Abstract
Objective
This study aimed to evaluate hepatitis B virus (HBV) reactivation and its effect on tumor response and survival outcomes in patients with HBV-related advanced hepatocellular carcinoma (HCC) undergoing lenvatinib plus camrelizumab treatment.
Methods
216 patients with HBV-related advanced HCC receiving lenvatinib and camrelizumab were enrolled. Overall survival (OS), progression-free survival, and tumor response were evaluated. Univariate and multivariate analyses were performed to determine risk factors for HBV reactivation.
Results
HBV reactivation occurred in 24 patients (11.1%). It was associated with poor survival and tumor response in these patients. Undetectable DNA levels, the absence of antiviral therapy, and high ALT levels were identified as vital risk factors for HBV reactivation. After receiving or adjusting the antiviral strategy, tumor response improved in patients with HBV reactivation.
Conclusions
HBV reactivation could occur in patients with HBV-related HCC, treated with lenvatinib and camrelizumab, worsening tumor response and patient survival. Regular monitoring of the indicators of HBV infection and effective antiviral treatments are recommended for these patients to prevent severe complications.
Introduction
Hepatocellular carcinoma (HCC) is the fourth most common cancer and the second leading cause of cancer-related deaths in China; 85% of HCC cases are associated with Hepatitis B virus (HBV) infection.1,2 Unfortunately, approximately half of the HCC cases are advanced at diagnosis, and systemic therapies such as immune checkpoint inhibitors (ICIs) and tyrosine kinase inhibitors (TKIs) are required at an advanced stage. 3 In the landmark IMbrave 150 trial, the combination of a PD-L1 antibody and a VEGF inhibitor demonstrated superior survival outcomes and improved tumor response compared to sorafenib. 4
HBV reactivation is characterized by the sudden resurgence or elevation of HBV DNA levels in patients with resolved or inactive chronic HBV infection. 5 This phenomenon often presents as a rapid increase in HBV load or reappearance of HBsAg. 6 HBV reactivation can cause severe complications including liver failure, interruption of anticancer treatment, and mortality. Recent studies have highlighted the occurrence of HBV reactivation in various HCC treatments such as immunotherapy, transarterial chemoembolization (TACE), hepatectomy, and radiofrequency ablation (RFA).7-10 Despite recommendations for patients with advanced HCC receiving systemic therapies to undergo antiviral prophylaxis, 6 the persistence of covalently closed circular (ccc) DNA in chronic HBV infection poses challenges, 11 leading to the risk of persistent HBV reactivation.
Lenvatinib, a novel TKI, was approved as a first-line treatment for advanced HCC in 2018 and has demonstrated non-inferiority to sorafenib in improving the overall survival (OS) of patients with HCC. 12 Camrelizumab (AiRuiKa™), a PD-1 antibody developed by Jiangsu Hengrui Medicine Co. Ltd, has received approval for the treatment of various malignancies, including HCC, exhibiting promising antitumor efficacy with manageable side effects.13-15
To the best of our knowledge, limited research has focused on HBV reactivation in patients with HBV-related advanced HCC receiving TKIs and ICIs, and no study has specifically addressed HBV reactivation in patients receiving lenvatinib and camrelizumab treatment. The incidence of HBV reactivation and its potential risk factors in patients undergoing combination therapy remain unclear. Moreover, the optimal clinical management following HBV reactivation remains elusive. Therefore, we conducted this retrospective study to assess HBV reactivation in patients with HBV-related advanced HCC who were treated with lenvatinib and camrelizumab.
Patients and Methods
Patients
This retrospective study enrolled 216 patients who underwent treatment with lenvatinib plus camrelizumab between January 2019 and January 2023 at the Wuhan Third Hospital and Wuhan Union Hospital. The inclusion criteria comprised: (1) histopathologically or clinically confirmed advanced HCC; (2) receipt of lenvatinib and camrelizumab; (3) age between 18 and 75 years; and (4) positivity for HBsAg or negativity for HBsAg but positivity for hepatitis B core antibody. The exclusion criteria were as follows: (1) coinfection with other viruses, such as hepatitis C virus (HCV); (2) concurrent use of other treatments, such as TACE and RFA; and (3) incomplete data. Patients experiencing HBV reactivation were categorized into the HBV reactivation group (HBVr), whereas the remaining patients were placed in the non-HBV reactivation group (non-HBVr). This study was conducted according to the STROBE checklist for cohort studies. 16 This study was approved by the Ethics Committee of the Wuhan Third Hospital. Owing to the retrospective nature of the study, the requirement for written informed consent was waived by the Ethics Committee of Wuhan Third Hospital (Approval Document Number: KY2023-035) and Wuhan union hospital (UHCT-IEC-SOP-017-08-03).
