Abstract
Introduction
The aim of this study was to determine the success rate of alternative chemotherapy regimens, and related factors, in Gestational Trophoblastic Neoplasia (GTN) patients who failed first-line Methotrexate (MTX) chemotherapy at Tu Du Hospital, Vietnam.
Methods
This was a retrospective cohort study of 124 patients with post-molar GTN resistant to MTX, who were treated between January 2018 and December 2023 at Tu Du Hospital. We used the log-rank test and Cox proportional hazard model to determine factors related to failure of alternative chemotherapy.
Results
The success rate of alternative chemotherapy was 89.52%. There were 13/124 cases requiring salvage chemotherapy; the salvage chemotherapy rate in the Act-D group was 16.67%, and in the EMA-CO group 3.44%. Based on the multivariate regression analysis model, the prognostic factors for failure of alternative chemotherapy were the number of MTX chemotherapy cycles < 4 (HR = 0.24, 95% CI = 0.07 – 0.83, p=0.024), and the alternative chemotherapy regimen being Act-D (HR = 9.51, 95% CI = 2.27 – 39.82, p=0.002). Regarding safety, EMA-CO was associated with significantly higher hematological toxicity. Grade 3-4 neutropenia and leukopenia occurred in 56.9% and 43.1% of the EMA-CO group, respectively, compared to 13.6% and 9.1% in the Act-D group (p<0.001).
Conclusion
The success rate of alternative chemotherapy was 89.52%; factors including the number of MTX chemotherapy cycles and the type of alternative chemotherapy regimen being Act-D were prognostic factors for failure.
Plain Language Summary
This research is a retrospective cohort study that utilized a total sampling technique, involving 124 patients diagnosed with Methotrexate-resistant Gestational Trophoblastic Neoplasia (GTN). It is important to highlight that at present in Vietnam, there is no widely accepted alternative treatment protocol available for cases of GTN that do not respond to Methotrexate (MTX). As a result, treatment decisions primarily depend on the judgment and experience of the healthcare professionals involved. According to research findings, the alternative treatment regimen known as EMA-CO has shown to be more effective compared to Actinomycin D (Act-D). Nevertheless, in particular circumstances, Act-D remains a viable treatment option for some patients.
Introduction
Gestational Trophoblastic Neoplasia (GTN) is a curable malignancy. The incidence of GTN in patients with initial Methotrexate (MTX) monotherapy resistance ranges from 10-30%. Most MTX-resistant patients respond to alternative monotherapy (Actinomycin D - Act-D), only 5-10% require multi-chemotherapy.1,2 Currently, there are no randomized clinical trials comparing the efficacy of monotherapy (Act-D) and multi-chemotherapy (EMA-CO, BEP) in the group of GTN patients who fail MTX. Experience at some centers has shown that patients with βhCG < 1,000 IU/L when switching regimens respond well to Act-D (93% success), and βhCG > 1,000 IU/L are often indicated for multi-chemotherapy. 3 However, Act-D is preferred due to its safety, fewer short- and long-term side effects, and lower risk of secondary cancers than etoposide-containing regimens (EMA-CO, BEP), which increase the risk of acute leukemia and early menopause.4-6
Studies show that the response rate of Act-D after MTX failure ranges from 76 - 87%, and failure of monotherapy is closely related to pre-chemotherapy βhCG levels.7-10 However, there is currently no consensus on the “βhCG level” threshold - the threshold to decide to switch to multi-chemotherapy in MTX-resistant GTN (ranging from 100-1,000 IU/L in different studies).
In Vietnam, Tu Du Hospital is one of the GTN treatment centers in the South of Vietnam, receiving about 200 GTN cases each year. Here, there is no research on the success rate of alternative chemotherapy regimens in GTN patients who fail MTX. The choice of alternative chemotherapy regimen (Act-D or EMA-CO) is mainly based on experience, there are no specific standards. Therefore, this research aims to determine the success rate of alternative chemotherapy regimens in GTN patients who fail MTX chemotherapy at Tu Du Hospital and related factors. The results will contribute to optimizing treatment strategies, reducing unnecessary toxicity from polychemotherapy.
