Abstract
Introduction
While gender is a known prognostic factor for many cancers, its specific role in pancreatic neuroendocrine tumors (PanNETs) survival remains poorly characterized. This study aimed to investigate gender-based differences in PanNETs prognosis using the Surveillance, Epidemiology, and End Results (SEER) database.
Methods
This retrospective cohort study included patients diagnosed with PanNETs between the years 2000 and 2020, extracted from the SEER database. Propensity score matching (PSM) was applied to mitigate potential selection bias. Overall survival (OS) was evaluated using Kaplan-Meier analysis and multivariable Cox regression.
Results
Among the 5155 patients included (2814 males, 2341 females), males showed significantly worse OS than females both before (hazard ratio [HR] 1.29, 95% CI 1.14-1.47, P < 0.001) and after (HR 1.19, 95% CI 1.02-1.38, P = 0.026) PSM. Subgroup analyses confirmed a consistent OS advantage for females across most categories. Multivariable analysis identified marital status, age, tumor grade, gender, year of diagnosis, N stage, M stage, and surgical intervention as independent predictors of OS. Similar predictors were found in males, whereas in females, marital status, age, grade, N stage, M stage, and surgery were specifically significant.
Conclusions
Female patients with PanNETs exhibit superior OS rates. Further research is needed to clarify the biological and clinical mechanisms underlying these gender-related disparities.
Introduction
Pancreatic neuroendocrine tumors (PanNETs), which originate from the neuroendocrine cells of the pancreas, constitute a unique category of tumors, accounting for approximately 3%-5% of all pancreatic neoplasms.1-3 Recent progress in diagnostic techniques and increased recognition of endocrine tumors have led to a rise in the number of identified PanNET cases. 4 According to the National Cancer Institute’s registry, the United States reports an estimated 1000 new cases of PanNETs each year. 5 Based on their clinical behavior, PanNETs are primarily classified into functional and non-functional types depending on hormone secretion. Additionally, histological grading—based on differentiation and proliferative indices—plays a crucial role in prognostic stratification.6-9 Previous research has identified numerous factors that can affect the prognosis of patients with PanNETs. These factors include clinical factors related to manifestations, laboratory tests, imaging, and treatment; pathological factors such as histology, classification, and grading; and molecular factors. 10 Specifically, the research by Miao et al 11 highlights sex, age, marital status, primary site, TNM stage, tumor grade, and therapy as risk factors impacting the overall survival (OS) of patients with PanNETs. To further elucidate the impact of different factors, Zhou et al12,13 conducted a detailed analysis focusing on the effects of racial disparities and marital status on the prognosis of patients with PanNETs.
In recent years, an increasing number of studies have emphasized gender as an important risk factor influencing disease prognosis, with the prognosis of various diseases being related to gender.14-16 Abdel et al 17 examined the association between gender and outcomes in gastroenteropancreatic-neuroendocrine neoplasms (GEP-NEN). However, it is important to note that gastrointestinal neuroendocrine tumors and PanNETs differ in their site of origin, clinical manifestations, diagnosis and treatment methods, and potentially in their prognosis. Few studies to date have specifically addressed how gender influences survival outcomes in patients with PanNETs. To address this gap, the present study analyzed population-level data from the Surveillance, Epidemiology, and End Results (SEER) database to evaluate the effect of gender on OS in patients with PanNETs. This work seeks to generate novel insights into gender-related prognostic factors, with the long-term goal of informing more tailored therapeutic approaches for male and female patients.
Methods
Study Population
Patient data were extracted from the SEER database, which provides publicly available, de-identified information representing approximately 28% of the U.S. population. This retrospective cohort study included individuals diagnosed with PanNETs between 2000 and 2020. Cases were identified based on the International Classification of Diseases for Oncology, 3rd Edition (ICD-O-3) histology code 8240/3 (carcinoid tumor, not otherwise specified). Patients without a confirmed histological diagnosis or complete survival follow-up were excluded. A flow diagram of the selection process is presented in Figure 1. Demographic and clinical variables collected included marital status, age at diagnosis, self-reported race, tumor grade, diagnostic year, sex, TNM stage, tumor size, and treatment details (surgery, chemotherapy, and radiotherapy). Age was dichotomized into <65 years (younger group) and ≥65 years (older group). Race was categorized as White, Black, or Other (including American Indian, Alaska Native, and Asian/Pacific Islander). Tumor staging followed the 7th edition of the American Joint Committee on Cancer (AJCC) Cancer Staging Manual. The variable “Radiation” in the SEER database refers to external beam radiation therapy or brachytherapy administered as part of primary treatment. It does not include peptide receptor radionuclide therapy (PRRT), which is captured under systemic therapy variables. OS was defined as the time from diagnosis to death from any cause or last known follow-up. This study used de-identified, publicly available data from the SEER database and did not directly involve human subjects, thus obviating the need for Institutional Review Board (IRB) approval and informed consent. The study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. Patients were identified from the SEER database based on the defined histological and temporal inclusion criteria, representing a population-based sample rather than a consecutive, random, or selectively recruited cohort. The reporting of this study conforms to the STROBE guidelines.
