Abstract
Introduction
Rare epidermal growth factor receptor (EGFR) mutations other than G719X, S768I, and L861Q are infrequently represented in clinical trials. The efficacy of tyrosine kinase inhibitors (TKIs) against these uncommon variants, either alone or in combination with common mutations, remains limited.
Methods
We retrospectively analyzed patients with advanced-stage non-small cell lung cancer (NSCLC) harboring non-major uncommon EGFR mutations who received first-line afatinib between January 2018 and October 2024. Patients with only TKI-sensitive mutations (Del19, L858 R, G719X, S768I, and L861Q) and without additional rare variants were excluded. The primary endpoints were the objective response rate (ORR) and progression-free survival (PFS). The secondary endpoint was the duration of response (DOR).
Results
Among the 44 patients, 36.4% had solitary non-major uncommon mutations. The overall ORR and the disease control rates were 65.9% and 86.4%, respectively. The ORR by subgroup was 75.0% for patients with non-major uncommon mutations plus EGFR-TKI-sensitive mutations, and 55.0% for patients with dual or solitary uncommon mutations. At a median follow-up time of 21.9 months, the median PFS was 11.5 months (95% CI: 7.5-22.6). Patients with non-major uncommon mutations co-occurring with TKI-sensitive mutations had a longer median PFS (17.7 months; 95% CI: 13.6–NR) than those harboring non-major mutations alone (either single or dual) (9.1 months; 95% CI: 4.6–NR), P = 0.04. The median duration of response was 16.1 months (95% CI: 10.3–NR) with a median follow-up time of 16.8 months.
Conclusion
Afatinib demonstrated encouraging efficacy in NSCLC patients with nonmajor uncommon EGFR mutations other than G719X, S768I, and L861Q, regardless of whether the mutations were solitary or compound. Comprehensive EGFR mutation profiling is crucial for identifying uncommon EGFR mutation patients likely to benefit significantly from afatinib.
Introduction
According to a local survey on genetic mutations in non-small cell lung cancer (NSCLC) patients in Vietnam, the frequency of epidermal growth factor receptor (EGFR) mutations is approximately 41%. Among them, exon 19 deletions (Del 19), L858R, uncommon, and compound mutations account for 55.4%, 27%, 17.4%, and 3.5%, respectively. 1 Numerous studies have also shown that Del 19 and L858R are the most prevalent EGFR gene mutations, accounting for approximately 90% of all mutations. 2 The remaining mutations are rare with various subtypes, either alone or in combination with other EGFR mutations. Among them, G719X, S768I, and L861Q are the most frequently observed and classified as major uncommon mutations, 3 whereas the others are considerably rare and are typically reported in case studies or small series of cases.
The second-generation EGFR tyrosine kinase inhibitor (TKI afatinib) is an irreversible tyrosine kinase inhibitor that improves both the objective response rate (ORR) and median progression-free survival (mPFS) compared to chemotherapy, consistent with both common mutations (Del 19 and L858R)3,4 and certain uncommon mutations, as evidenced by retrospective analysis of the LUX-Lung 2, 3, and 6 trials. 5 According to Yang et al, among 600 patients treated with afatinib across these trials, 75 patients (12%) harbored uncommon EGFR mutations (G719X, S768I, and L861Q). The ORR in this subgroup was 71.1% (95% CI 54.1-84.6), with an mPFS of 10.7 months (95% CI 5.6-14.7). 3 This study did not specifically address very rare mutations beyond the major uncommon ones (non-major uncommon mutations).
Several real-world studies on the efficacy of afatinib in NSCLC patients with non-major uncommon EGFR mutations have been published. Xiang Long et al reported favorable outcomes of first-line afatinib in a patient with the L833V/H835L double mutation in exon 21, with a PFS exceeding 10 months. 6 In another study, James Chih-Hsin Yang et al analyzed 1023 NSCLC patients with uncommon EGFR mutations, 15.9% of whom harbored non-major uncommon mutations. This subgroup had a median time to treatment failure (mTTF) of 10.7 months and an overall ORR of 63.9%, with encouraging outcomes for E709X (11.4 months; ORR of 84.6%), L747X (14.7 months; ORR of 80.0%), and compound mutations (11.5 months; ORR of 63.9%). Yang also noted that, despite the small sample size, afatinib showed promising activity in certain exon 20 mutations, including those at positions A763, M766, N771, and V769, as well as in osimertinib-resistant mutations such as G724S, L718X, and C797S. 7
However, the non-major uncommon mutation group—those beyond G719X, L861Q, and S768I—is highly heterogeneous and incompletely understood. More importantly, clinicians occasionally encounter mutations not yet reported in the literature. Hence, whether afatinib, other EGFR TKIs, or chemotherapy is the optimal treatment for a specific rare mutation remains a challenge. Thus, reports on the efficacy of therapies for these EGFR mutations are essential.
We conducted a retrospective study evaluating the effectiveness of afatinib in Vietnamese patients with NSCLC harboring non-major uncommon EGFR mutations.
