Abstract
Background and Objective
Cervical lymph node metastasis (CLNM) is considered a marker of papillar Fethicy thyroid cancer (PTC) progression and has a potential impact on the prognosis of PTC. The purpose of this study was to screen for predictors of CLNM in PTC and to construct a predictive model to guide the surgical approach in patients with PTC.
Methods
This is a retrospective study. Preoperative dual-energy computed tomography images of 114 patients with pathologically confirmed PTC between July 2019 and April 2023 were retrospectively analyzed. The dual-energy computed tomography parameters [iodine concentration (IC), normalized iodine concentration (NIC), the slope of energy spectrum curve (λHU)] of the venous stage cancer foci were measured and calculated. The independent influencing factors for predicting CLNM were determined by univariate and multivariate logistic regression analysis, and the prediction models were constructed. The clinical benefits of the model were evaluated using decision curves, calibration curves, and receiver operating characteristic curves.
Results
The statistical results show that NIC, derived neutrophil-to-lymphocyte ratio (dNLR), prognostic nutritional index (PNI), gender, and tumor diameter were independent predictors of CLNM in PTC. The AUC of the nomogram was .898 (95% CI: .829-.966), and the calibration curve and decision curve showed that the prediction model had good predictive effect and clinical benefit, respectively.
Conclusion
The nomogram constructed based on dual-energy CT parameters and inflammatory prognostic indicators has high clinical value in predicting CLNM in PTC patients.
Keywords
Introduction
Thyroid cancer is the most common endocrine tumor in the human body, with papillary carcinoma accounting for around 84% of occurrences. The prevalence of thyroid cancer is rising worldwide as screening technologies and diagnosis have improved in recent years.1,2 According to cancer statistics, there are around 580,000 thyroid cancer cases globally, with women having a 3-fold greater prevalence than men. 3 Although the overall prognosis of papillary thyroid cancer (PTC) is the best among all pathological types of thyroid cancer, it is prone to lymph node metastasis (LNM), particularly central lymph node metastasis and lateral cervical lymph node metastasis (CLNM). 1 CLNM is closely related to the prognosis of PTC and the method and scope of surgery. Although prophylactic lymph node dissection can reduce the risk of postoperative CLNM in patients with PTC, it may also lead to hypoparathyroidism and recurrent laryngeal nerve injury. 4 Therefore, it is necessary to evaluate the presence of enlarged lymph nodes in the neck through imaging methods before undergoing lymph node dissection. Imaging is an important tool for preoperative examination of CLNM, but these methods have limitations. For example, ultrasonography is the preferred method for screening superficial lymph nodes, but it is difficult to scan deep cervical lymph nodes, and the operator’s expertise and technique affect the accuracy and reliability of the examination results. 5 MRI is not sensitive to calcification and is easily affected by breathing and swallowing movements. Meanwhile, the results of the above imaging methods for identifying benign or malignant enlarged lymph nodes sometimes do not match the actual situation. Dual-energy CT (DECT) is a new imaging device that uses less radiation than traditional CT and can gather image data at various energy levels (40 KeV-140 KeV). Its optimum single-energy images not only optimise image quality but also give qualitative and quantitative information about tissues within the scan area by applying post-processing techniques, 6 which help to display micro lesions and identify benign and malignant lymph nodes.
The inflammatory microenvironment is a critical component of most tumors, and inflammatory responses mediated by inflammatory factors such as tumor necrosis factor, interleukin-1, and chemokines promote tumor angiogenesis and reduce anticancer effects, playing a key role in tumor invasion, progression, and metastasis.7,8 Derived neutrophil-to-lymphocyte ratio (dNLR), prognostic nutritional index (PNI), systemic immune-inflammation index (SII), and other inflammatory biomarkers have been widely used in the study of the relationship between inflammation and cancer interactions, and many studies have demonstrated that some inflammatory indicators are closely associated with the prognosis of cancer patients.
Although some researchers have investigated the relationship between DECT parameters and CLNM, no researchers have yet combined energy spectrum parameters and inflammatory indicators to predict CLNM in PTC patients. This study aimed to develop and validate a nomogram constructed by combining DECT parameters and inflammatory factors and to investigate the feasibility and clinical value of this model in predicting CLNM in PTC patients in order to improve the accuracy of clinicians’ judgement of CLNM, to implement precise and personalized treatment for patients, and to reduce unnecessary intraoperative injuries.