Treatment
Patients received intravenous administration of 200 mg of camrelizumab every 3 weeks, along with oral administration of lenvatinib once daily at a dose of 8 mg for individuals weighing less than 60 kg or 12 mg for those weighing ≥60 kg. Treatment cessation occurred in the absence of clinical benefits or due to unacceptable toxicity.
In cases of HBV reactivation, we instituted or adjusted antiviral therapy as follows: for patients not undergoing antiviral treatment, we recommended tenofovir alafenamide fumarate (TAF) at a dose of 25 mg per day (manufactured by Gilead); for patients receiving only entecavir (ETV) at a dose of 0.5 mg per day (manufactured by Bristol-Myers Squibb), we recommended a combination of ETV and TAF.
Outcome Assessments
The primary end point of this study was HBV reactivation. According to the Asian-Pacific Association for the Study of the Liver clinical practice guidelines on HBV reactivation, 17 it is defined as follows: For patients with chronic HBV infection (HBsAg-positive), it includes (1) a ≥2 log(100-fold) increase in HBV DNA levels compared to baseline; (2) detection of HBV DNA at levels >100 IU/mL in a person with undetectable HBV DNA at baseline. For patients with past HBV infection (HBsAg-negative and anti-HBc-positive), reactivation includes (1) reverse HBsAg seroconversion, where HBsAg-negative becomes HBsAg-positive, or (2) the appearance of HBV DNA in the absence of HBsAg, where HBV DNA-undetectable becomes HBV DNA-detectable.
Tumor responses were assessed using contrast-enhanced computed tomography (CT) or magnetic resonance imaging (MRI) based on the modified Response Evaluation Criteria for Solid Tumors. 18 The objective response rate (ORR) was determined as the sum of the incidences of complete response (CR) and partial response (PR). The disease control rate (DCR) included CR, PR, and stable disease (SD). Progression-free survival (PFS) was calculated from the initiation of treatment to the occurrence of tumor progression, death, or the last follow-up in censored data. OS was measured from the commencement of treatment to death or the last follow-up in the censored data.
HBV reactivation was assessed before treatment and then every 4-6 weeks after treatment. The tumor response was evaluated at the end of the third month of treatment. In the HBV reactivation group, the tumor response was reassessed 3 months after the initiation of a new antiviral treatment regimen.
Follow-Up
During follow-up, the patients underwent laboratory, imaging, and physical examinations every 4-6 weeks. Laboratory examinations included HBV DNA levels, HBeAg, alanine aminotransferase (ALT), and aspartate aminotransferase (AST), among others. Abdominal contrast-enhanced CT or MRI was performed to analyze tumor response, recurrence, and metastasis inside and outside the liver.
Statistical Analysis
Continuous variables were presented as medians (ranges), while categorical variables were expressed as frequencies (percentages). Categorical variables were analyzed using the chi-square test or Fisher’s exact test, whereas continuous variables were compared using the Student's t test or Mann–Whitney U test. Risk factors for HBV reactivation were identified using univariate and multivariate logistic regression analyses. Variables with a bilateral P-value <0.1 in the univariate analysis were included in the multivariate analysis. The use of a more lenient threshold of P < 0.1 in the univariate analysis was intended to capture potential risk factors that might show some association with HBV reactivation but did not meet the stricter conventional threshold of P < 0.05. This broader screening approach allowed for the consideration of additional variables that could be relevant. For all other analyses in this study, a bilateral P-value <0.05 was considered statistically significant. Statistical analyses were conducted using SPSS software version 24.0, and GraphPad Prism 8.0 was utilized to generate the necessary figures.