Materials and Methods
Study Design
This retrospective analysis included patients with advanced-stage NSCLC harboring non-major uncommon EGFR mutations who were treated at Vietnam National Cancer Hospital between January 2018 and October 2024. The primary endpoints were the ORR and PFS. The secondary endpoint was the duration of response (DOR). Informed consent was obtained in written form from the patient or, when not possible, from a Legally Authorized Representative. The study was approved by the Hanoi Medical University Institutional Ethical Review Board (HMU IRB), Hanoi, Vietnam (Approval Number: 905/GCN-HĐĐĐNCYSH-ĐHYHN, dated May 16, 2023).
Patients
Eligible participants were adult patients (≥18 years) with treatment-naive, advanced-stage non-small cell lung cancer (NSCLC) harboring non-major uncommon EGFR mutations (other than Del 19, L858R, T790M, G719X, S768I, L861Q, or any combination of these mutations). Patients with cooccurrences of non-major uncommon EGFR mutations and either common mutations (Del19, L858R) or major uncommon mutations (G719X, S768I, L861Q) were included and classified as having compound mutations with TKI-sensitive mutations. EGFR mutations were identified via next-generation sequencing using either pretreatment tumor biopsy or liquid biopsy samples. All participants were treated with first-line afatinib. Patients with asymptomatic or stable brain metastases were eligible. Additional inclusion criteria included an Eastern Cooperative Oncology Group Performance Status (ECOG PS) score of 3 or below.
The exclusion criteria were severe hepatic dysfunction (Child‒Pugh C), significant renal impairment (estimated glomerular filtration rate <15 mL/min/1.73 m2), or the presence of a concurrent malignancy. Patients who discontinued afatinib for reasons unrelated to disease progression or adverse effects were also excluded.
EGFR Mutation Analysis
EGFR mutations were identified using targeted next-generation sequencing (NGS) with the Gene2Me Cancercheck 100 panel (Gene2Me) on the DNBSEQ-G99 platform. The median sequencing depth was 1000× for Formalin-Fixed Paraffin-Embedded (FFPE) tumor tissue and 10000× for plasma/ctDNA samples. Variant filtering criteria included a variant allele frequency (VAF) threshold of ≥1% for tissue and ≥0.1% for plasma, with a minimum coverage of ≥1000× (tissue) and ≥10000× (plasma). Sequencing quality metrics were Q30 ≥ 96% (R1: 96.48%; R2: 96.02%) and on-target reads ≥94.5%. Detected variants were interpreted and classified according to the American College of Medical Genetics and Genomics/Association for Molecular Pathology (ACMG/AMP) guidelines.
Procedures
All eligible patients received afatinib once daily until disease progression or intolerable toxicity. The starting treatment dose was determined by the treating physician on the basis of patient characteristics, including age, sex, comorbidities, and ECOG PS. Thoracic radiotherapy prior to disease progression with EGFR-TKIs was not allowed.
Initial evaluations included a physical examination, computed tomography (CT) or magnetic resonance imaging (MRI), bone scintigraphy, positron emission tomography-computed tomography, if indicated, and hematologic and biochemical methods. After initiating afatinib treatment, patients underwent routine evaluations, with clinical examinations, laboratory tests, and chest CT or MRI scans, at an interval of 2 months (± 1 month) at the physician’s discretion. Primary tumor evaluation was conducted via contrast
CT, whereas assessment of brain lesions was performed via either contrast CT or brain MRI, according to the physician’s discretion.
Statistical Analyses
The data were analyzed via R software for Windows version 4.3.3 (https://cran.r-project.org/bin/windows/base/). Continuous variables are presented as the means ± standard deviations or medians with interquartile ranges; categorical variables are summarized as counts and percentages. Comparisons were performed via Fisher’s exact test and chi-square tests, as appropriate. PFS was estimated via the Kaplan–Meier method and compared via the log-rank test. The median follow-up was calculated via the reverse Kaplan‒Meier method. A two-sided test with P < 0.05 was considered to indicate statistical significance. An exploratory post-hoc power analysis for the unadjusted log-rank comparison was performed using Schoenfeld’s approximation based on the observed univariate Cox HR, number of events, and allocation. As a supplementary analysis, restricted mean survival time (RMST) was estimated up to a truncation time of 18 months, selected to ensure adequate numbers of patients at risk across comparison groups and to provide stable estimates.
PFS was defined as the duration from the initiation of afatinib treatment to either documented disease progression, as assessed by RECIST 1.1, or death from any cause, whichever occurred first. DOR was defined as the median time during which a patient maintained a response to treatment before disease progression.
The ORR, defined as the best response recorded across evaluations, was calculated as the proportion of patients achieving a complete response (CR) or partial response (PR) according to the RECIST 1.1 criteria, as assessed by the investigator. The disease control rate (DCR) was defined as the proportion of patients who achieved CR, PR, or stable disease (SD) as their best response.
Results
Study Population
Patient Characteristics

EGFR: epidermal growth factor receptor.
The detailed frequencies and types of uncommon EGFR mutations are summarized in Supplemental Table S1.
Objective Response Rate
Objective Response Rate by Mutation Subtypes

ORR: overall response rate, DCR: disease control rate; TKI-sensitive mutations: Del19/L858 R/G719X/L861Q/S768I.