Methodology
Study Design and Population
This is a retrospective cohort study with a total sampling method. Data collection was conducted from January 2025 to May 2025 at Tu Du Hospital. We retrospectively reviewed the medical records of patients diagnosed with Methotrexate-resistant GTN who were treated at the Gynecological Oncology Department between January 2018 and December 2023. Inclusion criteria were: (i) Patients diagnosed with GTN after molar abortion according to FIGO 2000 criteria and had a pathological result of curettage as a vesicular mole, (ii) Patients diagnosed with treatment failure with MTX. Cases were excluded: (i) Incomplete medical records, (ii) GTN after molar abortion without molar abortion and initial follow-up at the TD Hospital, (iii) GTN with pathological findings of trophoblastic tumor at the placenta, epithelial trophoblastic tumor and atypical nodule at the placenta, (iv) Patients did not comply with treatment or discontinued treatment.
Variable Measurement
Data collected from medical records include epidemiological information, GTN pathological characteristics, chemotherapy regimens, and treatment results of GTN. GTN pathological staging is based on FIGO criteria. 11 Chemotherapy regimens: Act-D: Administered at a dose of 0.5 mg intravenously daily for 5 days, repeated every 14 days; EMA-CO: Consisted of Etoposide (100 mg/m2), Methotrexate (300 mg/m2 with folinic acid rescue), and Actinomycin-D (0.5 mg) on Days 1 and 2, followed by Cyclophosphamide (600 mg/m2) and Vincristine (1 mg/m2) on Day 8. Cycles were repeated every 14 days. Response to alternative chemotherapy is assessed according to Goldstein and Berkowitz criteria. Healing is achieved when all criteria are met: (i) βhCG/blood < 5 mIU/mL 3 consecutive times, (ii) Normal uterus and 2 adnexa, (iii) Metastases disappear. Failure occurs when one of the following criteria is met, requiring switching to salvage chemotherapy: (i) hCG levels do not respond adequately after 2 consecutive chemotherapy cycles. No response to chemotherapy when β-hCG concentration plateaus (change in β-hCG concentration ± 10% after 1 chemotherapy cycle) or resistance to chemotherapy (β-hCG concentration increases > 10% after 1 cycle or plateaus over 2 chemotherapy cycles), (ii) Clinical progression: new metastases are recorded, (iii) Paraclinical: new metastases on ultrasound, X-ray or MRI if available, (iv) Toxicity in organs: bone marrow, liver, kidney, digestive tract still not improved after a maximum of 2 weeks of rest and supportive treatment. 12
The method of collecting total sampling was applied from January 2018 to December 2023. During chemotherapy, patients were monitored for the following characteristics: (1) signs of vaginal bleeding, uterine size, corpus luteum cysts, vaginal metastases if any, (2) β-hCG concentration chart, (3) blood test, liver function, kidney function, electrolytes and (4) detection of side effects of chemotherapy. After discharge, patients were re-examined and monitored for β-hCG concentration in the blood every month for 6 months, then every 2 for the next 6 months, and every 3 months for 12 months, then every 6 months. The follow-up period lasted for 5 years.
Sample Size
The sample size calculation formula pertinent to survival studies was used. The threshold for the minimum number of failure cases with alternative chemotherapy is at least
Statistical Analysis
We analyzed the data using Stata 17.0 software (Stata Corp LLC, Lakeway Drive College Station, April 2021-TX, USA). Time to salvage chemotherapy (in weeks) was defined as the time interval from the start of alternative chemotherapy to the switch to salvage chemotherapy. Descriptive analyses using percentages and median values were performed to examine the epidemiological and pathological characteristics of GTN. We used the life table method to estimate the cumulative probability of needing salvage chemotherapy. Comparisons of time to salvage chemotherapy between groups were performed using the Log-rank test. We used Cox proportional hazard regression models to examine the relationship between salvage chemotherapy and associated factors in univariate and multivariate analyses. Variables included in the multivariate model were selected based on statistically significant associations in univariate analysis (p < 0.25). Statistical significance was achieved when p < 0.05.