Material and Method
Patients
This is a retrospective study. We retrospectively analyzed the imaging and clinical data of 150 patients with PTC who received preoperative DECT scans of the neck at our institution between July 2019 and April 2023. According to the surgical pathological findings, the patients were divided into a lymph node metastasis group and a non-metastasis group. Inclusion criteria: (1) all patients underwent cervical lymph node dissection, and postoperative pathology confirmed the patient as papillary thyroid cancer; (2) a preoperative cervical DECT scan and a venous phase enhancement scan. Exclusion criteria: (1) a history of other malignant tumors; (2) previous therapy for thyroid cancer; (3) poor quality of CT images; (4) incomplete clinical data of patients. 114 cases were eventually included in the study based on the inclusion and exclusion criteria, and patients were randomly assigned to training and validation groups in a 2:1 ratio. Figure 1 depicts the flow chart of the study population. The study followed relevant Equator guidelines. The reporting of this study conforms to TRIPOD guidelines.
9
Individual consent for this retrospective analysis was waived, and all patient data were de-identified. Flow chart for patient inclusion and data analysis.
Dual-Energy CT Scanning Method
All patients underwent CT on a 64-MDCT scanner (Discovery CT750HD [fifth-generation spectral CT], GE Healthcare) with the gemstone spectral imaging (GSI) mode. The scan ranged from the skull base to the level of the aortic arch. Scanning parameters: 80-140 kVp fast tube voltage switching (.25 ms); 260 mA tube current; 40 mm collimation width; 0.7 s rotation speed; .984:1 pitch. A contrast dose of 1.2 mL/kg (Omnapaque 350 mg I/ml) was injected through the right elbow vein at a flow rate of 3.1 mL/s in the single-phase contrast-enhanced scan, and images of the venous phase were acquired 50 s after the start of contrast injection. After scanning, single-energy images with a reconstituted layer thickness of 1.25 mm in the venous phase were transferred to a workstation (AW4.7, GE Healthcare).
Dual-Energy CT Quantitative Parametric Analysis
The intravenous DECT images were examined by 2 radiologists using a double-blind procedure. GSI software (GSI Viewer 4.7, GE Healthcare) was used to evaluate iodograms and energy spectrum curves (Figure 2). The ROI was plotted on the solid section of the lesion on a 70 keV enhanced image of 1.25 mm while avoiding areas of hemorrhage, necrosis, cystic lesions, and calcification as much as possible. When a primary lesion has multiple cancer foci, the largest cancer foci are selected for measurement, and at least 2 levels are chosen for each lesion, with each level being measured once, the iodine concentration (IC) of the cancer foci and the common carotid artery was obtained. Normalized iodine concentration (NIC) = IC lesion/IC common carotid artery, the slope of energy spectrum curve (λHU) = (CT 40 keV − CT 100 keV)/(100-40). Measurement of dual-energy CT parameters in venous phase. Draw the region of interest for iodine concentration measurement (A). L1: Measurement of iodine concentration in thyroid papillary cancer; L2: Measurement of iodine concentration in the common carotid artery (B). Diagram of spectrum curve measurement (C).
Clinical Data Analysis
All of the patients in this study had complete clinical data. The clinical data collected included gender (male or female), age (≥45 years or <45 years), tumor diameter (≥10 mm or <10 mm), number of lesions (single or multiple), calcification (present or absent), TSH (thyroid stimulating hormone), TGAb (thyroglobulin antibody), body mass index (BMI), neutrophil count, lymphocyte count, platelet count, and serum albumin count. Note: A single lesion is defined as the number of thyroid cancer foci <2, whereas a multiple lesion is defined as the number of cancer foci in 1 or different lobes of the thyroid ≥2; in the case of multiple lesions, the diameter was measured using the largest lesion. Inflammatory index calculation: dNLR = neutrophil count/(white blood cell count - neutrophil count); PNI = albumin (g/L) + 5× lymphocyte count (×109/L); platelet-to-lymphocyte ratio (PLR) = platelet count/lymphocyte count; SII = neutrophil × platelet/lymphocyte count. PIS: Patients with a low neutrophil-lymphocyte ratio (<3) and high serum albumin (≥40 g/L) were scored as 2; patients with a high neutrophil-lymphocyte ratio (≥3) or low serum albumin levels (<40 g/L) were scored as 1.