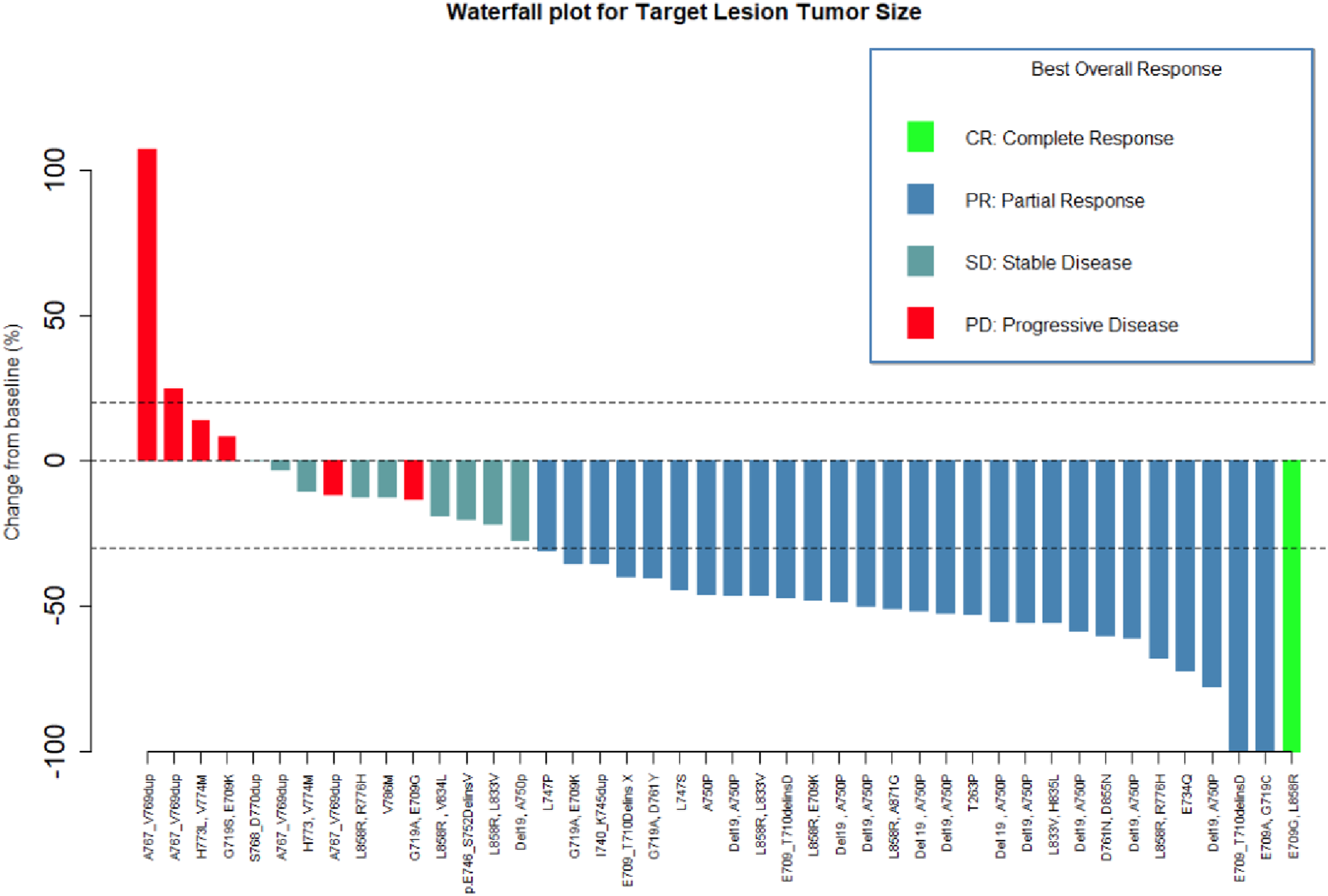
Objective Response – Waterfall Plot for Target Lesion
Patients harboring compound mutations involving Del19, L858R, or G719X together with a non-major uncommon mutation demonstrated the highest treatment benefit, with an ORR of 75.0% and a DCR of 91.7% (no patients harbored compound mutations involving L786Q or S768I). This group also accounted for the only CR observed (4.2%) and the highest PR rate (70.8%). In contrast, patients with single or dual non-major uncommon mutations had an ORR of 55.0% and a DCR of 80.0%, with no complete responses observed. PD was more common in this group (20.0%) than in the TKI-sensitive plus uncommon mutation group (8.3%).
Progression-Free Survival
After a median follow-up of 21.9 months (95% CI: 12.3–NR), the median PFS of the overall population was 11.5 months (95% CI: 7.5-22.6) (Figure 2A). The individual progression status and PFS for each patient are shown in Figure 3. Subgroup analyses of mPFS based on sex, age (<65 vs ≥65 years), performance status, smoking history, baseline brain metastasis status, starting dose, and co-occurrence with common EGFR mutations or G719X are reported in Table 3. Kaplan–Meier Curves for Progression Free Survival (A) and Duration of Response (B) Individual Progression Status and Duration of Progression-free Survival Progression – Free Survival and Related Factors aUnivariate Cox regression analysis. bMultivariate Cox regression analysis. RMST: Restricted Mean Survival Time, calculated up to τ (18 months), robust to non-proportional hazards. p_per: Two-sided permutation P-value (10 000 resamplings). EGFR: epidermal growth factor receptor; TKI-sensitive mutations: Del19/L858 R/G719X/L861Q/S768I.