18
Study inclusion flowchart
Multiple Imputation
Several variables contained missing values, with proportions as follows: marital status (5.5%), race (0.9%), grade (12.2%), T stage (5.0%), N stage (5.0%), M stage (5.0%), surgery status (0.4%), and tumor size (7.6%). To address this, we performed multiple imputation (MI) using a random forest-based approach implemented in R software (version 4.1.0), thereby improving the robustness of subsequent statistical analyses.
Statistical Analysis
Categorical data were presented as counts and percentages, and chi-square tests were utilized for group comparisons. Continuous variables not following a normal distribution are presented as median (interquartile range, IQR) and compared using the Mann-Whitney U test. To account for baseline differences between sexes, 1:1 propensity score matching (PSM) was performed with a caliper of 0.01. The propensity model incorporated marital status, age, race, tumor grade, year of diagnosis, sex, TNM stage, tumor size, and treatment information (surgery, chemotherapy, radiation). OS was estimated via the Kaplan-Meier method, with between-group differences assessed by the log-rank test. Univariate and multivariable Cox proportional hazards models were used to calculate hazard ratios (HRs) and 95% confidence intervals (CIs). Variables showing statistical significance (P < .05) in univariate analysis were entered into the multivariable model. All analyses were conducted in R (version 4.1.0), with P < .05 considered statistically significant.
Results
Patient Characteristics
Demographic and Clinical Characteristics of Patients Before and After PSM

PSM: propensity score matching; Others: American Indian, Alaska Native, Asian/Pacifc Islander; IQR: interquartile range; bold values indicate P < .05.
Survival Analysis of Patients With PanNETs
Before PSM, males showed significantly worse OS than females (median OS: males 33.0 months [IQR 13.0-59.0], females 35.0 months [IQR 14.0-63.0]; HR 1.29, 95% CI 1.14-1.47, P < .001) (Figure 2A). After PSM, median OS was 35.0 months (IQR 14.0-61.0) for males and 38.5 months (IQR 16.0-65.0) for females, with males still exhibiting worse OS (HR 1.19, 95% CI 1.02-1.38, P = .026) (Figure 2B). Comparison of overall survival between male and female patients (A) before and (B) after propensity score matching (PSM)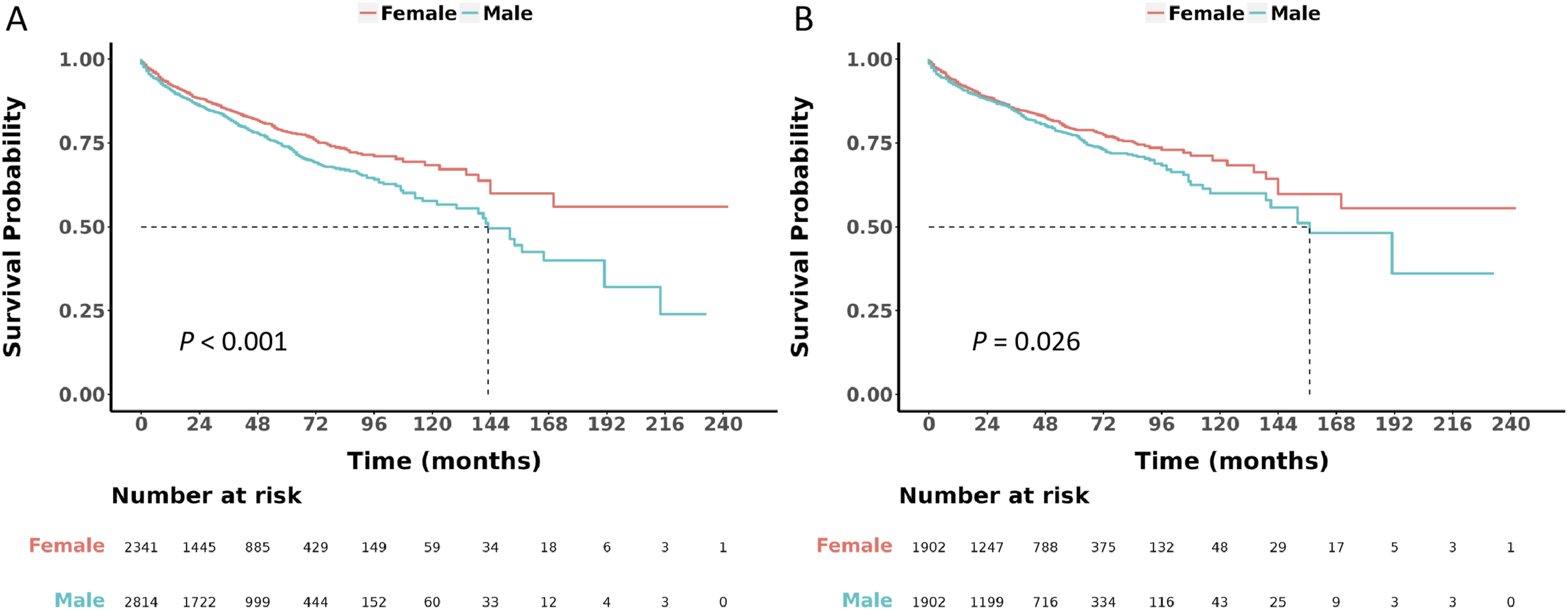
Univariate and Multivariate Cox Regression
Univariate and Multivariate Cox Regression for Analyzing the Overall Survival for Patients With Pancreatic Neuroendocrine Tumors

HR: hazard ratios; CI: confidence interval; Others: American Indian, Alaska Native, Asian/Pacifc Islander; bold values indicate P < .05.
Univariate and Multivariate Cox Regression for Analyzing the Overall Survival for Male Patients With Pancreatic Neuroendocrine Tumors

HR: hazard ratios; CI: confidence interval; Others: American Indian, Alaska Native, Asian/Pacifc Islander; bold values indicate P < .05.