Ethical Considerations
This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. Ethics approval for the study was obtained from the Institutional Review Board of the University of Medicine and Pharmacy at Ho Chi Minh City (No: 3206/UMP-Board, 30th October 2024). The informed consent was waived due to the retrospective design of the study. Data was kept anonymous and confidential during all stages of the study.
The reporting of this study conforms to the STROBE guidelines. 14
Results
From January 2018 to December 2023, 675 cases were diagnosed with trophoblastic tumor (ICD-10 D39.2) at the Department of Gynecological Oncology, Tu Du Hospital. After reviewing the records, we recorded 134 cases of GTN after vesicular mole requiring a change in chemotherapy regimen after chemotherapy with MTX, of which 124 cases met the inclusion criteria, the remaining 10 cases were excluded because the patient discontinued treatment or did not have enough necessary information during data collection (Figure 1). Patient flow diagram
Characteristics of GTN Failed With MTX

*Clinical classification of high-risk molar pregnancy according to WHO 1983.
**The p-value was calculated using the logistic regression model.
Characteristics of the 2nd Chemotherapy
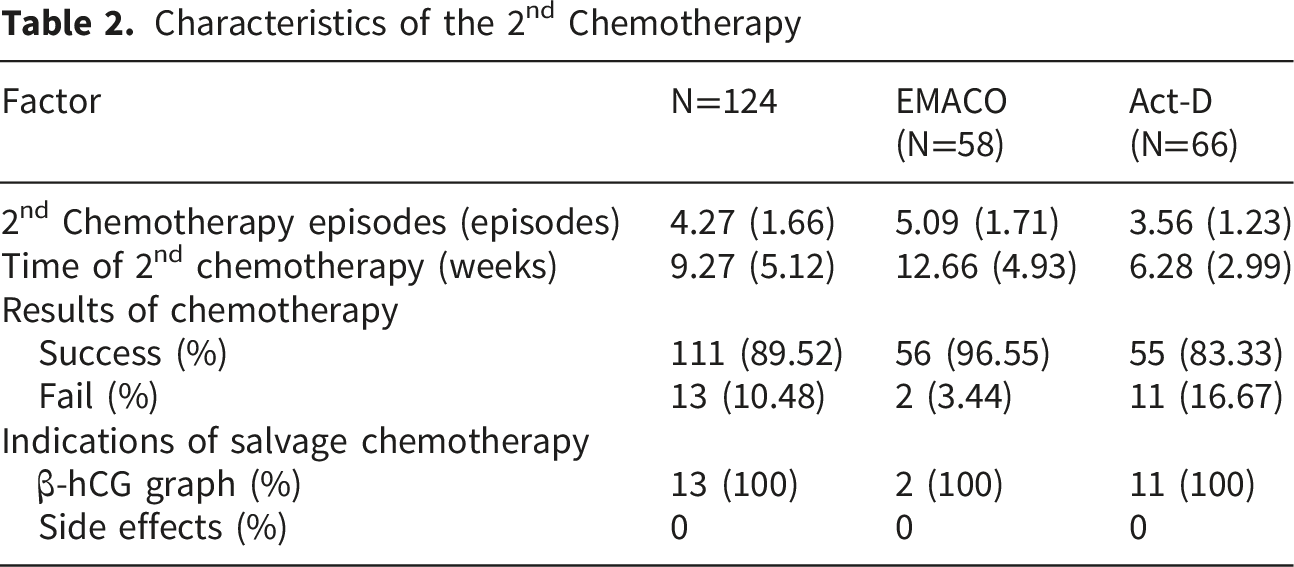
Comparing the frequency of salvage chemotherapy over time between the Act-D and EMACO groups, we found that the frequency of needing salvage chemotherapy was lower in the EMACO group than in the Act-D group. The time of failure of alternative chemotherapy in the EMACO group was later than in the Act-D group. Specifically, 2 cases of failure with EMACO alternative chemotherapy occurred at 6 weeks and 7 weeks; while 11 cases of failure with Act-D occurred at 5 weeks. Among the 11 patients who failed Act-D, 10 achieved remission with EMACO, and 1 required further salvage with EM-EP (Etoposide, Methotrexate, Etoposide, Cisplatin) to achieve remission. The 2 patients who failed second-line EMACO were successfully treated with EM-EP. Ultimately, all patients (100%) achieved complete remission.