Results
Patient Characteristics
In total, 282 patients with HCC received lenvatinib plus camrelizumab during the follow-up period, and 66 patients were excluded for the following reasons: five patients had co-infection with HCV, 39 were receiving this therapy in combination with other treatments, and 22 had incomplete data. Finally, 216 patients were enrolled in this study (Figure 1). Of the total patients, 24 experienced HBV reactivation. The detailed baseline characteristics of the patients with and without HBV reactivation are summarized in Table 1. Flow chart illustrating the selection of patients. Baseline Characteristics of Patients. BCLC Barcelona Clinic Liver Cancer; HBV, hepatitis B virus; HBeAg, hepatitis B e antigen; AFP, a-fetoprotein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TB, total bilirubin; NLR, neutrophil-to-lymphocyte ratio; PLT, platelet.

HBV Reactivation
In this study, 24 patients (11.1%) experienced HBV reactivation with a median reactivation time of 2.4 months (range 1.0-8.4 months). In addition, 15 patients had an elevated HBV load; however, the viral load did not meet the HBV reactivation criteria. Five of 24 patients with HBV reactivation received antiviral prophylaxis before or during treatment. When the patients experienced HBV reactivation, the use of ETV was adjusted to include ETV and TAF. The remaining 19 patients failed to receive antiviral therapy because of undetectable HBV DNA; however, after HBV reactivation, they chose to undergo antiviral therapy (TAF).
Predicting Factors for HBV Reactivation
Univariate and Multivariate Analysis of Prognostic Factors for HBV Reactivation.
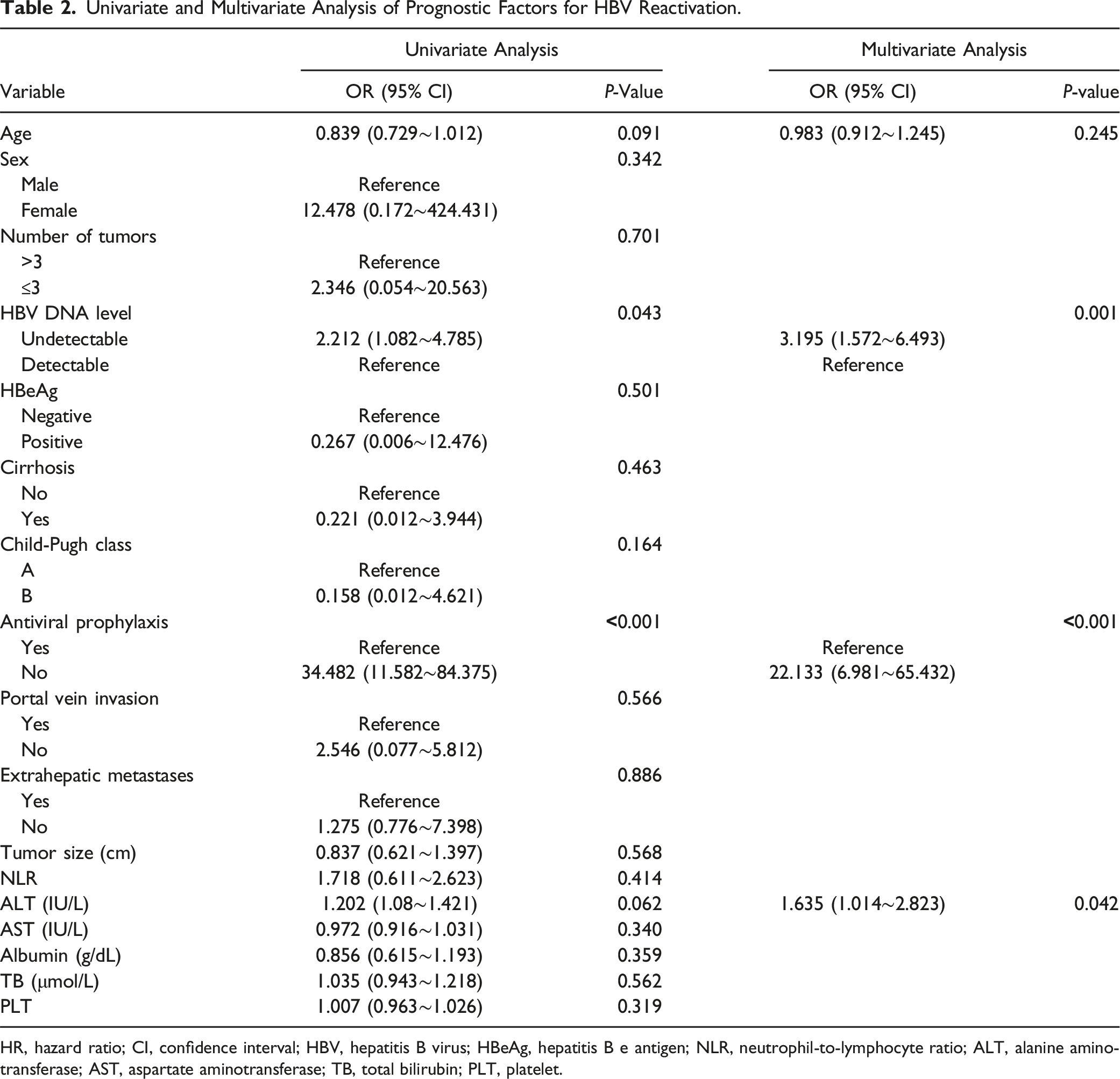
HR, hazard ratio; CI, confidence interval; HBV, hepatitis B virus; HBeAg, hepatitis B e antigen; NLR, neutrophil-to-lymphocyte ratio; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TB, total bilirubin; PLT, platelet.
