Abstract
Introduction
Therapeutic intervention with chemopreventive agents (CPA) represents a promising avenue for cancer prevention; however, additional data on patient adherence are needed. We developed the Colorectal Cancer Chemoprevention Acceleration Platform (CRC-CHAMP) and examined the feasibility of real-world CPA intervention in persons with an increased risk for CRC. Herein, we describe the recruitment, uptake, and adherence within this pilot study.
Methods
For this single-arm prospective pilot study (NCT05402124), we recruited individuals from the Forzani and MacPhail Colon Cancer Screening Centre (CCSC) in Calgary, Alberta, Canada. Eligible individuals were between the ages of 50-59 and had a history of high-risk adenomatous polyps diagnosed at the CCSC in the preceding 12 months. After consenting, each participant was provided open-label acetylsalicylic acid (ASA) at 81 mg every day for 90 days. Participants were contacted by telephone every 30 days to collect current adherence (primary outcome) and occurrence of any adverse events (secondary outcome).
Results
Throughout recruitment, 823 potential participants were identified, with 69 (53.1%) being eligible and 67 (51.5%) finally enrolling in the study. The median age of the participants was 56 yr. At the follow-ups, 60 participants (89.6%) were adherent at 30 days, 54 participants (80.6%) were adherent at 60 days, and 55 (82.1%) participants were adherent at 90 days. A total of 9 (13.4%) participants experienced minor adverse events likely unrelated to the ASA.
Conclusion
Participants recruited to this pilot study had a high level of adherence throughout the 90-day period with minimal side effects. Challenges to recruitment included low response rate by mail and a high number of those contacted already taking non-steroidal anti-inflammatory drugs. This study suggests that individuals post-polypectomy of large polyp may be interested in CRC chemoprevention. Additionally, this study provides a framework for future evaluation of additional therapeutic interventions.
Introduction
Colorectal cancer (CRC) is the fourth most diagnosed cancer and the second leading cause of cancer-related mortality in Canada. 1 In Canada, CRC screening programs are commonly used approaches to prevent and to detect CRC early and have led to decreases in incidence and mortality rates. However, adherence to CRC screening can vary considerably across provinces across Canada (ranging from 16.6% to 47.7%). 2 These adherence rates fall short of the national target of ≥60%. 3 Therefore, additional approaches should be considered to compliment CRC screening programs to further reduce CRC incidence and mortality. One potential approach is the use of chemoprevention agents (CPAs). 4 The term ‘chemoprevention’ also referred to ‘therapeutic intervention’ or ‘preventive therapy’ which involves the use of specific agents or substances to lower the risk of disease development.
CPA has generated interest as it may be more sustainable and cost-effective than interventions targeting long-term lifestyle modifications. 4 Interest in chemoprevention has been further bolstered by a push for ‘precision prevention’, which would tailor prevention strategies to individuals rather than populations. 5 As an example of this shift, greater emphasis has been placed on utilizing chemoprevention for individuals with family history of CRC or high-risk genetic profiles. 6 We previously conducted a systematic review and a network meta-analysis of the efficacy of known or suspected CPAs on the incidence of colorectal adenoma. 7 Trials of acetylsalicylic acid (ASA), difluoromethylornithine plus sulindac, and metformin has the strongest evidence of efficacy as CPAs. 7 Despite these results, evidence is insufficient to recommend population-based distribution of CPAs for CRC prevention. 7 Specifically, further evidence is required to assess potential adverse events and patient uptake of these interventions. 7
Notable reduction in CRC risk would require those at risk to adequately participate and adhere to CPAs. Many trials have been performed with Lynch syndrome participants, but greater evidence is needed among individuals in average risk screening programs. 8 To further evaluate the potential to use chemoprevention in a population undergoing screening, we developed a pilot study to evaluate the feasibility of large-scale chemoprevention trials and real-world chemoprevention studies for CRC prevention within a large CRC endoscopy screening center. In this study, we evaluated the feasibility of a real-world CPA intervention (open-label ASA) in persons at increased risk for CRC (one or more high-risk adenomas removed during colonoscopy) based on adherence (days taking ASA), and adverse events. ASA was used due to its wide availability promising results in previous studies. 8 Additionally, we evaluated factors related to adherence to ASA using validated surveys and interviews. Herein we described the baseline characteristics of the participants and various factors related to recruitment and subsequent adherence to ASA over a 90-day period.