Side Effects of the 2nd Chemotherapy
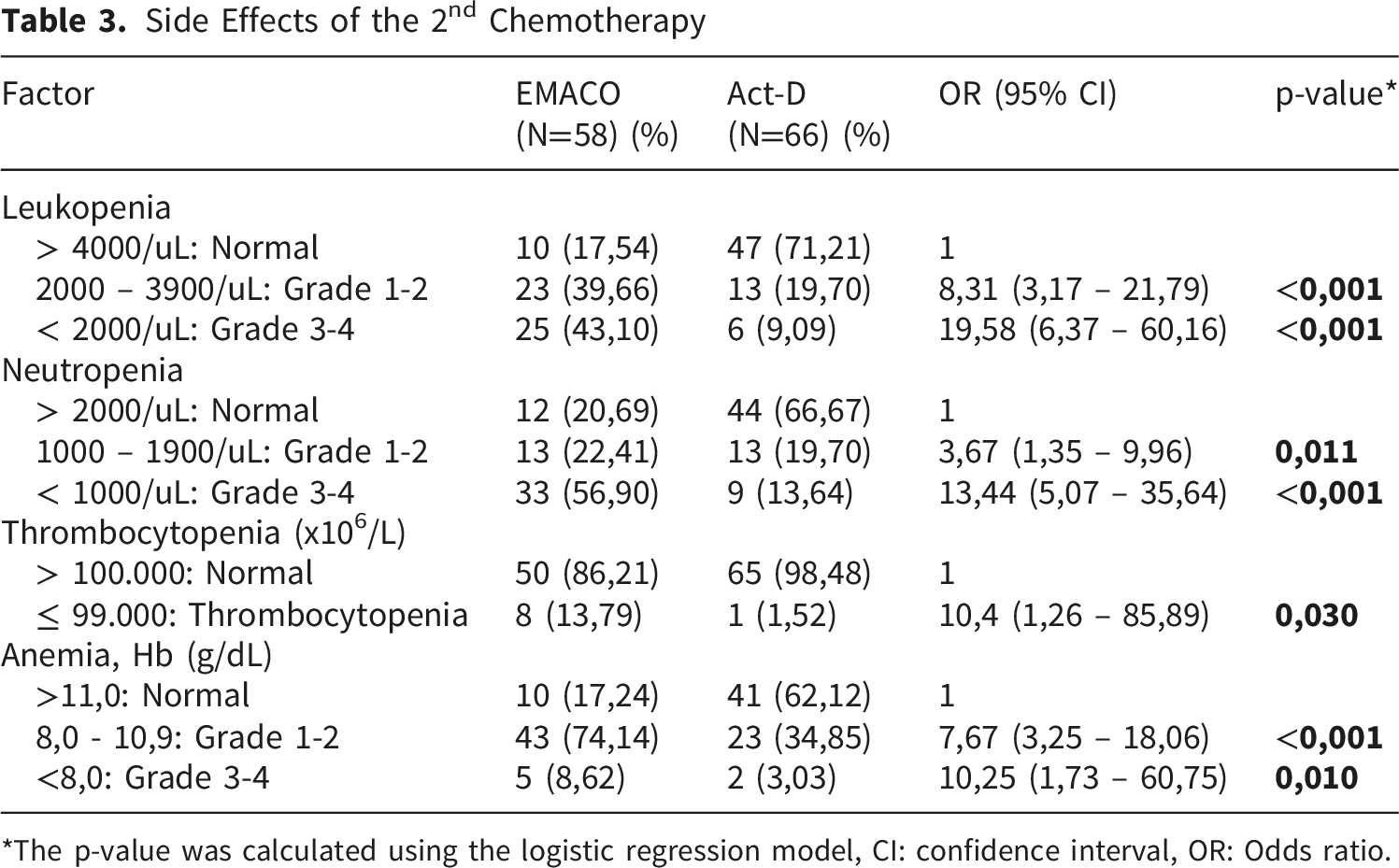
*The p-value was calculated using the logistic regression model, CI: confidence interval, OR: Odds ratio.