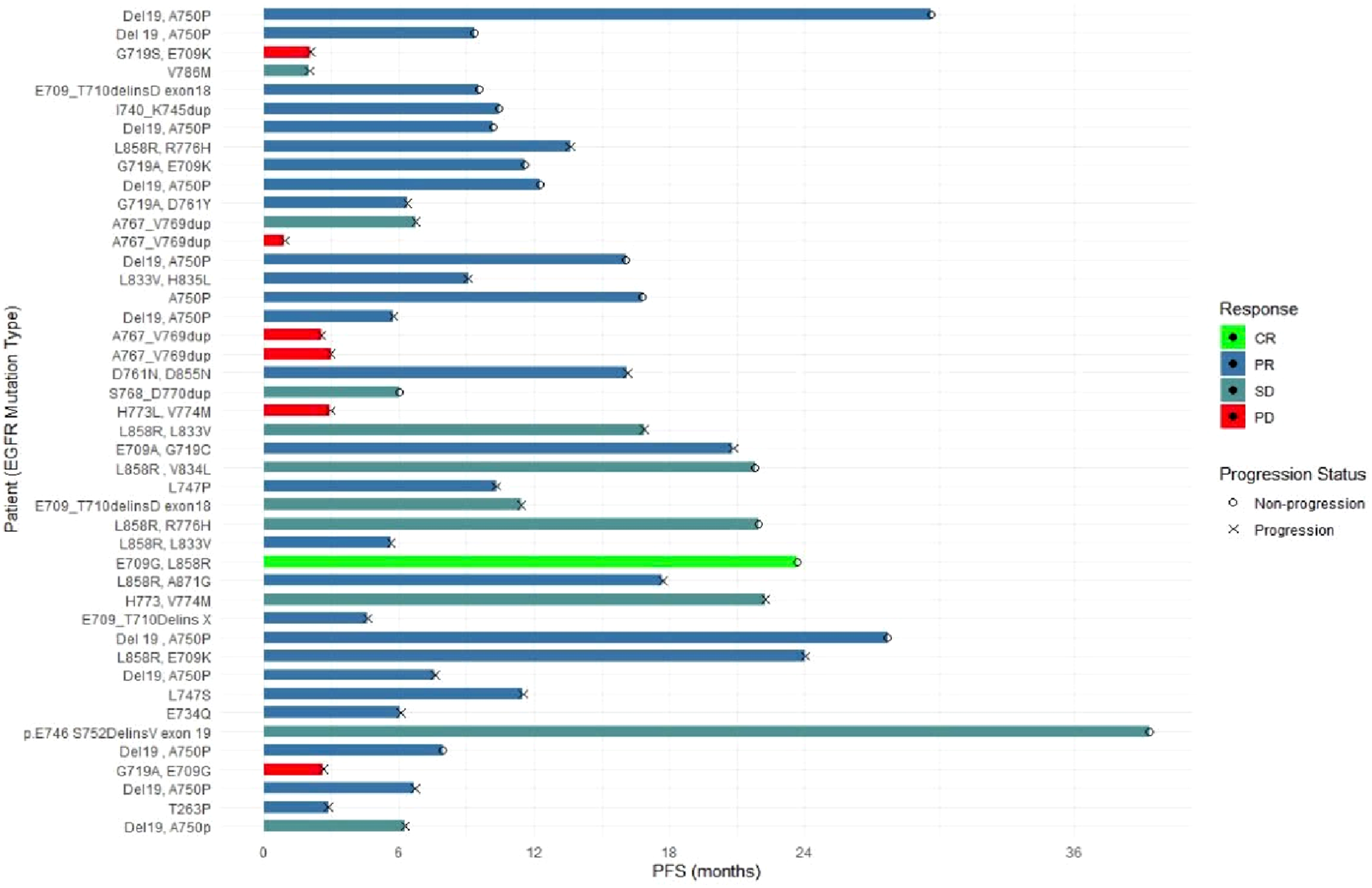

In univariate analysis, patients with non-major uncommon mutations co-occurring with TKI-sensitive mutations (Del19/L858 R/G719X/L861Q/S768I) had a significantly longer mPFS than those harboring non-major mutations alone (either single or dual) (17.7 vs 9.1 months; log-rank P = 0.044, RMST permutation P = 0.039) (Table 3). The details of the RMST by baseline characteristics are presented in Supplemental Table S2. In the multivariable Cox model, co-occurrence of Del19/L858 R/G719X/L861Q/S768I yielded an adjusted HR of 0.50 (95% CI, 0.22-1.11; P = 0.087). No other covariates showed statistically significant associations with PFS. An exploratory post-hoc power analysis using Schoenfeld’s approximation indicated that, with 28 PFS events among 44 patients and an observed Cox univariate HR of 0.47 between mutation subgroups, the estimated power was ∼51% at α = 0.05.
Duration of Response
The median DOR in NSCLC patients with non-major uncommon EGFR mutations in this study was 16.1 months (95% CI: 10.3–NR), with a median follow-up of 16.8 months (95% CI: 11.6–NR) (Figure 2B).