Methods
Cohort Description and Eligibility Criteria
We selected patients from the Forzani and MacPhail Colon Cancer Screening Centre (CCSC) in Calgary, Alberta, Canada. The CCSC offers screening-related colonoscopies to asymptomatic patients within screening eligibility age. Participants were recruited between October of 2022 and September of 2023. Study recruitment and data collection were completed in April of 2024. Potentially eligible CCSC patients were identified from the Centre’s pathology database and were invited by mail through the CCSC to participate in the study. Eligible persons were between the ages of 50-59 at the time of consent and had a documented history of high-risk adenomatous polyps diagnosed at the CCSC within the previous 12 months. Indications for a patient’s index colonoscopy were gathered via electronic medical records. A patient was considered to have a history of high-risk polyps if they had at least one villous or tubulovillous polyp of any size, at least one polyp with high-grade dysplasia of any size, an adenomatous polyp measuring ≥1 cm, more than two tubular adenomas <1 cm in size, or any combination of these findings. Any of these findings would indicate an increased risk of future CRC and would alter follow-up surveillance. 9 We were primarily interested in chemoprevention for individuals increased CRC risk, so we included those who would likely receive a more intensive surveillance. Exclusion criteria included allergy or hypersensitivities to ASA, pre-existing regular use of ASA or other non-steroidal anti-inflammatory drugs (NSAIDs) or current use of corticosteroid/methotrexate/valproic acid/digoxin, daily use of anti-cancer/anti-platelet drugs. This was to ensure that those involved in the study would not have any medication-related obstacles to continued use of ASA for chemoprevention. We also excluded those with a daily use of greater than 3 units of alcohol, anticipated surgery in the next month, current or past history of GI ulcers or bleeding, cirrhosis or hepatic impairment, bleeding disorders, asthma, nasal polyps, history of colorectal cancer, platelet count <120K or >450K per microlitre, renal insufficiency or history of congestive heart failure. These criteria were to ensure that participants did not have any existing co-morbidities or risk factors which may conflict with ASA administration or increase risk of adverse events.
Study Design
The study was a single-arm (non-randomized), open-label prospective cohort pilot study (NCT05402124) which was divided into three phases (see Table S1). The study began with a 14-day recruitment phase. During this phase, participants gave written consent to being enrolled in the study and to the use of ASA for a 90-day period. After the completion of the recruitment phase, a 90-day active treatment phase was conducted. Persons who agreed to participate were instructed to take 81 mg of ASA daily for 90-days. Participants were contacted by telephone at 30, 60, and 90 days to examine levels of adherence and adverse events. At the end of the 90-day period, participants completed follow-up questionnaires. For 90-days following the active treatment phase, participants were not provided with ASA through the study and were not specifically instructed to continue treatment. No specific advice regarding future use of ASA was given. All participants were contacted via telephone at the end of this period (180 days after initiation) to determine the prevalence of continued use of ASA on their own. During each of these telephone calls, participants were asked to report any adverse events that they may have experienced. Each adverse event was given a score by one physician investigator indicating whether it was unrelated, unlikely, possible, probably or definitely related. We used a standardized form which indicated specific symptoms required for an event to be deemed related to the administration of ASA. Events labelled as unrelated or unlikely were designated as “unrelated” for the purposes of this study. This scale was based on the Common Terminology Criteria for Adverse Events (CTCAE) 5.0 system. 10 This study conforms to STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 11
Questionnaire Descriptions and Designs
A total of seven questionnaires were administered to participants throughout the study. A brief description of each questionnaire can be found in Table S2. For the purposes of this report, we focused on data obtained from the Health and Lifestyle Questionnaire (HLQ), adherence, and adverse events reported by participants. The HLQ contains questions regarding personal and family health history, cancer screening behaviour, diet, and sociodemographic characteristics. Questions focused on cancer and colorectal polyp history of the participants and their extended family. All patient information has been de-identified.