Factors Associated With Second-Line Chemotherapy Failure
Cumulative Salvage Chemotherapy
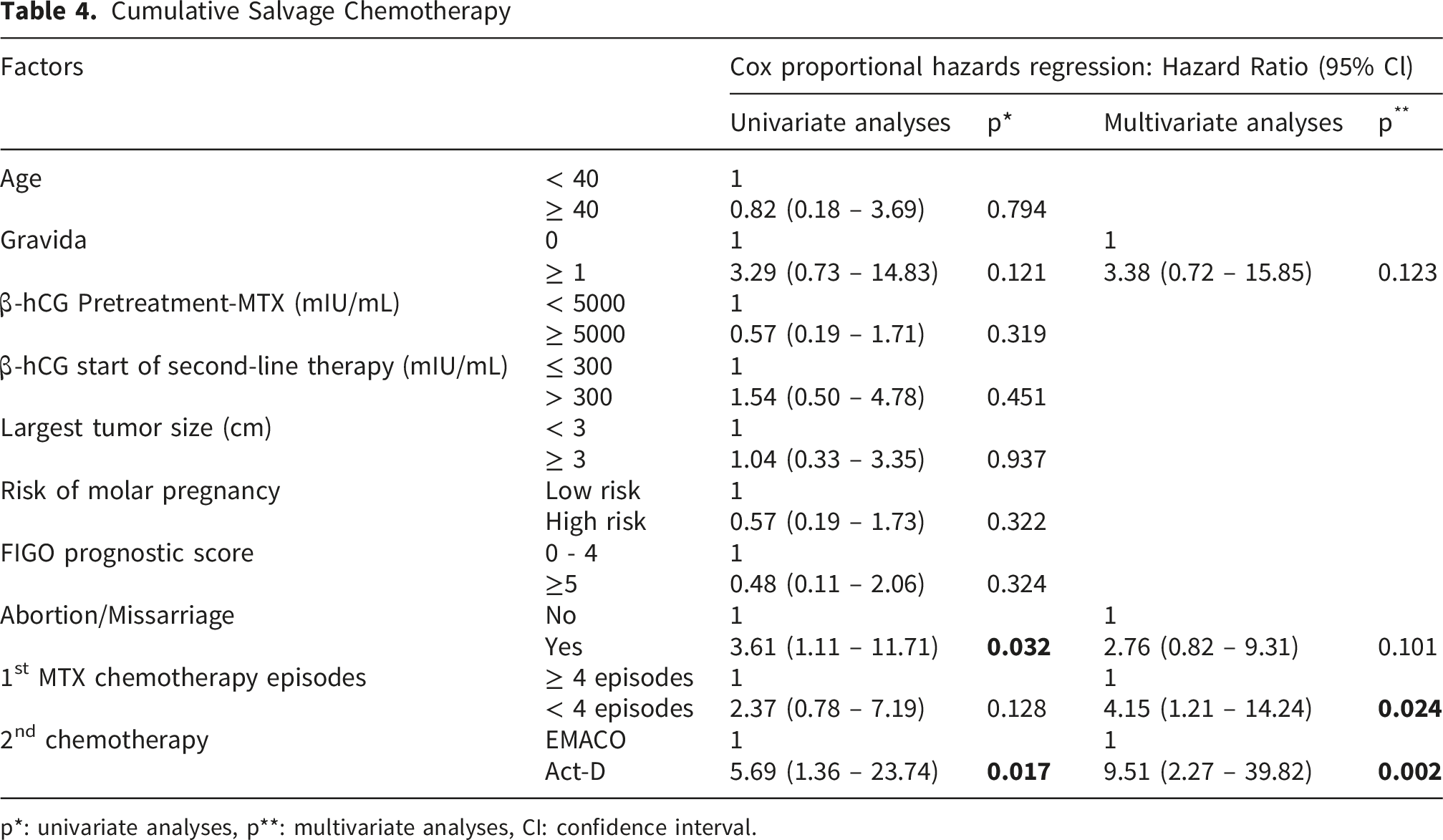
p*: univariate analyses, p**: multivariate analyses, CI: confidence interval.