Toxicity Profile
In our cohort, afatinib was generally well tolerated. No grade 4 toxicities were observed
Discussion
This study enrolled 44 patients with advanced-stage NSCLC and non-major uncommon EGFR mutations. Compound mutations appeared in 63.6% of the cases, most often a non-major uncommon mutation co-occurring with a TKI-sensitive mutation (Del19, L858R, G719X, S768I, and L861Q). Within our cohort, no cases of S768I or L861Q co-occurring with other variants were identified; consequently, these mutations were not represented in the “TKI-sensitive” subgroup. Previous retrospective analyses related to rare, dual, and complex mutations revealed considerable heterogeneity due to numerous factors such as ethnicity, testing methods, and types of genetic testing samples.8,9 In our study, although both tissue and plasma were eligible, all 44 patients were ultimately tested using tissue specimens (43 FFPE tumor samples and 1 FFPE cell block), with no plasma-based liquid biopsy performed. This uniformity in sample type may partly reduce variability, yet it is important to acknowledge that assay platforms, pre-specified thresholds (eg, variant allele fraction cut-offs), and specimen quality can influence detection sensitivity and specificity, thereby contributing to differences across published studies. However, most studies report that the most common form of complex EGFR mutations consists of TKI-sensitive alterations (eg, Del19, L858R, or G719X) co-occurring with rare mutations whose clinical significance remains uncertain. 10 Importantly, the location of EGFR mutations within the kinase domain also plays a pivotal role in determining TKI sensitivity, with certain regions conferring strong responsiveness and others associated with resistance.11,12 This positional effect further highlights the heterogeneity of clinical outcomes and the need for individualized therapeutic strategies.
Although this study focused on non-major uncommon mutations with variable and poorly defined sensitivity to EGFR-TKIs, afatinib achieved favorable outcomes in this population, including a high ORR and DCR and promising median PFS and DOR. Among the 44 patients, the ORR was 65.9%, with a CR of 2.3% and a DCR of 86.4%. The median PFS and DOR were 11.5 months and 16.8 months, respectively. These outcomes are consistent with the findings of the LUX-Lung 2, 3, and 6 trials, which were retrospective analyses of patients with major uncommon EGFR mutations and demonstrated that first-line afatinib achieved an ORR of 71.1%, DCR of 84.2%, and median PFS of 10.7 months. 7 Additionally, a meta-analysis of 693 cases further supports the efficacy of afatinib in this population and reinforces its role as a viable treatment option. 3 In contrast, first-generation TKIs showed lower efficacy in a study by Beau-Faller et al, in which patients with exon 18 and 20 mutations had an ORR of 15% and a DCR of 47%, 13 highlights the limitations of early generation TKIs. Notably, even third-generation osimertinib, despite its superiority in common EGFR mutations, 14 exhibited comparable efficacy only for uncommon mutations in Yusuke Okuma’s study (ORR 55.0%, median PFS 9.4 months, and median DOR 22.7 months). 15 These results support those of Attili’s meta-analysis of 40 global studies, which revealed that patients harboring uncommon EGFR mutations derived superior clinical benefits from second- or third-generation EGFR-TKIs. 16 Our findings reinforce this perspective and emphasize the need for optimally tailored treatment selection for extremely rare EGFR mutations.
In our study, 63.6% of patients with non-major uncommon EGFR mutations harbored co-occurring Del19, L858R, or G719X mutations. Del19 and L858R mutations are highly sensitive to EGFR-TKIs because of their structural features, which enhance their binding affinity and inhibitory activity. 13 G719X, a major and uncommon mutation located in exon 18, involves the substitution of glycine at position 719 with serine, alanine, or cysteine. This alteration destabilizes the inactive αC conformation, thereby enhancing the kinase activity and rendering the receptor more amenable to TKI inhibition. 17 Nevertheless, rare EGFR mutations can alter the structure of the tyrosine kinase domain, leading to heterogeneous sensitivity to TKIs. 18 The coexistence of multiple mutations creates a complex molecular background that makes the efficacy of EGFR-TKIs unpredictable. Afatinib is an irreversible tyrosine kinase inhibitor that targets the ErbB receptor family and blocks the growth and survival signaling in cancer cells. This irreversible binding mechanism also enables a broader inhibitory spectrum against diverse tumor subclones. 19 Preclinical studies have further demonstrated the wide inhibitory activity of afatinib across a range of complex and uncommon EGFR mutations, with 62 of 69 variants being sensitive.16,18,20,21 In our study, patients harboring non-major uncommon mutations co-occurring with Del19, L858R, or G719X achieved an ORR of 75% and a median PFS of 17.7 months, suggesting that afatinib is effective against common sensitive mutations, even when combined with rare variants. Although the adjusted association in the multivariate analyses did not reach conventional statistical significance, the point estimate (HR≈0.5) was consistent with the univariate finding of longer PFS in patients with co-occurring Del19/L858R/G719X. The wide confidence interval crossing unity likely reflects the limited sample size and number of events; therefore, this result should be interpreted as a hypothesis and requires confirmation in larger cohorts.