Tumor Response and Patient Survival
Tumor Response in HBVr Group and Non-HBVr Group.

Data are presented as n (%), CR complete response, PR partial response, SD stable disease, PD progressive disease, ORR objective response rate, DCR disease control rate, HBVr group, patients with HBV reactivation, no-HBVr group, and patients without HBV reactivation.

Typical CT images of the HBV reactivation group and the non-reactivation groups. (A): a 56-year-old male with HBV reactivation (CT images before and 3 months after treatment); (B): a 62-year-old male without HBV reactivation (CT images before and 3 months after treatment).
Tumor Response in HBVr (+) Group at HBV Reactivation and After Adjusted Antiviral Therapy.

Data are presented as n (%). CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; ORR, objective response rate; DCR, disease control rate.

The CT data of a 58-year-old male receiving new antiviral treatment in the HBV reactivation group. (A): the CT image before treatment; (B): the CT image after HBV reactivation; (C): the CT image after adjustment of antiviral treatment (ETV to ETV + TAF).
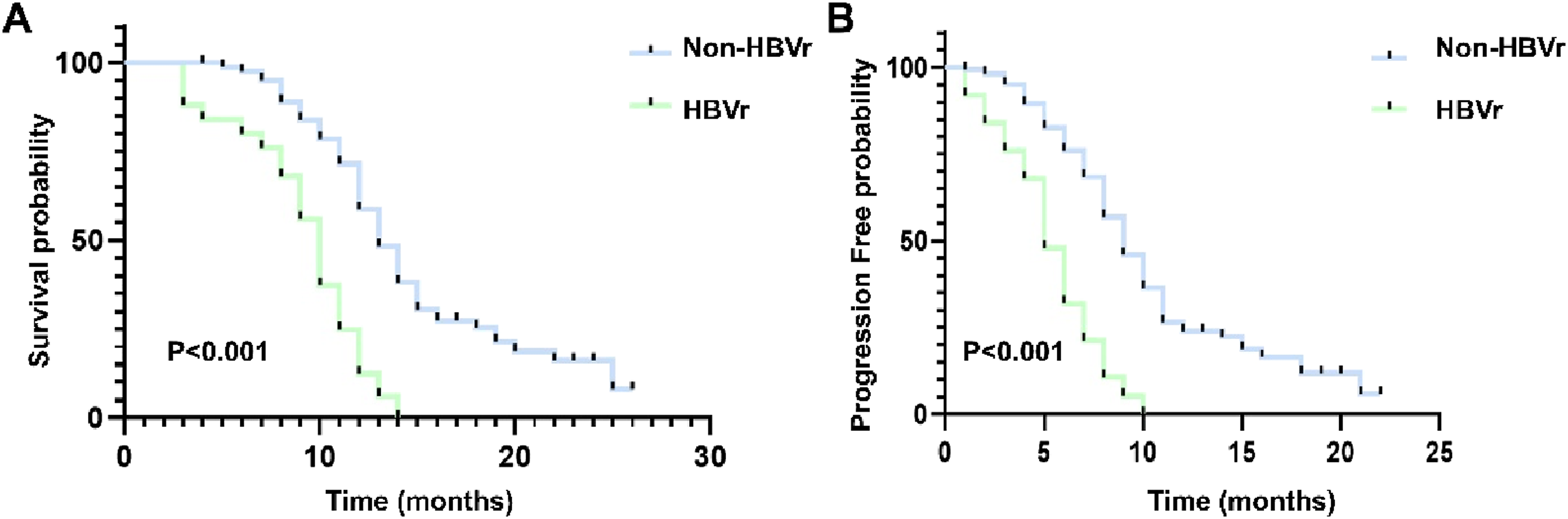
The Kaplan–Meier (KM) curves for HBV-related advanced HCC patients who received the treatment of lenvatinib plus camrelizumab: (A) the KM curves of overall survival time; (B) the KM curves of progression-free time.