Univariate and Multivariate Cox Regression for Analyzing the Overall Survival for Female Patients With Pancreatic Neuroendocrine Tumors

HR: hazard ratios; CI: confidence interval; Others: American Indian, Alaska Native, Asian/Pacifc Islander; bold values indicate P < .05.
Subgroup Analyses of Gender on OS of Patients With PanNETs
Subgroup analysis showed that, before PSM, the prognosis of the female group was generally better than that of the male group across most subgroups, with this advantage being particularly pronounced in the married group, the group aged under 65 years, the White race group, the well-grade group, the T1 stage group, the group without lymph node metastasis and distant metastasis, and the surgery group (Figure 3). Following PSM, while the prognoses of male and female groups were comparable in some subgroups, the prognosis for females was notably superior to that of males in the age less than 65 years group, the White race group, the well-grade group, the T1 stage group, the non-lymph node metastasis and non-distant metastasis group, the surgery group, the non-radiotherapy and non-chemotherapy group, and the tumor size less than 2.0 cm group (Figure 4). Subgroup analysis of overall survival by sex prior to propensity score matching Subgroup analysis of overall survival by sex following propensity score matching

Discussion
The pancreas plays a vital role in both the digestive and endocrine systems. 19 In the latter system, islet cells secrete hormones and peptides such as insulin, glucagon, somatostatin, and pancreatic polypeptide. Gastrin may be secreted in the setting of functional gastrinomas, a subtype of PanNETs, but is not typically produced by normal islet cells. These hormones and peptides are crucial for regulating blood sugar levels, gastrointestinal function, and ensuring pancreatic health.20,21 PanNETs arise from neuroendocrine cells of the pancreas, which may include hormone-producing islet cells as well as other neuroendocrine cell types. Functional PanNETs secrete hormones, whereas non-functional tumors do not exhibit hormone hypersecretion.1,7 Previous research has indicated that multiple factors can impact the outcome of PanNETs. 10 The impact of gender on survival outcomes in PanNETs remains underexplored. Using data from the SEER registry, this study assessed prognostic differences by sex. Among 5155 patients (2814 men, 2341 women), men consistently showed poorer overall survival than women, both before and after PSM. Multivariable Cox analysis identified marital status, age, grade, sex, year of diagnosis, N stage, M stage, and surgery as independent survival predictors in the overall cohort. In sex-stratified models, similar factors were significant for men, while for women the independent predictors were marital status, age, grade, N stage, M stage, and surgical intervention. These findings collectively indicate that female sex is associated with more favorable survival in PanNETs.