To control for confounding and co-occurring factors, we included variables with p<0.25 in the univariate analysis in the multivariate regression model. After examining the correlation between variables and excluding highly correlated variables, we included 4 factors in the model including: number of births, number of miscarriages, number of MTX chemotherapy cycles and alternative chemotherapy regimen to analyze prognostic factors for salvage chemotherapy. Based on the multivariate regression analysis model, it was noted that GTN patients with MTX chemotherapy <4 cycles had a 4.15-fold increased risk of failure with alternative chemotherapy compared to the group with MTX chemotherapy ≥ 4 cycles, p<0.05. GTN patients who failed with MTX and received Act-D alternative chemotherapy had a 9.51-fold higher risk of salvage chemotherapy than the EMACO alternative chemotherapy group, p<0.05. No statistically significant differences in the risk of salvage chemotherapy were noted among the remaining factors (Table 4).
Discussion
After switching to alternative chemotherapy, 13 cases still did not respond to treatment. This number ensured the sample power for analysis for the main objective of the study, as mentioned in the sample size calculation section (Figure 1). Our study on 124 GTN patients who failed with first-line chemotherapy MTX recorded a success rate with alternative chemotherapy of 89.52%, of which 83.33% for the Act-D group and 96.55% for the EMACO multi-chemotherapy group. This result is almost equivalent to the study of Cortés-Charry et al. (2021), 3 the overall success rate of alternative chemotherapy including Act-D and EMACO was 93.10%. 3 When referring to the prognostic factors for alternative chemotherapy failure requiring salvage chemotherapy, our study noted: the Act-D alternative chemotherapy regimen increased the risk of salvage chemotherapy 9.51 times higher than the EMACO alternative chemotherapy group and the group with the number of MTX chemotherapy <4 cycles increased the risk of failure with alternative chemotherapy 4.15 times higher than the group with the number of MTX chemotherapy cycles ≥ 4 cycles.
EMACO chemotherapy is a commonly chosen regimen for high-risk GTN patients or after failure with single chemotherapy. Nida Jareemit et al.’s study (2023) included 335 GTN patients receiving EMACO chemotherapy, of which 266 patients received EMACO chemotherapy from the beginning and 69 patients received EMACO chemotherapy after failure with single chemotherapy. The results recorded in the EMACO chemotherapy group as alternative chemotherapy, the success rate was 87% and the average number of EMACO cycles to achieve remission was 3 cycles. 15 In our study, the success rate of the EMACO regimen was up to 96.55% with 56 patients, higher than the report of Nida Jareemit, while the failure rate was 3.44% with 2 patients. This may be explained by the fact that the rate of patients with trophoblastic disease undergoing hysterectomy at TuDu is quite high, and hysterectomy is one of the protective factors that helps reduce the risk of chemotherapy resistance. 16 In addition, our study only included cases of post-molar pregnancy, so the prognosis would be better for cases of previous pregnancies that were not molar pregnancies. In our study, the cause of chemotherapy failure in these 2 patients was abnormal hCG progression. These 2 patients continued to receive salvage chemotherapy with the EA-EP regimen and achieved remission. Thus, our results showed that the chemotherapy group with the alternative EMACO regimen achieved 100% remission. 15
When analyzing the Act-D alternative chemotherapy group, we recorded the success rate of the Act-D alternative chemotherapy group reaching 83.33% with 55 patients and the failure rate was 16.67% with 11 patients. This rate is similar to the previous report of Nguyen HBV. at the National Obstetrics Hospital, Hanoi at 85%, 13 the study of Cortés-Charry at 92.20% 3 and the study of Wu at 82.02%. 9 However, the success rate with Act-D alternative chemotherapy was recorded lower in the study of Woo Dae Kang at 28/37 patients (75.7%), 17 the study of Catherine Prouvot at 75/103 patients (72.82%), 8 and the study of YM Hoeijmakers at 30/45 patients (66.67%). 