Discussion
Patients with Advanced HBV-related HCC are predisposed to HBV reactivation, which is a poor prognostic factor for HCC. 19 Furthermore, HBVr can deteriorate liver function and interrupt antineoplastic therapy, further worsening survival.20,21 Currently, the combination of TKIs and ICIs is pivotal for the treatment of advanced HCC.22,23 However, the rate of HBV reactivation and potential prognostic factors for HBVr in patients treated with TKIs and ICIs remain unclear, and there is no consensus regarding the clinical management of HBV reactivation. To the best of our knowledge, this is the first study to focus on HBV reactivation in patients with HCC treated with lenvatinib and camrelizumab; 11.1% of the patients experienced HBV reactivation after combination treatment.
Patients with HBV reactivation had a poor tumor response rate, with a lower ORR (16.7% vs 54.8%, P < 0.001) and DCR (45.8% vs 93.7%, P < 0.001) than those of patients without HBV reactivation. Addition or adjustment of antiviral treatments can significantly increase tumor response rates. Thus, regular monitoring of HBV reactivation indicators and the timely use of new antiviral protocols are crucial for these patients. However, our results demonstrated that HBV reactivation significantly worsened OS and PFS in patients treated with lenvatinib plus camrelizumab despite timely adjustment in the antiviral protocol. The effectiveness of the new protocol may be limited, as evidenced by the fact that nearly 35% of the patients showed decreased HBV DNA levels, 40 maintained stable HBV DNA levels, and 25% had uncontrolled HBV DNA levels. This highlights the need for more effective antiviral treatments. Although the ongoing monitoring of HBV DNA is imperative, combination therapy remains safe for patients with HBV-related advanced HCC with a low reactivation rate.
In the univariate analyses, age, undetectable HBV DNA levels, lack of antiviral prophylaxis, and high ALT levels were risk predictors of HBV reactivation. Multivariate analyses confirmed that undetectable HBV DNA levels, lack of antiviral prophylaxis, and high ALT levels were risk predictors of HBV reactivation. Among patients who experienced HBV reactivation, 83.3% had undetectable HBV DNA levels at baseline and discontinued antiviral treatment. They might have received effective antiviral therapy in the past or HBV was inactive before treatment. However, antiviral drugs can effectively suppress HBV replication without eradicating the cccDNA. 24 cccDNA serves as a template for HBV RNA transcription and persists in infected hepatocytes. 25 Viral replication can persist indefinitely, resulting in chronic HBV infection, which can cause varying degrees of liver damage. Even if there is spontaneous ‘sero-virological recovery,’ intrahepatic cccDNA may persist, potentially leading to HBV reactivation. Thus, even in HCC patients with a past HBV infection, it is necessary to regularly monitor the HBV viral load and ensure the timely use of antiviral drugs, especially for those with undetectable HBV DNA.
This study had certain limitations. This was a single-center retrospective study with a relatively small sample size, which should be further confirmed in future prospective studies. Furthermore, the patients were exclusively from endemic areas with HBV genotypes distinct from those in other populations. Thus, the application of these results to the general population requires further investigation.
Conclusion
HBV reactivation can occur in patients with HBV-related HCC treated with lenvatinib and camrelizumab and worsen tumor response and patient survival. Regular monitoring of the indicators of HBV infection and effective antiviral treatments are recommended for these patients to prevent severe complications.
Footnotes
Author Contributions
J.W designed research; D.W., B.S. collected and analyzed the data; B.S., J.W., D.W. wrote the paper. All the authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