Male patients presented with more advanced tumors and underwent surgery less frequently, which contributed to their poorer survival. However, male sex remained an independent adverse prognostic factor even after adjusting for tumor stage and surgical intervention. This indicates that beyond differences in clinical presentation, inherent sex-related factors—such as hormonal influences, tumor biology, or health behaviors—likely impact outcomes. While more advanced disease in males explains part of the survival gap, these residual disparities highlight the need for further investigation into the biological and clinical drivers of sex-specific outcomes in PanNETs.
Consistent with our findings, numerous previous studies have utilized the SEER database to assess the prognostic risk factors in patients with PanNETs, revealing that female patients tend to have a better prognosis than male patients.11,12,22-24 Furthermore, Li et al 25 performed a competing risks analysis of prognosis in patients with PanNETs, which also suggested that male patients experienced a poorer prognosis compared to female patients. Another investigation also highlighted the prognostic significance of gender among older patients with PanNETs, revealing that elderly women tend to experience more favorable outcomes compared to their male counterparts. 26 Although these studies examined various factors when analyzing prognostic risk factors in patients with PanNETs, their emphasis on the prognostic impact of a specific factor (such as gender) may not be adequately thorough or detailed. Due to the simultaneous consideration of multiple factors in these analyses, the independent effect of a single factor could be influenced by others, resulting in less conclusive outcomes. By directly comparing survival outcomes between male and female patients with PanNETs, our study yields clearer and more clinically actionable conclusions. These findings can help clinicians better appreciate the influence of sex on prognosis and support the design of sex-tailored management strategies. To our knowledge, this analysis also constitutes the largest cohort to date examining gender-based prognostic differences in PanNETs.
The impact of gender on disease outcomes is likely to involve intricate interactions among genetic factors, hormonal disparities, body composition, as well as behavioral and social elements. Although our study design precludes the investigation of etiological factors, our findings offer valuable insights for exploring how gender differences influence the treatment and outcomes of PanNETs. Several potential mechanisms may explain why female patients with PanNETs tend to have better survival outcomes than males. Firstly, hormonal disparities between men and women could potentially affect patient prognosis. Arnason et al 27 discovered that gastrointestinal neuroendocrine tumors with elevated progesterone receptor expression exhibit a more favorable clinical prognosis and lower disease stage. Another study revealed that PanNETs characterized by good prognostic features have increased expression of estrogen receptor β. 28 It is plausible that the improved survival seen in female patients with PanNETs relates to elevated expression of estrogen and progesterone receptors, though this possibility warrants additional study. Secondly, higher rates of smoking and alcohol use among men may contribute to poorer outcomes, as these behaviors are established risk factors for worse prognosis in multiple cancers.29,30 Both smoking and alcohol can impair immune response and metabolic homeostasis, creating conditions that may accelerate tumor progression and spread. However, Leoncini et al 31 conducted a systematic review and meta-analysis and found that the impact of smoking and alcohol consumption on neuroendocrine tumors is comparatively minor. Nonetheless, prospective studies are still warranted to further elucidate the influence of smoking and alcohol consumption on the prognosis of patients with PanNETs. Thirdly, female patients may exhibit a greater inclination to seek medical assistance and demonstrate better adherence to treatment plans. This proactive approach to treatment and behavior may contribute to an improved prognosis. Fourthly, sex-related differences in drug metabolism could influence treatment response and clinical outcomes. For example, certain targeted agents may undergo faster clearance in males, potentially reducing drug exposure and compromising therapeutic efficacy.