Statistical Analysis
The demographic characteristics of the participants were summarized using descriptive statistics. Continuous variables were evaluated with means and standard deviations (SD), and categorical variables were evaluated with frequency counts and percentages. We assessed adherence to the medication intervention over the 90-day active treatment phase by patient characteristics. Separate visuals for adherence and adverse events were generated using R. All analyses were conducted using R (version 4.5.0).
Ethics and Regulatory Approval
This study was conducted in compliance with the protocol approved by the Health Research Ethics Board of Alberta (HREBA), International Council for Harmonization of Technical Requirements for Human Use - Good Clinical Practice (ICH GCP), and according to Health Canada regulatory requirements. The project was approved under HREBA.CC-22-0062. This project is registered with ClinicalTrials.gov under registration identification number NCT05402124.
Results
Study invitation letters were sent to 823 potentially eligible participants over 11 months (September 2022 to July 2023), from which 130 responses were received with 69 (53.1%) being eligible and 67 (51.5%) enrolling in the study. Of the participants who sent back recruitment letters but were not enrolled in this study, the current use of ASA (n = 11; 8.4%) was noted as the most common reason for ineligibility apart from declining to participate (n = 14; 10.8%) or not following up with communications (n = 15; 11.5%). Other reasons for exclusion are described in Figure 1. Consort diagram depicting recruitment in the CRC-Champ study. 823 total mailouts were initially sent to potential participants, and 67 were successfully recruited
Participant Demographics
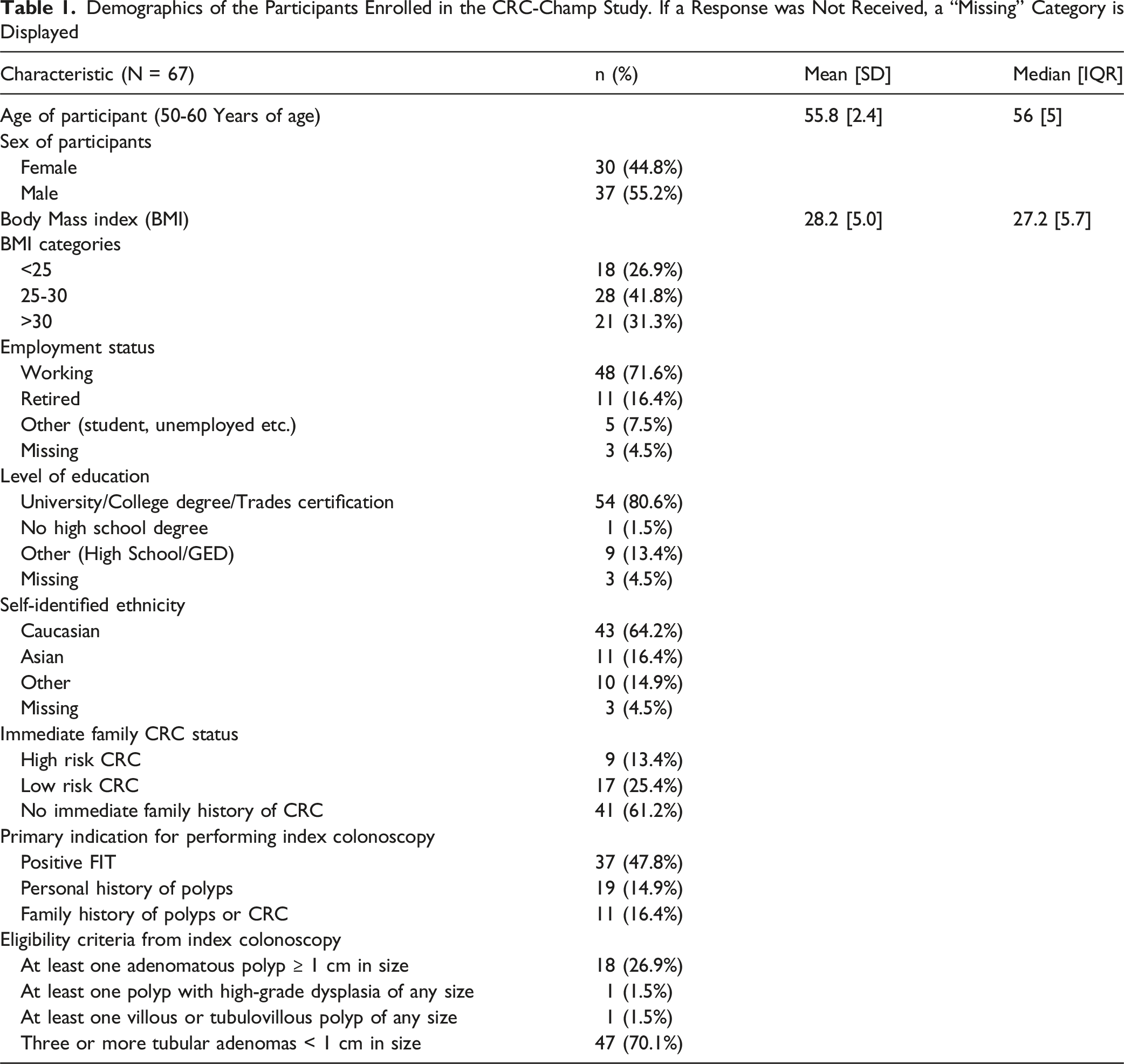
Demographics of the Participants Enrolled in the CRC-Champ Study. If a Response was Not Received, a “Missing” Category is Displayed
Patient Adherence and Adverse Events
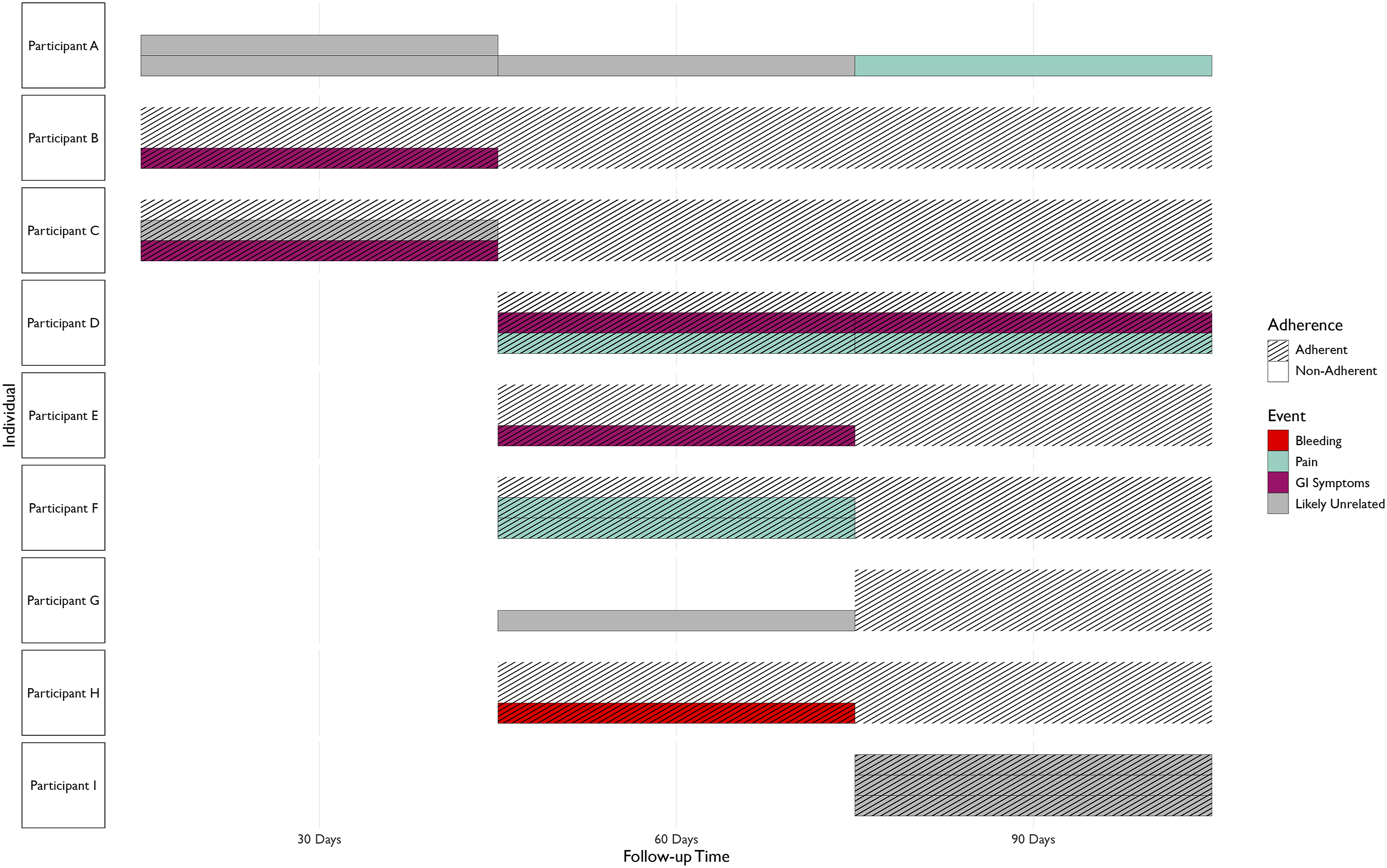
The study began with 67 participants taking ASA. After the first 30 days of active treatment phase, 60 (89.6%) participants reported that they were still taking ASA, while three participants discontinued treatment (4.5%) and four did not respond (6%) (Figure 2). Among these 63 people, within the first 30 days, there were five reported adverse events, including