment, thereby alleviating myeloid cell-mediated inhibition of anti-tumor immunity and partially reversing ICIs resistance associated with bone metastases. Preclinical studies have demonstrated that denosumab combined with ICIs can markedly increase the infiltration of CD8+ and CD4+ T cells within tumor tissues and enhance anti-tumor immune responses.11,12 Clinically, in melanoma patients receiving dual ICIs plus denosumab, an increase in circulating CD8+ T cells and elevated levels of pro-inflammatory cytokines in the serum were observed, consistent with the trends seen in preclinical studies. 16 Moreover, although direct evidence remains limited, the addition of denosumab may favorably remodel the bone metastatic microenvironment. For example, it may inhibit osteoclast activity via RANKL blockade, potentially normalizing the vascular network and alleviating hypoxia at metastatic sites; since the RANK-RANKL axis can promote the activity or accumulation of immunosuppressive myeloid cells within the bone marrow, denosumab may reduce the enrichment of these cells. These potential effects, however, require further investigation for confirmation.
Based on the ability of denosumab to improve the bone metastatic microenvironment and partially reverse resistance to ICIs, the sequence of administering denosumab before ICIs could theoretically provide greater clinical benefit. However, as discussed above, current evidence does not establish which treatment sequence is optimal, and this question remains to be further investigated.
Potential Biomarkers for Predicting the Efficacy of ICI With or Without Denosumab in Bone Metastases From Solid Tumors
A key focus is the identification of specific biomarkers that can predict responses to immunotherapy, thereby optimizing patient selection. Previous studies have highlighted the potential of various biomarkers, including protein expression profiles of primary tumors—such as the combined expression of macrophage capping protein and GIPC PDZ domain-containing protein—as well as gene expression signatures and circulating tumor cells. 37 However, research on predictive biomarkers for ICIs in the treatment of bone metastases remains ongoing, and further studies are needed to establish reliable indicators for therapeutic response. Alberto et al. demonstrated that a pre-treatment neutrophil-to-lymphocyte ratio (NLR) ≤5 was associated with a favorable OS in NSCLC patients with bone metastases receiving ICIs, with or without bone-targeted therapy. 22 Asano et al. developed two novel predictive scoring systems—one incorporating dynamic changes in serum inflammatory markers of NLR and C-reactive protein (CRP) levels, and another (IMMUNO-SCORE) incorporating serum inflammatory and nutritional markers such as CRP, NLR, albumin, and the prognostic nutritional index, which both revealed significant correlations with clinical outcomes during ICI treatment in NSCLC with bone metastases. 38 Some retrospective studies have identified potential predictive factors, such as HBTB, PD-L1 tumor proportion score, and an ICI plus denosumab treatment duration of more than three months.23,26 Overall, the current evidence regarding predictive biomarkers for ICI combined with or without denosumab in bone metastases remains low-level. Further research is needed to refine patient stratification based on the likelihood of benefiting from immunotherapy, ultimately advancing toward a more personalized treatment approach.
Strengths and Limitations
This review has summarized the current state of bone metastases from multiple perspectives, including epidemiology, disease diagnosis, clinical characteristics, pathogenesis, and the clinical application of ICI therapy combined with denosumab. As a narrative review, it provides an overview and synthesis of the existing evidence, with an emphasis on integrating clinical and mechanistic findings. However, several limitations should be acknowledged. First, the literature search was conducted using PubMed, Web of Science, and Google Scholar, while other databases were not systematically searched, which may have resulted in the omission of relevant studies. Nevertheless, as a narrative review, study selection was performed in a thoughtful and purposive manner, with explicit and transparent justification for inclusion, aiming to provide a balanced and comprehensive overview of the available evidence. Then, no quantitative synthesis or meta-analysis was performed, and publication bias cannot be excluded.
Conclusion
Bone remains one of the most common metastatic sites, particularly in breast cancer, prostate cancer, and lung cancer. Although ICIs have demonstrated remarkable success in treating primary tumors and certain distant metastases, and denosumab has shown promising bone-protective effects, existing clinical evidence preliminarily suggests that their combination may improve survival outcomes in patients with bone metastases; however, the therapeutic effect has not yet reached the expected level. It must be emphasized that the synergistic efficacy of this combination in bone metastases remains in the preliminary exploration stage. Most available evidence originates from retrospective studies, which are inherently subject to bias and heterogeneity, while prospective studies remain limited, often involving small sample sizes or single-arm designs. Therefore, the synergistic effect must be confirmed through high-quality prospective trials before being incorporated into clinical implementation.
Significant knowledge gaps remain regarding the combined use of ICIs and denosumab, highlighting the urgent need for randomized controlled trials in baseline-balanced populations and for clarifying whether denosumab primarily functions as a bone-protective agent or an immune modulator in exerting synergistic effects. Future research should prioritize: (i) designing rigorous prospective randomized controlled trials to determine the efficacy of the combination therapy and to explore the optimal sequencing of administration; (ii) exploring bone metastasis-specific predictive biomarkers for patient stratification; (iii) establishing expert consensus on toxicity management for combination regimens; and (iv) further elucidating the molecular and immunological mechanisms underlying the combined effects to inform optimized therapeutic strategies. Until such high-level evidence becomes available, this combination strategy must be approached with caution. In current clinical practice, denosumab may be used in patients receiving ICIs according to standard indications for SRE prevention, but it should not yet be considered a routine immune-enhancing agent. Clinicians should carefully balance patient survival, disease status, and economic factors while ensuring the best possible quality of life for patients.
Footnotes
Ethical Considerations
This manuscript is a review article and does not involve a research protocol requiring approval by the relevant institutional review board or ethics committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the 1. Gansu Science and Technology Project (24JRRA314). 2. Excellent Young Talents Project from Gansu Provincial Department of Health (GSWSQN2024-10). 3. Chronic Disease Management Research Project of National Health Commission Capacity Building and Continuing Education Center (GWJJMB202510022031).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