Furthermore, this retrospective study included various uncommon mutations, some of which have scarcely been reported previously or mentioned only in case series. Among these, treatment efficacy in patients harboring only single/dual non-major uncommon mutations was lower than that in patients with co-occurring TKI-sensitive mutations; however, it remained encouraging, with an ORR of 50% and a median PFS of 9.1 months (95% CI: 4.6–NR). Preclinical studies have indicated that the sensitivity of these rare mutations to EGFR-TKIs is significantly heterogeneous, reflecting differences in receptor dimerization and phosphorylation capabilities.22,23 The wide range of rare mutations presents substantial clinical challenges in identifying the treatment of choice. The extensive diversity of uncommon EGFR mutations, each occurring at a low frequency and rarely represented in clinical trials, poses substantial challenges in selecting optimal treatment strategies. A frequent clinical issue is whether treatment should be initiated with a TKI or chemotherapy, and if a TKI is selected, which agent represents the most appropriate choice. A retrospective Chinese study comparing platinum-based chemotherapy to first-generation TKIs in patients with uncommon mutations reported similarly modest median PFS of 7.1 vs 6.1 months (P = 0.893). 24 In the global UpSwinG real-world cohort of EGFR-mutant NSCLC, afatinib demonstrated a superior median time to treatment failure (TTF) of 11.3 months (95% CI: 8.5-14.9), compared with 8.8 months (95% CI: 6.4-10.7) for first-generation EGFR-TKIs. 9 Data on third-generation EGFR-TKIs, such as osimertinib, in this setting are limited. The prospective UNICORN trial (n = 40-42) reported an ORR of 55% and a median PFS of 9.4 months in previously untreated patients with uncommon EGFR mutations, although the outcomes for non-major uncommon mutations were not specified. 15
This study collected 3 patients with germline mutations: T263P, V786M and R776H. Germline EGFR mutations are ubiquitous from birth. While many act only as predisposing factors requiring additional somatic drivers (eg, Del19, L858R), certain germline variants have been shown to independently confer kinase activation and sensitivity to EGFR-TKIs.25-27 Firstly, the rare EGFR mutation T263P (exon 7) has been previously shown to be sensitive to first- and second-generation EGFR-TKIs28,29; however, in our patient, afatinib yielded only a short-lived response, highlighting the complexity of rare mutation responses and necessitating further investigation to identify the optimal treatment approach. Two other germline mutations on exon 20 of the EGFR gene were identified in our study, namely V786M and R776H. V786M is a germline mutation in exon 20 of the EGFR gene, with some reported cases thus far.30,31 Structural analysis indicated that the V786M mutation may facilitate EGFR dimerization without interfering with TKI binding. The only available information regarding TKI treatment for this mutation is related to gefitinib. 30 Ba/F3 cell assays using the MANO method demonstrated that the V786M mutation is likely to be more susceptible to second- or third-generation EGFR-TKIs than to first-generation agents. 30 Our patient is the first documented case of afatinib treatment. Given its broader inhibitory profile than that of first-generation EGFR-TKIs and its reported efficacy against various uncommon EGFR mutations, afatinib is considered a rational treatment option. However, in the present case, the patient did not achieve a clinical response or meaningful disease control. Together with previous reports, these findings suggest that V786M may confer resistance to EGFR-TKIs and could be better managed with alternatives such as gefitinib or chemotherapy. R776H is a germline mutation in exon 20 of the EGFR gene that frequently co-occurs with other EGFR mutations, such as L858 R, L861Q, and G719A. 32 This co-occurrence suggests that R776H may not be independently oncogenic but could enhance tumorigenicity when combined with other mutations. 32 Clinical studies and case reports have supported this hypothesis. A retrospective study of 44 patients harboring the R776H mutation indicated that this variant may confer sensitivity to certain EGFR tyrosine kinase inhibitors (TKIs), including erlotinib, afatinib, and osimertinib, although the clinical responses vary depending on the coexisting mutations. 33 Guo et al described a case of L861Q NSCLC that responded to afatinib, and Kobayashi et al reported 3 R776H cases: 2 with L858R and 1 with G724S, all of which responded to erlotinib or afatinib.34,35 Consistent with these findings, our patient with the R776H/L858R compound mutation achieved a partial response when treated with afatinib and maintained an ongoing clinical benefit for 16 months. Collectively, these cases suggest that the presence of R776H, even when accompanied by a common activating mutation, does not preclude sensitivity to EGFR-TKIs.
In exon 18 of EGFR, E709-T710delins is classified as a complex insertion–deletion (indel) mutation, typically occurring as a single mutation. Its sensitivity to EGFR-TKIs has been reported to be heterogeneous, with some clinical cases suggesting greater sensitivity to afatinib than to first-generation TKIs. 36 By contrast, also located in exon 18, E709X is a missense mutation that typically occurs in the compound form. Single E709X mutations show poor responses to first-generation TKIs, whereas compound forms with L858R or G719X mutations respond favorably (approximate ORR, 69%). 37 Although limited data are available regarding the efficacy of afatinib, individual case reports have indicated positive outcomes. 38 In our cohort, 3 patients harboring E709-T710delins responded well to afatinib treatment; notably, 1 of them achieved a partial response with complete disappearance of the target lesion, and PFS remained at the time of analysis. Additionally, 3 patients with compound mutations involving E709X (in combination with L858R or G719A) received afatinib therapy. Two patients (E709K/L858R and E709G/L858R) responded well, including 1 with a PR lasting 16 months. Our results suggest that afatinib may be beneficial for treating patients with compound E709X mutations, despite heterogeneous responses.