constipation, acid reflux (GI symptoms) and three unrelated events among three individuals (4.5%) (Figure 3). Two out of these three participants had discontinued the ASA. Between 30 and 60 days, 54 participants were adherent (80.6%), six participants were not (9.0%), with seven participants not responding (10.4%) (Figure 2). During this period, among these 60 people, there were eight adverse events among six people (9.0%) (including abdominal pain/heartburn, bloating, bleeding due to abrasions, bruising/pain, and two unrelated events) (Figure 3). At the end of the follow-up period (90 days), 55 participants were adherent (82.1%), with seven non-adherent participants (10.4%) and five missing responses (7.4%) (Figure 2). Out of these 62 people, there were six adverse events in three participants (one case of bruising, one case of abdominal pain/heartburn and three unrelated events) (Figure 3). Out of the three participants with adverse events at the 90-day follow-up, two were not adherent. Throughout the entire period, 48 (71.6%) participants were adherent at all follow-ups. Overall, out of the 67 participants recruited, nine (13.4%) experienced some adverse event, seven of which had events which were likely related to ASA (Figure 3). The final follow-up at 180 days also revealed that 6 out of 39 (15.4%) participants (who responded to the follow-up), continued to take ASA after the conclusion of the active treatment period. Adherence of 67 persons taking ASA in this study, between 1 and 3 months. Adherence is displayed as a Sankey diagram, where some participants responded as “Adherent” or “Not Adherent”, while some participants did not respond Adherence and adverse events of persons taking ASA in this study, between 1 and 3 months. Adverse events labelled as “unrelated” or “unlikely” related are indicated with a grey category. Black dashes indicate that the participant was not adherent during a month. Events are separated based on whether the participant was adherent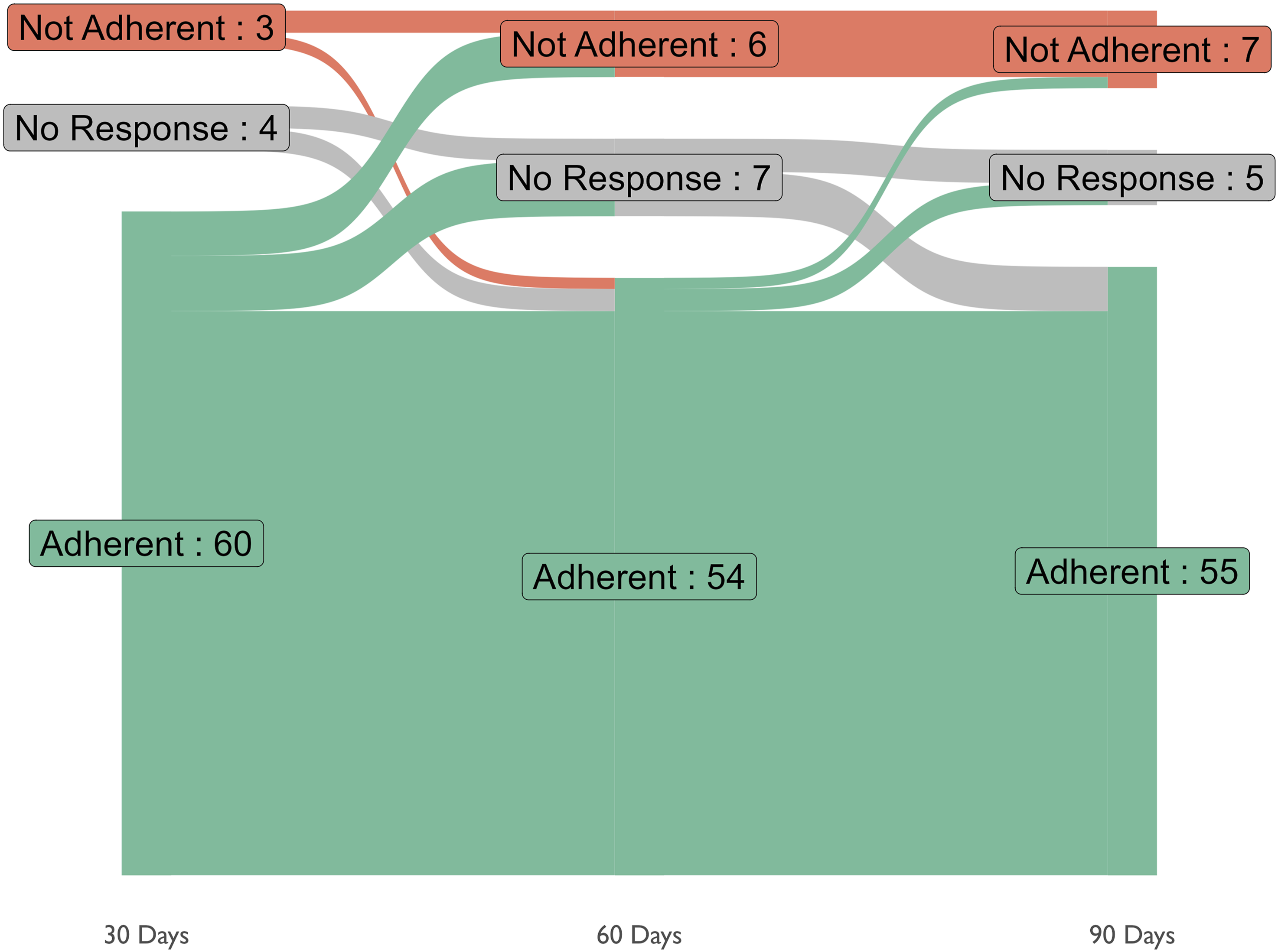
Discussion
Overall, during this study, we recruited 67 people and collected data regarding their use of ASA. At each of the 30-day follow-ups, over 80% of participants reported adherence to the treatment. At the end of this 90-day period, 9 participants experienced a minor adverse event (many of which were deemed unrelated to ASA).