Statistical Analyses
Statistical analysis was performed using SPSS 26.0 and R 4.2.2: (1) Continuous variables were transformed into binary variables by ROC curve analysis and calculating the best cut-off value; (2) Independent influences factors on predicting LNM in PTC were screened using univariate and multivariate logistic regression analysis; (3) The results of multivariate logistic regression (variables with P < .05) were used to construct nomogram; (4) The nomogram was evaluated by calculating the consistency index (C-index) and plotting the receiver operating characteristic curve (ROC), calibration curve, and decision curve; (5) Finally, Bootstrap validation was performed to internally validate the model and brought into the validation group for external validation.
Results
Baseline Characteristics of the Included Patients
Baseline Information of 114 Patients With Papillary Thyroid Cancer.

IC: iodine concentration; NIC: normalized iodine concentration; λHU: the slope of energy spectrum curve; BMI: body mass index; dNLR: derived neutrophil-to-lymphocyte ratio; PNI: prognostic nutritional index; SII: systemic immune-inflammation index; PLR: platelet-to-lymphocyte ratio.
Mean Value of Dual-Energy CT Parameters.

IC: iodine concentration; NIC: normalized iodine concentration; λHU: the slope of energy spectrum curve; CT: computed tomography.
Result of Logistic Regression Analysis
Univariate and Multivariate Logistic Regression Analysis of CLNM in Patients With PTC in Training Group.
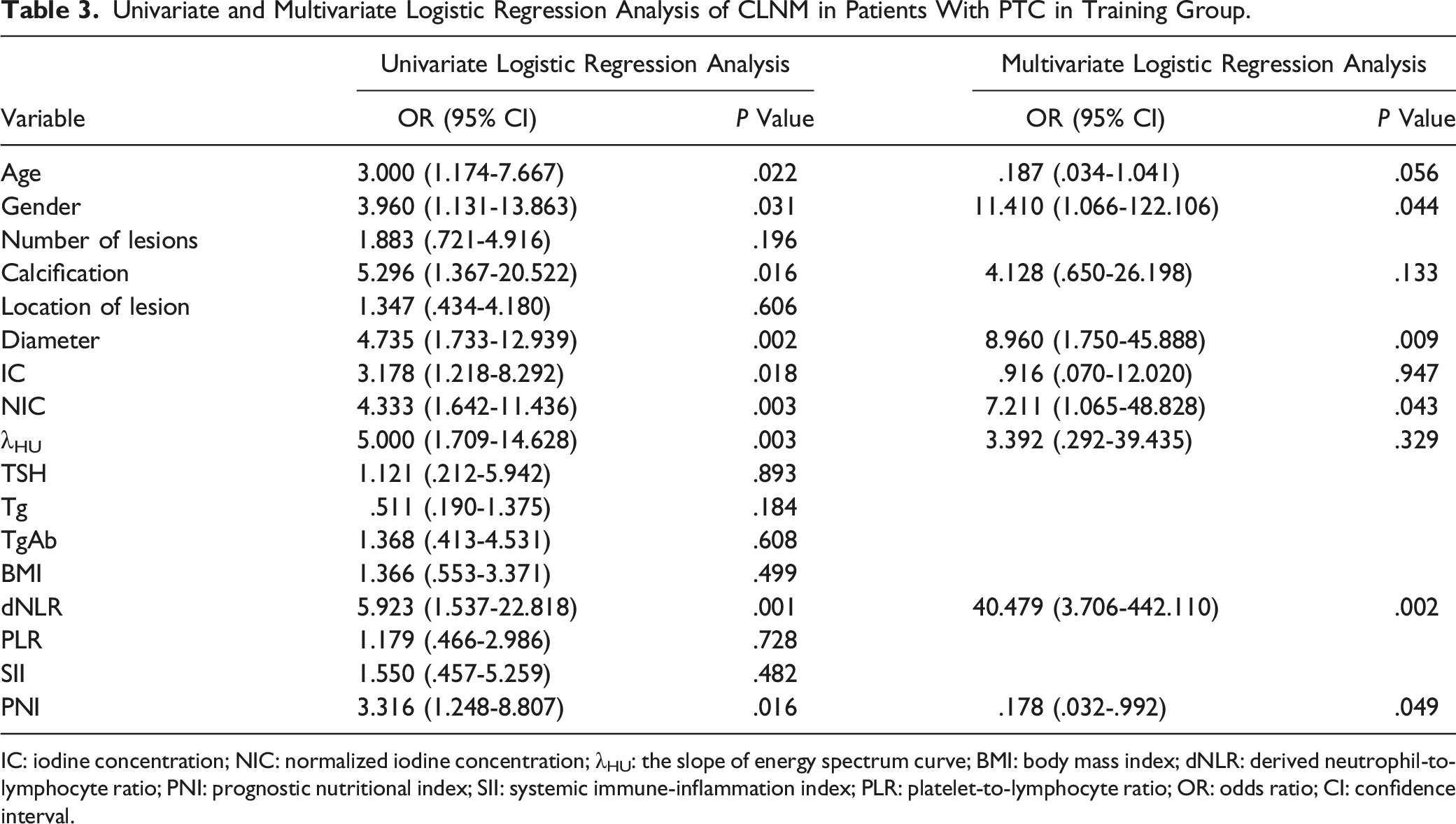
IC: iodine concentration; NIC: normalized iodine concentration; λHU: the slope of energy spectrum curve; BMI: body mass index; dNLR: derived neutrophil-to-lymphocyte ratio; PNI: prognostic nutritional index; SII: systemic immune-inflammation index; PLR: platelet-to-lymphocyte ratio; OR: odds ratio; CI: confidence interval.