10 This difference may be because in these studies the authors followed patients for at least 12 months, and failure factors included chemotherapy resistance and relapse. Our study and that of author Nguyen HBV. were retrospective, did not follow up patients, and did not include relapse in the failure factor. In our study, among 11 patients who failed Act-D replacement chemotherapy due to abnormal hCG levels, no cases of Act-D failure due to side effects were recorded. Our results showed that the average number of chemotherapy cycles in the Act-D chemotherapy group was 3.56 ± 1.23 cycles. The average treatment duration for the Act-D group was 6.29 ± 2.99 weeks, with the shortest duration being 1 week and the longest being 13 weeks. The results were similar to Nguyen HBV with the average number of Act-D chemotherapy cycles being 4 ± 1.1, the shortest being 1 cycle and the longest being 7 cycles. 13 When the Act-D alternative chemotherapy fails, the patient will continue with salvage chemotherapy with the EMACO regimen. The success rate of EMACO treatment after failure of Act-D alternative chemotherapy is 90.91% with 10 patients succeeding and 1 (9.09%) patient failing. This patient who failed with EMACO then continued chemotherapy and achieved remission with the EM-EP regimen for 6 cycles in 13 weeks. In the study of Hoeijmakers et al., among 15 patients who failed with Act-D, they continued with the EMACO chemotherapy regimen, of which 1 patient had to change to the 3rd and 4th line regimen and died after stem cell transplantation. The remission rate in this study was 97.78%. 10 Thus, the results of the above studies are quite similar to our study regarding the remission rate in the group of patients receiving chemotherapy with the Act-D alternative regimen. Regarding fertility preservation, while this study did not assess long-term reproductive outcomes, current literature suggests that cytotoxic chemotherapy, particularly methotrexate and actinomycin-D, has a transient effect on ovarian function. A recent study highlights that patients treated with second-line chemotherapy can still achieve successful pregnancies, although counseling regarding the timing of conception is crucial. 18 Future studies at our center will aim to track these long-term reproductive outcomes.
Limitations
This is a retrospective study, and data collected based on medical records may lead to information bias. Second, the study only evaluated patients up to the time they were recorded as being in remission, without follow-up, leading to data loss due to patients not returning for follow-up. The data collection period coincided with the COVID-19 pandemic, affecting follow-up. Finally, from late 2018 to early 2019, the hospital had a shortage of Act-D, which may have affected the decision to choose a treatment regimen.
Conclusions
There are 2 options for alternative chemotherapy regimens in MTX-resistant GTN depending on the clinical situation. The success rate of alternative chemotherapy is 89.52%, in which factors including the number of MTX chemotherapy cycles and the type of alternative chemotherapy regimen Act-D are predictors of failure. Although the risk of failure is higher with Act-D alternative chemotherapy, it can still be treated well in patients with internal medical conditions such as anemia and liver disease because of limited side effects.
Footnotes
Acknowledgement
We are indebted to the participants for making this research possible and to all physicians and staffs of the Oncological Gynecology unit of Tudu hospital, Ho Chi Minh city, Vietnam.
Ethical Considerations
This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. Ethics approval for the study was obtained from the Institutional Review Board of the University of Medicine and Pharmacy at Ho Chi Minh City (No: 3206/UMP-Board, 30th October 2024). The informed consent was waived due to the retrospective design of the study. Data was kept anonymous and confidential during all stages of the study.
Author Contributions
T.V., K.L, and N.N. made substantial contributions to the conception and design of the study. T.V., K.L, and N.N. were involved in data acquisition, analysis, and interpretation. T.V., K.L, N.N and C.L. contributed to drafting the manuscript. All authors critically reviewed the manuscript for important intellectual content and approved the final version. All authors have reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