There is increasing interest in CPAs as a strategy for preventing CRC. Several network meta-analyses (NMAs) of randomized controlled trials (RCTs) have evaluated the efficacy and safety of CPAs on CRC or colorectal adenoma (CRAs) prevention.7,12-19 With growing evidence on the potential of CPAs for CRC prevention in RCTs, there is a critical need to understand how this evidence will translate to adherence and feasibility of chemoprevention in real-world populations. In recent years, there has been an epidemiological shift in the incidence rates of CRC in younger Canadians, surpassing historical levels, further stressing the importance of enhanced cancer control strategies including prevention programs. 20
Our study population had a sex distribution similar to those in large RCTs that evaluated aspirin for the chemoprevention of CRC, such as the Aspirin/Folate Polyp Prevention Study and the Aspirin in Reducing Events in the Elderly (ASPREE) Trial.21,22 The median age was 56 years (IQR of 5), which is representative of the target population for CRC screening. Our study population was also comparable to a biorepository at the CCSC in Calgary, which reflects the screening-eligible population in Calgary and surrounding areas. 23 Within the biorepository, 54% of participants were male and 46% were female, which is comparable to our recruitment of 55.2% male and 44.8% female participants.
Among the 67 participants enrolled in this pilot study, we observed a high level of adherence to ASA at the end of the 90-day follow-up period. Our adherence rate was similar to the ASPREE trial, which reported an average compliance of 72.7% for their aspirin group. 22 This adherence dropped to 62% in the final 12 months of the trial’s intervention phase. In contrast, the Aspirin/Folate Polyp Prevention Study reported higher adherence rates, with 94.1% of participants taking virtually all study tablets during the first year, and 88.3% maintaining this level of adherence even in the final year. 21 However, these higher adherence rates may be attributed to the more rigorous monitoring and structured follow-up processes involved in these RCTs, which may help to ensure participants remain adherent to taking study medication. Our study’s adherence rate is noteworthy, given that it was achieved in a real-world setting with passive follow-up. This finding is encouraging for future studies involving real-world applications where extensive monitoring is not feasible.
Another aspect of this study that must be considered is the rate of adverse events. No serious adverse events occurred within the course of this study. However, as reported here 9/67 (13.4%) of the participants recruited to this study experienced some type of minor adverse event (Figure 3). Most are likely unrelated to the treatment were general symptoms with a high prevalence in this age group, even in the absence of treatment. 24 Our results can be compared to the Aspirin/Folate Polyp Prevention study, that evaluated the effect of aspirin on the risk of colorectal polyps. 22 This study tracked the occurrence of various adverse events and found few events overall. Additionally, the events that were reported occurred at similar rates in the placebo and aspirin groups, indicating that there may be no association. Another study conducted was the Cancer Prevention Programme (CAPP2), which recruited 861 participants with Lynch syndrome from several countries and prescribed aspirin to 427 of them. This study also found similar numbers of adverse events between the aspirin and placebo interventions. 25 Our results agree with these previous studies to suggest that CPA with NSAIDs does not increase the risk of serious adverse events or increase the rate of minor GI symptoms beyond those observed in this age group.
The data from this pilot study suggest that participants are generally interested in the potential use of ASA as a CPA and would consider using the medication for prolonged periods of time should the medication decrease rates of colorectal polyps. Furthermore, ASA is already widely used in the eligible population. We observed 11 out of 61 individuals who declined to participate in the study reported already taking ASA (Figure 1).
Limitations
This pilot study highlighted a limitation of physical mail-based recruitment into research studies. The response rate to our mailouts to potential participants was 15.8% (n = 130/823), which may suggest reconsidering mail-based recruitment for future studies and potentially highlight a need for means of digital recruitment approaches, such as through email or social media. In alignment with our ethics-approved protocol, a detailed recruitment letter was used to inform participants about the study’s background and purpose. The letter included details such as the target population, intervention, and rationale, information on data collection and eligibility, and assurances that the study would not affect or delay their ongoing care.