Construction and Evaluation of Nomogram
According to the ROC curves (Figure 3), the combined prediction model outperforms the dual-energy CT parameters and inflammatory indicators alone. Drawing nomogram (Figure 4(A)) and its forest plot (Figure 4(B)) based on the results of multivariate logistic regression analysis. The C-index of the nomogram predicting CLNM was .898 (95% CI: .830-.965), and the area under the curve (AUC) for Bootstrap validation was .896 (95% CI: .891-.900). Further introducing nomogram into the validation group, the C-index was .853 (95% CI: .727-.978), and the AUC was .854 (95% CI: .845-.863). Calibration plots (Figure 4(C)) and DCA curve (Figure 4(D)) showed good predictive effect and clinical benefit of the nomogram, respectively. ROC curve for predicting cervical lymph node metastasis in thyroid papillary cancer. The AUC of the combined prediction model was .898 (95% CI: .829-.966), which was the largest. AUC, the area under the curve; ROC, receiver operating characteristic curve; NIC, normalized iodine concentration; dNLR, derived neutrophil-to-lymphocyte ratio; PNI, prognostic nutritional index. Nomogram (A) and its forest plot (B), decision curve (C), calibration curve (D) for predicting cervical lymph node metastasis in papillary thyroid cancer.

Discussion
The thyroid gland is an iodine-rich organ, and iodine in the thyroid gland is mainly stored in follicular epithelial cells as thyroglobulin. When the gland becomes malignant, the follicular epithelial cells are replaced by cancer cells, and iodine uptake and storage are reduced, resulting in malignant thyroid nodules with lower overall iodine concentration than normal tissue. In this study, iodograms were obtained by GSI workstation, and the IC of parenchymal components of the lesion was measured quantitatively. Filling tissue microvasculature with iodine contrast is the foundation of iodine basemap imaging. The IC measured by DECT is highly consistent with the actual amount of iodine, so the measured iodine concentration can represent the blood supply to the lesion indirectly,10,11 ie, the higher the iodine concentration, the richer the blood supply to the lesion. Currently, many studies have shown that dual-energy CT parameters can be used as biomarkers for predicting LNM in cancer. Li et al, 12 in their findings for predicting LNM in gastric cancer noted that the IC of cancer foci was closely associated with LNM. Su et al 13 indicated significant differences in NIC in esophageal cancer foci in patients with metastatic and non-metastatic lymph. This study included 3 DECT parameters of IC, NIC, and λHU of intravenous phase PTC, and all 3 parameters were correlated with CLNM of PTC by univariate statistical analysis (P < .05), and the results of the multivariate analysis showed that only intravenous phase NIC was an independent predictor of CLNM, and when NIC ≥.68, patients with PTC had an increased risk of developing CLNM. The findings of this study are similar to those of Liu et al 14 However, these scholars conducted their study on arterial phase NIC. Meanwhile, the results of this study showed that the IC, NIC, and λHU of the primary cancer lesions in the patients with metastatic cervical lymph nodes were 33.18 ± 6.89, .71 ± .14, and 3.82 ± .84, respectively, while the IC, NIC, and λHU of the non-metastatic group were 31.20 ± 7.43, .66 ± .13, and 3.60 ± .87, respectively, that is, the DECT parameters in the metastatic group were higher than those in the non-metastatic group. The reason for this result may be due to the increased metabolic rate of cancer cells during the process of cervical lymph node metastasis, as well as the increase of tumor blood vessels in the cancer foci to provide rich nutrients to maintain the growth and proliferation of cancer cells, so the IC and NIC of the cervical lymph node metastasis group were higher than those of the non-metastasis group. This is also consistent with Zou’s findings that differences in iodine intake may lead to differences in the capacity of LNM. 15 The λHU reflects the decay of HU values of tissues in the 40-140 keV range and can be used to quantify the different tissue iodine content in different tissues.16,17 So, it is reasonable to explain that the λHU is higher in the metastatic group than in the non-metastatic group based on the above.