Our findings that female PanNET patients exhibit better OS than males, even after adjusting for tumor stage and other prognostic factors, raise the question of whether this survival advantage is specific to PanNETs or part of a broader gender-based survival pattern observed across many diseases. Indeed, epidemiological studies have consistently shown that women tend to have longer life expectancy and better outcomes in several cancer types and chronic conditions, potentially due to hormonal, genetic, immunological, and behavioral differences. In our cohort, the higher proportion of females with early-stage (T1, N0, M0) disease may partially contribute to their superior survival. However, the persistence of a significant gender effect after multivariable adjustment and propensity score matching suggests that factors beyond stage distribution are at play. This indicates that while some of the survival advantage may align with general gender trends, PanNETs may also possess unique biological or clinical characteristics that modulate gender-specific outcomes. Future research should aim to disentangle disease-specific mechanisms from general survival patterns, potentially through comparative studies across different tumor types or integrated omics analyses.
Our findings are consistent with a growing body of literature highlighting sex-based outcome disparities in pancreatic surgery and cancer. Recent multicenter and national cohort studies have shown that female patients experience fewer postoperative complications, lower mortality, and higher rates of achieving an “ideal outcome” after pancreatectomy compared to males, even after adjusting for comorbidities and surgical extent.32,33 These differences extend beyond PanNETs to pancreatic adenocarcinoma, where female sex, particularly in combination with obesity, has been associated with reduced liver recurrence and improved overall survival after curative surgery. 34 Additionally, emerging preclinical and clinical evidence suggests that sex hormones and their receptors—such as estrogen, progesterone, and androgen receptors—may modulate tumor biology and treatment response in neuroendocrine neoplasms, including PanNETs. 35 Although our study did not directly examine hormonal or molecular mechanisms, these parallel findings reinforce the notion that sex-related factors—whether biological, hormonal, or behavioral—exert a significant influence on pancreatic disease outcomes across histologic subtypes. Future studies integrating clinical, molecular, and hormonal data are warranted to elucidate the underlying drivers of these disparities and to inform sex-tailored management strategies.
Our study has several limitations that should be acknowledged. Firstly, akin to other evaluations of population-based cancer database information, a multitude of patient-specific characteristics are absent. These encompass treatment specifics (such as detailed protocols for surgical interventions, chemotherapy, and radiotherapy), postoperative complications, and the Ki-67/mitotic index, which holds significance in determining the World Health Organization’s grading system. Secondly, the retrospective nature of this SEER-based analysis carries inherent risks of data omissions and biases. MI and PSM were applied to mitigate the impact of missing information and selection bias. Thirdly, as the SEER registry primarily covers the U.S. population, the generalizability of our findings to other regions may be limited. Finally, it should be emphasized that this study is exploratory and hypothesis-generating in nature. While our findings suggest significant sex-based survival differences in PanNETs, they do not test a predefined biological or clinical hypothesis, and further prospective or mechanistic studies are needed to confirm and elucidate these observations. Despite these constraints, the present study provides a valuable contribution by leveraging the large, diverse, and longitudinally-followed SEER cohort—representing the largest sample to date—to comprehensively assess gender-associated prognostic differences in PanNETs.
Conclusion
In summary, this retrospective SEER-based analysis identifies significant sex-based prognostic differences in PanNETs, with female patients exhibiting better overall survival than males. Multivariable models confirm that marital status, age, tumor grade, year of diagnosis, N stage, M stage, and surgical intervention are independent predictors of survival. These results emphasize the value of sex-specific risk assessment in clinical management and point to the need for further investigation into the biological and behavioral drivers of these survival disparities.
Supplemental Material
Supplemental Material - Gender Differences in Prognosis of Pancreatic Neuroendocrine Tumors: A Retrospective Cohort Study Based on the SEER Database
Supplemental Material for Gender Differences in Prognosis of Pancreatic Neuroendocrine Tumors: A Retrospective Cohort Study Based on the SEER Database by Yining Li, Shuaiyang Zhou, Qiao Qiao, Ke Ji, Qin Li, Zhenguo Qiao, Ping Zhang in Cancer Control
Footnotes
Ethical Considerations
This study used de-identified, publicly available data from the SEER database and did not directly involve human subjects, thus obviating the need for Institutional Review Board (IRB) approval and informed consent. The study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024.
Consent to Participate
This study used de-identified, publicly available data from the SEER database and did not directly involve human subjects, thus obviating the need for Institutional Review Board approval and informed consent.
Author Contributions
Conception and design: Yining Li, Ping Zhang. Administrative support: Qiao Qiao. Provision of study materials or patients: Zhenguo Qiao, Ke Ji. Collection and assembly of data: Shuaiyang Zhou. Data analysis and interpretation: Qin Li. Manuscript writing: All authors. Final approval of manuscript: All authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