Future efforts could refine the recruitment materials with additional visual approaches, which may enhance its appeal to potential study participants. While we attempted to target a potentially eligible population, approximately half (53.1%; n = 69/130) of the respondents were deemed eligible after expressing interest. The largest number of people who were not eligible were already taking a non-steroidal anti-inflammatory drug.
In the future, we may also implement strategies to regularly check-in with participants to examine factors which may lead to a lack of adherence. Additionally, future research may attempt to gather further details regarding a participant’s decision to decline chemoprevention. Other factors leading to a lack of response or a refusal to participate may inform future health initiatives.
This study may also not be representative of the general population, as recruitment only took place within a single screening center in Alberta. This approach may have led to under-representation across ethnic and socioeconomic groups. Additionally, the sample size of 67 is too small for thorough statistical analysis, somewhat limiting analysis of various trends and behaviors regarding chemoprevention. The issues discussed may limit the generalizability to other populations. These insights will be useful for refining recruitment strategies and streamlining future enrollment processes. Finally, this study only included a single NSAID over a relatively short follow-up period and was single-arm, meaning there was no other treatment or placebo used as a comparator. The processes developed in the study can be expanded to evaluate a wider range of potential CPAs and compare patient reception, efficacy and level of adverse events.
Strengths
To our knowledge, this pilot study is among the first to evaluate the potential for chemoprevention in a real-world population undergoing screening in Canada. The few occurrences of minor adverse events underscore the tolerability of ASA in this population, making it a viable option for future use. Importantly, the high level of adherence observed indicates that participants are willing to engage in chemoprevention and may have positive perceptions of these interventions. Overall, our findings suggest that real-world implementation of chemoprevention for CRC is feasible, and our platform could be expanded to include other CPAs, such as vitamin D or GLP-1 receptor agonists for broader application. Future longer-term studies are needed to explore adherence rates among persons using ASA as a CPA in addition to adverse events when using ASA for prolonged periods of time.
Conclusion
In this pilot study utilizing ASA, participants indicated an interest in chemoprevention and indicated high adherence rates in the short term with minimal safety issues. However, given our low initial response rates, larger future studies will be required to identify novel digital recruitment approaches to increase recruitment to proceed to larger, longitudinal studies.
Supplemental Material
Supplemental Material - The Colorectal Cancer Chemoprevention Acceleration and Improvement Platform (CRC-CHAMP) – Cohort Description
Supplemental Material for The Colorectal Cancer Chemoprevention Acceleration and Improvement Platform (CRC-CHAMP) – Cohort Description by Darren R. Brenner, Xuanhao Feng, Courtney Maxwell, John M. Hutchinson, Chantelle Carbonell, Reynaldo Nambayan, Linda Rabeneck, Jill Tinmouth, Nauzer Forbes, Steven J. Heitman, Khara M. Sauro, Colleen Cuthbert, Dylan E. O’Sullivan, Robert J. Hilsden in Cancer Control
Footnotes
Acknowledgements
We are grateful for the participants who have donated their time for this study. In addition, we are grateful to the support and assistance of all of the nurses and endoscopists at the Forzani & MacPhail Colon Cancer Screening Centre.
Ethical Considerations
This study was conducted in compliance with the protocol approved by the Health Research Ethics Board of Alberta (HREBA), International Council for Harmonization of Technical Requirements for Human Use - Good Clinical Practice (ICH-GCP), and according to Health Canada regulatory requirements.
Consent to Participate
All participants were provided with a consent form, which described this study and provided sufficient information for participants to make an informed decision regarding their participation in the study.
Author Contributions
DRB, RJH, LR, JT, NF, DO, SH and CM contributed to the conception and design of the project. XF, CM, RN and RJH contributed to the acquisition and analysis of the data. XF, JH and CC drafted the manuscript. All authors reviewed and approved the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Canadian Institutes of Health Research & Canadian Cancer Society (CCS/CIHR Action Grants – 2022 grant (#707227 – awarded January 2022).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The relevant data has been included within the manuscript document.
Clinical Trial Registration
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