Research on the relationship between inflammation and various cancers has continued to heat up in recent years. Numerous previous studies have shown that cancer-associated inflammatory responses play a crucial role in the tumor microenvironment and are key mediators of cancer development, progression and metastasis. 7 Cancer cells induce an inflammatory response, altering peripheral blood leukocyte and platelet levels and releasing pro-inflammatory molecules, which in turn drive cancer development.18-20 The major inflammatory factors include IL-1, TNF-a, etc. Cancer cells can secrete vascular endothelial growth factor (VEGF) to promote tumor microvasculature and lymphatic vessel production, and the probability of CLNM is increased when VEGF is overexpressed. 21 Despite many studies describing the interaction between cancer and inflammation-mediated tumor microenvironment, determining the status of cancer-associated inflammation in clinical practice remains problematic. According to the literature, NLR can be a key predictor of thyroid cancer survival and LNM, with higher NLR indicating a more aggressive tumor and a worse prognosis.22-24 In this study, we investigated the connection between inflammatory markers and CLNM in PTC. dNLR and PNI were shown to be independent predictors of CLNM in patients with PTC. PNI not only reflects the nutritional and immune status of cancer patients but also predicts prognosis.25,26 PTC patients with low preoperative PNI were more likely to develop CLNM in the current study. It could be due to the high metabolic rate of cancer cells and the tumor’s aggressiveness during lymph node metastasis, which causes malnutrition and reduced immunological function, resulting in a lower PNI.
Furthermore, in this study, gender and tumor diameter were also independent influencing factors for CLNM in PTC patients. The significance of gender in CLNM may be due to the higher hormone level in women than men in general, which has a potential influence on the metastasis of cancer cells. The larger the tumor diameter, the more likely it is to break through the envelope and invade the surrounding lymphatic vessels and lymph nodes, and when the diameter exceeds 10 mm, CLNM is more likely.
Currently, the nomogram has shown excellent results in predicting the prognosis of various tumors.27,28 Based on the findings of the preceding study, we attempted to construct a nomogram for predicting CLNM in PTC by combining 5 independent influencing factors (NIC, dNLR, PNI, gender, and tumor diameter). The C-index of this nomogram was .898. The calibration curve showed that the probability of the nomogram predicting CLNM and the actual occurrence of metastasis were in good agreement. The decision curves showed that the model had the greatest net clinical benefit over the most reasonable threshold probability ranges. In the nomogram, each factor is assigned a weighted number of points, and the corresponding probability value for the occurrence of CLNM is obtained by calculating the total number of points for each patient.
In conclusion, NIC, dNLR, PNI, gender, and diameter can be used as independent predictors of CLNM in PTC patients. The nomogram constructed by combining the above 5 factors has higher accuracy and clinical benefit in predicting CLNM in PTC patients, and by scoring the system, it can provide clinicians with a visualized prediction result, which may be beneficial for clinicians to develop personalized treatment plans for patients and avoid unnecessary injury to patients due to prophylactic dissection of the lymph nodes.
This research has the following deficiencies: (1) when patients had multiple cancer foci, only the largest cancer foci were selected for measurement, and the remaining foci were not analyzed. (2) the DECT parameters of the venous phase were only measured and analyzed in this study, and DECT parameters in the arterial phase will be added later for further study. (3) the number of samples used to develop and validate the model in this study was small, and it’s currently not possible to increase the sample size. (4) We did not perform power calculations when estimating the sample size selected for the study. (5) Loss of thyroglobulin information in individual patients.
Footnotes
Acknowledgments
All authors sincerely thank all patients who participated in this study.
Author Contributions
DJN and WYY led the study. WLY conducted the data analysis; BJ, SMJ and FBY implemented the methodology; WLY and XBZ collected the data and discussed the results, WLY and ZY prepared the original draft, WLY and DJN reviewed and edited the final manuscript. The author(s) read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article is supported by Key Specialties in Clinical Medicine Construction Funding Project of Anhui Province (No. 2019sjlczdzk), Joint Fund for Medical Artificial Intelligence (No. MAI2023Q032) and The National Cancer Center Climbing Foundation for Clinical Research (No. NCC201912B03).