The point mutations in exon 19 included I740_K745dup, A750P, L747X, and p.E746_S752delinsV. One patient with an uncommon exon 19 insertion mutation (I740_K745dup) achieved PR with afatinib. Exon 19 insertions are extremely rare (approximately 0.14% of all EGFR mutations), with I740_K745dup being the most common (88%). 39 Previous reports have indicated the limited efficacy of gefitinib, with a PFS of 4 months, but improved outcomes with afatinib (median PFS of 13.4 months). 40 At the time of data collection for analysis (after 5 months of treatment), our patient maintained ongoing PR without progression, supporting afatinib as a favorable treatment option.
The non-major uncommon exon 19 point mutation A750P, which involves the substitution of alanine with proline, differs from classic Del19 mutations in terms of EGFR-TKI sensitivity. 41 Although no direct structural data currently exist for the A750P variant, mechanistic modeling based on structurally analogous mutations suggests that the substitution of proline at the β3–αC loop may enhance afatinib binding without significantly disrupting the EGFR kinase domain conformation. 42 This provides a plausible explanation for the clinically observed TKI sensitivity for A750P, particularly favoring afatinib over first- or third-generation inhibitors. In a retrospective analysis of 14 429 Chinese patients with lung cancer (2017-2019), all of whom underwent molecular profiling using targeted next-generation sequencing (NGS) of tissue and/or liquid biopsy samples, the A750P mutation was identified in 0.51% of cases and classified as EGFR-TKI-sensitive. 43 Previous molecular studies have reported the limited efficacy of gefitinib.40,44 In our cohort of 12 patients (11 with combined A750P/Del19 and 1 with A750P alone), afatinib treatment resulted in a partial response (PR) in eleven of them, including single patients (Figure 1). The remaining patients achieved SD, with a DCR of 100%. One patient with a single A750P mutation did not exhibit disease progression, even after 16 months of afatinib treatment. These findings strongly support the use of afatinib to treat patients with A750P mutations.
L747X includes a group of exon 19 EGFR mutations, such as L747-P753 > S, L747-A750 > P, and L747-S752, in which leucine at position 747 is altered. These mutations result in varying degrees of sensitivity to EGFR-TKIs. For example, L747-A750 > P is sensitive to afatinib but is only modestly inhibited by erlotinib or osimertinib.10,42,44 In our study, 3 patients with L747X mutations achieved disease control: 1 had a PR lasting 13 months before progression, whereas the other 2 had SD; 1 progressed after 10 months, and the other remained progression-free at 14 months.
Similarly, p.E746_S752delinsV is another rare mutation located in exon 19 of EGFR, with limited clinical data. Previous case reports have reported encouraging responses to first-generation EGFR-TKIs. A report by Yamin Meng documented a partial response lasting 19 months to icotinib (a first-generation EGFR-TKI) in advanced NSCLC. 45 Another study by Weitao Zhuang et al reported a complete response to gefitinib in a male patient with stage IIIB squamous cell carcinoma harboring this mutation. 46 In our study, however, the patient with this mutation progressed early during afatinib treatment, suggesting that gefitinib may be a more effective option for this variant.
In our study, 2 patients harboring the rare complex EGFR mutation H733L/V774M (exon 20) showed no response or stable disease with afatinib, which is consistent with previous reports indicating the limited efficacy of afatinib for this mutation. 47 Conversely, osimertinib previously demonstrated favorable outcomes for similar mutations, suggesting that osimertinib might be preferable. 48
Exon 20 insertions account for approximately 1-10% of EGFR mutations, with the most common variants being Asp770_Asn771ins and Val769_Asp770ins.49,50 These mutations confer primary resistance to EGFR-tyrosine kinase inhibitors (EGFR-TKIs). A retrospective analysis by Yang et al. from the Lux-Lung study revealed poor outcomes with afatinib in patients harboring isolated exon 20 insertions, with an ORR of 2.7%, mPFS of 2.7 months, and mOS of 9.2 months. 7 Although newer therapies, such as amivantamab, have been approved for these patients, 51 access remains limited in Vietnam, and some patients decline or are ineligible for platinum-based chemotherapy. In contrast to the general resistance of patients to exon 20 insertions, emerging evidence suggests that certain EGFR subtypes retain EGFR-TKI sensitivity. 11 Notably, the A763_Y764insFQEA variant has a 10-fold greater binding affinity for first-generation EGFR-TKIs than other exon 20 insertions. 52 Additionally, case reports have described afatinib sensitivity in patients with mutations such as H773dup, H773_V774insNPH, and N771delinsKG, although the mechanisms involved remain unclear.53,54 In a study of 42 patients, James et al reported that insertions at A763, M766, N771, and V769 responded to afatinib, with TTFs of 8.0-39.0 months and ORRs of 50-100%. 55 In the present study, exon 20 insertion mutations were included only if specific subtypes could be identified. Among the patients with exon 20 insertions, 3 with A767_V769dup mutations experienced disease progression. The other 2, S768_D770dup and A767_V769dup, showed a stable disease (Figure 1). Mutations such as A767_V769dup and S768_D770dup were associated with poor TKI response, consistent with previous studies. These findings highlight the need for precise exon 20 insertion subtyping using next-generation sequencing (NGS) and emphasize the limited efficacy of second-generation TKIs in this subgroup.
These findings highlight the need for precise exon 20 insertion subtyping using next-generation sequencing (NGS) and emphasize the limited efficacy of first-generation TKIs in this subgroup, driven by the inclusion of extremely rare EGFR mutations. Additionally, the extended enrollment period led to variable follow-up durations, which limited long-term outcome assessments. Future studies with larger cohorts and longer follow-up periods are crucial to obtain more detailed subgroup analyses and to draw stronger clinical conclusions.
A871G is a rare exon 21 EGFR mutation often found in combination with L858R, forming compound mutations that may influence EGFR-TKI sensitivity. Reports suggest that patients with the A871G/L858R mutation may exhibit poor responses to erlotinib, leading to rapid disease progression, whereas alternative EGFR TKIs, such as icotinib and gefitinib, have demonstrated better efficacy.56,57 In our study, a patient with A871G/L858R achieved PR with afatinib and maintained disease control for 19 months, emphasizing the complexity of TKI sensitivity in patients with compound EGFR mutations.
L833V, a rare exon 21 EGFR mutation, often coexists with other mutations, such as H835L or V774M. Reports have suggested encouraging responses to gefitinib, osimertinib, and almonertinib, with a Chinese case demonstrating the efficacy of afatinib combined with anlotinib in L833V/H835L patients.58-61 In our cohort, 2 patients with L833V (1 with L858R and 1 with H835L) were treated with afatinib monotherapy and achieved PR for 6 and 9 months, respectively. Although the response times were shorter than those observed in patients with common EGFR mutations, our findings indicated that afatinib could be considered a viable treatment option over chemotherapy.
Other non-major, uncommon EGFR mutations include E734Q (exon 18), D761Y/D761N (exon 19), and D855N (exon 21), with limited data on their prevalence and TKI sensitivity.62-64 In our study, the patient with the E734Q mutation partially responded to afatinib treatment. Another patient with D761 N/D855N achieved a partial response and remained progression-free for 15 months, whereas a D761Y/G719A patient presented with SD for 9 months. These cases expand our current understanding of second-generation EGFR-TKI activity in patients with rare EGFR mutations.
Rare EGFR mutations are biologically heterogeneous with unclear oncogenic roles, diverse responses to EGFR-TKIs, and uncertain treatment sequences after resistance, creating major challenges in clinical practice.11,12 Multiomics approaches offer a promising solution by integrating genomic, transcriptomic, proteomic, and metabolomic data to refine mutation classifications, uncover resistance mechanisms, and guide molecular feature–based treatment strategies beyond traditional mutation typing.65-68
Adverse Events Related to Treatment

This study had some limitations. First, as a single-center retrospective cohort study, it may be affected by selection bias from referral patterns and clinicians’ tendency to prescribe afatinib to patients deemed suitable. Second, information bias is possible due to reliance on medical records, non-blinded investigator-assessed RECIST 1.1 responses, and ±1-month imaging intervals. Third, confounding by both measured and unmeasured factors (eg, ECOG status, tumor burden, brain metastasis, co-alterations, dosing, and subsequent therapies) is possible. The modest sample size and heterogeneous EGFR subtypes limited robust multivariate adjustment; therefore, the subgroup findings were exploratory. Our exploratory post-hoc power analysis, based on Schoenfeld’s approximation, indicated only ∼51% power (α = 0.05) with 28 PFS events among 44 patients and an observed HR of 0.47. This limited statistical power highlights the weakness of subgroup analyses in our study. Fourth, the results from this Vietnamese tertiary center may not be generalizable to settings with different EGFR variant profiles, testing platforms, TKI access, dosing strategies, or supportive care. Finally, a shorter follow-up for patients enrolled later could bias time-to-event outcomes by using the reverse Kaplan–Meier to report median follow-up and emphasize ORR, which is less time-sensitive. Overall, these limitations suggest that our findings are hypothesis-generating and should be validated in larger, multi-center cohorts with standardized testing and independent review.
Conclusion
Our study analyzed non-major and uncommon EGFR mutations in NSCLC and their association with afatinib treatment outcomes, including ORR, PFS, and DOR. These findings highlight the heterogeneity of mutation patterns and treatment outcomes and emphasize the importance of personalized therapeutic strategies. Overall, our findings suggest that afatinib remains a viable treatment option for select patients with non-major uncommon EGFR mutations, although further research is needed to guide treatment in subgroups with limited TKI responsiveness.
Supplemental Material
Supplemental Material - Real-World Evidence for First-Line afatinib in Advanced Non-Small Cell Lung Cancer With Uncommon Epidermal Growth Factor Receptor Mutations Other than G719X/L861Q/S768I
Supplemental Material for Real-World Evidence for First-Line afatinib in Advanced Non-Small Cell Lung Cancer With Uncommon Epidermal Growth Factor Receptor Mutations Other than G719X/L861Q/S768I by Thanh Ha Vu, Hoa Thai Thi Nguyen, To Van Ta, Minh Cong Truong, Thang Bui Xuan, Anh Tuan Nguyen, Cao Van Nguyen, Hang Thuy Nguyen, Tuyet Thi Doan, Thanh Thi Vu, Dung Khac Dinh, Giang Vinh Le, Hung Huy Hoang, Chi Khanh Duong, Linh Khanh Dao in Cancer Control.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
